Submitted:
17 April 2026
Posted:
20 April 2026
You are already at the latest version
Abstract
Phycobiliproteins (PBPs) are a family of pigment-proteins renowned for their exceptional light-harvesting, fluorescent, and antioxidant properties. Among cyanobacteria, Spirulina stands out as one of the richest natural sources of PBPs, particularly phycocyanin (PC) and allophycocyanin (APC), yet the large-scale production of analytical-grade PBPs remains hampered by an inherently complex downstream process that relies on multiple purification steps, compromising both yield and scalability. This work presents a streamlined strategy to obtain analytical-grade PC, combining ultrasound-assisted extraction (UAE) with an aqueous ionic liquid (IL) solution and a single hydrophobic interaction chromatography (HIC) step, integrated within a biorefinery framework. The proposed approach yielded analytical-grade PC with a recovery of up to 50.44% and enhanced APC purity up to 10.57-fold. Therefore, the IL was successfully reused in both extraction and purification steps without compromising yield or purity. The environmental performance of the proposed process was assessed through a cradle-to-gate life cycle assessment (LCA), with system boundaries encompassing the following biorefinery stages: cultivation, harvesting and drying, PC extraction and purification, post-processing, and spent biomass valorization via anaerobic digestion. The LCA identified the main environmental hotspots and guided the proposal of targeted process improvements—particularly HIC salt substitution and increased IL recovery—which reduced environmental impacts by 65.9–89.8% across most categories. The proposed strategy was further benchmarked against two model scenarios for analytical-grade PC production, one conventional and one innovative, revealing its relative advantages and limitations. Overall, this work demonstrates a viable pathway for producing high-purity PC that balances process efficiency with environmental sustainability, supporting the development of greener microalgae-based bioprocesses.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. PC-APC Extraction
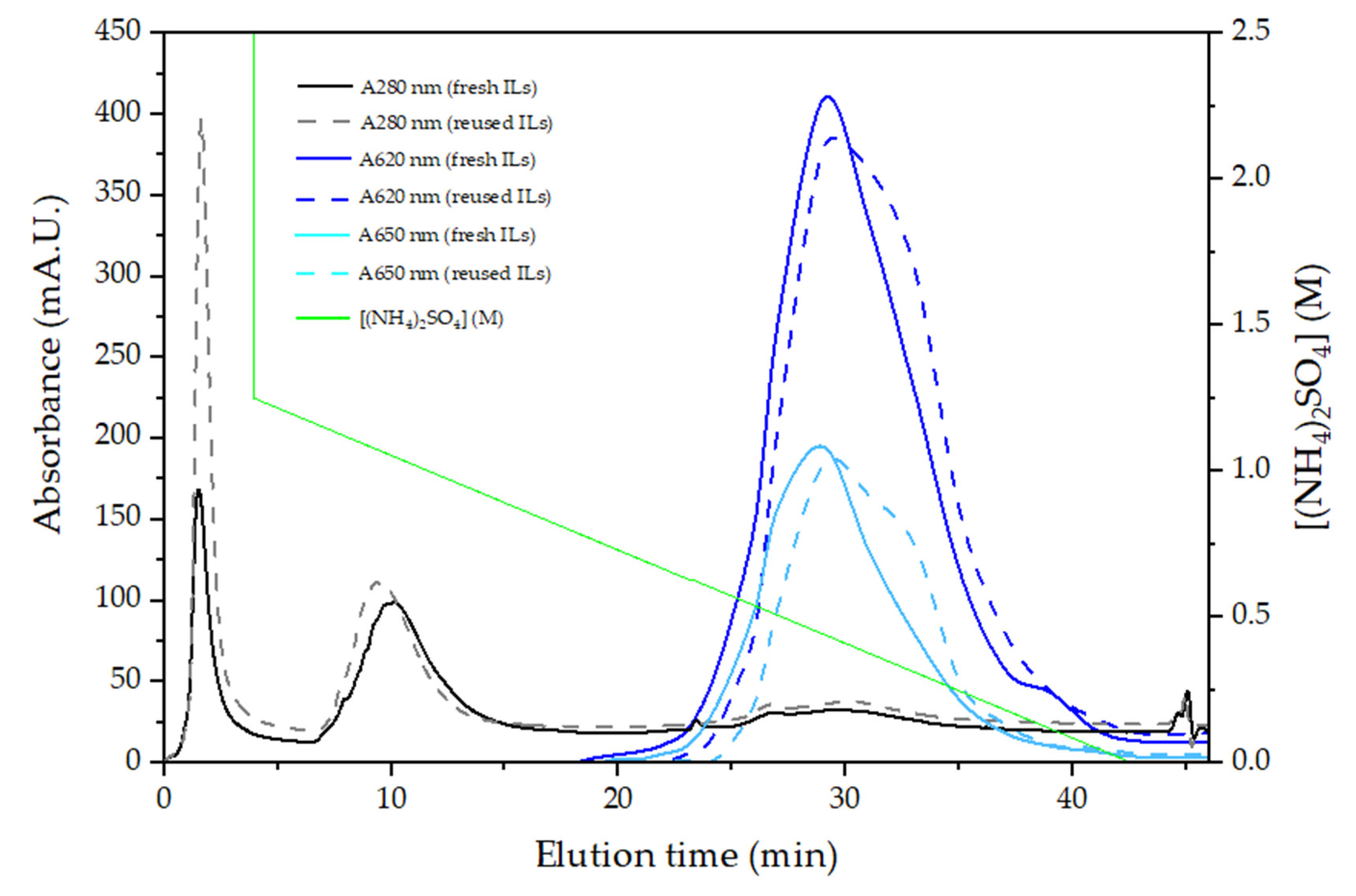
2.2.2. PC-APC Purification
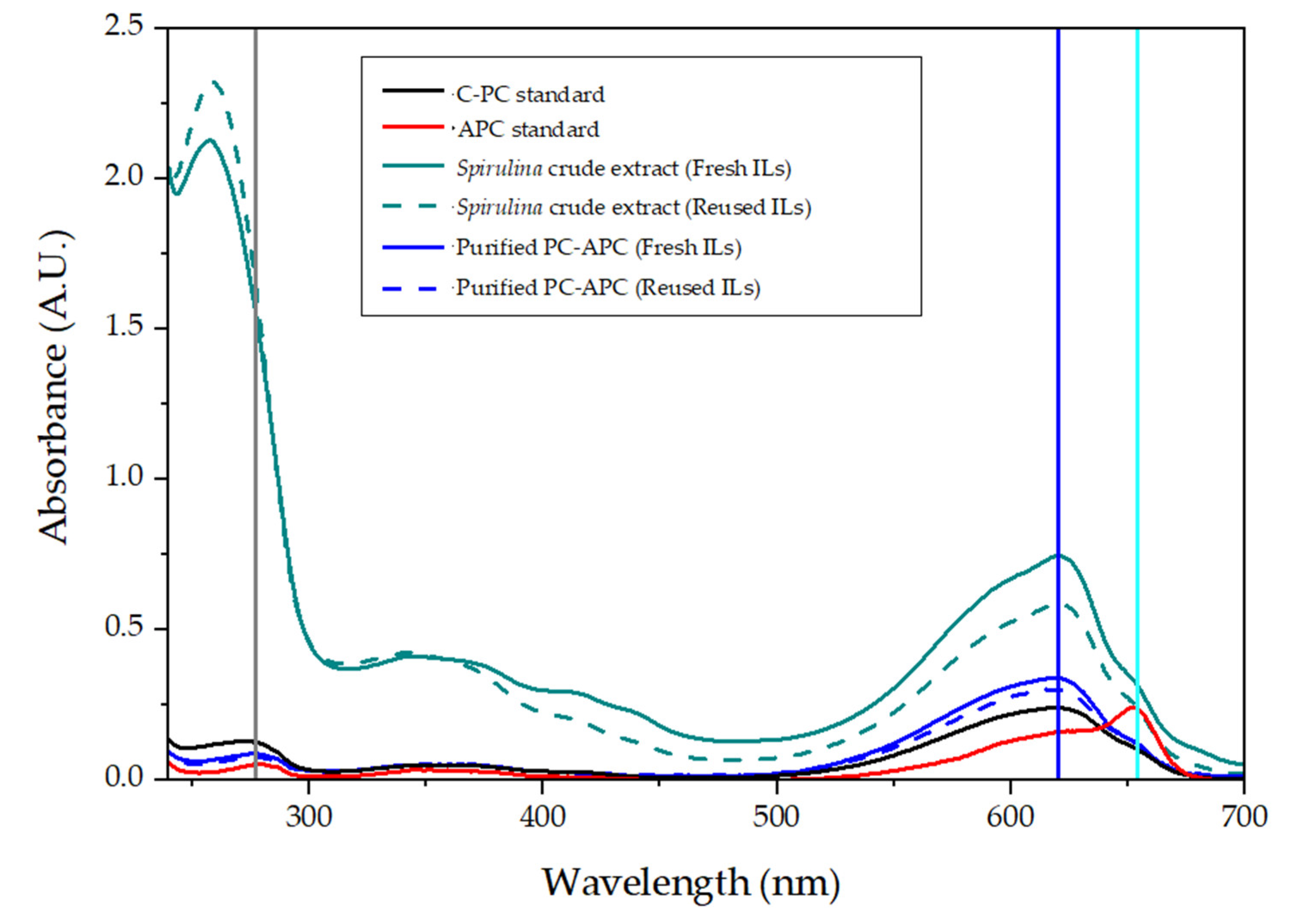
2.2.3. PC and APC Characterization
2.2.4. IL Recovery
2.2.5. Goal and Scope of the LCA Study
2.2.6. System Boundaries
2.2.7. Life Cycle Inventory (LCI)
2.2.8. Modeling Biorefinery Process Stages
2.2.9. Methodology of Environmental Impact Assessment
3. Results
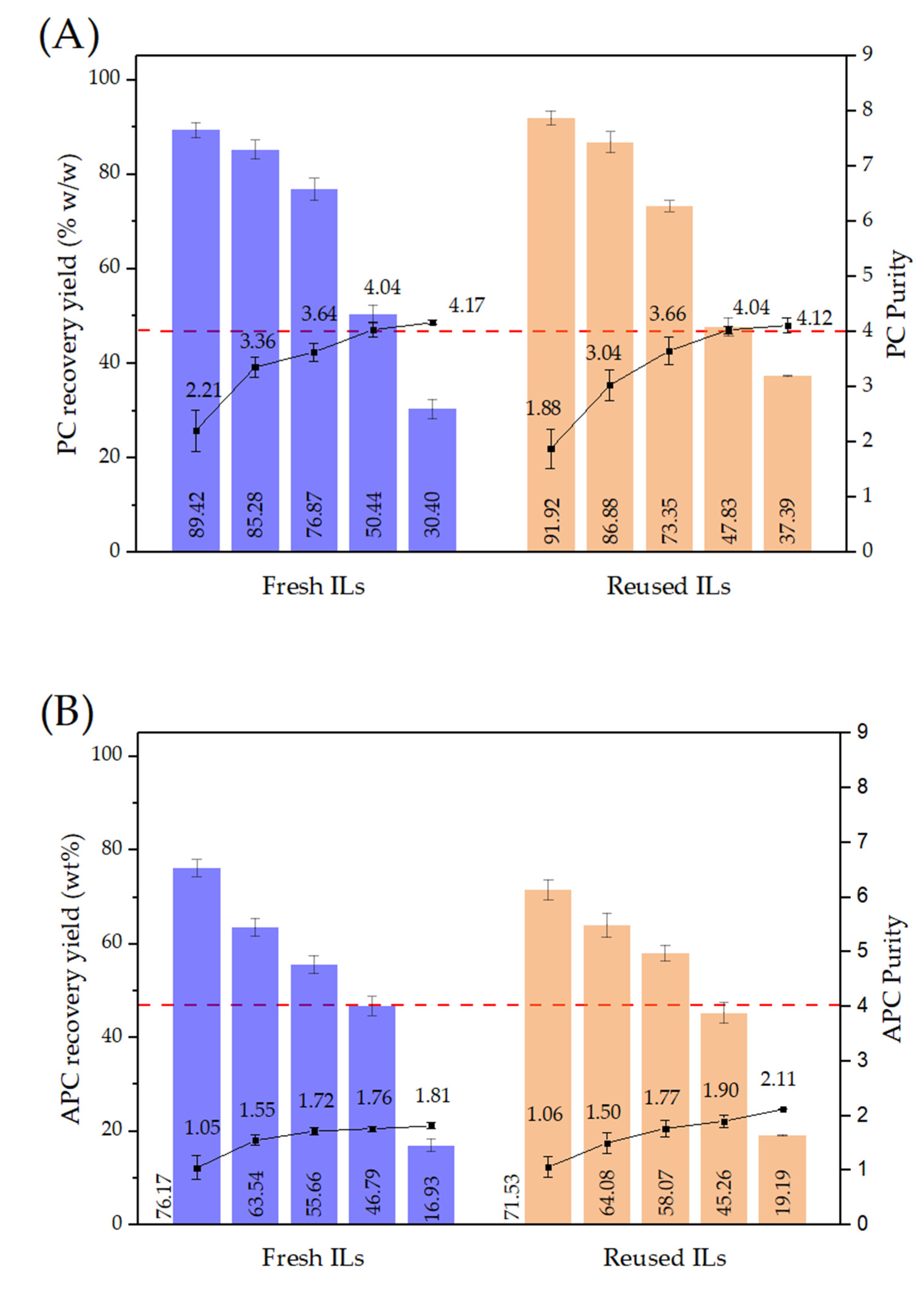
3.1. PC-APC Extraction
3.2. PC-APC Purification
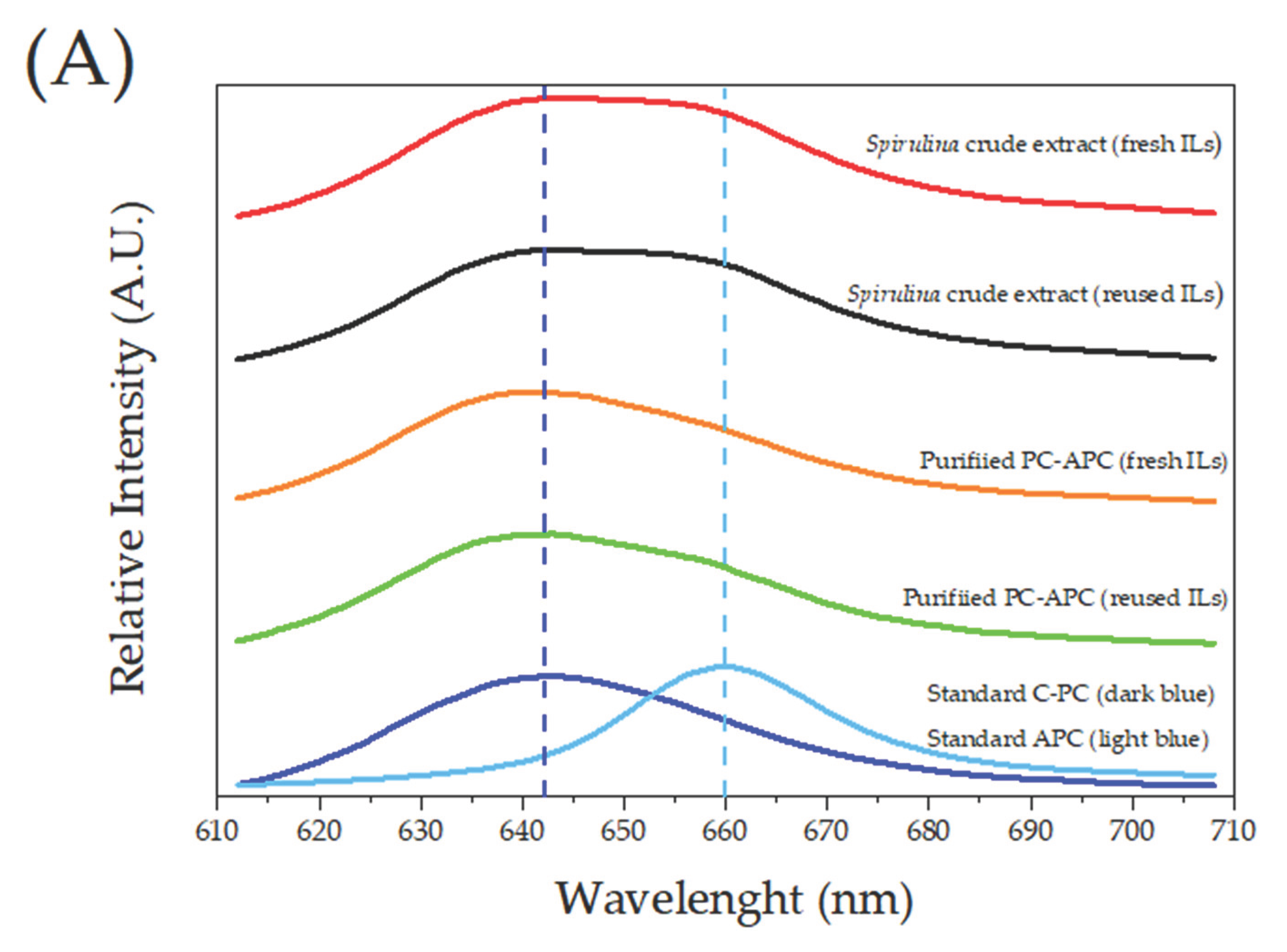
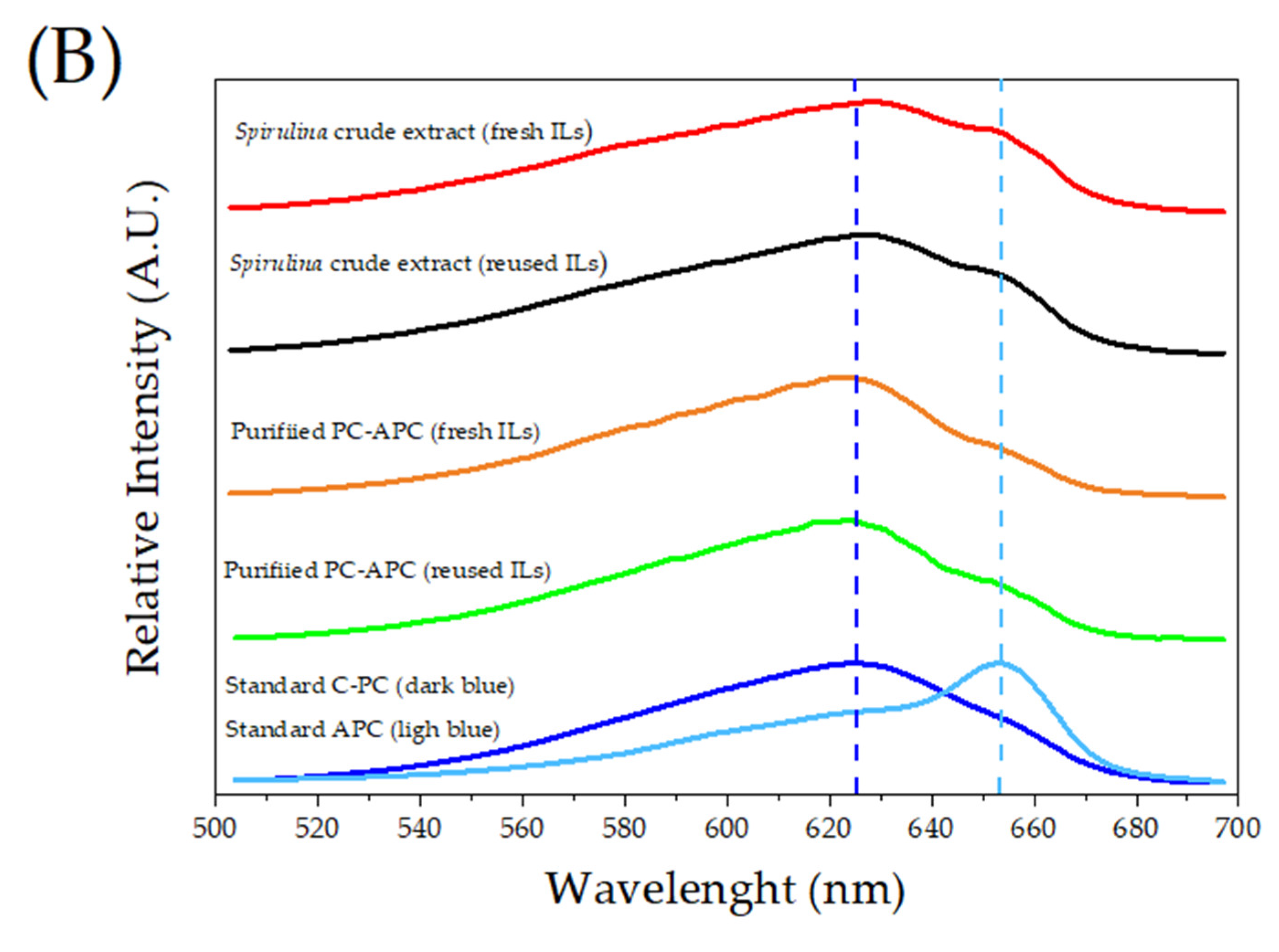
3.3. PC and APC Characterization
3.4. Life Cycle Assessment (LCA)
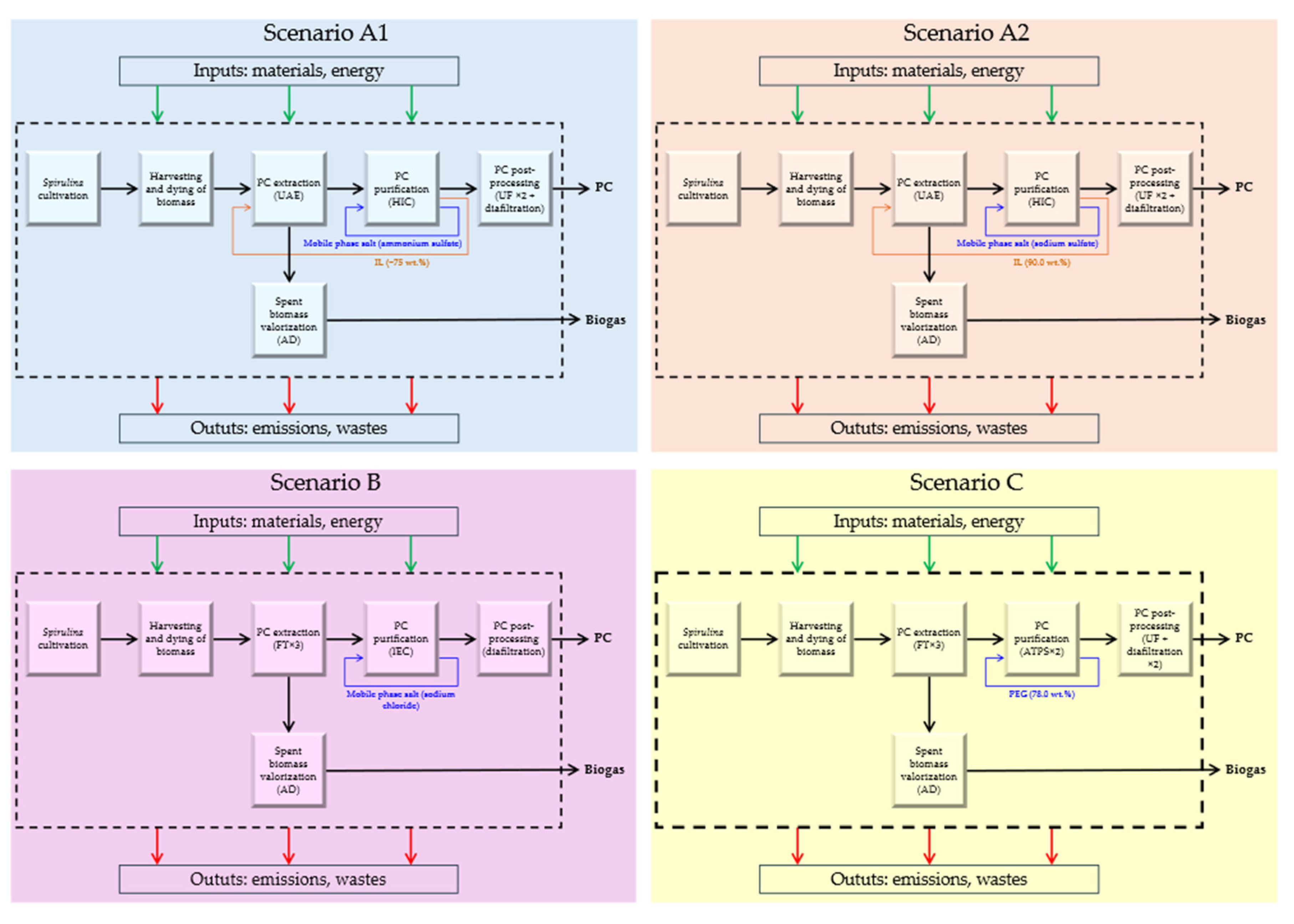
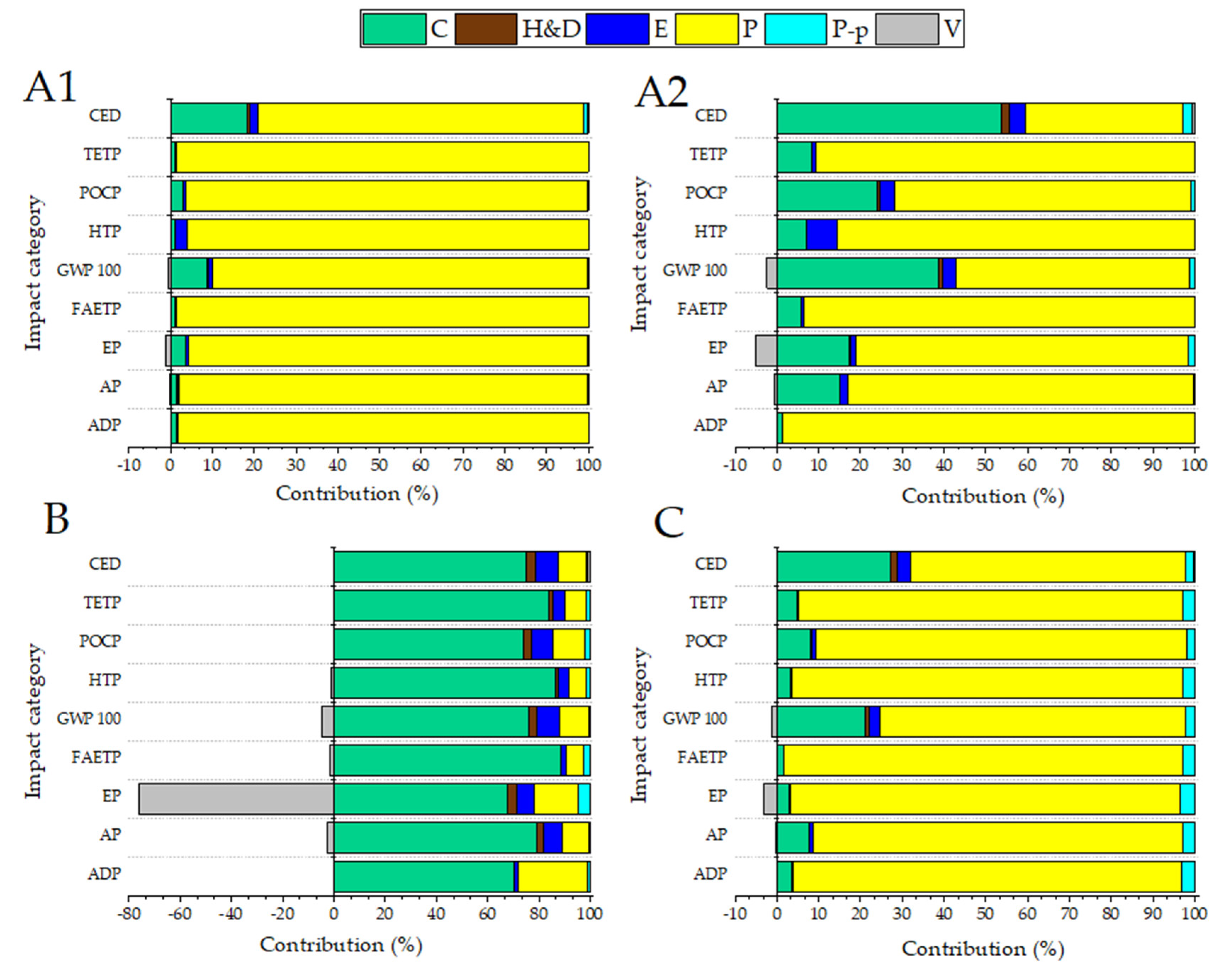
3.4.1. Scenarios A1 and A2
3.4.2. Scenarios B and C
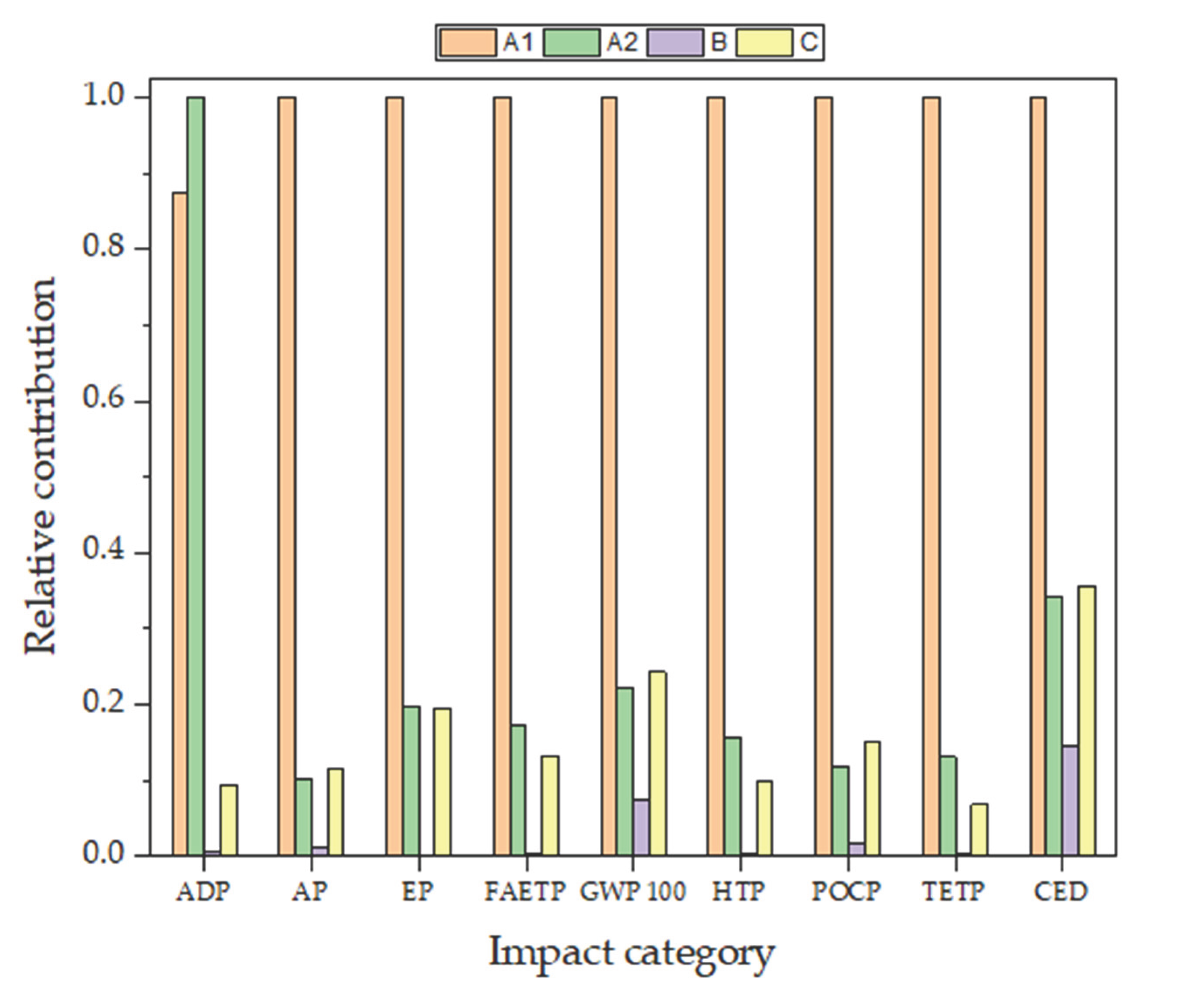
3.4.3. Comparison of Scenarios
4. Discussion
4.1. PC-APC Extraction and Purification
4.2. LCA
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AD | Anaerobic digestion |
| ADP | Abiotic depletion potential |
| AP | Acidification potential |
| APC | Allophycocyanin |
| ATPS | Aqueous two-phase system |
| BV | Bed volume |
| C | Cultivation stage |
| CED | Cumulative energy demand |
| EPC | Phycocyanin extraction stage |
| EAPC | Extracted allophycocyanin |
| EP | Eutrophication potential |
| EPC | Extracted phycocyanin |
| FAETP | Freshwater aquatic ecotoxicity |
| FT | Freeze and thaw |
| GWP 100 | 100-year global warming potential |
| H&D | Harvesting and drying stage |
| HIC | Hydrophobic interaction chromatography |
| HTP | Human toxicity potential |
| IEC | Ionic exchange chromatography |
| IL | Ionic liquid |
| LCA | Life cycle assessment |
| LCI | Life cycle inventory |
| OD | Optical density |
| P | Phycocyanin purification stage |
| PAPC | Purity of allophycocyanin |
| PC | Phycocyanin |
| PEG | Polyethylene glycol |
| PF | Purification factor |
| POCP | Photochemical ozone creation potential |
| P-p | Phycocyanin post-processing stage |
| PPC | Purity of phycocyanin |
| TETP | Terrestrial ecotoxicity potential |
| UAE | Ultrasound-assisted extraction |
| UF | Ultrafiltration |
| V | Spent biomass valorization |
References
- Costa, 1 J.A.V.; Freitas, B.C.B.; Rosa, G.M.; Moraes, L.; Morais, M.G.; Mitchell, B.G. Operational and economic aspects of Spirulina-based biorefinery. Bioresour. Technol. 2019, 292, 121946. [Google Scholar] [CrossRef]
- Braud, 2 L.; McDonnell, K.; Murphy, F. Beyond phycocyanin: Environmental life cycle assessment of a European pilot scale Spirulina biorefinery. Bioresour. Technol. Rep. 2025, 29, 102027. [Google Scholar] [CrossRef]
- Thevarajah, 3 B.; Nishshanka, G.K.S.H.; Premaratne, M.; Nimarshana, P.H.V.; Nagarajan, D.; Chang, J.-S.; Ariyadasa, T.U. Large-scale production of Spirulina-based proteins and c-phycocyanin: A biorefinery approach. Biochem. Eng. J. 2022, 185, 108541. [Google Scholar] [CrossRef]
- Ashaolu, 4 T.J.; Samborska, K.; Lee, C.C.; Tomas, M.; Capanoglu, E.; Tarhan, Ö; Taze, B.; Jafari, S.M. Phycocyanin, a super functional ingredient from algae; properties, purification characterization, and applications. Int. J. Biol. Macromol. 2021, 193, 2320–2331. [Google Scholar] [CrossRef]
- Fernández-Rojas, 5 B.; Hernández-Juárez, J.; Pedraza-Chaverri, J. Nutraceutical properties of phycocyanin. J. Funct. Foods 2014, 11, 375–392. [Google Scholar] [CrossRef]
- Moraes, 6 C.C.; Kalil, S.J. Strategy for a protein purification design using C-phycocyanin extract. Bioresour. Technol. 2009, 100, 5312–5317. [Google Scholar] [CrossRef]
- de Amarante, 7 M.C.A.; Braga, A.R.C.; Sala, L.; Moraes, C.C.; Kalil, S.J. Design strategies for C-phycocyanin purification: Process influence on purity grade. Sep. Purif. Technol. 2020, 252, 117453. [Google Scholar] [CrossRef]
- Chaiklahan, 8 R.; Chirasuwan, N.; Loha, V.; Tia, S.; Bunnag, B. Separation and purification of phycocyanin from Spirulina sp. using a membrane process. Bioresour. Technol. 2011, 102, 7159–7164. [Google Scholar] [CrossRef]
- Lauceri, 9 R.; Chini Zittelli, G.; Torzillo, G. A simple method for rapid purification of phycobiliproteins from Arthrospira platensis and Porphyridium cruentum biomass. Algal Res. 2019, 44, 101685. [Google Scholar] [CrossRef]
- Tang, 10 Z.; zhao, Jilu; Ju, B.; Li, W.; Wen, S.; Pu, Y.; Qin, S. One-step chromatographic procedure for purification of B-phycoerythrin from Porphyridium cruentum. Protein Expr. Purif. 2016, 123, 70–74. [Google Scholar] [CrossRef]
- Mogany, 11 T.; Kumari, S.; Swalaha, F.M.; Bux, F. Extraction and characterisation of analytical grade C-phycocyanin from Euhalothece sp. J. Appl. Phycol. 2019, 31, 1661–1674. [Google Scholar] [CrossRef]
- Roy, 12 D.; Pabbi, S. A simple improved protocol for purification of C-phycocyanin from overproducing cyanobacteria and its characterization. J. Appl. Phycol. 2022, 34, 799–810. [Google Scholar] [CrossRef]
- Patil, 13 G.; Chethana, S.; Sridevi, A.S.; Raghavarao, K.S.M.S. Method to obtain C-phycocyanin of high purity. J. Chromatogr. A 2006, 1127, 76–81. [Google Scholar] [CrossRef]
- Zang, 14 F.; Qin, S.; Ma, C.; Li, W.; Lin, J. Preparation of high-purity R-phycoerythrin and R-phycocyanin from Pyropia yezoensis in membrane chromatography. J. Appl. Phycol. 2020, 32, 3411–3418. [Google Scholar] [CrossRef]
- Lauceri, 15 R.; Cavone, C.; Chini Zittelli, G.; Kamburska, L.; Musazzi, S.; Torzillo, G. High purity grade phycocyanin recovery by decupling cell lysis from the pigment extraction: an innovative approach. Food Bioproc. Tech. 2023, 16, 111–121. [Google Scholar] [CrossRef]
- Scorza, 16 L.C.T.; Simon, U.; Wear, M.; Zouliatis, A.; Dimartino, S.; McCormick, A.J. Evaluation of novel 3D-printed monolithic adsorbers against conventional chromatography columns for the purification of c-phycocyanin from Spirulina. Algal Res. 2021, 55, 102253. [Google Scholar] [CrossRef]
- O'Connor, 17 B.F.; Cummins, P.M. Hydrophobic interaction chromatography. In Protein Chromatography, Methods in Molecular Biology; Walls, D., Loughran, S.T., Eds.; Humana Press (New York), 2017; vol. 1485, pp. 355–363. [Google Scholar] [CrossRef]
- 18. H.P. Jennissen, Hydrophobic interaction chromatography. In Encyclopedia of life sciences; Wiley (Chichester), 2016; pp. 1–12. [CrossRef]
- 19. J.T. McCue, Theory and use of hydrophobic interaction chromatography in protein purification applications. In Methods in Enzymology; Burgess, R.R., Deutscher, M.P., Eds.; Academic Press (New York), 2009; Volume 463, pp. 405–414. [Google Scholar] [CrossRef]
- Ebrahimi, 20; Pazuki, G.; Mozaffarian, M.; Ahsaie, F.G.; Abedini, H. Separation and purification of C-Phycocyanin from Spirulina platensis using aqueous two-phase systems based on triblock thermosensitive copolymers. Food Bioproc. Tech. 2023, 16, 2582–2597. [Google Scholar] [CrossRef]
- Porav, 21 A.S.; Bocăneală, M.; Fălămaş, A.; Bogdan, D.F.; Barbu-Tudoran, L.; Hegeduş, A.; Dragoş, N. Sequential aqueous two-phase system for simultaneous purification of cyanobacterial phycobiliproteins. Bioresour. Technol. 2020, 315, 123794. [Google Scholar] [CrossRef] [PubMed]
- Patil, 22 G.; Raghavarao, K.S.M.S. Aqueous two phase extraction for purification of C-phycocyanin. Biochem. Eng. J. 2007, 34, 156–164. [Google Scholar] [CrossRef]
- Arashiro, 23 L.T.; Boto-Ordóñez, M.; Van Hulle, S.W.H.; Ferrer, I.; Garfí, M.; Rousseau, D.P.L. Natural pigments from microalgae grown in industrial wastewater. Bioresour. Technol. 2020, 303, 122894. [Google Scholar] [CrossRef]
- Cruz, 24 J.D.; Vasconcelos, V. Legal aspects of microalgae in the European food sector. Foods 2023, 13, 124. [Google Scholar] [CrossRef]
- Barros, 25 A.I.; Gonçalves, A.L.; Simões, M.; Pires, J.C.M. Harvesting techniques applied to microalgae: A review. Renewable and Sustainable Energy Reviews 2015, 41, 1489–1500. [Google Scholar] [CrossRef]
- Zhou, 26 S.; Lin, M.; Zhang, X.; Zhan, L.; Li, R.; Wu, Y. Study of life cycle assessment: Transforming microalgae to biofuel through hydrothermal liquefaction and upgrading in organic or aqueous medium. J. Clean. Prod. 2024, 444, 140942. [Google Scholar] [CrossRef]
- Maity, 27 S.; Mallick, N. Unraveling C-phycocyanin extraction by dark incubation from marine cyanobacterium Leptolyngbya valderiana, Sustain. Chem. Pharm. 2023, 31, 100929. [Google Scholar] [CrossRef]
- Kovaleski, 28 G.; Kholany, M.; Dias, L.M.S.; Correia, S.F.H.; Ferreira, R.A.S.; Coutinho, J.A.P.; Ventura, S.P.M. Extraction and purification of phycobiliproteins from algae and their applications. Front. Chem. 2022, 10. [Google Scholar] [CrossRef]
- Alreshidi, 29 M.A.; Yadav, K.K.; Gunasekaran, S.; Gacem, A.; Sambandam, P.; Subbiah, G.; Bhutto, J.K.; Palanivel, S.; Fallatah, A.M.; Abo El-Khair, M.A.; Almalawi, J.F.; Alam, M.W.; Perumal, T.; Annadurai, S. A review on the evolution of ionic liquids: Sustainable synthesis, applications, and future prospects. Materials Today Sustainability 2025, 31, 101160. [Google Scholar] [CrossRef]
- Martins, 30 M.; Vieira, F.A.; Correia, I.; Ferreira, R.A.S.; Abreu, H.; Coutinho, J.A.P.; Ventura, S.P.M. Recovery of phycobiliproteins from the red macroalga Gracilaria sp. using ionic liquid aqueous solutions. Green Chemistry 2016, 18, 4287–4296. [Google Scholar] [CrossRef]
- Antecka, 31; Szeląg, R.; Ledakowicz, S. A novel two-step purification process for highly stable C-phycocyanin of analytical grade purity and its properties. Appl. Microbiol. Biotechnol. 2025, 109 72. [Google Scholar] [CrossRef] [PubMed]
- Patil, 32 G.; Chethana, S.; Madhusudhan, M.C.; Raghavarao, K.S.M.S. Fractionation and purification of the phycobiliproteins from Spirulina platensis. Bioresour. Technol. 2008, 99, 7393–7396. [Google Scholar] [CrossRef] [PubMed]
- Fabre, 33 J.-F.; Niangoran, N.U.F.; Gaignard, C.; Buso, D.; Mouloungui, Z.; Valentin, R. Extraction, purification and stability of C-phycocyanin from Arthrospira platensis. European Food Research and Technology 2022, 248, 1583–1599. [Google Scholar] [CrossRef]
- 34. Jungbauer, N. Walch, Buffer recycling in downstream processing of biologics. Curr. Opin. Chem. Eng. 2015, 10, 1–7. [CrossRef]
- Wellhoefer, 35 M.; Sprinzl, W.; Hahn, R.; Jungbauer, A. Continuous processing of recombinant proteins: Integration of refolding and purification using simulated moving bed size-exclusion chromatography with buffer recycling. J. Chromatogr. A 2014, 1337, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Isaksson, 36 M.; Andersson, N.; Nilsson, B. Improving the sustainability of biopharmaceutical downstream processing through buffer recycling. J. Chromatogr. A 2025, 1740, 465545. [Google Scholar] [CrossRef] [PubMed]
- Imura, 37 Y.; Tagawa, T.; Miyamoto, Y.; Nonoyama, S.; Sumichika, H.; Fujino, Y.; Yamanouchi, M.; Miki, H. Washing with alkaline solutions in protein A purification improves physicochemical properties of monoclonal antibodies. Sci. Rep. 2021, 11, 1827. [Google Scholar] [CrossRef]
- Grönberg, 38; Eriksson, M.; Ersoy, M.; Johansson, H.J. A tool for increasing the lifetime of chromatography resins. MAbs 2011, 3, 192–202. [Google Scholar] [CrossRef]
- Zhao, 39 L.; Peng, Y.; Gao, J.; Cai, W. Bioprocess intensification: an aqueous two-phase process for the purification of C-phycocyanin from dry Spirulina platensis. European Food Research and Technology 2014, 238, 451–457. [Google Scholar] [CrossRef]
- Zeng, 40 W.; Luo, J.; Grimi, N. Maximizing C-phycocyanin purification efficiency from Spirulina: A synergistic strategy combining CaCl2 precipitation and membrane diafiltration. Sep. Purif. Technol. 2025, 361, 131575. [Google Scholar] [CrossRef]
- Boi, 41 C.; Malavasi, A.; Carbonell, R.G.; Gilleskie, G. A direct comparison between membrane adsorber and packed column chromatography performance. J. Chromatogr. A 2020, 1612, 460629. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Laso, 42 J.; Piera, A.; Vicente, G.; Bautista, L.F.; Rodríguez, R.; Espada, J.J. A successful method for phycocyanin extraction from Arthrospira platensis using [Emim] [EtSO4] ionic liquid. Biofuels, Bioproducts and Biorefining 2021, 15, 1638–1649. [Google Scholar] [CrossRef]
- Sánchez-Laso, 43 J.; Espada, J.J.; Rodríguez, R.; Vicente, G.; Bautista, L.F. Novel biorefinery approach for phycocyanin extraction and purification and biocrude production from Arthrospira platensis. Ind. Eng. Chem. Res. 2023, 62, 5190–5198. [Google Scholar] [CrossRef]
- Piera, 44; Sainz-Urruela, C.; Espada, J.J.; Morales, V.; Vicente, G.; Bautista, L.F. One-step purification process to obtain analytical grade phycoerythrin from Porphyridium sp. using hydrophobic interaction chromatography (HIC). Algal Res. 2026, 104596. [Google Scholar] [CrossRef]
- 45. Bennett, L. Bogorad, Complementary Chromatic Adaptation in a Filamentous Blue-Green Alga, J. Cell Biol. 1973, 58, 419–435. [CrossRef] [PubMed]
- García, 46; Longo, E.; Murillo, M.; Bermejo, R. Using a B-phycoerythrin extract as a natural colorant: application in milk-based products. Molecules 2021, 26, 297. [Google Scholar] [CrossRef]
- Bermejo Román, 47 R.; Alvárez-Pez, J.M.; Acién Fernández, F.G.; Grima, E. Molina. Recovery of pure B-phycoerythrin from the microalga Porphyridium cruentum. J. Biotechnol. 2002, 93, 73–85. [Google Scholar] [CrossRef]
- 48. A.N. Glazer, Phycobiliproteins — a family of valuable, widely used fluorophores. J. Appl. Phycol. 1994, 6, 105–112. [CrossRef]
- 49. R. Bermejo, Phycocyanins, in: Cyanobacteria. In Wiley; 2014; pp. 209–225. [CrossRef]
- de Amarante, 50 M.C.A.; Corrêa Júnior, L.C.S.; Sala, L.; Kalil, S.J. Analytical grade C-phycocyanin obtained by a single-step purification process. Process Biochemistry 2020, 90, 215–222. [Google Scholar] [CrossRef]
- Sivasankari, 51 S.; Vinoth, M.; Ravindran, D.; Baskar, K.; Alqarawi, A.A.; Abd_Allah, E.F. Efficacy of red light for enhanced cell disruption and fluorescence intensity of phycocyanin. Bioprocess Biosyst. Eng. 2021, 44, 141–150. [Google Scholar] [CrossRef]
- Patel, 52; Mishra, S.; Pawar, R.; Ghosh, P.K. Purification and characterization of C-Phycocyanin from cyanobacterial species of marine and freshwater habitat. Protein Expr. Purif. 2005, 40, 248–255. [Google Scholar] [CrossRef]
- Zhang, 53 Y.-M.; Chen, F. A simple method for efficient separation and purification of c-phycocyanin and allophycocyanin from Spirulina platensis. Biotechnology Techniques 1999, 13, 601–603. [Google Scholar] [CrossRef]
- Yan, 54 S.-G.; Zhu, L.-P.; Su, H.-N.; Zhang, X.-Y.; Chen, X.-L.; Zhou, B.-C.; Zhang, Y.-Z. Single-step chromatography for simultaneous purification of C-phycocyanin and allophycocyanin with high purity and recovery from Spirulina (Arthrospira) platensis. J. Appl. Phycol. 2011, 23, 1–6. [Google Scholar] [CrossRef]
- Raoof, 55; Kaushik, B.D.; Prasanna, R. Formulation of a low-cost medium for mass production of Spirulina. Biomass Bioenergy 2006, 30, 537–542. [Google Scholar] [CrossRef]
- Affan, 56 M.-A.; Lee, D.-W.; Al-Harbi, S.M.; Kim, H.-J.; Abdulwassi, N.I.; Heo, S.-J.; Oh, C.; Park, H.-S.; Ma, C.W.; Lee, H.-Y.; Kang, D.-H. Variation of Spirulina maxima biomass production in different depths of urea-used culture medium. Brazilian Journal of Microbiology 2015, 46, 991–1000. [Google Scholar] [CrossRef]
- 57. M. Al hattab, Microalgae Harvesting Methods for Industrial Production of Biodiesel: Critical Review and Comparative Analysis. Journal of Fundamentals of Renewable Energy and Applications 2015, 5. [CrossRef]
- Sinetova, 58 M.A.; Kupriyanova, E. V.; Los, D.A. Spirulina/Arthrospira/Limnospira—Three Names of the Single Organism. Foods 2024, 13, 2762. [Google Scholar] [CrossRef] [PubMed]
- Lao, 59 I.K.M.; Edullantes, B. Growth, productivity, and size structure of Spirulina strain under different salinity levels: implications for cultivation optimization. Phycology 2025, 5 31. [Google Scholar] [CrossRef]
- Andreeva, 60; Budenkova, E.; Babich, O.; Sukhikh, S.; Ulrikh, E.; Ivanova, S.; Prosekov, A.; Dolganyuk, V. Production, purification, and study of the amino acid composition of microalgae proteins. Molecules 2021, 26, 2767. [Google Scholar] [CrossRef]
- Ronda, 61; Martín-Lara, M.A.; Osegueda, O.; Castillo, V.; Blázquez, G. Scale-up of a packed bed column for wastewater treatment. Water Science and Technology 2018, 77, 1386–1396. [Google Scholar] [CrossRef]
- Babu, 62 B.V.; Gupta, S. Modeling and simulation of fixed bed adsorption column: effect of velocity variation. i-Manager’s Journal on Future Engineering and Technology 1 2005, 60–66. [Google Scholar] [CrossRef]
- Fernandes, 63 R.; Campos, J.; Serra, M.; Fidalgo, J.; Almeida, H.; Casas, A.; Toubarro, D.; Barros, A.I.R.N.A. Exploring the benefits of phycocyanin: From Spirulina Cultivation to its widespread applications. Pharmaceuticals 2023, 16, 592. [Google Scholar] [CrossRef]
- Zhou, 64 Y.; Huang, Z.; Liu, Y.; Li, B.; Wen, Z.; Cao, L. Stability and bioactivities evaluation of analytical grade C-phycocyanin during the storage of Spirulina platensis powder. J. Food Sci. 2024, 89, 1442–1453. [Google Scholar] [CrossRef]
- Huijbregts, 65 M.A.J.; Hellweg, S.; Frischknecht, R.; Hendriks, H.W.M.; Hungerbühler, K.; Hendriks, A.J. Cumulative energy demand as predictor for the environmental burden of commodity production. Environ. Sci. Technol. 2010, 44, 2189–2196. [Google Scholar] [CrossRef]
- Helbling, 66 E.W.; Barbieri, E.S.; Sinha, R.P.; Villafañe, V.E.; Häder, D.-P. Dynamics of potentially protective compounds in Rhodophyta species from Patagonia (Argentina) exposed to solar radiation. J. Photochem. Photobiol. B 2004, 75, 63–71. [Google Scholar] [CrossRef]
- 67. D.A. Bryant, Phycoerythrocyanin and phycoerythrin: properties and occurrence in cyanobacteria. Microbiology 1982, 128, 835–844. [CrossRef]
- J.R. MacColl, Fluorescence studies on R-phycoerythrin and C-phycoerythrin. Fluoresc., 1991; 1, pp. 135–140. [CrossRef]
- Li, 69; Wu, H.; Xiang, W.; Wu, H.; Wang, N.; Wu, J.; Li, T. Comparison of production and fluorescence characteristics of phycoerythrin from three strains of Porphyridium. Foods 2022, 11, 2069. [Google Scholar] [CrossRef]
- Abalde, 70 J.; Betancourt, L.; Torres, E.; Cid, A.; Barwell, C. Purification and characterization of phycocyanin from the marine cyanobacterium Synechococcus sp. IO9201. Plant Science 1998, 136, 109–120. [Google Scholar] [CrossRef]
- Soni, 71; Trivedi, U.; Madamwar, D. A novel method of single step hydrophobic interaction chromatography for the purification of phycocyanin from Phormidium fragile and its characterization for antioxidant property. Bioresour. Technol. 2008, 99, 188–194. [Google Scholar] [CrossRef]
- Chen, 72 K.-H.; Wang, S.S.-S.; Show, P.-L.; Lin, G.-T.; Chang, Y.-K. A rapid and efficient technique for direct extraction of C-phycocyanin from highly turbid Spirulina platensis algae using hydrophobic interaction chromatography in stirred fluidized bed. Biochem. Eng. J. 2018, 140, 47–56. [Google Scholar] [CrossRef]
- Su, 73 H.-N.; Xie, B.-B.; Chen, X.-L.; Wang, J.-X.; Zhang, X.-Y.; Zhou, B.-C.; Zhang, Y.-Z. Efficient separation and purification of allophycocyanin from Spirulina (Arthrospira) platensis. J. Appl. Phycol. 2010, 22, 65–70. [Google Scholar] [CrossRef]
- Kempen, 74 T.; Cadang, L.; Fan, Y.; Zhang, K.; Chen, T.; Wei, B. Online native hydrophobic interaction chromatography-mass spectrometry of antibody-drug conjugates. MAbs 2025, 17. [Google Scholar] [CrossRef]
- Xiu, 75 L.; Valeja, S.G.; Alpert, A.J.; Jin, S.; Ge, Y. Effective protein separation by coupling hydrophobic interaction and reverse phase chromatography for top-down proteomics. Anal. Chem. 2014, 86, 7899–7906. [Google Scholar] [CrossRef]
- Freitas, 76 S.S.; Santos, J.A.L.; Prazeres, D.M.F. Plasmid purification by hydrophobic interaction chromatography using sodium citrate in the mobile phase. Sep. Purif. Technol. 2009, 65, 95–104. [Google Scholar] [CrossRef]
- Kreusser, 77 J.; Jirasek, F.; Hasse, H. Influence of salts on the adsorption of lysozyme on a mixed-mode resin. Adsorption Science & Technology 2021. [Google Scholar] [CrossRef]
- Koehnlein, 78 W.; Kastenmueller, E.; Meier, T.; Treu, T.; Falkenstein, R. The beneficial impact of kosmotropic salts on the resolution and selectivity of Protein A chromatography. J. Chromatogr. A 2024, 1715, 464585. [Google Scholar] [CrossRef] [PubMed]
- More, 79 K.S.; Maree, J.P.; Mahlangu, M. Optimising Salt Recovery—Four-year operational insights into Na2SO4 recovery from saline waters using pipe freeze-crystallization. Water (Basel) 2025, 17, 101. [Google Scholar] [CrossRef]
- Shen, 80; Zhao, B.; Du, H.; Ren, Y.; Tang, J.; Liu, Y.; Hua, Q.; Wang, B. Influence of organic impurities on fractional crystallization of NaCl and Na2SO4 from high-salinity coal chemical wastewater: thermodynamics and nucleation kinetics analysis. Molecules 2024, 29, 1928. [Google Scholar] [CrossRef]
- Beaudor, 81 M.; Vauchel, P.; Pradal, D.; Aljawish, A.; Phalip, V. Comparing the efficiency of extracting antioxidant polyphenols from spent coffee grounds using an innovative ultrasound-assisted extraction equipment versus conventional method. Chemical Engineering and Processing - Process Intensification 2023, 188, 109358. [Google Scholar] [CrossRef]
- Gharibzahedi, 82 S.M.T.; Altintas, Z. Ultrasound-assisted alcoholic extraction of lesser mealworm larvae oil: process optimization, physicochemical characteristics, and energy consumption. Antioxidants 2022, 11, 1943. [Google Scholar] [CrossRef]
- Berrouane, 83 N.E.H.; Attal, F.-S.; Benchabane, A.; Saghour, I.; Bitam, A.; Gachovska, T.; Amiali, M. Freeze–thaw-, enzyme-, ultrasound- and pulsed electric field-assisted extractions of C-phycocyanin from Spirulina platensis dry biomass. Journal of Food Measurement and Characterization 2022, 16, 1625–1635. [Google Scholar] [CrossRef]
- Piera, 84; Espada, J.J.; Morales, V.; Rodríguez, R.; Vicente, G.; Bautista, L.F. Optimised phycoerythrin extraction method from Porphyridium sp. combining imidazolium-based ionic liquids. Heliyon 2024, 10, e34957. [Google Scholar] [CrossRef]
- Costa, 85 S.P.F.; Pinto, P.C.A.G.; Lapa, R.A.S.; Saraiva, M.L.M.F.S. Toxicity assessment of ionic liquids with Vibrio fischeri: An alternative fully automated methodology. J. Hazard. Mater. 2015, 284, 136–142. [Google Scholar] [CrossRef]
- Liu, 86 J.; Li, Z.; Chen, J.; Xia, C. Synthesis, properties and catalysis of novel methyl- or ethyl- sulfate-anion-based acidic ionic liquids. Catal. Commun. 2009, 10, 799–802. [Google Scholar] [CrossRef]
| Solvent | PPC | EPC (mg g-1) | PAPC | EAPC (mg·g-1) |
|---|---|---|---|---|
| IL aqueous solution (fresh IL) | 0.57 ± 0.01 | 87.6 ± 1.7 | 0.27 ± 0.01 | 30.2 ± 1.4 |
| IL aqueous solution (reused IL) | 0.41 ± 0.01 | 84.5 ± 0.9 | 0.20 ± 0.01 | 29.3 ± 0.3 |
| Impact Category | Unit | C | H&D | E | P | P-p | V | Total |
|---|---|---|---|---|---|---|---|---|
| ADP | kg Sb eq. | 1.36·10-4 | 3.93·10-7 | 2.31·10-5 | 9.74·10-3 | 1.09·10-6 | -3.74·10-8 | 9.90·10-3 |
| AP | kg SO2 eq. | 5.48·10-2 | 9.71·10-4 | 1.41·10-2 | 3.52 | 1.19·10-3 | -2.07·10-3 | 3.59 |
| EP | kg PO43- eq. | 3.50·10-2 | 3.76·10-4 | 7.69·10-3 | 9.32·10-1 | 3.00·10-3 | -1.04·10-2 | 9.68·10-1 |
| FAETP | kg DCB eq. | 3.78 | 3.17·10-3 | 1.46 | 3.80·102 | 1.18·10-2 | -2.69·10-2 | 3.86·102 |
| GWP 100 | kg CO2 eq. | 2.57·101 | 7.85·10-1 | 3.36 | 2.64·102 | 9.41·10-1 | -1.61 | 2.93·102 |
| HTP | kg DCB eq. | 9.79 | 4.73·10-2 | 2.63·101 | 8.58·102 | 6.14·10-2 | -5.11·10-2 | 8.94·102 |
| POCP | kg Ethene eq. | 5.96·10-3 | 1.57·10-4 | 1.72·10-3 | 2.00·10-1 | 1.85·10-4 | 1.34·10-5 | 2.08·10-1 |
| TETP | kg DCB eq. | 8.17·10-2 | 5.37·10-4 | 2.06·10-2 | 7.33 | 9.83·10-4 | -1.70·10-4 | 7.43 |
| CED | MJ | 1.11·103 | 3.98·101 | 1.16·102 | 4.72·103 | 4.60·101 | 1.41·101 | 6.04·103 |
| Impact Category | Unit | C | H&D | E | P | P-p | V | Total |
|---|---|---|---|---|---|---|---|---|
| ADP | kg Sb eq. | 1.36·10-4 | 3.93·10-7 | 9.50·10-6 | 1.12·10-2 | 1.09·10-6 | -3.74·10-8 | 1.13·10-2 |
| AP | kg SO2 eq. | 5.48·10-2 | 9.71·10-4 | 6.42·10-3 | 3.04·10-1 | 1.19·10-3 | -2.07·10-3 | 3.65·10-1 |
| EP | kg PO43- eq. | 3.50·10-2 | 3.76·10-4 | 3.18·10-3 | 1.61·10-1 | 3.00·10-3 | -1.04·10-2 | 1.92·10-1 |
| FAETP | kg DCB eq. | 3.78 | 3.17·10-3 | 5.83·10-1 | 6.25·101 | 1.18·10-2 | -2.69·10-2 | 6.68·101 |
| GWP 100 | kg CO2 eq. | 2.57·101 | 7.85·10-1 | 1.98 | 3.71·101 | 9.41·10-1 | -1.61 | 6.49·101 |
| HTP | kg DCB eq. | 9.79 | 4.73·10-2 | 1.05·101 | 1.20·102 | 6.14·10-2 | -5.11·10-2 | 1.40·102 |
| POCP | kg Ethene eq. | 5.96·10-3 | 1.57·10-4 | 8.11·10-4 | 1.76·10-2 | 1.85·10-4 | 1.34·10-5 | 2.47·10-2 |
| TETP | kg DCB eq. | 8.17·10-2 | 5.37·10-4 | 8.63·10-3 | 8.90·10-1 | 9.83·10-4 | -1.70·10-4 | 9.82·10-1 |
| CED | MJ | 1.11·103 | 3.98·101 | 7.92·101 | 7.74·102 | 4.60·101 | 1.41·101 | 2.06·103 |
| Impact Category | Unit | C | H&D | E | P | P-p | V | Total |
|---|---|---|---|---|---|---|---|---|
| ADP | kg Sb eq. | 4.38·10-5 | 1.97·10-7 | 8.58·10-7 | 1.69·10-5 | 5.81·10-7 | -5.09·10-8 | 6.23·10-5 |
| AP | kg SO2 eq. | 3.57·10-2 | 1.29·10-3 | 3.29·10-3 | 4.55·10-3 | 2.64·10-4 | -1.23·10-3 | 4.38·10-2 |
| EP | kg PO43- eq. | 6.16·10-3 | 3.16·10-4 | 6.25·10-4 | 1.54·10-3 | 4.35·10-4 | -6.90·10-3 | 2.17·10-3 |
| FAETP | kg DCB eq. | 9.23·10-1 | 2.43·10-3 | 2.25·10-2 | 6.73·10-2 | 2.74·10-2 | -1.79·10-2 | 1.02 |
| GWP 100 | kg CO2 eq. | 1.73·101 | 7.90·10-1 | 1.99 | 2.58 | 1.11·10-1 | -1.03 | 2.17·101 |
| HTP | kg DCB eq. | 3.09 | 4.69·10-2 | 1.45·10-1 | 2.46·10-1 | 4.98·10-2 | -3.14·10-2 | 3.55 |
| POCP | kg Ethene eq. | 2.87·10-3 | 1.16·10-4 | 3.21·10-4 | 5.01·10-4 | 6.29·10-5 | 4.89·10-6 | 3.87·10-3 |
| TETP | kg DCB eq. | 2.81·10-2 | 5.22·10-4 | 1.60·10-3 | 2.83·10-3 | 5.10·10-4 | -6.12·10-5 | 3.35·10-2 |
| CED | MJ | 6.64·102 | 3.07·101 | 7.70·101 | 9.78·101 | 3.95 | 8.99 | 8.83·102 |
| Impact Category | Unit | C | H&D | E | P | P-p | V | Total |
|---|---|---|---|---|---|---|---|---|
| ADP | kg Sb eq. | 3.87·10-5 | 1.85·10-7 | 1.96·10-6 | 9.82·10-4 | 3.31·10-5 | -5.26·10-8 | 1.06·10-3 |
| AP | kg SO2 eq. | 3.15·10-2 | 1.21·10-3 | 3.39·10-3 | 3.70·10-1 | 1.16·10-2 | -1.09·10-3 | 4.16·10-1 |
| EP | kg PO43- eq. | 5.44·10-3 | 2.89·10-4 | 6.28·10-4 | 1.82·10-1 | 6.97·10-3 | -6.09·10-3 | 1.89·10-1 |
| FAETP | kg DCB eq. | 8.15·10-1 | 2.26·10-3 | 7.27·10-2 | 4.88·101 | 1.49 | -1.58·10-2 | 5.11·101 |
| GWP 100 | kg CO2 eq. | 1.53·101 | 7.42·10-1 | 1.83 | 5.26·101 | 1.64 | -9.07·10-1 | 7.12·101 |
| HTP | kg DCB eq. | 2.73 | 4.40·10-2 | 2.17·10-1 | 8.25·101 | 2.48 | -2.78·10-2 | 8.79·101 |
| POCP | kg Ethene eq. | 2.53·10-3 | 1.09·10-4 | 2.85·10-4 | 2.78·10-2 | 6.01·10-4 | 3.96·10-6 | 3.13·10-2 |
| TETP | kg DCB eq. | 2.48·10-2 | 4.90·10-4 | 1.81·10-3 | 4.67·10-1 | 1.42·10-2 | -6.27·10-5 | 5.08·10-1 |
| CED | MJ | 5.86·102 | 2.88·101 | 7.01·101 | 1.41·103 | 4.00·101 | 7.91 | 2.15·103 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




