Submitted:
18 April 2026
Posted:
21 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Soils Material
2.2. Experimental Design
2.3. Measurement Indicators and Methods
2.3.1. Selenium Speciation Determination in Soil
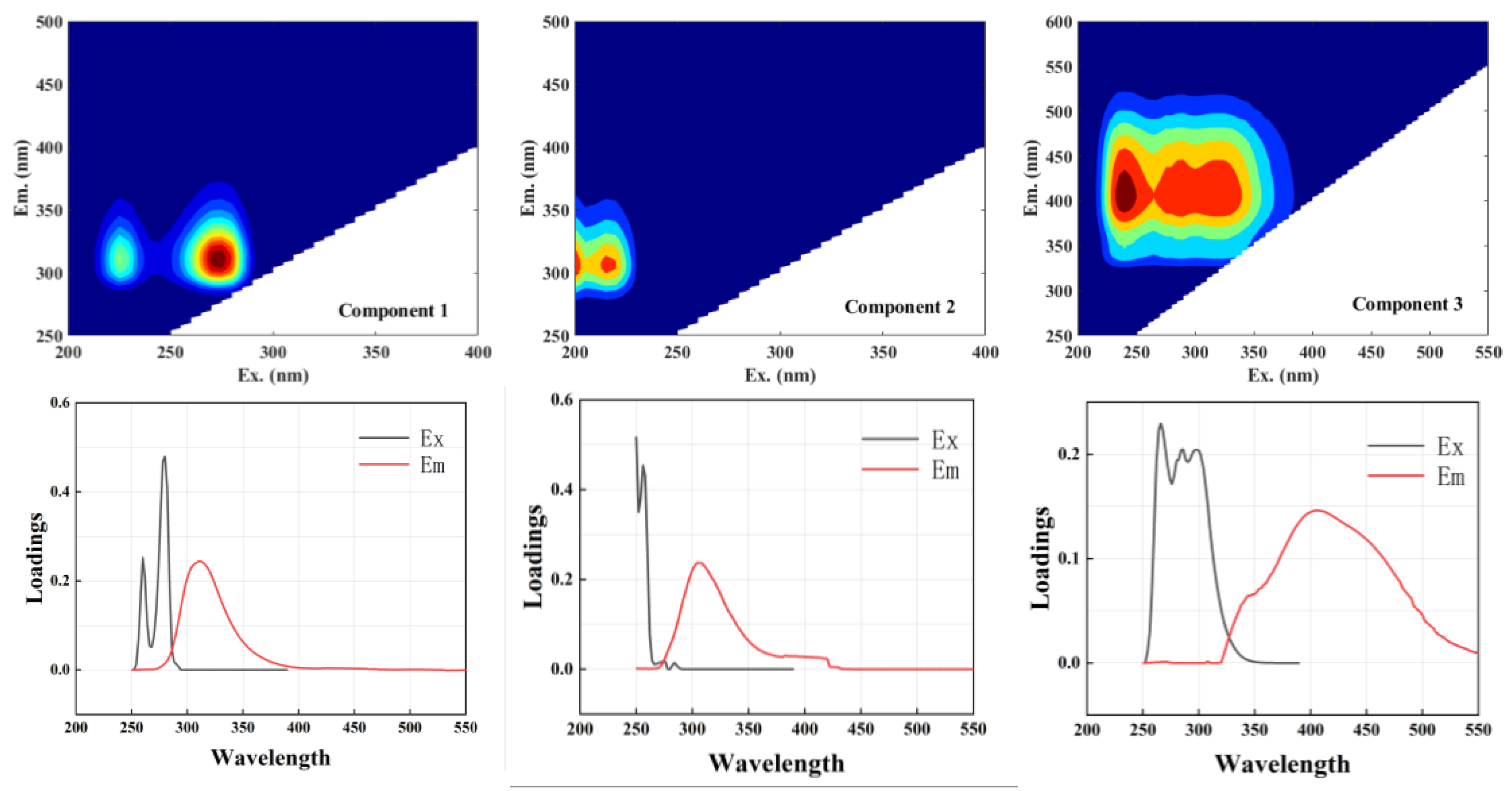
2.3.2. DOM Extraction and EEM Analysis
2.4. Data Analysis
3. Results
3.1. Soluble Se in Soil After LMWOAs Treatment
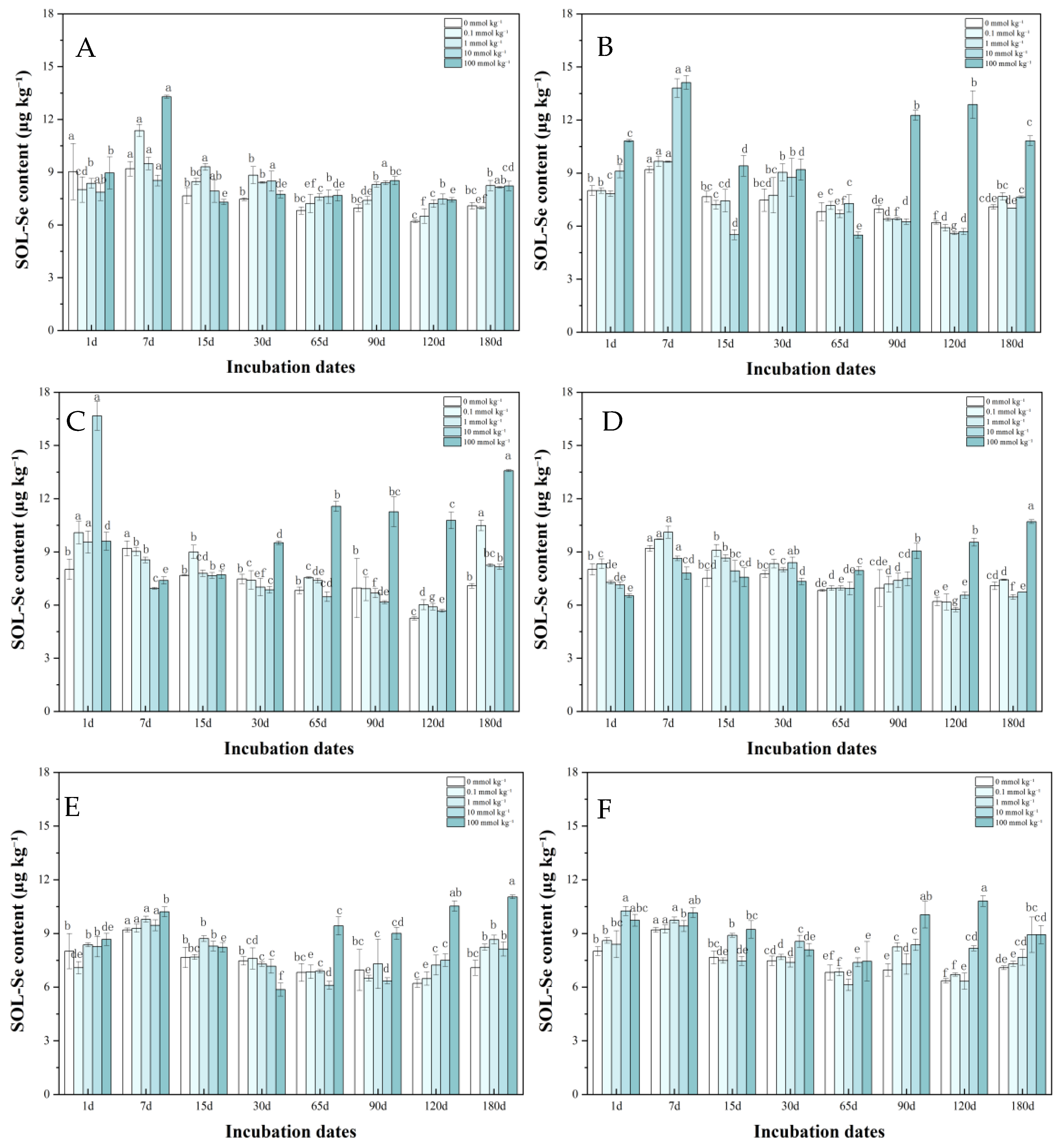
3.1.1. Exchangeable Se in Soil After LMWOAs Treatment

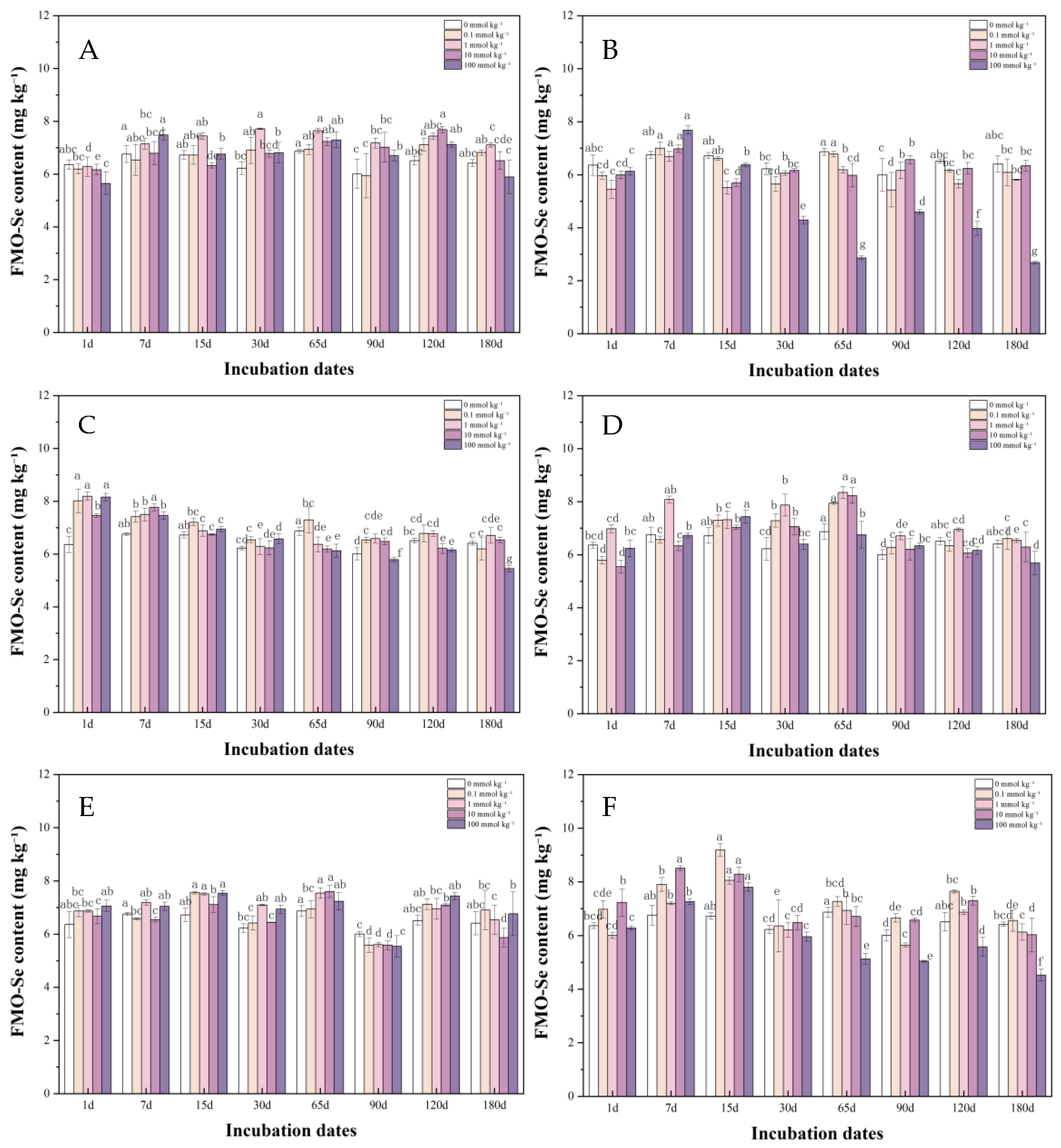
3.1.2. Iron-Manganese Oxide-Bound Se in Soil After LMWOAs Treatment
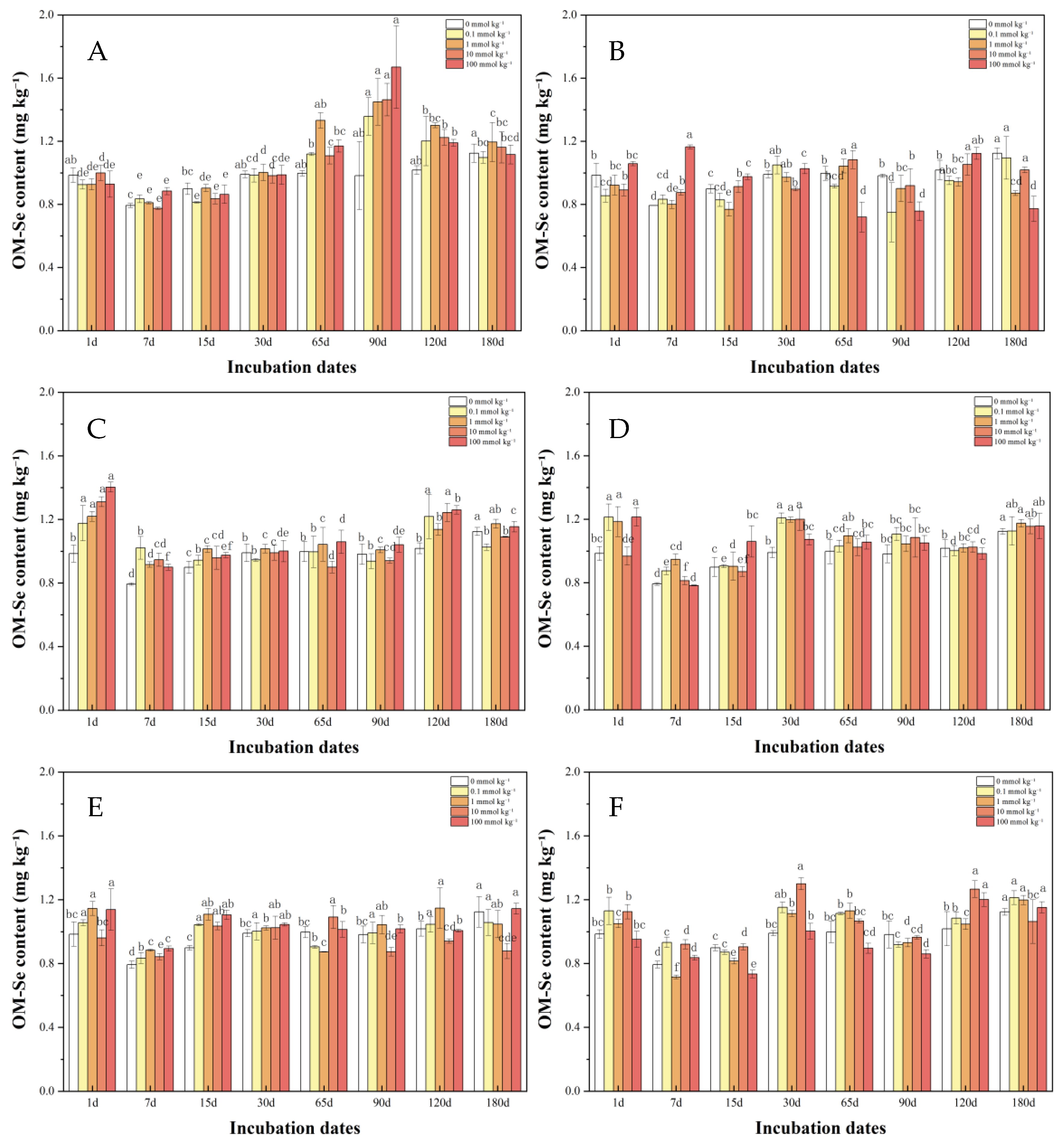
3.1.3. Organically Bound Se in Soil After LMWOAs Treatment
3.2. Correlation Analysis of Soil DOM Components and Se Morphology
4. Discussion
4.1. Mechanisms and Regional Specificity of Low-Molecular-Weight Organic Acids in Regulating Soil SOL-Se and FMO-Se
4.2. Dual effects of Exchangeable Se in Selenium-Rich Red Soils
4.3. Differential Regulation and Specific Mechanisms of Low-Molecular-Weight Organic Acids on Soil OM-Se
4.4. Correlation Between Fluorescent Components and Soil Se
5. Conclusion
Supplementary Materials
Funding
Data Availability Statement
Declarations
References
- Zhai, H.; Kleawsampanjai, P.; Wang, R.; Zhou, F.; Xie, J.; Hao, X.; Liu, Y.; Yao, D.; Xiao, H.; Liang, D. Effect of alternating wetting and drying cycles on selenium aging in three soils amended with selenate/selenite. Journal of Soils and Sediments 2025, 25(5), 1532–1543. [Google Scholar] [CrossRef]
- Li, Z.; Man, N.; Wang, S.; Liang, D.; Liu, J. Selenite adsorption and desorption in main Chinese soils with their characteristics and physicochemical properties. Journal of Soils and Sediments 2015, 15(5), 1150–1158. [Google Scholar] [CrossRef]
- Alfthan, G.; Eurola, M.; Ekholm, P.; Venäläinen, E.-R.; Root, T.; Korkalainen, K.; Hartikainen, H.; Salminen, P.; Hietaniemi, V.; Aspila, P.; Aro, A. Effects of nationwide addition of selenium to fertilizers on foods, and animal and human health in finland: From deficiency to optimal selenium status of the population. Journal of Trace Elements in Medicine and Biology 2015, 31, 142–147. [Google Scholar] [CrossRef]
- Natasha; Shahid, M.; Niazi, N.K.; Khalid, S.; Murtaza, B.; Bibi, I.; Rashid, M.I. A critical review of selenium biogeochemical behavior in soil-plant system with an inference to human health. Environmental Pollution 2018, 234, 915–934. [Google Scholar] [CrossRef]
- Panchal, P.; Miller, A.J.; Giri, J. Organic acids: Versatile stress-response roles in plants. Journal of Experimental Botany 2021, 72(11), 4038–4052. [Google Scholar] [CrossRef] [PubMed]
- Adeleke, R.; Nwangburuka, C.; Oboirien, B. Origins, roles and fate of organic acids in soils: A review. South African Journal of Botany 2017, 108, 393–406. [Google Scholar] [CrossRef]
- Potysz, A.; Grybos, M.; Kierczak, J.; Guibaud, G.; Fondaneche, P.; Lens, P.N.L.; Van Hullebusch, E.D. Metal mobilization from metallurgical wastes by soil organic acids. Chemosphere 2017, 178, 197–211. [Google Scholar] [CrossRef] [PubMed]
- Favorito, J.E.; Eick, M.J.; Grossl, P.R. Adsorption of selenite and selenate on ferrihydrite in the presence and absence of dissolved organic carbon. Journal of Environmental Quality 2018, 47(1), 147–155. [Google Scholar] [CrossRef]
- Chen, Z.C.; Liao, H. Organic acid anions: An effective defensive weapon for plants against aluminum toxicity and phosphorus deficiency in acidic soils. Journal of Genetics and Genomics 2016, 43(11), 631–638. [Google Scholar] [CrossRef]
- Zhang, Hongyu.; Xie, S.; Bao, Z.; Tian, H.; Carranza, E.J.M.; Xiang, W.; Yao, L.; Zhang, Hai. Underlying dynamics and effects of humic acid on selenium and cadmium uptake in rice seedlings. Journal of Soils and Sediments 2020, 20(1), 109–121. [Google Scholar] [CrossRef]
- Fernández-Martínez, A.; Charlet, L. Selenium environmental cycling and bioavailability: A structural chemist point of view. Reviews in Environmental Science and Bio/Technology 2009, 8(1), 81–110. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, X.; Whalen, J.K.; Cao, Y.; Quan, Z.; Lu, C.; Shi, Y. Kinetics of inorganic and organic phosphorus release influenced by low molecular weight organic acids in calcareous, neutral and acidic soils. Journal of Plant Nutrition and Soil Science 2015, 178(4), 555–566. [Google Scholar] [CrossRef]
- Agnello, A.C.; Huguenot, D.; Van Hullebusch, E.D.; Esposito, G. Enhanced phytoremediation: A review of low molecular weight organic acids and surfactants used as amendments. Critical Reviews in Environmental Science and Technology 2014, 44(22), 2531–2576. [Google Scholar] [CrossRef]
- Bolan, N.; Kunhikrishnan, A.; Thangarajan, R.; Kumpiene, J.; Park, J.; Makino, T.; Kirkham, M.B.; Scheckel, K. Remediation of heavy metal(loid)s contaminated soils – to mobilize or to immobilize? Journal of Hazardous Materials 2014, 266, 141–166. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Liang, D.; Wang, D.; Wei, W.; Fu, D.; Lin, Z. Selenium fractionation and speciation in agriculture soils and accumulation in corn (zea mays L.) under field conditions in shaanxi province, China. Science of The Total Environment 2012, 427-428, 159–164. [Google Scholar] [CrossRef]
- Hong, H.; Wu, S.; Wang, Q.; Dai, M.; Qian, L.; Zhu, H.; Li, Junwei.; Zhang, J.; Liu, J.; Li, Jian.; Lu, H.; Yan, C. Fluorescent dissolved organic matter facilitates the phytoavailability of copper in the coastal wetlands influenced by artificial topography. Science of the Total Environment 2021, 790, 147855. [Google Scholar] [CrossRef]
- Murphy, K.R.; Stedmon, C.A.; Graeber, D.; Bro, R. Fluorescence spectroscopy and multi-way techniques. PARAFAC. Analytical Methods 2013, 5(23), 6557. [Google Scholar] [CrossRef]
- D’Andrilli, J.; Junker, J.R.; Smith, H.J.; Scholl, E.A.; Foreman, C.M. DOM composition alters ecosystem function during microbial processing of isolated sources. Biogeochemistry 2019, 142(2), 281–298. [Google Scholar] [CrossRef]
- Painter, S.C.; Lapworth, D.J.; Woodward, E.M.S.; Kroeger, S.; Evans, C.D.; Mayor, D.J.; Sanders, R.J. Terrestrial dissolved organic matter distribution in the north sea. Science of the Total Environment 2018, 630, 630–647. [Google Scholar] [CrossRef]
- Yamashita, Y.; Boyer, J.N.; Jaffé, R. Evaluating the distribution of terrestrial dissolved organic matter in a complex coastal ecosystem using fluorescence spectroscopy. Continental Shelf Research 2013, 66, 136–144. [Google Scholar] [CrossRef]
- Derrien, M.; Choi, H.; Jardé, E.; Shin, K.-H.; Hur, J. Do early diagenetic processes affect the applicability of commonly-used organic matter source tracking tools? An assessment through controlled degradation end-member mixing experiments. Water Research 2020, 173, 115588. [Google Scholar] [CrossRef]
- Derrien, M.; Shin, K.-H.; Hur, J. Biodegradation-induced signatures in sediment pore water dissolved organic matter: Implications from artificial sediments composed of two contrasting sources. Science of the Total Environment 2019, 694, 133714. [Google Scholar] [CrossRef]
- Chen, M.; Jung, J.; Lee, Y.K.; Hur, J. Surface accumulation of low molecular weight dissolved organic matter in surface waters and horizontal off-shelf spreading of nutrients and humic-like fluorescence in the chukchi sea of the arctic ocean. Science of the Total Environment 2018, 639, 624–632. [Google Scholar] [CrossRef]
- Sharma, P.; Laor, Y.; Raviv, M.; Medina, S.; Saadi, I.; Krasnovsky, A.; Vager, M.; Levy, G.J.; Bar-Tal, A.; Borisover, M. Compositional characteristics of organic matter and its water-extractable components across a profile of organically managed soil. Geoderma 2017, 286, 73–82. [Google Scholar] [CrossRef]
- Li, J.; Peng, Q.; Liang, D.; Liang, S.; Chen, J.; Sun, H.; Li, S.; Lei, P. Effects of aging on the fraction distribution and bioavailability of selenium in three different soils. Chemosphere 2016, 144, 2351–2359. [Google Scholar] [CrossRef] [PubMed]
- Antoniadis, V.; Levizou, E.; Shaheen, S.M.; Ok, Y.S.; Sebastian, A.; Baum, C.; Prasad, M.N.V.; Wenzel, W.W.; Rinklebe, J. Trace elements in the soil-plant interface: Phytoavailability, translocation, and phytoremediation–a review. Earth-science Reviews 2017, 171, 621–645. [Google Scholar] [CrossRef]
- Martin, D.P.; Seiter, J.M.; Lafferty, B.J.; Bednar, A.J. Exploring the ability of cations to facilitate binding between inorganic oxyanions and humic acid. Chemosphere 2017, 166, 192–196. [Google Scholar] [CrossRef]
- Peel, H.R.; Martin, D.P.; Bednar, A.J. Extraction and characterization of ternary complexes between natural organic matter, cations, and oxyanions from a natural soil. Chemosphere 2017, 176, 125–130. [Google Scholar] [CrossRef]
- Nakamaru, Y.M.; Sekine, K. Sorption behavior of selenium and antimony in soils as a function of phosphate ion concentration. Soil Science & Plant Nutrition 2008, 54(3), 332–341. [Google Scholar] [CrossRef]
- Fang, D.; Wei, S.; Xu, Y.; Xiong, J.; Tan, W. Impact of low-molecular weight organic acids on selenite immobilization by goethite: Understanding a competitive-synergistic coupling effect and speciation transformation. Science of The Total Environment 2019, 684, 694–704. [Google Scholar] [CrossRef]
- Fordyce, F.M. Selenium deficiency and toxicity in the environment[M]//Selinus O. In Essentials of Medical Geology; Springer Netherlands: Dordrecht, 2013; pp. 375–416. [Google Scholar] [CrossRef]
- Guo, Q.; Ye, J.; Zeng, J.; Chen, L.; Korpelainen, H.; Li, C. Selenium species transforming along soil–plant continuum and their beneficial roles for horticultural crops. Horticulture Research 2023, 10(2), uhac270. [Google Scholar] [CrossRef]
- Saha, U.K.; Kozak, L.M.; Huang, P.M. Kinetics of citrate-induced selenite desorption from montmorillonite as affected by complexation with hydroxyaluminum and hydroxyaluminosilicate ions. Clays and Clay Minerals 2007, 55(1), 71–88. [Google Scholar] [CrossRef]
- El-Said, A.G.; Badawy, N.A.; Abdel-Aal, A.Y.; Garamon, S.E. Optimization parameters for adsorption and desorption of Zn(II) and Se(IV) using rice husk ash: kinetics and equilibrium. Ionics 2011, 17(3), 263–270. [Google Scholar] [CrossRef]
- Adeleke, R.; Nwangburuka, C.; Oboirien, B. Origins, roles and fate of organic acids in soils: A review. South African Journal of Botany 2017, 108, 393–406. [Google Scholar] [CrossRef]
- He, J.; Shi, Y.; Yang, X.; Zhou, W.; Li, Y.; Liu, C. Influence of fe(II) on the se(IV) sorption under oxic/anoxic conditions using bentonite. Environmental Research 2018, 164, 288–301. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Xue, M.; Wang, Y.; Zhou, D.; Tang, L.; Cao, S.; Wei, Y.; Yang, C.; Liang, D.-L. Effects of straw amendment on selenium aging in soils: Mechanism and influential factors. Science of The Total Environment 2019, 657, 871–881. [Google Scholar] [CrossRef] [PubMed]
- Vermeiren, C.; Ceulemans, J.; Thiry, Y.; Smolders, E. Increased soil pH and enhanced microbial activity stimulate the gradual immobilisation of selenate added to soils. Soil Biology & Biochemistry 2025, 202, 109688. [Google Scholar] [CrossRef]
- Kader, M.; Lamb, D.T.; Megharaj, M.; Naidu, R. Sorption parameters as a predictor of arsenic phytotoxicity in australian soils. Geoderma 2016, 265, 103–110. [Google Scholar] [CrossRef]
- He, J.; Shi, Y.; Yang, X.; Zhou, W.; Li, Y.; Liu, C. Influence of fe(II) on the se(IV) sorption under oxic/anoxic conditions using bentonite. Chemosphere 2018, 193, 376–384. [Google Scholar] [CrossRef]
- Bruggeman, C.; Maes, A.; Vancluysen, J. The interaction of dissolved boom clay and gorleben humic substances with selenium oxyanions (selenite and selenate). Applied Geochemistry 2007, 22(7), 1371–1379. [Google Scholar] [CrossRef]
- Jordan, N.; Marmier, N.; Lomenech, C.; Giffaut, E.; Ehrhardt, J.-J. Competition between selenium (IV) and silicic acid on the hematite surface. Chemosphere 2009, 75(1), 129–134. [Google Scholar] [CrossRef] [PubMed]
- Tolu, J.; Thiry, Y.; Bueno, M.; Jolivet, C.; Potin-Gautier, M.; Le Hécho, I. Distribution and speciation of ambient selenium in contrasted soils, from mineral to organic rich. Science of The Total Environment 2014, 479-480, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Dinh, Q.T.; Li, Z.; Tran, T.A.T.; Wang, D.; Liang, D. Role of organic acids on the bioavailability of selenium in soil: A review. Chemosphere 2017, 184, 618–635. [Google Scholar] [CrossRef] [PubMed]
| Se Speciation | OA Treatment | DOM Components | |||||||
| C1(%) | C2(%) | C3(%) | |||||||
| R | P | R | P | R | P | ||||
| SOL-Se | 0 mmol kg⁻¹ | -0.3631 | >0.05 | 0.2587 | >0.05 | 0.3618 | >0.05 | ||
| 0.1 mmol kg⁻¹ | 0.6371 | >0.05 | 0.1619 | >0.05 | -0.1531 | >0.05 | |||
| 1 mmol kg⁻¹ | 0.4918 | >0.05 | -0.1257 | >0.05 | -0.5218 | >0.05 | |||
| 10 mmol kg⁻¹ | 0.6419 | >0.05 | 0.1241 | >0.05 | -0.4377 | >0.05 | |||
| 100 mmol kg⁻¹ | -0.0295 | >0.05 | -0.1931 | >0.05 | -0.0296 | >0.05 | |||
| EXC-Se | 0 mmol kg⁻¹ | -0.2015 | >0.05 | 0.1243 | >0.05 | 0.1917 | >0.05 | ||
| 0.1 mmol kg⁻¹ | 0.4645 | >0.05 | 0.2527 | >0.05 | -0.2485 | >0.05 | |||
| 1 mmol kg⁻¹ | 0.0015 | >0.05 | -0.3963 | >0.05 | -0.068 | >0.05 | |||
| 10 mmol kg⁻¹ | 0.7959 | <0.05 | 0.437 | >0.05 | 0.0106 | >0.05 | |||
| 100 mmol kg⁻¹ | -0.2208 | >0.05 | 0.2413 | >0.05 | 0.0912 | >0.05 | |||
| FMO-Se | 0 mmol kg⁻¹ | 0.2145 | >0.05 | 0.0402 | >0.05 | -0.3563 | >0.05 | ||
| 0.1 mmol kg⁻¹ | -0.2486 | >0.05 | -0.033 | >0.05 | -0.3703 | >0.05 | |||
| 1 mmol kg⁻¹ | -0.6041 | >0.05 | -0.4662 | >0.05 | 0.1517 | >0.05 | |||
| 10 mmol kg⁻¹ | -0.0523 | >0.05 | -0.3753 | >0.05 | -0.1683 | >0.05 | |||
| 100 mmol kg⁻¹ | -0.1571 | >0.05 | 0.1462 | >0.05 | 0.2137 | >0.05 | |||
| OM-Se | 0 mmol kg⁻¹ | 0.5184 | >0.05 | 0.3105 | >0.05 | -0.1523 | >0.05 | ||
| 0.1 mmol kg⁻¹ | -0.2911 | >0.05 | 0.1255 | >0.05 | 0.5144 | >0.05 | |||
| 1 mmol kg⁻¹ | -0.3614 | >0.05 | 0.2478 | >0.05 | 0.2926 | >0.05 | |||
| 10 mmol kg⁻¹ | 0.0621 | >0.05 | -0.1704 | >0.05 | 0.1566 | >0.05 | |||
| 100 mmol kg⁻¹ | 0.7975 | < 0.05 | 0.974 | <0.001 | 0.8077 | <0.05 | |||
| Se Speciation | CA Treatment | DOM Components | |||||||
| C1(%) | C2(%) | C3(%) | |||||||
| R | P | R | P | R | P | ||||
| SOL-Se | 0 mmol kg⁻¹ | -0.2657 | >0.05 | -0.0108 | >0.05 | 0.1975 | >0.05 | ||
| 0.1 mmol kg⁻¹ | -0.1585 | >0.05 | -0.8235 | <0.05 | 0.6572 | >0.05 | |||
| 1 mmol kg⁻¹ | -0.1668 | >0.05 | -0.6180 | >0.05 | -0.5315 | >0.05 | |||
| 10 mmol kg⁻¹ | -0.6890 | <0.05 | -0.0016 | >0.05 | -0.4679 | >0.05 | |||
| 100 mmol kg⁻¹ | 0.7618 | >0.05 | -0.0779 | >0.05 | -0.5374 | >0.05 | |||
| EXC-Se | 0 mmol kg⁻¹ | -0.2014 | >0.05 | 0.1242 | >0.05 | 0.1916 | >0.05 | ||
| 0.1 mmol kg⁻¹ | -0.8193 | <0.05 | -0.4823 | >0.05 | 0.1814 | >0.05 | |||
| 1 mmol kg⁻¹ | -0.8545 | <0.05 | -0.8037 | <0.05 | -0.7725 | <0.05 | |||
| 10 mmol kg⁻¹ | -0.7226 | <0.05 | -0.3598 | >0.05 | -0.2583 | >0.05 | |||
| 100 mmol kg⁻¹ | 0.4152 | >0.05 | -0.0418 | >0.05 | -0.4253 | >0.05 | |||
| FMO-Se | 0 mmol kg⁻¹ | 0.2147 | >0.05 | 0.0401 | >0.05 | -0.3569 | >0.05 | ||
| 0.1 mmol kg⁻¹ | -0.7224 | <0.05 | -0.1457 | >0.05 | 0.5389 | >0.05 | |||
| 1 mmol kg⁻¹ | -0.2318 | >0.05 | -0.3251 | >0.05 | -0.1405 | >0.05 | |||
| 10 mmol kg⁻¹ | 0.3931 | >0.05 | 0.2427 | >0.05 | -0.4687 | >0.05 | |||
| 100 mmol kg⁻¹ | 0.2871 | >0.05 | -0.0443 | >0.05 | -0.5129 | >0.05 | |||
| OM-Se | 0 mmol kg⁻¹ | 0.5184 | >0.05 | 0.3106 | >0.05 | -0.5234 | >0.05 | ||
| 0.1 mmol kg⁻¹ | 0.0663 | >0.05 | -0.0994 | >0.05 | -0.5616 | >0.05 | |||
| 1 mmol kg⁻¹ | -0.1070 | >0.05 | 0.1048 | >0.05 | -0.1523 | >0.05 | |||
| 10 mmol kg⁻¹ | -0.1994 | >0.05 | -0.5234 | >0.05 | -0.2795 | >0.05 | |||
| 100 mmol kg⁻¹ | 0.4025 | >0.05 | -0.5616 | >0.05 | -0.0251 | >0.05 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




