Submitted:
17 April 2026
Posted:
21 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
| Product Type | Number of Samples |
|---|---|
| Salami-type | 70 |
| Frankfurter-type | 30 |
| Total | 100 |
2.1. Sample Production and Thermal Processing
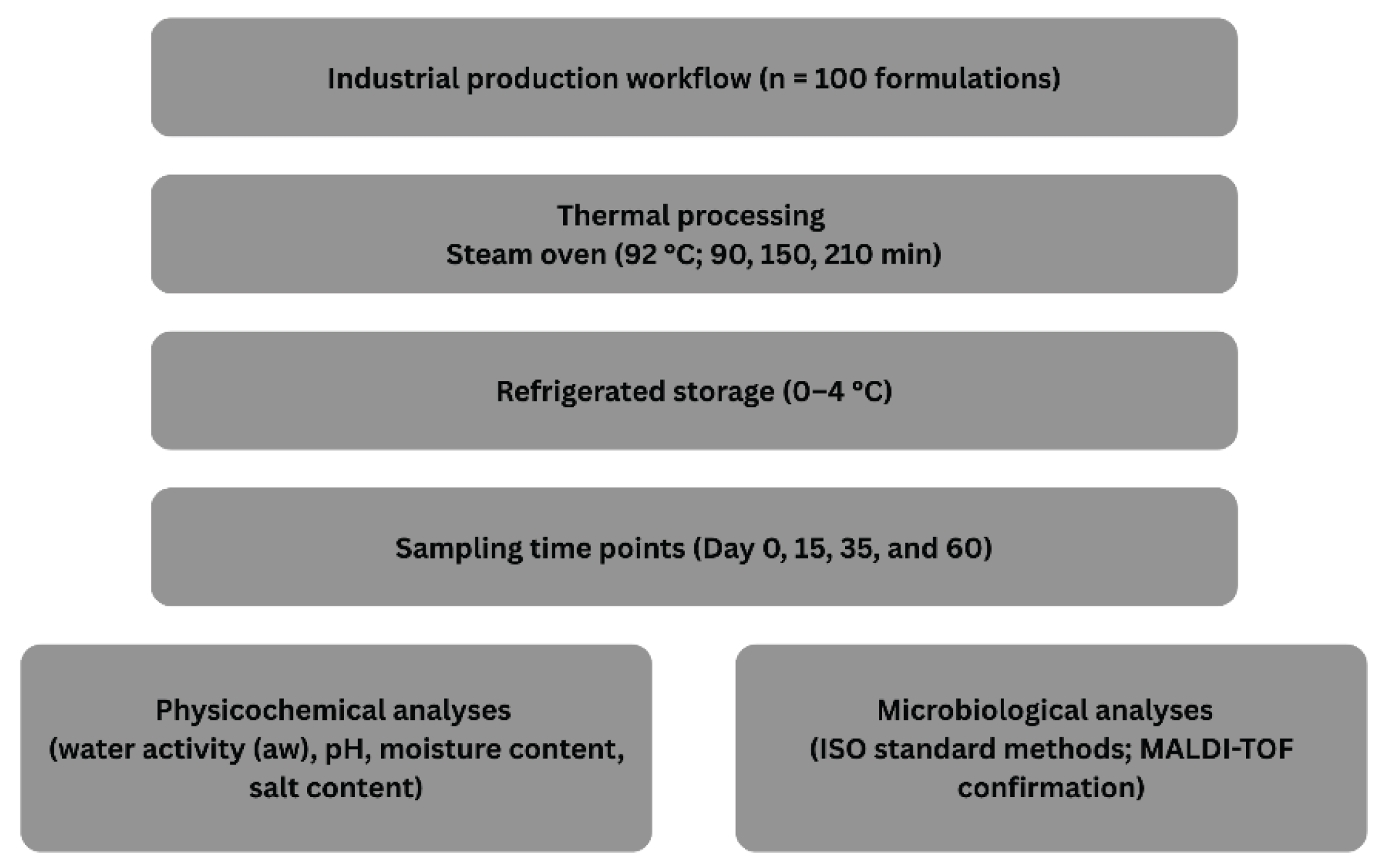
2.2. Physicochemical Analyses
2.2.1. pH Measurement
2.2.2. Water Activity (aw)
2.2.3. Moisture Content
2.2.4. Salt Content
2.3. Microbiological Analyses
2.3.1. Enumeration and Confirmation of Indicator Microorganisms
2.3.2. Qualitative Detection and Identification of Selected Foodborne Pathogens
2.4. MALDI-TOF Confirmation
2.5. Statistical Analysis
3. Results
3.1. Physicochemical Results
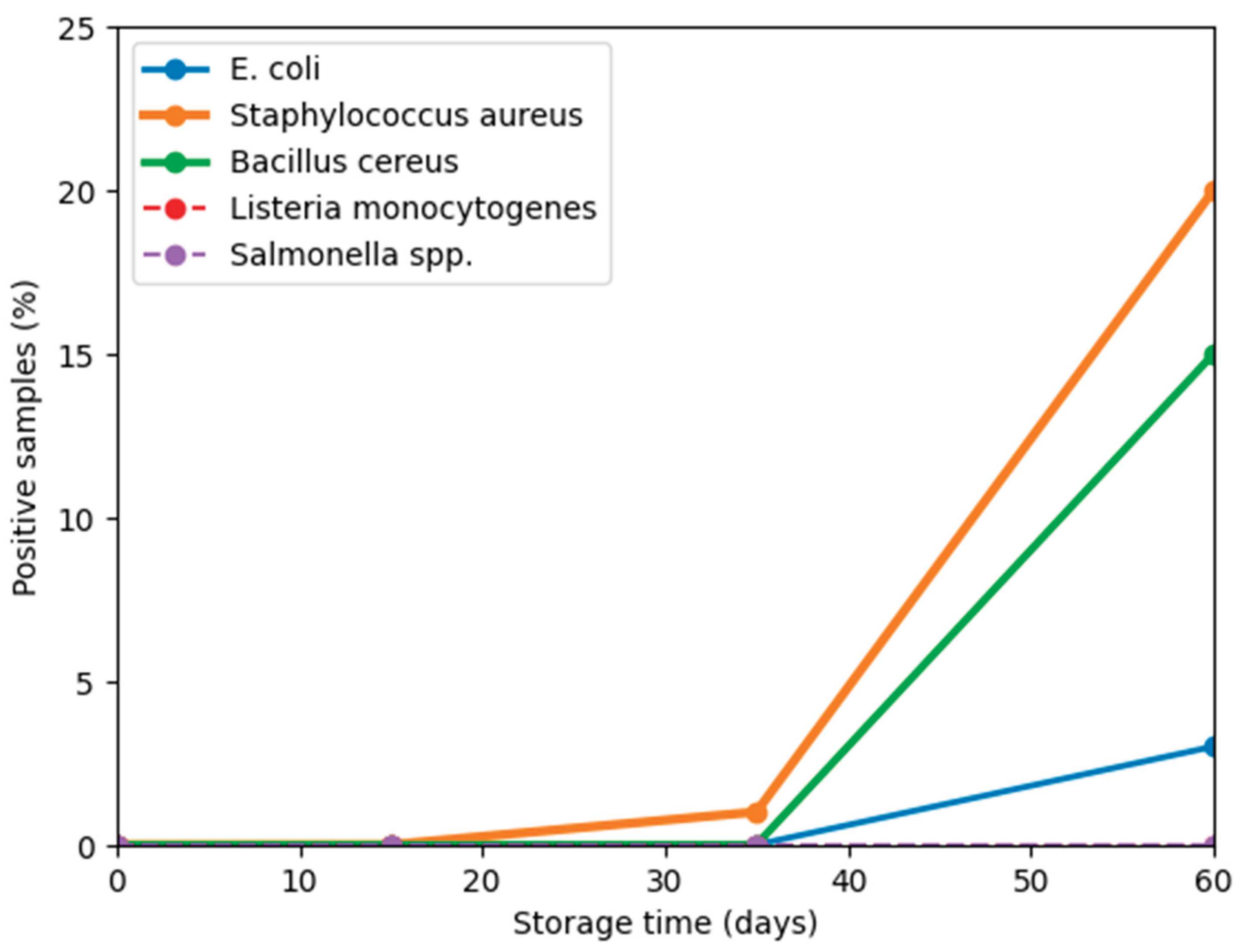
3.2. Microbiological Results
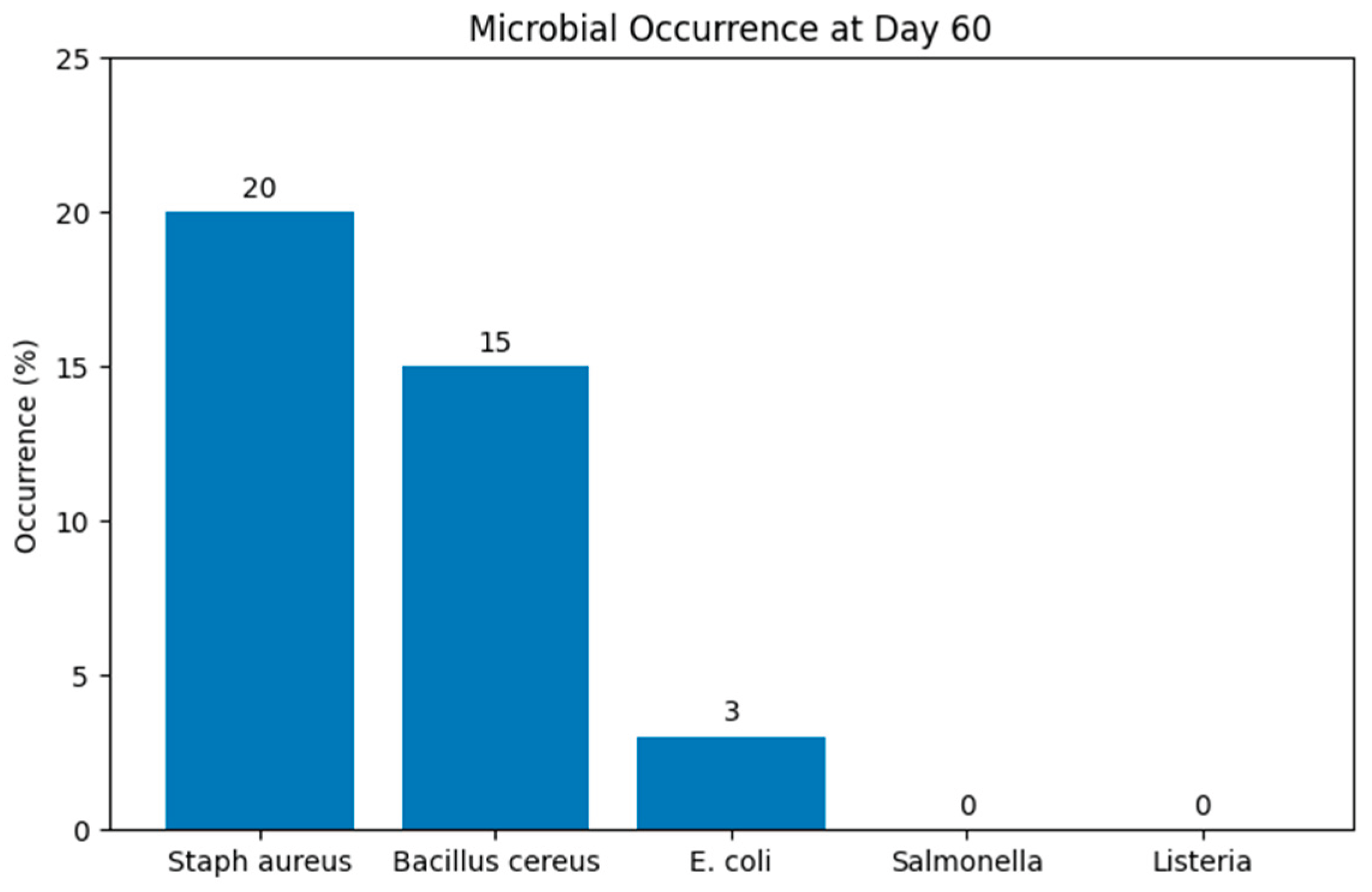
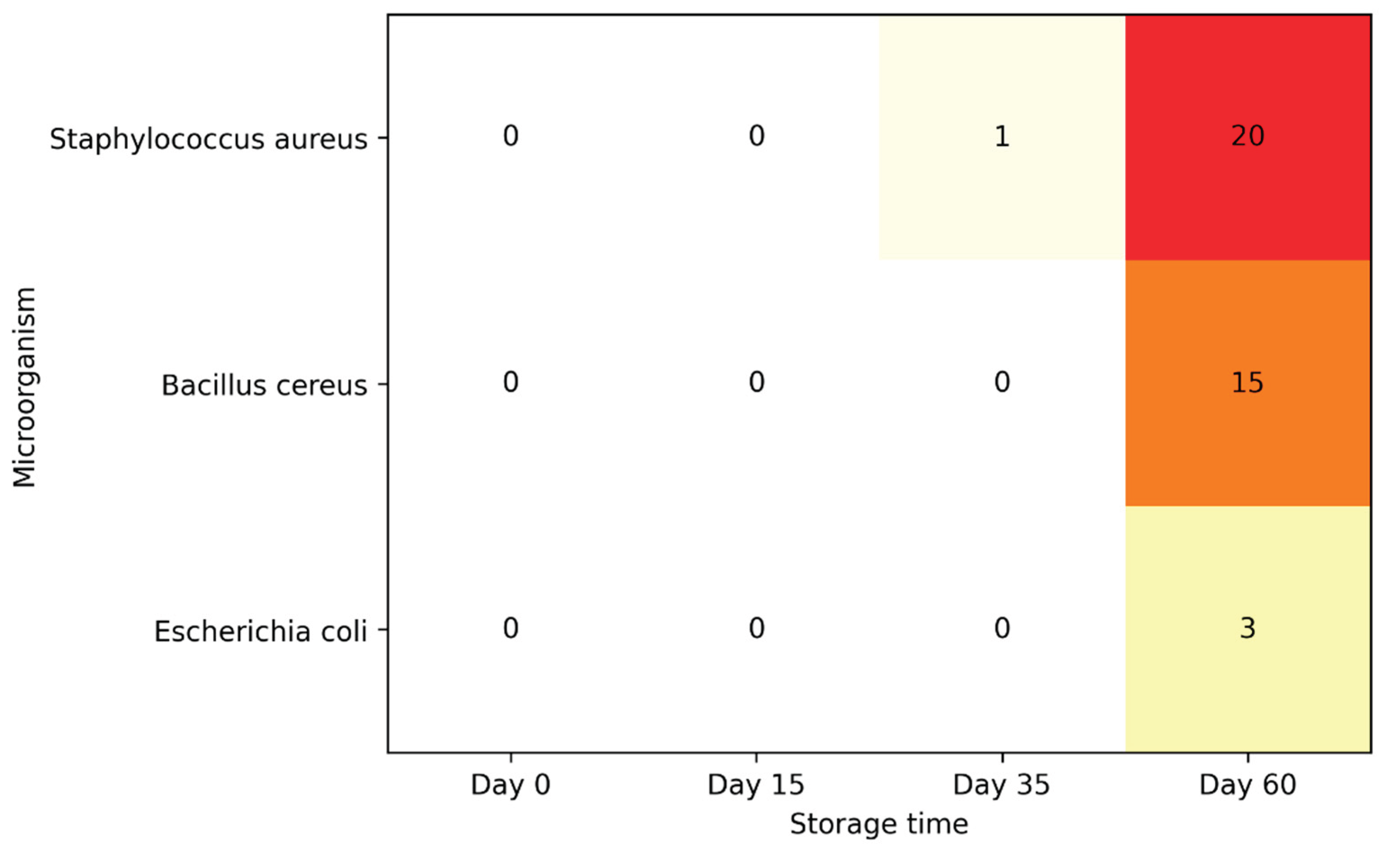
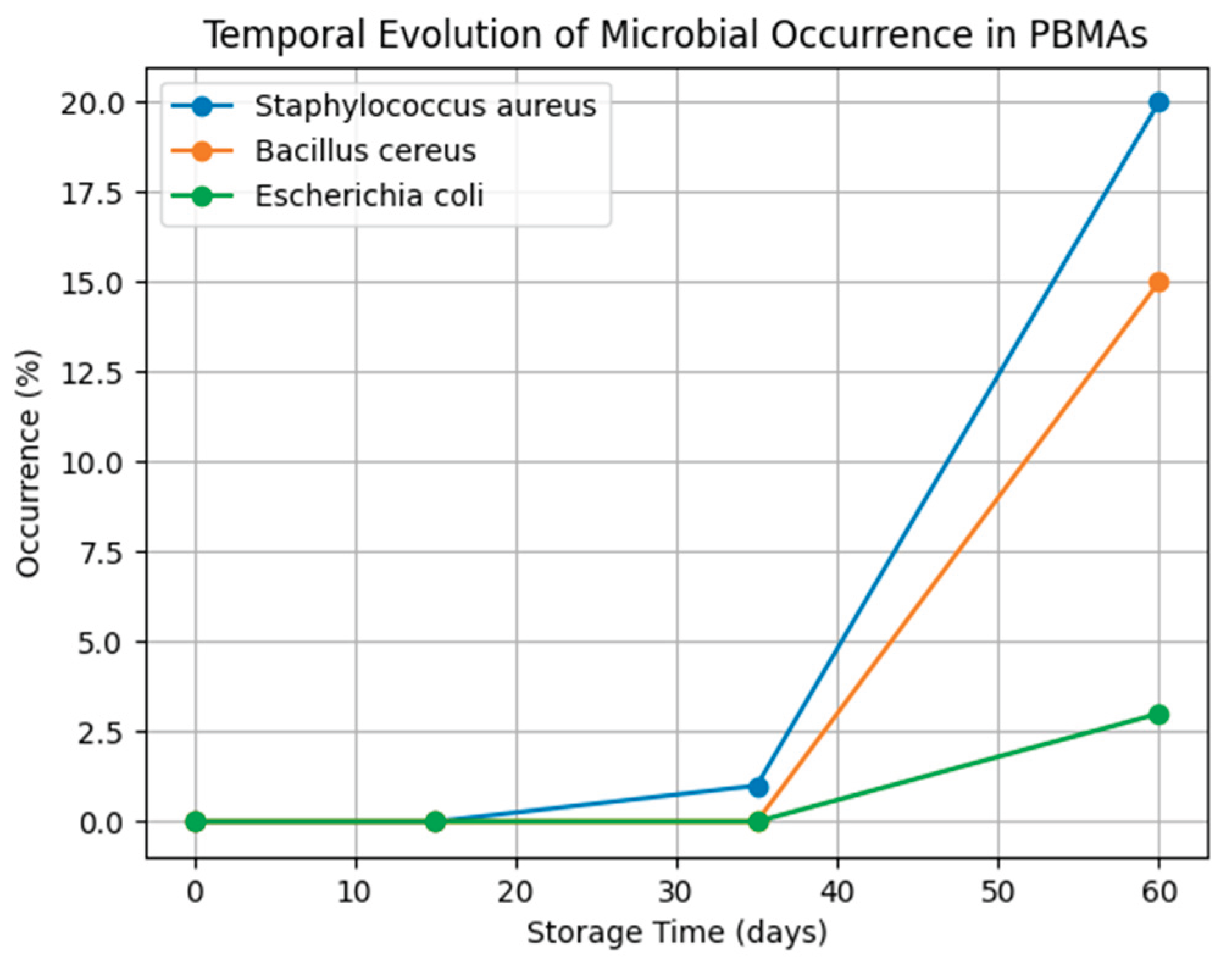
| Microorganism | Day 0 | Day 15 | Day 35 | Day 60 |
|---|---|---|---|---|
| Salmonella spp. | ND | ND | ND | ND |
| Listeria monocytogenes | ND | ND | ND | ND |
| Escherichia coli | ND | ND | ND | 3% |
| Enterobacteriaceae | ND | ND | ND | ND |
| Staphylococcus aureus | ND | ND | 1% | 20% (103 – 105 CFU/g) |
| Bacillus cereus | ND | ND | ND | 15% |
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| PBMA | Plant-Based Meat Alternatives |
References
- Choudhury, D.; Singh, S.; Seah, H.S.J.; Yeo, D.C.H.; Tan, P.L. Commercialization of plant-based meat alternatives. Trends Food Sci. Technol. 2020, 105, 233-249 . [CrossRef]
- Jang, J.; Lee, D.-W. Advancements in plant-based meat analogs enhancing sensory and nutritional attributes. npj Sci.Food 2024, 8, 50 . [CrossRef]
- Bohrer, B.M. An investigation of the formulation and nutritional composition of modern meat analogue products. Food Sci. Hum. Wellness 2019, 8, 320-329 . [CrossRef]
- Dušková, M.; Dorotíková, K.; Bartáková, K.; Králová, M.; Šedo, O.; Kameník, J. The microbial contaminants of plant-based meat analogues from the retail market. Int. J. Food Microbiol. 2024, 425, 110869 . [CrossRef]
- Kyriakopoulou, K.; Keppler, J.K.; van der Goot, AJ. Functionality of ingredients and additives in plant-based meat analogues. Foods 2021, 10, 600. [CrossRef]
- Capiani, S.; Brown, L.P.; Carrillo, C.D.; Lau, C.H. Bacterial microbiota associated with raw plant-based meat analogue products and their influences on selective enrichment for Escherichia coli O157:H7. Curr. Res. Food Sci. 2024, 10, 100944. [CrossRef]
- Roch, F.-F.; Dzieciol, M.; Quijada, N.M.; et al. Microbial community structure of plant-based meat alternatives. npj Sci. Food 2024, 8, 27. [CrossRef]
- Capiani, S.; Brown, L.P.; Carrillo, C.D.; Lau, C.H. Bacterial microbiota associated with raw plant-based meat analogue products. Curr. Res. Food Sci. 2024, 10, 100944. [CrossRef]
- Kyriakopoulou, K.; Keppler, J.K.; van der Goot, A.J. Functionality of ingredients in plant-based meat analogues. Foods 2021, 10, 600. [CrossRef]
- Mehta, N.; Kumar, P.; et al. Microbial quality of plant-based meat analogs. Discover Food 2026, 6, 31. [CrossRef]
- Dušková, M.; Dorotíková, K.; et al. Microbial contaminants of plant-based meat analogues. Int. J. Food Microbiol. 2024, 425, 110869. [CrossRef]
- Dogan, M.; Mann, D.A.; Deng, X. Characterization of spoilage microbial communities in plant-based meat alternatives. Microbiol. Spectr. 2025. [CrossRef]
- Ishaq, A.; Irfan, S.; Sameen, A.; Khalid, N. Plant-based meat analogs: A review with reference to formulation and gastrointestinal fate. Curr.Res.Food Sci. 2022, 5, 973-983. [CrossRef]
- Barmettler, K.; Waser, S.; Stephan, R. Microbiological quality of plant-based meat alternative products collected at retail in level in Switzerland. J.Food.Prot 2025, 88, 100402. [CrossRef]
- ISO (International Organization for Standardization). Microbiology of food and animal feeding stuffs – Horizontal method for the enumeration of coagulase-positive staphylococci (Staphylococcus aureus and other species) – Part 2: Technique using rabbit plasma fibrinogen agar medium – Amendment 1: Inclusion of an alternative confirmation test; ISO 6888-2:1999/Amd.1:2023; ISO: Geneva, Switzerland, 2003.
- ISO (International Organization for Standardization). Microbiology of food and animal feeding stuffs – Horizontal methods for the enumeration of β-glucuronidase-positive Escherichia coli – Part 2: Colony-count techniques at 44 °C using 5-bromo-4-chloro-3-indolyl β-D-glucuronide; ISO 16649-2:2001 (reaffirmed 2022); ISO: Geneva, Switzerland, 2001.
- ISO (International Organization for Standardization). Microbiology of the food chain – Horizontal method for the detection and enumeration of Enterobacteriaceae – Part 2: Colony-count techniques; ISO 21528-2:2017; ISO: Geneva, Switzerland, 2017.
- ISO (International Organization for Standardization). Microbiology of food and animal feeding stuffs – Horizontal method for the enumeration of presumptive Bacillus cereus – Colony-count technique at 30 °C; ISO 7932:2004; ISO: Geneva, Switzerland, 2004.
- ISO (International Organization for Standardization). Microbiology of the food chain – Horizontal method for the detection, enumeration and serotyping of Salmonella – Part 1: Detection of Salmonella spp.; ISO 6579-1:2017; ISO: Geneva, Switzerland, 2017.
- ISO (International Organization for Standardization). Microbiology of the food chain – Horizontal method for the detection, enumeration and serotyping of Salmonella – Part 3: Guidelines for serotyping of Salmonella spp.; ISO 6579-3:2014; ISO: Geneva, Switzerland, 2014.
- ISO (International Organization for Standardization). Microbiology of the food chain – Horizontal method for the detection and enumeration of Listeria monocytogenes and of Listeria spp. – Part 1: Detection method; ISO 11290-1:2017; ISO: Geneva, Switzerland, 2017.
- Juneja, V.K.; Eblen, B.S.; Marks, H.M. Thermal inactivation of Salmonella spp. in poultry products. Journal of Food Protection 2001, 64, 80-87.
- Li, X.; Sheldon, B.W.; Walker, J.T.; Slavik, M.F. Thermal inactivation of Salmonella spp., Escherichia coli O157:H7, and Listeria monocytogenes in ready-to-eat meat products. Journal of Food Science 2005, 70, M112-M118.
- Kennedy, J.; Blair, I.S.; McDowell, D.A.; Bolton, D.J. An investigation of the thermal resistance of Staphylococcus aureus in foods. International Journal of Food Microbiology 2005, 99, 257-262.
- Doyle, M.P.; Buchanan, R.L. Food Microbiology: Fundamentals and Frontiers, 4th ed.; ASM Press: Washington, DC, USA 2013.
- Jay, J.M.; Loessner, M.J.; Golden, D.A. Modern Food Microbiology, 7th ed.; Springer: New York, NY, USA 2005.
- Mession, J.; Sok, N.; Assifaoui, A.; Saurel, R. Thermal denaturation of pea globulins (Pisum sativum L.): Molecular interactions leading to heat-induced protein aggregation. Food Chem. 2013, 139, 587-594. [CrossRef]
- Bulgaru, V.; Sensory, I.; Netreba, N.; Gurev, A.; Altanlas, U.; Paiu, S.; Dragancea, V.; Monsanu, A. Qualitative and antioxidant evaluation of high-moisture plant-based meat analogs obtained by extrusion. Foods 2025, 14, 2939. [CrossRef]
- Bainy, E.; Corredig, M.; Poysa, V.; Woodrow, L.; Tash, S. Assessment of the effects of soy protein isolates with different protein compositions on gluten thermosetting gelation. Food Hydrocoll. 2010, 24, 168-175. [CrossRef]
- Jang, J.; Lee, Y.; Kim, H. Advancements in plant-based meat analogs enhancing texture, flavor, and nutritional profiles. Nat. Food 2024, 5, 50.
- Cebrián, G.; Condón, S.; Mañas, P. Physiology of the inactivation of vegetative bacteria by thermal treatments: Mode of action, influence of environmental factors and inactivation kinetics. International Journal of Food Microbiology 2017, 266, 1-11. [CrossRef]
- Parniakov, O.; Barba, F.J.; Grimi, N.; Lebovka, N. Microbiological quality and safety of plant-based meat alternatives at retail level. Foods 2023, 12, 1123.
- Heir, E.; Bjerke, G.A.; Hånninen, M.-L.; Møretrø, T.; Langsrud, S. Microbiological hazards associated with plant-based meat alternatives. Microorganisms 2023, 11, 2432.
- Russell, N.J. The effects of chill storage and food processing on microbial growth. International Journal of Food Microbiology 2002, 79, 1-9.
- Hennekinne, J-A.; De Buyser, M-L.; Dragacci, S. Staphylococcus aureus and its food poisoning toxins: Characterization and outbreak investigation. FEMS Microbiology Reviews 2012, 36, 815-836. [CrossRef]
- Argudín, M.Á.; Mendoza, M.C.; Rodicio, M.R. Food poisoning and Stpahylococcus aureus enterotoxins. Toxins 2010, 2, 1751-1773. [CrossRef]
- Kadariya, J.; Smith, T.C.; Thapaliya, D. Staphylococcus aureus and staphylococcal food borne disease: An ongoing challenge in public health. BioMed Research International 2014, 827965. [CrossRef]
- European Commission. Commission Regulation (EC) No 2073.2005 on microbiological criteria for foodstuffs. Official Journal of the European Union 2005, L338, 1-26.
- Liu, Y.; Chen, Q.; Zhang, X.; Wang, J.; Li, H. Detection of Staphylococcus aureus enterotoxins in food: Recent advances and challenges. Comprehensive Reviews in Food Science and Food Safety 2023, 22, 1234-1256.
- ICMSF. Microorganisms in Foods 6: Microbial Ecology of food Commodities; Springer: Boston, MA, USA, 2005.
- EFSA BIOHAZ Panel. Scientific Opinion on the public health risks of Staphylococcus aureus toxins in food. EFSA Journal 2015, 13, 3994.
- U.S. FDA. Bad Bug Book: Foodborne Pathogenic Microorganisms and Natural Toxins Handbook, 2nd ed.; Silver Spring, MD, USA, 2012.
- Gutiérrez, D.; Delgado, S.; Vázquez-Sánchez, D.; Martínez, B., Rodríguez, A.; García, P. Incidence of Staphylococcus aureus and analysis of associated bacterial communities on food industry surfaces. Applied and Environmental Microbiology 2012, 78, 8547, 8554. [CrossRef]
- Barmettler, K.; Waser, S.; Stephan, R. Microbiological Quality of Plant-Based Meat-Alternative Products Collected at Retail Level in Switzerland. Journal of Food Protection 2025, 88, 100402. [CrossRef]
- Kabisch, J.; Joswig. G.; Böhnlein, C.; Fiedler, G.; Franz, C.M.A.P. Microbiological Status of Vegan Ground Meat Products from German Retail. Journal of Consumer Protection and Food Safety 2024, 19, 33-40. [CrossRef]
- Leistner, L.; Gould, G.W. Hurdle Technologies: Combination Treatments for Food Stability, Safety and Quality; Springer: Boston, MA, USA 2002.
- International Commission on Microbiological Specifications for Foods (ICMSF). Microorganisms in Foods 5: Characteristics of Microbial Pathogens; Springer: Boston, MS, USA, 1996.
- Jay, J.M.; Loessner, M.L.; Golde, D.A. Modern Food Microbiology, 7th ed,; Springer, New York, NY, USA, 2005.
- Barbosa-Cánovas, G.V.; Fontana, A.J.; Schmidt, S.J.; Labuza, T.P. Water Activity in Foods: Fundamentals and Applications; Blackwell Publishing: Ames, IA, USA, 2007.
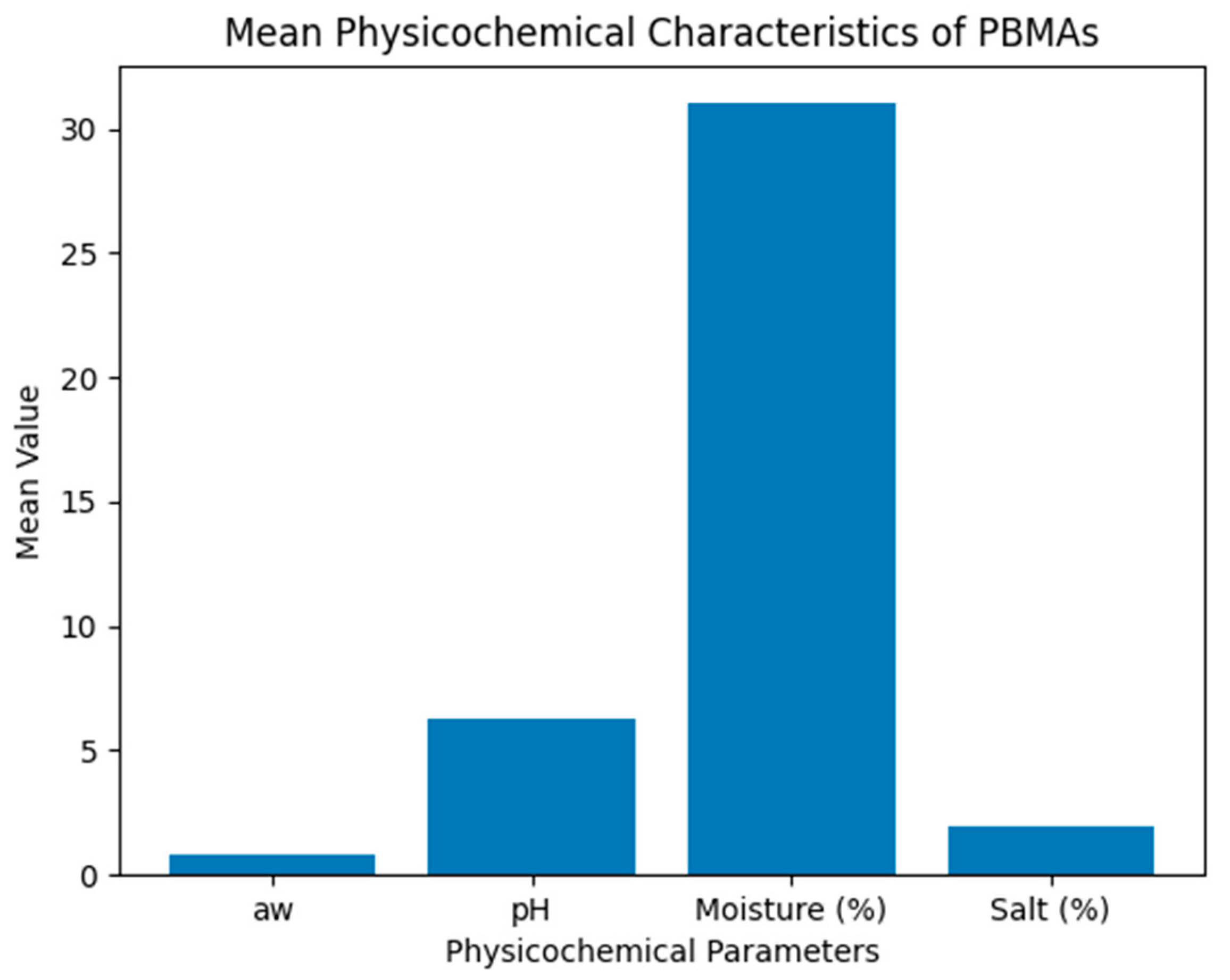
| Parameter | Minimum | Maximum | Mean ± SD | Range |
|---|---|---|---|---|
| aw | 0.68 | 1.0 | 0.85 ± 0.06 | 0.32 |
| pH | 5.3 | 7.1 | 6.3 ± 0.38 | 1.8 |
| Moisture (%) | 24% | 45% | 31% ± 5.5 | 21% |
| Salt (%) | 0.98% | 4.85% | 1.95% ± 0.55 | 3.87% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




