Submitted:
17 April 2026
Posted:
21 April 2026
You are already at the latest version
Abstract
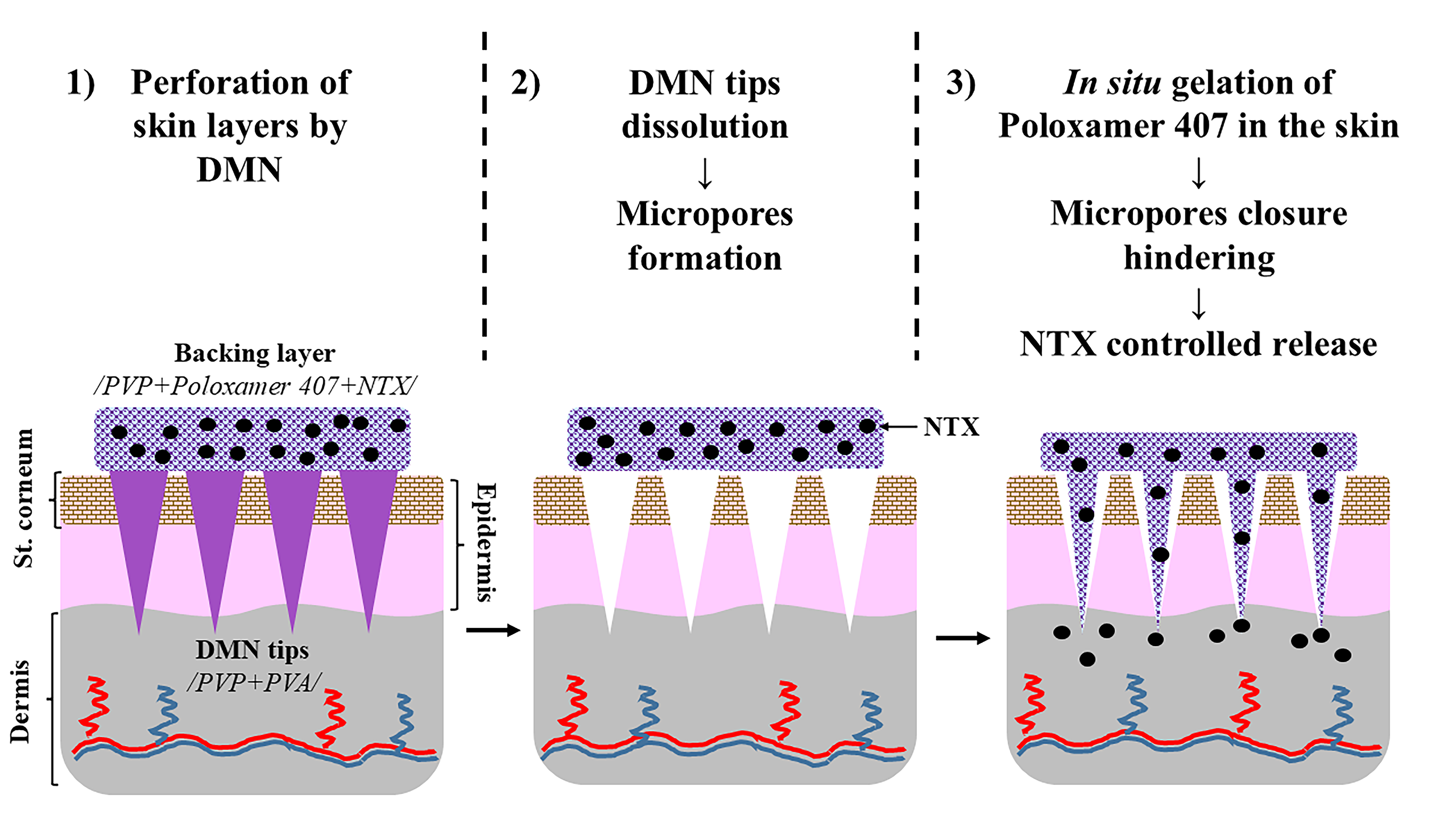
Keywords:
1. Introduction
2. Results and Discussion
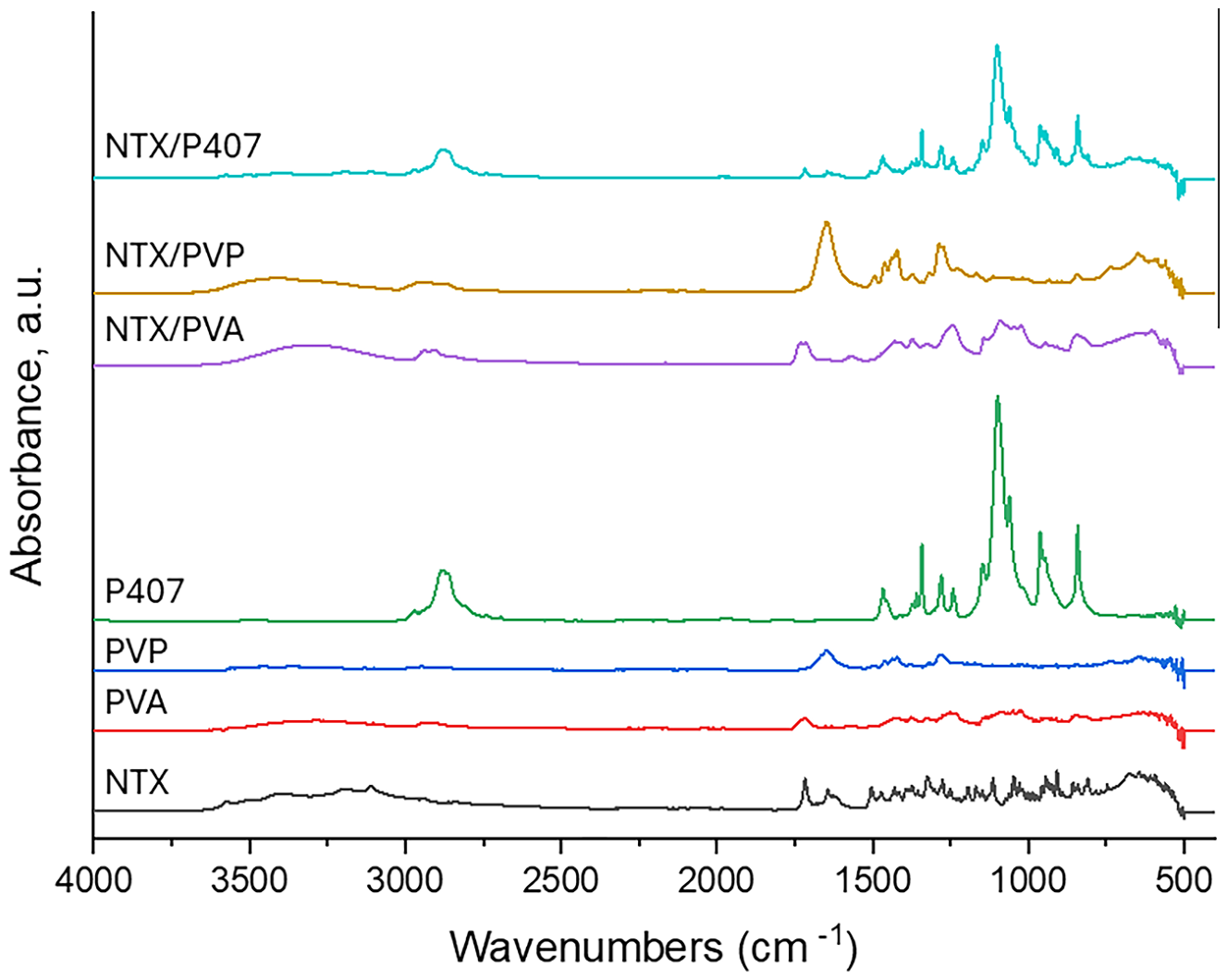
2.1. Fourier Transform Infrared (FT-IR) Spectroscopy
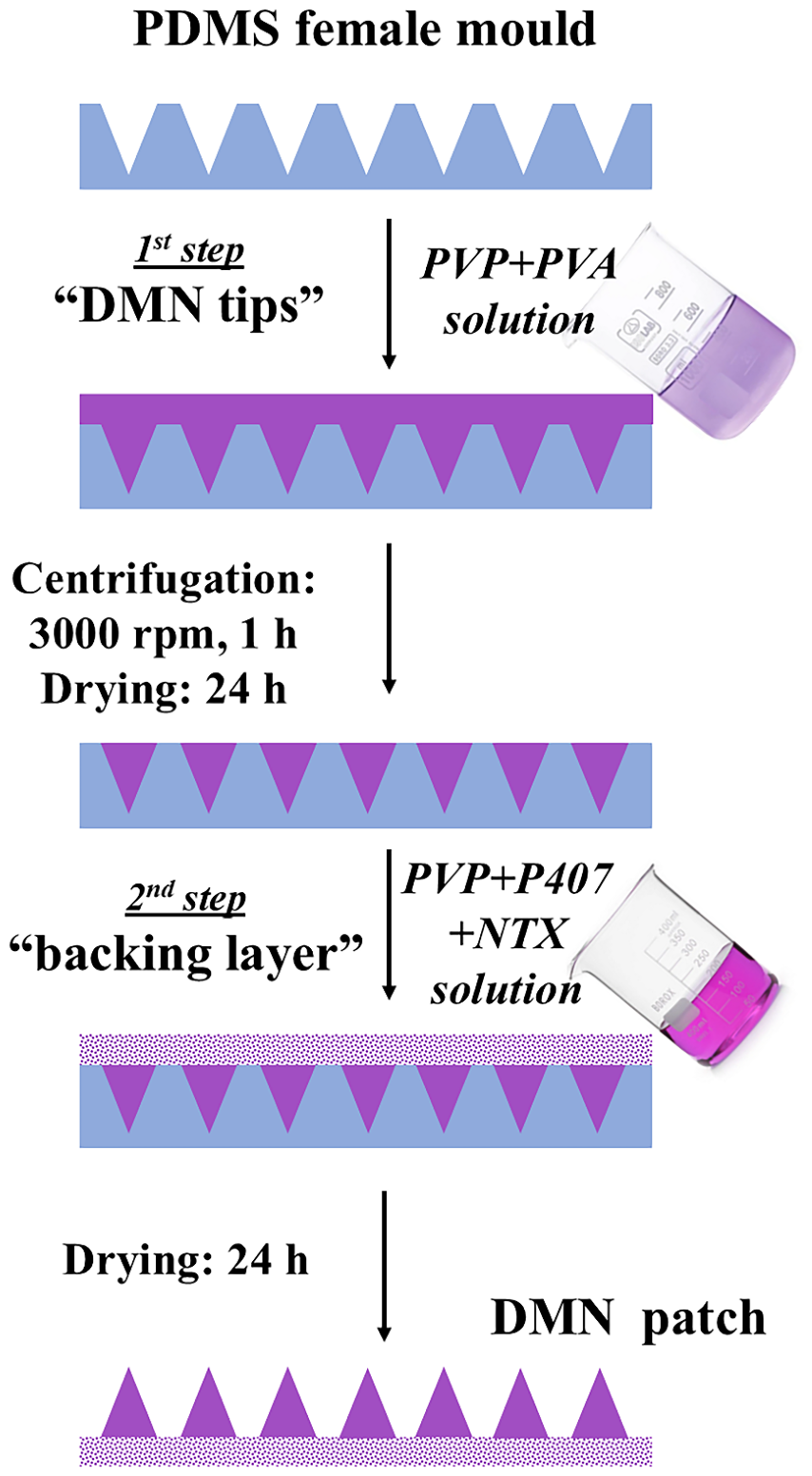
2.2. Preparation of Dissolving Microneedle Patches (DMN Patches) and Simple Matrix Patches (SP Patches)
2.3. Scanning Electron Microscopy (SEM) of DMN Patches
2.4. The Assay of API
2.5. In Vitro Gelation Temperature and Gelling Time
2.6. Mechanical Properties
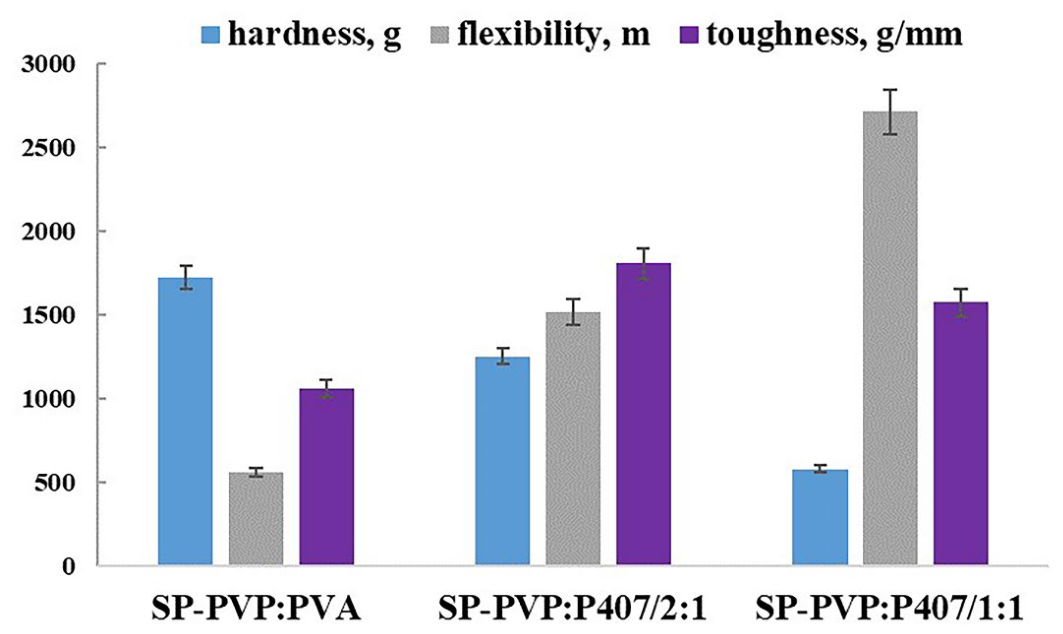
2.6.1. Three-Point Bend Testing of SP Patches
2.6.2. Mechanical Strength of DMN Patches (Fracture Force Test)
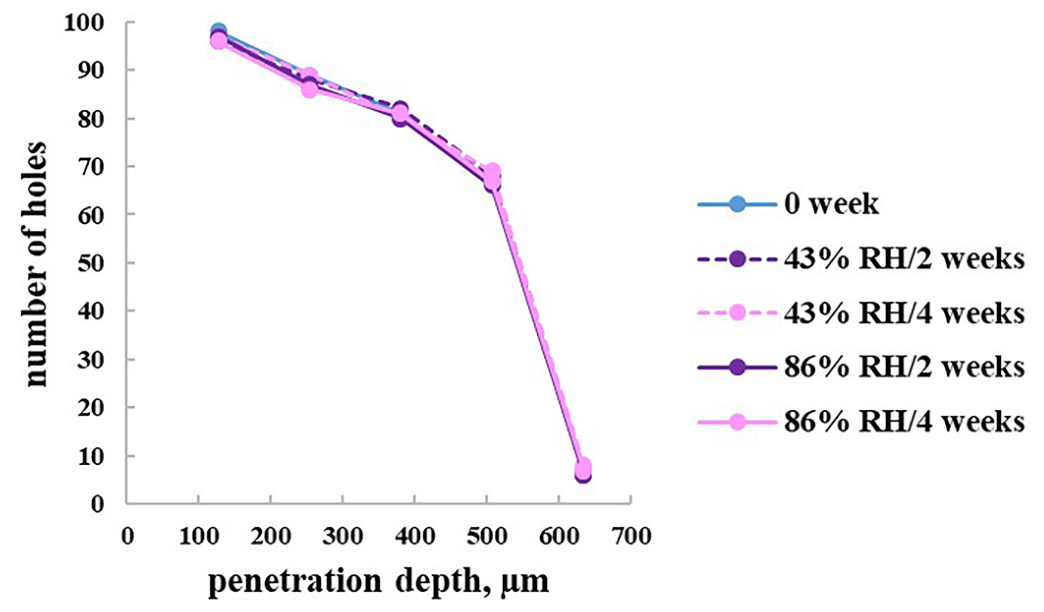
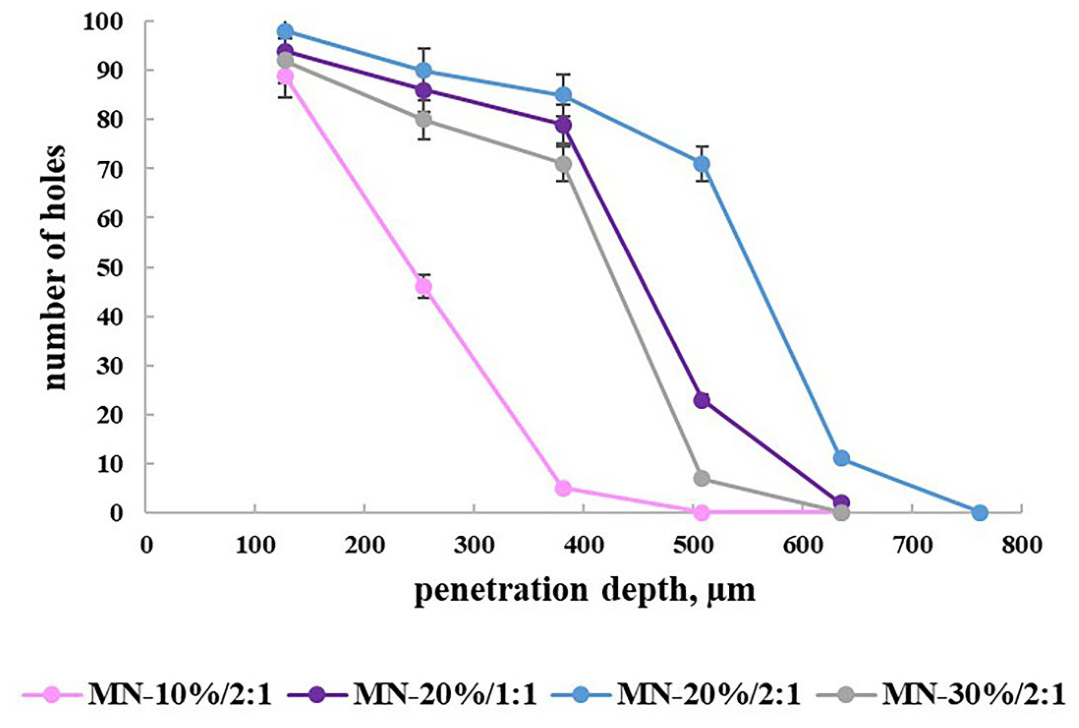
2.6.3. Parafilm Penetration Test of DMN Patches
2.7. In Vitro Release Study
2.8. In Vitro Permeability Study
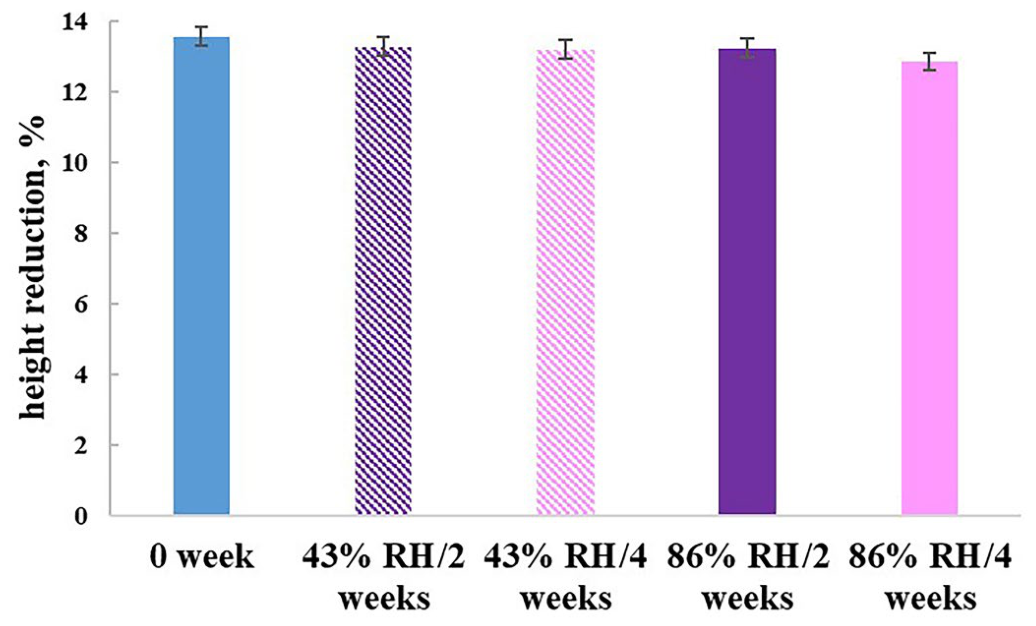
2.9. Stability Study
3. Materials and Methods
3.1. Materials
3.2. Fourier-Transform Infrared (FTIR) Spectroscopy
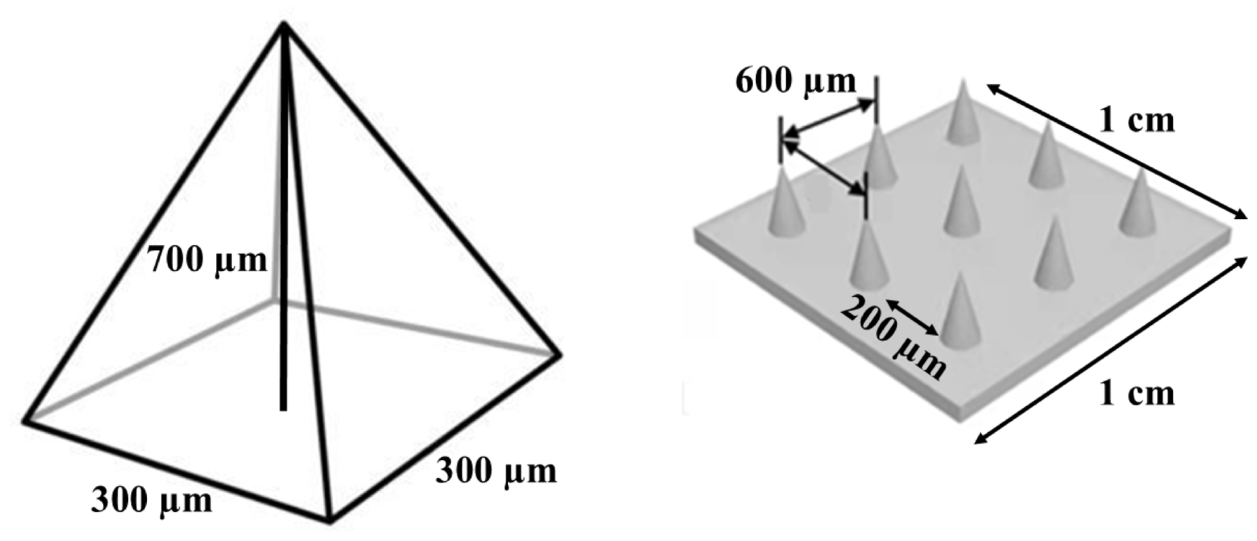
3.3. Preparation of Dissolving Microneedle Patches (DMN Patches)
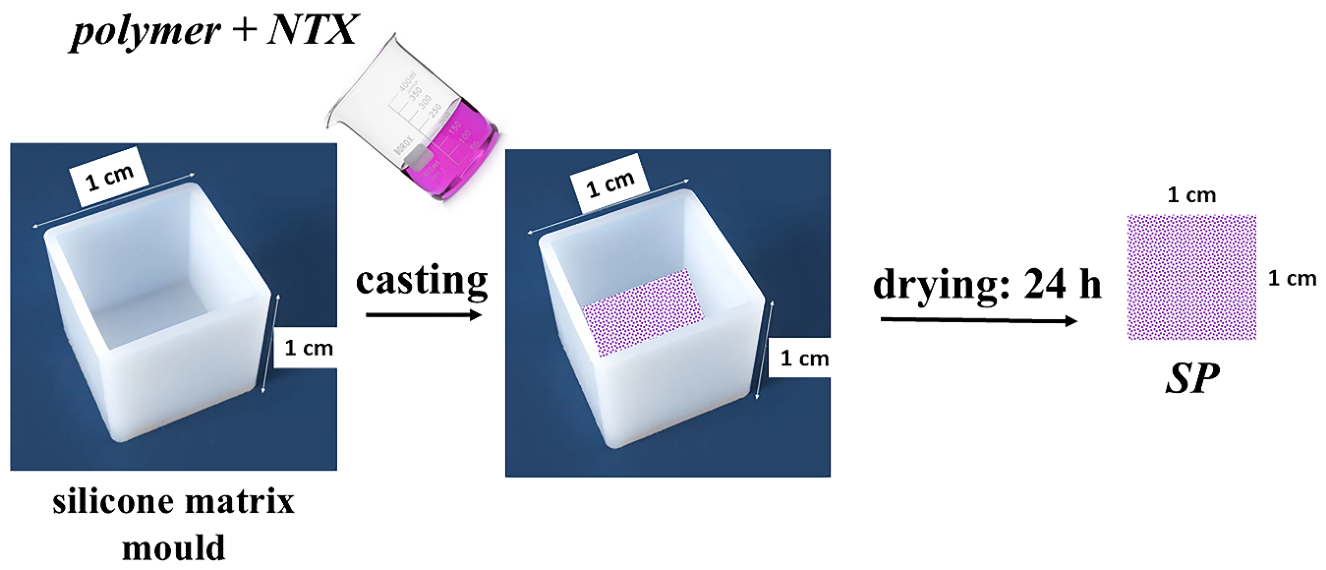
3.4. Preparation of Simple Matrix Patches (SP Patches)
3.5. Scanning Electron Microscopy (SEM) of DMN Patches
3.6. The Assay of API
3.7. In Vitro Gelation Temperature and Gelling Time
3.8. Mechanical Properties
3.8.1. Three-Point Bend Testing of Simple Matrix Patches
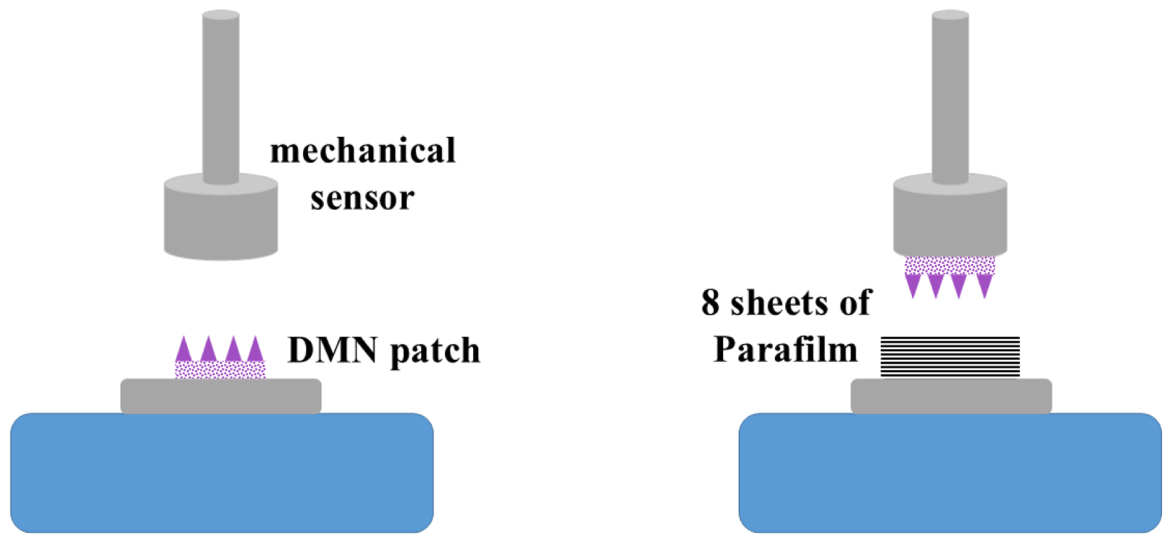
3.8.2. Mechanical Strength of DMN Patches (Fracture Force Test)
3.8.3. Parafilm Penetration Test of DMN Patches
3.9. In Vitro Drug Release Study
3.10. In Vitro Permeability Study
3.11. Stability Study
4. Conclusions
5. Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| API | Active pharmaceutical ingredient |
| DMN | Dissolving microneedles |
| DMN patch | Dissolving microneedle patch |
| FT-IR | Fourier Transform Infrared |
| MN | Microneedles |
| NTX | Naltrexone hydrochloride |
| P407 | Poloxamer 407 |
| PNIPAM | Poly(N-isopropylacrylamide) |
| PVA | Polyvinyl alcohol |
| PVP | Polyvinylpyrrolidone |
| SD | Standard deviation |
| SEM | Scanning electron microscopy |
References
- Singh, D.; Saadabadi, A. Naltrexone. In StatPearls; StatPearls Publishing: Treasure Island (FL), 2025. [Google Scholar]
- Kim, P.S.; Fishman, M.A. Low-Dose Naltrexone for Chronic Pain: Update and Systemic Review. Curr Pain Headache Rep 2020, 24, 64. [Google Scholar] [CrossRef]
- Leiber, K.K.; Parker, R.W. Therapeutic Uses and Efficacy of Low-Dose Naltrexone: A Scoping Review. Cureus 2025. [Google Scholar] [CrossRef]
- Stancil, S.L.; Abdel-Rahman, S.; Wagner, J. Developmental Considerations for the Use of Naltrexone in Children and Adolescents. J Pediatr Pharmacol Ther 2021, 26, 675–695. [Google Scholar] [CrossRef]
- Gonzalez, J.P.; Brogden, R.N. Naltrexone. Drugs 1988, 35, 192–213. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.W.; Fujioka, K. Naltrexone for the Treatment of Obesity: Review and Update. Expert Opinion on Pharmacotherapy 2009, 10, 1841–1845. [Google Scholar] [CrossRef] [PubMed]
- New Drugs 2. In Therapeutics Letter; Perry, T., Ed.; Therapeutics Initiative: Vancouver (BC), 1994. [Google Scholar]
- Shenkoya, B.; Gopalakrishnan, M.; Eke, A.C. Physiologically Based Pharmacokinetic Modeling of Long-Acting Extended-Release Naltrexone in Pregnant Women with Opioid Use Disorder. CPT: Pharmacometrics & Systems Pharmacology 2024, 13, 1939–1952. [Google Scholar] [CrossRef]
- Sudakin, D. Naltrexone: Not Just for Opioids Anymore. J Med Toxicol 2016, 12, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Santos, R.A.; Rae, M.; Dartora, V.F.M.C.; Matos, J.K.R.; Camarini, R.; Lopes, L.B. Bioresponsive Nanostructured Systems for Sustained Naltrexone Release and Treatment of Alcohol Use Disorder: Development and Biological Evaluation. International Journal of Pharmaceutics 2020, 585, 119474. [Google Scholar] [CrossRef]
- Shulman, M.; Greiner, M.G.; Tafessu, H.M.; Opara, O.; Ohrtman, K.; Potter, K.; Hefner, K.; Jelstrom, E.; Rosenthal, R.N.; Wenzel, K.; et al. Rapid Initiation of Injection Naltrexone for Opioid Use Disorder: A Stepped-Wedge Cluster Randomized Clinical Trial. JAMA Netw Open 2024, 7, e249744. [Google Scholar] [CrossRef]
- Syed, Y.Y.; Keating, G.M. Extended-Release Intramuscular Naltrexone (VIVITROL®): A Review of Its Use in the Prevention of Relapse to Opioid Dependence in Detoxified Patients. CNS Drugs 2013, 27, 851–861. [Google Scholar] [CrossRef]
- Sofuoglu, M.; DeVito, E.E.; Carroll, K.M. Pharmacological and Behavioral Treatment of Opioid Use Disorder. Psychiatr Res Clin Pract 2018, 1, 4–15. [Google Scholar] [CrossRef]
- Nalluri, B.N.; Milligan, C.; Chen, J.; Crooks, P.A.; Stinchcomb, A.L. In Vitro Release Studies on Matrix Type Transdermal Drug Delivery Systems of Naltrexone and Its Acetyl Prodrug. Drug Dev Ind Pharm 2005, 31, 871–877. [Google Scholar] [CrossRef]
- Milewski, M.; Pinninti, R.R.; Stinchcomb, A.L. Naltrexone Salt Selection for Enhanced Transdermal Permeation through Microneedle-Treated Skin. J Pharm Sci 2012, 101, 2777–2786. [Google Scholar] [CrossRef]
- Paudel, K.S.; Nalluri, B.N.; Hammell, D.C.; Valiveti, S.; Kiptoo, P.; Hamad, M.O.; Crooks, P.A.; Stinchcomb, A.L. Transdermal Delivery of Naltrexone and Its Active Metabolite 6-Beta-Naltrexol in Human Skin in Vitro and Guinea Pigs in Vivo. J Pharm Sci 2005, 94, 1965–1975. [Google Scholar] [CrossRef]
- Siamidi, A.; Dedeloudi, A.; Vlachou, M. Probing the Release of Bupropion and Naltrexone Hydrochloride Salts from Biopolymeric Matrices of Diverse Chemical Structures. Polymers (Basel) 2021, 13, 1456. [Google Scholar] [CrossRef]
- Vaseem, R.S.; D’cruz, A.; Shetty, S.; -, H.; Vardhan, A.; R, S.S.; Marques, S.M.; Kumar, L.; Verma, R. Transdermal Drug Delivery Systems: A Focused Review of the Physical Methods of Permeation Enhancement. Adv Pharm Bull 2024, 14, 67–85. [Google Scholar] [CrossRef]
- Guillot, A.J.; Cordeiro, A.S.; Donnelly, R.F.; Montesinos, M.C.; Garrigues, T.M.; Melero, A. Microneedle-Based Delivery: An Overview of Current Applications and Trends. Pharmaceutics 2020, 12, 569. [Google Scholar] [CrossRef]
- Jia, B.; Xia, T.; Wang, X.; Xu, Y.; Guo, Z. Morphology Design of Polymer Microneedle Arrays: Key Factors from the Application Perspective. Journal of Drug Delivery Science and Technology 2023, 88, 104883. [Google Scholar] [CrossRef]
- Ferreira, L.M.; Sari, M.H.M.; Azambuja, J.H.; da Silveira, E.F.; Cervi, V.F.; Marchiori, M.C.L.; Maria-Engler, S.S.; Wink, M.R.; Azevedo, J.G.; Nogueira, C.W.; et al. Xanthan Gum-Based Hydrogel Containing Nanocapsules for Cutaneous Diphenyl Diselenide Delivery in Melanoma Therapy. Invest New Drugs 2020, 38, 662–674. [Google Scholar] [CrossRef]
- Stinchcomb, A.; Banks, S.; Pinninti, R. Transdermal Delivery of Naltrexone Hydrochloride, Naltrexol Hydrochloride, and Bis(Hydroxy-Methyl)Propionyl-3-0 Ester Naltrexone Using Microneedles 2008.
- Tobin, K.V.; Fiegel, J.; Brogden, N.K. Thermosensitive Gels Used to Improve Microneedle-Assisted Transdermal Delivery of Naltrexone. Polymers 2021, 13, 933. [Google Scholar] [CrossRef]
- V. Tobin, K.; K. Brogden, N. Thermosensitive Biomaterial Gels with Chemical Permeation Enhancers for Enhanced Microneedle Delivery of Naltrexone for Managing Opioid and Alcohol Dependency. Biomaterials Science 2023, 11, 5846–5858. [Google Scholar] [CrossRef]
- Milewski, M.; Paudel, K.S.; Brogden, N.K.; Ghosh, P.; Banks, S.L.; Hammell, D.C.; Stinchcomb, A.L. Microneedle-Assisted Percutaneous Delivery of Naltrexone Hydrochloride in Yucatan Minipig: In Vitro-in Vivo Correlation. Mol Pharm 2013, 10. [Google Scholar] [CrossRef] [PubMed]
- Wermeling, D.P.; Banks, S.L.; Hudson, D.A.; Gill, H.S.; Gupta, J.; Prausnitz, M.R.; Stinchcomb, A.L. Microneedles Permit Transdermal Delivery of a Skin-Impermeant Medication to Humans. Proc Natl Acad Sci U S A 2008, 105, 2058–2063. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, P.; Brogden, N.; Stinchcomb, A. Effect of Formulation PH on Transport of Naltrexone Species and Pore Closure in Microneedle-Enhanced Transdermal Drug Delivery. Molecular pharmaceutics 2013, 10. [Google Scholar] [CrossRef] [PubMed]
- Priya, S.; Tomar, Y.; Desai, V.M.; Singhvi, G. Enhanced Skin Drug Delivery Using Dissolving Microneedles: A Potential Approach for the Management of Skin Disorders. Expert Opin Drug Deliv 2023, 20, 721–738. [Google Scholar] [CrossRef]
- Moawad, F.; Pouliot, R.; Brambilla, D. Dissolving Microneedles in Transdermal Drug Delivery: A Critical Analysis of Limitations and Translation Challenges. Journal of Controlled Release 2025, 383, 113794. [Google Scholar] [CrossRef]
- Zhuo, Y.; Wang, F.; Lv, Q.; Fang, C. Dissolving Microneedles: Drug Delivery and Disease Treatment. Colloids and Surfaces B: Biointerfaces 2025, 250, 114571. [Google Scholar] [CrossRef]
- Lee, J.W.; Park, J.-H.; Prausnitz, M.R. Dissolving Microneedles for Transdermal Drug Delivery. Biomaterials 2008, 29, 2113–2124. [Google Scholar] [CrossRef]
- Wang, Z.; Liu, L.; Liu, E.; Chen, R.; Huang, Y.; Li, Q. Carrier Polymer-Free Dissolvable Microneedles Enable Superhigh Drug Payload for Percutaneous Protein Delivery. ACS Materials Lett. 2024, 6, 4980–4987. [Google Scholar] [CrossRef]
- Li, J.Y.; Feng, Y.H.; He, Y.T.; Hu, L.F.; Liang, L.; Zhao, Z.Q.; Chen, B.Z.; Guo, X.D. Thermosensitive Hydrogel Microneedles for Controlled Transdermal Drug Delivery. Acta Biomaterialia 2022, 153, 308–319. [Google Scholar] [CrossRef]
- Roussel, S.; Udabe, J.; Bin Sabri, A.; Calderón, M.; Donnelly, R. Leveraging Novel Innovative Thermoresponsive Polymers in Microneedles for Targeted Intradermal Deposition. Int J Pharm 2024, 652, 123847. [Google Scholar] [CrossRef]
- Khan, S.; Minhas, M.U.; Tekko, I.A.; Donnelly, R.F.; Thakur, R.R.S. Evaluation of Microneedles-Assisted in Situ Depot Forming Poloxamer Gels for Sustained Transdermal Drug Delivery. Drug Deliv. and Transl. Res. 2019, 9, 764–782. [Google Scholar] [CrossRef]
- Khan, S.; Minhas, M.U. Micro Array Patch Assisted Transdermal Delivery of High Dose, Ibuprofen Sodium Using Thermoresponsive Sodium Alginate/Poly (Vinylcaprolactam) in Situ Gels Depot. International Journal of Biological Macromolecules 2023, 252, 126464. [Google Scholar] [CrossRef]
- Khan, S.; Minhas, M.U.; Singh Thakur, R.R.; Aqeel, M.T. Microneedles Assisted Controlled and Improved Transdermal Delivery of High Molecular Drugs via in Situ Forming Depot Thermoresponsive Poloxamers Gels in Skin Microchannels. Drug Development and Industrial Pharmacy 2022, 48, 265–278. [Google Scholar] [CrossRef]
- Menze, A.R.; Sinnott, J.P.; Nazarenko, A.Y. Crystal Structure of Naltrexone Chloride Solvates with Ethanol, Propan-2-Ol, and 2-Methyl propan-2-Ol. Acta Crystallogr E Crystallogr Commun 2017, 73, 1013–1020. [Google Scholar] [CrossRef]
- Tavakol, H.; Esfandyari, M.; Taheri, S.; Heydari, A. Investigation of Structure, Vibrational and NMR Spectra of Oxycodone and Naltrexone: A Combined Experimental and Theoretical Study. Spectrochim Acta A Mol Biomol Spectrosc 2011, 79, 574–582. [Google Scholar] [CrossRef] [PubMed]
- D’Amelia, R.P.; Huang, L.; Mancuso, J. Quantitative Analysis of Polyvinyl Alcohol-Polyethylene (PVOH-PE) Copolymers and Polyvinyl Pyrrolidone-Polyvinyl Acetate (PVP-PVAc) Copolymers and Blends Using Fourier Transform Infrared Spectroscopy and Elemental Analysis. World Journal of Chemical Education 2019, 7, 1–11. [Google Scholar] [CrossRef]
- Mansur, H.S.; Sadahira, C.M.; Souza, A.N.; Mansur, A.A.P. FTIR Spectroscopy Characterization of Poly (Vinyl Alcohol) Hydrogel with Different Hydrolysis Degree and Chemically Crosslinked with Glutaraldehyde. Materials Science and Engineering: C 2008, 28, 539–548. [Google Scholar] [CrossRef]
- Karolewicz, B.; Gajda, M.; Pluta, J.; Górniak, A. Dissolution Study and Thermal Analysis of Fenofibrate–Pluronic F127 Solid Dispersions. J Therm Anal Calorim 2016, 125, 751–757. [Google Scholar] [CrossRef]
- Giuliano, E.; Paolino, D.; Fresta, M.; Cosco, D. Mucosal Applications of Poloxamer 407-Based Hydrogels: An Overview. Pharmaceutics 2018, 10. [Google Scholar] [CrossRef]
- Sullivan, S.P.; Koutsonanos, D.G.; del Pilar Martin, M.; Lee, J.W.; Zarnitsyn, V.; Choi, S.-O.; Murthy, N.; Compans, R.W.; Skountzou, I.; Prausnitz, M.R. Dissolving Polymer Microneedle Patches for Influenza Vaccination. Nat Med 2010, 16, 915–920. [Google Scholar] [CrossRef] [PubMed]
- Tong, R.; Kohane, D.S. Shedding Light on Nanomedicine. WIREs Nanomedicine and Nanobiotechnology 2012, 4, 638–662. [Google Scholar] [CrossRef]
- Jin, S.Y.; Park, E.J.-J.; Kwon, S.M.; Jung, H.-S.; Kim, D.W. Rapidly Dissolving Microneedles Incorporating Lidocaine Hydrochloride: A PVP/PVA-Based Approach for Local Anesthesia. Pharmaceutics 2025, 17. [Google Scholar] [CrossRef]
- Justyna, K.; Patrycja, Ś; Krzysztof, M.; Rafał, W. Dissolving Microneedles Fabricated from 3D-Printed Master Molds for Application in Veterinary Medicine. Sci Rep 2025, 15, 14102. [Google Scholar] [CrossRef]
- Ramadon, D.; Ulayya, F.; Qur’ani, A.S.; Iskandarsyah, I.; Harahap, Y.; Anjani, Q.K.; Aileen, V.; Hartrianti, P.; Donnelly, R.F. Combination of Dissolving Microneedles with Nanosuspension and Co-Grinding for Transdermal Delivery of Ketoprofen. Pharmaceuticals 2023, 16, 378. [Google Scholar] [CrossRef]
- Chen, Y.; Lee, J.-H.; Meng, M.; Cui, N.; Dai, C.-Y.; Jia, Q.; Lee, E.-S.; Jiang, H.-B. An Overview on Thermosensitive Oral Gel Based on Poloxamer 407. Materials 2021, 14. [Google Scholar] [CrossRef]
- Hennig, M. Microneedles Assisted Controlled and Improved Transdermal Delivery of High Molecular Drugs via in Situ Forming Depot Thermorespo. Pharma Excipients 2022. [Google Scholar]
- Alrimawi, B.H.; Lee, J.Y.; Ng, K.W.; Goh, C.F. In Vitro Evaluation of Microneedle Strength: A Comparison of Test Configurations and Experimental Insights. RSC Pharm. 2024, 1, 227–233. [Google Scholar] [CrossRef]
- La Malfa, F.; van Hulst, I.A.; Ossendorp, F.; Staufer, U.; van der Maaden, K. A Comprehensive Design-to-Skin Pipeline to Fabricate Polymeric Microneedles Using Ultrahigh-Resolution 3D Printing. Pharm Res 2025, 42, 2125–2143. [Google Scholar] [CrossRef]
- Chellathurai, M.S.; Ling, V.W.; Palanirajan, V.; Chellathurai, M.S.; Ling, V.W.; Palanirajan, V. Fabrication and Evaluation of Transdermal Microneedles for a Recombinant Human Keratinocyte Growth Factor. Turkish Journal of Pharmaceutical Sciences 2021. [Google Scholar] [CrossRef]
- Yang, S.; Feng, Y.; Zhang, L.; Chen, N.; Yuan, W.; Jin, T. A Scalable Fabrication Process of Polymer Microneedles. Int J Nanomedicine 2012, 7, 1415–1422. [Google Scholar] [CrossRef]
- Hennessy, M.G.; Craster, R.V.; Matar, O.K. Drying-Induced Stresses in Poroelastic Drops on Rigid Substrates. Phys. Rev. E 2022, 105, 054602. [Google Scholar] [CrossRef]
- Dave, R.; Shinde, S.; Kalayil, N.; Budar, A. Engineering Microscopic Delivery Systems: A Review of Dissolving Microneedle Design, Fabrication, and Function. Micro and Nano Syst Lett 2024, 12, 14. [Google Scholar] [CrossRef]
- Zhang, Z.; Du, G.; Sun, X.; Zhang, Z. Viscoelastic Properties of Polymeric Microneedles Determined by Micromanipulation Measurements and Mathematical Modelling. Materials 2023, 16. [Google Scholar] [CrossRef]
- Eltayib, E.M. Fabrication of Porous Polymeric Microneedles: A Concise Overview. RSC Adv 15 18697–18714. [CrossRef]
- Smith, E.; Lau, W.M.; Abdelghany, T.M.; Vukajlovic, D.; Novakovic, K.; Ng, K.W. Vac-and-Fill: A Micromoulding Technique for Fabricating Microneedle Arrays with Vacuum-Activated, Hands-Free Mould-Filling. International Journal of Pharmaceutics 2024, 650, 123706. [Google Scholar] [CrossRef]
- Yu, W.; Gu, J.; Li, Z.; Ruan, S.; Chen, B.; Shen, C.; Lee, L.J.; Wang, X. Study on the Influence of Microinjection Molding Processing Parameters on Replication Quality of Polylactic Acid Microneedle Array Product. Polymers 2023, 15. [Google Scholar] [CrossRef]
- Rad, Z.F.; Prewett, P.D.; Davies, G.J. An Overview of Microneedle Applications, Materials, and Fabrication Methods. Beilstein J. Nanotechnol. 2021, 12, 1034–1046. [Google Scholar] [CrossRef]
- Xiu, X.; Gao, G.; Liu, Y.; Ma, F. Drug Delivery with Dissolving Microneedles: Skin Puncture, Its Influencing Factors and Improvement Strategies. Journal of Drug Delivery Science and Technology 2022, 76, 103653. [Google Scholar] [CrossRef]
- Hennig, M. Alteration of Gel Point of Poloxamer 338 Induced by Pharmaceutical Actives and Excipients. Pharma Excipients 2025. [Google Scholar]
- Morita, T.; Torii, Y.; Imamura, H.; Kadota, T.; Sakuma, F.; Higashi, K.; Sumi, T. Pair Potential between Poloxamer 407 Micelles in 14 Wt % Aqueous Solution Clarifying the Sol–Gel–Sol Transition by Temperature Rise. J. Phys. Chem. Lett. 2024, 15, 7909–7915. [Google Scholar] [CrossRef]
- Balu, P.; Srikanth, S.; Gnandhas, D.P.; Durai, R.D.; Ulaganathan, V.; B Narayanan, V.H. Development and Optimization of an Injectable In-Situ Gel System for Sustained Release of Anti-Tuberculosis Drugs. Sci Rep 2025, 15, 21383. [Google Scholar] [CrossRef]
- Sakuma, F.; Higashi, K.; Ueda, K.; Morita, T.; Iohara, D.; Hirayama, F.; Moribe, K. Effect of Acetaminophen on Poloxamer 407 Micelles and Hydrogels: The Relationship between Structural and Physical Properties. Langmuir 2024, 40, 15610–15620. [Google Scholar] [CrossRef]
- Smith, F.; Fiedler, B.; Elkassas, K.; Mohamed, R.; Cheung, K.; Zelzer, M.; Crean, A.; Chowdhury, F.; Segal, J.; Rawson, F.; et al. Optimisation of Backing Layer Formulations via Rational Polymer Selection to Improve the Insertion of Dissolving Microneedles Into Skin. Mol. Pharmaceutics 2026, 23, 757–767. [Google Scholar] [CrossRef]
- Dołowacka-Jóźwiak, A.; Nawrot-Hadzik, I.; Matkowski, A.; Nowakowski, P.; Dudek-Wicher, R.; Markowska, D.; Adamski, R.; Krzyżanowska-Gołąb, D.; Karolewicz, B. Optimization of Cellulose Derivative-, PVA-, and PVP-Based Films with Reynoutria Japonica Extract to Improve Periodontal Disease Treatment. Materials 2024, 17. [Google Scholar] [CrossRef]
- Jamasri, J.; Yudhanto, F.; Yudha, V.; Syafri, E. Mechanical, Physical and Thermal Characterization of PVA (Polyvinyl Alcohol)/Chitosan Bioplastic Film. International Journal of Heat and Technology 2023, 41, 687–693. [Google Scholar] [CrossRef]
- Mondal, D.; Mollick, Md.M.R.; Bhowmick, B.; Maity, D.; Bain, M.K.; Rana, D.; Mukhopadhyay, A.; Dana, K.; Chattopadhyay, D. Effect of Poly(Vinyl Pyrrolidone) on the Morphology and Physical Properties of Poly(Vinyl Alcohol)/Sodium Montmorillonite Nanocomposite Films. Progress in Natural Science: Materials International 2013, 23, 579–587. [Google Scholar] [CrossRef]
- Sallal, A.A.; Salman, S.A.; Habeeb, A.A. STUDYING OF SOME DIELECTRIC AND MECHANICAL PROPERTIES OF (PVA:PVP) POLYMER BLEND. International Journal of Current Research 2018, 10. [Google Scholar]
- Rao, X.; Zhou, Q.; Wen, Q.; Ou, Z.; Fu, L.; Gong, Y.; Du, X.; Huo, C. High-Performance and Water Resistant PVA-Based Films Modified by Air Plasma Treatment. Membranes 2022, 12. [Google Scholar] [CrossRef]
- Yang, T.; Wu, J.; Yao, Y.; Wang, K.; Zhang, Q.; Fu, Q. Towards the Toughness-Strength Balance of Poly(Vinyl Alcohol) Films via Synergic Plasticization. Polymer 2024, 301, 127031. [Google Scholar] [CrossRef]
- Lu, Z.; Geng, B.; Ma, Q.; Ning, D.; Zhao, R.; Kong, F.; E, S. Polymer Induced Strengthening and Toughening of Aramid Nanofiber Film: The Importance of Densification and Hydrogen Bonding. Applied Surface Science 2023, 607, 155045. [Google Scholar] [CrossRef]
- Deng, C.; Dong, H.; Kou, Y.; Liu, H.; Sun, K.; Jian, W.; Shi, Q. Flexible Self-Healing Phase Change Film with High Transition Enthalpy for Thermal Management. Journal of Energy Storage 2023, 62, 106873. [Google Scholar] [CrossRef]
- Jantrawut, P.; Chaiwarit, T.; Jantanasakulwong, K.; Brachais, C.H.; Chambin, O. Effect of Plasticizer Type on Tensile Property and In Vitro Indomethacin Release of Thin Films Based on Low-Methoxyl Pectin. Polymers 2017, 9. [Google Scholar] [CrossRef]
- Tarique, J.; Sapuan, S.M.; Khalina, A. Effect of Glycerol Plasticizer Loading on the Physical, Mechanical, Thermal, and Barrier Properties of Arrowroot (Maranta Arundinacea) Starch Biopolymers. Sci Rep 2021, 11, 13900. [Google Scholar] [CrossRef]
- Van Zee, N.J.; Peroutka, A.S.; Hillmyer, M.A.; Lodge, T.P. Effect of Poloxamer Binding on the Elasticity and Toughness of Model Lipid Bilayers. Langmuir 2023, 39, 7258–7267. [Google Scholar] [CrossRef]
- Abdeltawab, H.; Svirskis, D.; Hill, A.G.; Sharma, M. Increasing the Hydrophobic Component of Poloxamers and the Inclusion of Salt Extend the Release of Bupivacaine from Injectable In Situ Gels, While Common Polymer Additives Have Little Effect. Gels 2022, 8. [Google Scholar] [CrossRef]
- Tahir, M.; Vicini, S.; Jędrzejewski, T.; Wrotek, S.; Sionkowska, A. New Composite Materials Based on PVA, PVP, CS, and PDA. Polymers 2024, 16. [Google Scholar] [CrossRef]
- Jeong, W.Y.; Kwon, M.; Choi, H.E.; Kim, K.S. Recent Advances in Transdermal Drug Delivery Systems: A Review. Biomaterials Research 2021, 25, 24. [Google Scholar] [CrossRef]
- Teodorescu, M.; Bercea, M.; Morariu, S. Biomaterials of PVA and PVP in Medical and Pharmaceutical Applications: Perspectives and Challenges. Biotechnology Advances 2019, 37, 109–131. [Google Scholar] [CrossRef]
- Ando, D.; Miyatsuji, M.; Sakoda, H.; Yamamoto, E.; Miyazaki, T.; Koide, T.; Sato, Y.; Izutsu, K. Mechanical Characterization of Dissolving Microneedles: Factors Affecting Physical Strength of Needles. Pharmaceutics 2024, 16, 200. [Google Scholar] [CrossRef] [PubMed]
- Chanabodeechalermrung, B.; Chaiwarit, T.; Chaichit, S.; Udomsom, S.; Baipaywad, P.; Worajittiphon, P.; Jantrawut, P. HPMC/PVP K90 Dissolving Microneedles Fabricated from 3D-Printed Master Molds: Impact on Microneedle Morphology, Mechanical Strength, and Topical Dissolving Property. Polymers 2024, 16, 452. [Google Scholar] [CrossRef]
- Donnelly, R.F.; Singh, T.R.R.; Morrow, D.I.J.; Woolfson, A.D. Microneedle-Mediated Transdermal and Intradermal Drug Delivery; John Wiley & Sons, 2012; ISBN 978-1-119-95966-3. [Google Scholar]
- Du, G.; Zhang, Z.; He, P.; Zhang, Z.; Sun, X. Determination of the Mechanical Properties of Polymeric Microneedles by Micromanipulation. Journal of the Mechanical Behavior of Biomedical Materials 2021, 117, 104384. [Google Scholar] [CrossRef]
- Larrañeta, E.; Moore, J.; Vicente-Pérez, E.M.; González-Vázquez, P.; Lutton, R.; Woolfson, A.D.; Donnelly, R.F. A Proposed Model Membrane and Test Method for Microneedle Insertion Studies. International Journal of Pharmaceutics 2014, 472, 65–73. [Google Scholar] [CrossRef]
- Putri, H.E.; Utami, R.N.; Aliyah; Wahyudin, E.; Oktaviani, W.W.; Mudjahid, M.; Permana, A.D. Dissolving Microneedle Formulation of Ceftriaxone: Effect of Polymer Concentrations on Characterisation and Ex Vivo Permeation Study. J Pharm Innov 2022, 17, 1176–1188. [Google Scholar] [CrossRef]
- Fatema, K. Microneedle Arrays for Pain-Free Transdermal Drug Delivery: Formulation and Characterisation.
- Lee, J.W.; Park, J.-H.; Prausnitz, M.R. Dissolving Microneedles for Transdermal Drug Delivery. Biomaterials 2008, 29, 2113–2124. [Google Scholar] [CrossRef]
- Waghule, T.; Singhvi, G.; Dubey, S.K.; Pandey, M.M.; Gupta, G.; Singh, M.; Dua, K. Microneedles: A Smart Approach and Increasing Potential for Transdermal Drug Delivery System. Biomedicine & Pharmacotherapy 2019, 109, 1249–1258. [Google Scholar] [CrossRef]
- Permana, A.D.; Paredes, A.J.; Volpe-Zanutto, F.; Anjani, Q.K.; Utomo, E.; Donnelly, R.F. Dissolving Microneedle-Mediated Dermal Delivery of Itraconazole Nanocrystals for Improved Treatment of Cutaneous Candidiasis. European Journal of Pharmaceutics and Biopharmaceutics 2020, 154, 50–61. [Google Scholar] [CrossRef]
- Permana, A.D.; McCrudden, M.T.C.; Donnelly, R.F. Enhanced Intradermal Delivery of Nanosuspensions of Antifilariasis Drugs Using Dissolving Microneedles: A Proof of Concept Study. Pharmaceutics 2019, 11. [Google Scholar] [CrossRef]
- Volpe-Zanutto, F.; Ferreira, L.T.; Permana, A.D.; Kirkby, M.; Paredes, A.J.; Vora, L.K.; P. Bonfanti, A.; Charlie-Silva, I.; Raposo, C.; Figueiredo, M.C.; et al. Artemether and Lumefantrine Dissolving Microneedle Patches with Improved Pharmacokinetic Performance and Antimalarial Efficacy in Mice Infected with Plasmodium Yoelii. Journal of Controlled Release 2021, 333, 298–315. [Google Scholar] [CrossRef]
- Hirun, N.; Kraisit, P.; Santhan, S. Mixed Micellar Gel of Poloxamer Mixture for Improved Solubilization of Poorly Water-Soluble Ibuprofen and Use as Thermosensitive In Situ Gel. Pharmaceutics 2024, 16. [Google Scholar] [CrossRef] [PubMed]
- Almeida, H.; Amaral, M.H.; Lobão, P.; Lobo, J.M.S. Pluronic® F-127 and Pluronic Lecithin Organogel (PLO): Main Features and Their Applications in Topical and Transdermal Administration of Drugs. Journal of Pharmacy & Pharmaceutical Sciences 2012, 15, 592–605. [Google Scholar] [CrossRef] [PubMed]
- Bercea, M.; Constantin, M.; Plugariu, I.-A.; Oana Daraba, M.; Luminita Ichim, D. Thermosensitive Gels of Pullulan and Poloxamer 407 as Potential Injectable Biomaterials. Journal of Molecular Liquids 2022, 362, 119717. [Google Scholar] [CrossRef]
- Dumortier, G.; Grossiord, J.L.; Agnely, F.; Chaumeil, J.C. A Review of Poloxamer 407 Pharmaceutical and Pharmacological Characteristics. Pharm Res 2006, 23, 2709–2728. [Google Scholar] [CrossRef]
- Fakhari, A.; Corcoran, M.; Schwarz, A. Thermogelling Properties of Purified Poloxamer 407. Heliyon 2017, 3, e00390. [Google Scholar] [CrossRef]
- Ur-Rehman, T.; Tavelin, S.; Gröbner, G. Effect of DMSO on Micellization, Gelation and Drug Release Profile of Poloxamer 407. Int J Pharm 2010, 394, 92–98. [Google Scholar] [CrossRef]
- Bouwer, F.; Brits, M.; Viljoen, J.M. Cracking the Skin Barrier: Models and Methods Driving Dermal Drug Delivery. Pharmaceutics 2025, 17. [Google Scholar] [CrossRef]
- Kichou, H.; Bonnier, F.; Dancik, Y.; Bakar, J.; Michael-Jubeli, R.; Caritá, A.C.; Perse, X.; Soucé, M.; Rapetti, L.; Tfayli, A.; et al. Strat-M® Positioning for Skin Permeation Studies: A Comparative Study Including EpiSkin® RHE, and Human Skin. International Journal of Pharmaceutics 2023, 647, 123488. [Google Scholar] [CrossRef]
- Neupane, R.; Boddu, S.H.S.; Renukuntla, J.; Babu, R.J.; Tiwari, A.K. Alternatives to Biological Skin in Permeation Studies: Current Trends and Possibilities. Pharmaceutics 2020, 12, 152. [Google Scholar] [CrossRef]
- Sohn, J.S.; Choi, J.-S. Development of a Tadalafil Transdermal Formulation and Evaluation of Its Ability to in Vitro Transdermal Permeate Using Strat-M® Membrane. European Journal of Pharmaceutical Sciences 2024, 192, 106615. [Google Scholar] [CrossRef]
- Nguyen, H.X.; Nguyen, C.N. Microneedle-Mediated Transdermal Delivery of Biopharmaceuticals. Pharmaceutics 2023, 15. [Google Scholar] [CrossRef]
- Yu Li, Q.; Nan Zhang, J.; Zhi Chen, B.; Lei Wang, Q.; Dong Guo, X. A Solid Polymer Microneedle Patch Pretreatment Enhances the Permeation of Drug Molecules into the Skin. RSC Advances 2017, 7, 15408–15415. [Google Scholar] [CrossRef]
- Stahl, J.; Wohlert, M.; Kietzmann, M. Microneedle Pretreatment Enhances the Percutaneous Permeation of Hydrophilic Compounds with High Melting Points. BMC Pharmacol Toxicol 2012, 13, 5. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Peng, Z.; Song, Y.; Dou, M.; Lu, X.; Li, M.; Zhai, X.; Gu, Y.; Mamujiang, R.; Du, S.; et al. Study of the Permeation-Promoting Effect and Mechanism of Solid Microneedles on Different Properties of Drugs. Drug Deliv 30 2165737. [CrossRef]
- Nguyen, H.X.; Kipping, T.; Banga, A.K. Polymeric Microneedles Enhance Transdermal Delivery of Therapeutics. Pharmaceutics 2024, 16, 845. [Google Scholar] [CrossRef]
- Patel, K.K.; Brogden, N.K. Impact of Formulation and Microneedle Length on Transdermal Metronidazole Permeation through Microneedle-Treated Skin. Pharm Res 2024, 41, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Milewski, M.; Brogden, N.K.; Stinchcomb, A.L. Current Aspects of Formulation Efforts and Pore Lifetime Related to Microneedle Treatment of Skin. Expert Opin Drug Deliv 2010, 7, 617–629. [Google Scholar] [CrossRef] [PubMed]
- Larrañeta, E.; Lutton, R.E.M.; Brady, A.J.; Vicente-Pérez, E.M.; Woolfson, A.D.; Thakur, R.R.S.; Donnelly, R.F. Microwave-Assisted Preparation of Hydrogel-Forming Microneedle Arrays for Transdermal Drug Delivery Applications. Macromolecular Materials and Engineering 2015, 300, 586–595. [Google Scholar] [CrossRef]
- McCrudden, M.T.C.; McAlister, E.; Courtenay, A.J.; González-Vázquez, P.; Raj Singh, T.R.; Donnelly, R.F. Microneedle Applications in Improving Skin Appearance. Experimental Dermatology 2015, 24, 561–566. [Google Scholar] [CrossRef]
- Zhang, C.; Vora, L.K.; Tekko, I.A.; Volpe-Zanutto, F.; Peng, K.; Paredes, A.J.; McCarthy, H.O.; Donnelly, R.F. Development of Dissolving Microneedles for Intradermal Delivery of the Long-Acting Antiretroviral Drug Bictegravir. International Journal of Pharmaceutics 2023, 642, 123108. [Google Scholar] [CrossRef]
- Donnelly, R.F.; Singh, T.R.R.; Garland, M.J.; Migalska, K.; Majithiya, R.; McCrudden, C.M.; Kole, P.L.; Mahmood, T.M.T.; McCarthy, H.O.; Woolfson, A.D. Hydrogel-Forming Microneedle Arrays for Enhanced Transdermal Drug Delivery. Advanced Functional Materials 2012, 22, 4879–4890. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Carrier, A.; Chen, Y.; Lin, S.; Wang, J.; Cui, S.; Zhang, X. Polymeric Microneedles for Controlled Transdermal Drug Delivery. Journal of Controlled Release 2019, 315, 97–113. [Google Scholar] [CrossRef]
- Wei, G.; Xu, H.; Ding, P.T.; Li, S.M.; Zheng, J.M. Thermosetting Gels with Modulated Gelation Temperature for Ophthalmic Use: The Rheological and Gamma Scintigraphic Studies. Journal of Controlled Release 2002, 83, 65–74. [Google Scholar] [CrossRef]
- Bhalerao, H.; Koteshwara, K.B.; Chandran, S. Brinzolamide Dimethyl Sulfoxide In Situ Gelling Ophthalmic Solution: Formulation Optimisation and In Vitro and In Vivo Evaluation. AAPS PharmSciTech 2020, 21, 69. [Google Scholar] [CrossRef]
- Aidoo, E.K.; Sumaila, A.; Jahan, M.; Li, G.; Mensah, P. Thermomechanical Properties of Sustainable Polymer Composites Incorporating Agricultural Wastes. Journal of Manufacturing and Materials Processing 2025, 9. [Google Scholar] [CrossRef]
- Kida, D.; Konopka, T.; Jurczyszyn, K.; Karolewicz, B. Technological Aspects and Evaluation Methods for Polymer Matrices as Dental Drug Carriers. Biomedicines 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- Lobita, M.C.; El-Sayed, N.; Pinto, J.F.; Santos, H.A. Development of Fast Dissolving Polymer-Based Microneedles for Delivery of an Antigenic Melanoma Cell Membrane. Int J Pharm 2023, 642, 123143. [Google Scholar] [CrossRef] [PubMed]
- González-Vázquez, P.; Larrañeta, E.; McCrudden, M.T.C.; Jarrahian, C.; Rein-Weston, A.; Quintanar-Solares, M.; Zehrung, D.; McCarthy, H.; Courtenay, A.J.; Donnelly, R.F. Transdermal Delivery of Gentamicin Using Dissolving Microneedle Arrays for Potential Treatment of Neonatal Sepsis. Journal of Controlled Release 2017, 265, 30–40. [Google Scholar] [CrossRef]
- Singh, A.; Bali, A. Formulation and Characterization of Transdermal Patches for Controlled Delivery of Duloxetine Hydrochloride. J Anal Sci Technol 2016, 7, 25. [Google Scholar] [CrossRef]
- Larrañeta, E.; Stewart, S.; Fallows, S.J.; Birkhäuer, L.L.; McCrudden, M.T.C.; Woolfson, A.D.; Donnelly, R.F. A Facile System to Evaluate in Vitro Drug Release from Dissolving Microneedle Arrays. International Journal of Pharmaceutics 2016, 497, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Quality of Transdermal Patches - Scientific Guideline | European Medicines Agency (EMA). Available online: https://www.ema.europa.eu/en/quality-transdermal-patches-scientific-guideline (accessed on 11 March 2026).
- Arce, F.J.; Asano, N.; See, G.L.; Itakura, S.; Todo, H.; Sugibayashi, K. Usefulness of Artificial Membrane, Strat-M®, in the Assessment of Drug Permeation from Complex Vehicles in Finite Dose Conditions. Pharmaceutics 2020, 12. [Google Scholar] [CrossRef] [PubMed]
- Kaur, L.; Singh, K.; Paul, S.; Singh, S.; Singh, S.; Jain, S.K. A Mechanistic Study to Determine the Structural Similarities Between Artificial Membrane Strat-MTM and Biological Membranes and Its Application to Carry Out Skin Permeation Study of Amphotericin B Nanoformulations. AAPS PharmSciTech 2018, 19, 1606–1624. [Google Scholar] [CrossRef]
- Liu, X.; Shen, B.; Shen, C.; Zhong, R.; Wang, X.; Yuan, H. Nanoparticle-Loaded Gels for Topical Delivery of Nitrofurazone: Effect of Particle Size on Skin Permeation and Retention. Journal of Drug Delivery Science and Technology 2018, 45, 367–372. [Google Scholar] [CrossRef]
- Zheng, J.; Shen, C.-Y.; Pang, J.-Y.; Xu, F.-C.; Liao, W.-B.; Hu, C.-X.; Xu, P.-H.; Han, J.; Yuan, H.-L. Preparation of tanshinone ⅡA loaded nanostructured lipid carrier and its in vitro transdermal permeation characteristics. Zhongguo Zhong Yao Za Zhi 2016, 41, 3232–3238. [Google Scholar] [CrossRef]
- Altameemi, K.K.A.; Abd-Alhammid, S.N. Anastrozole Nanoparticles for Transdermal Delivery through Microneedles: Preparation and Evaluation. Journal of Pharmaceutical Negative Results 2022, 13, 974–980. [Google Scholar] [CrossRef]
- Hutton, A.R.J.; Quinn, H.L.; McCague, P.J.; Jarrahian, C.; Rein-Weston, A.; Coffey, P.S.; Gerth-Guyette, E.; Zehrung, D.; Larrañeta, E.; Donnelly, R.F. Transdermal Delivery of Vitamin K Using Dissolving Microneedles for the Prevention of Vitamin K Deficiency Bleeding. International Journal of Pharmaceutics 2018, 541, 56–63. [Google Scholar] [CrossRef] [PubMed]

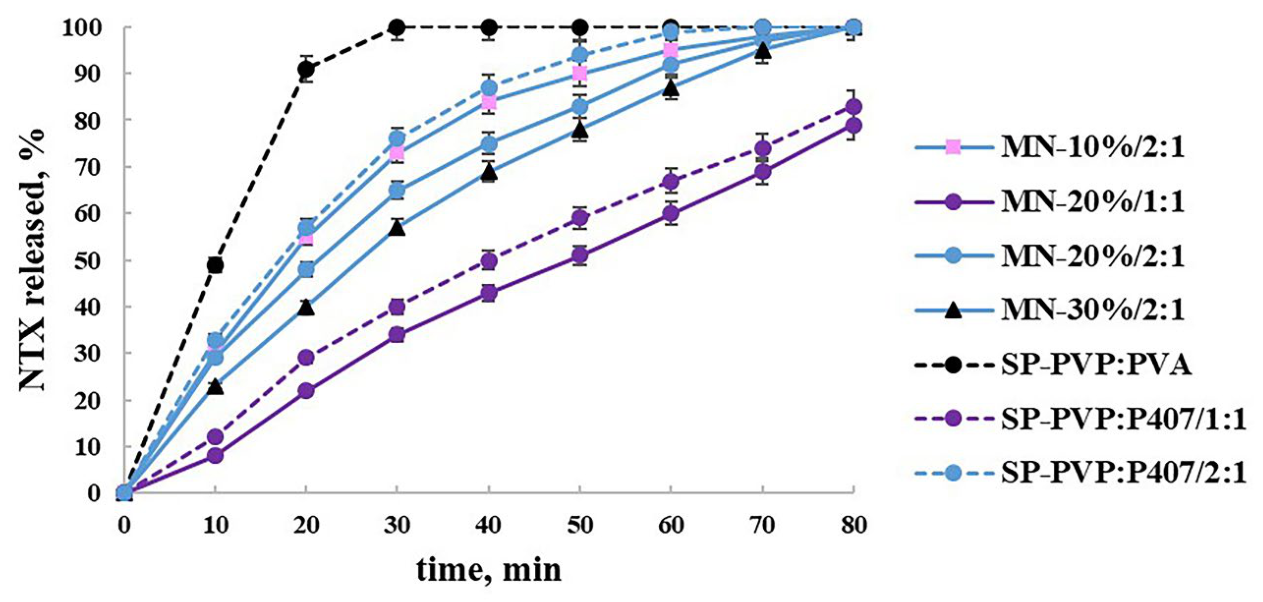
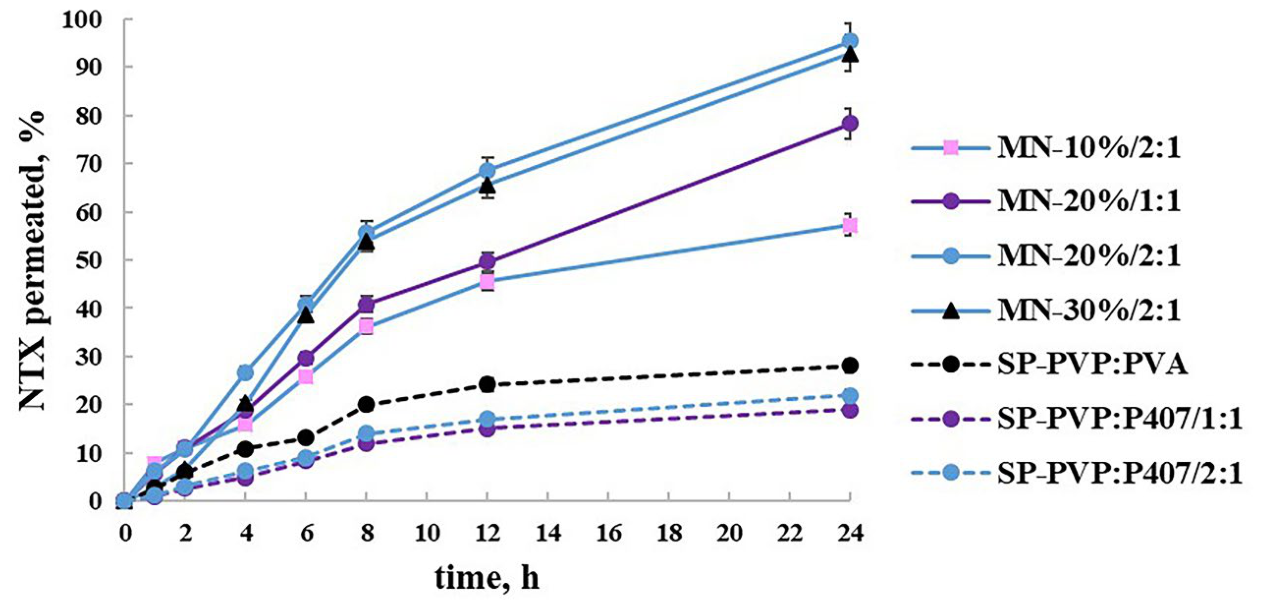
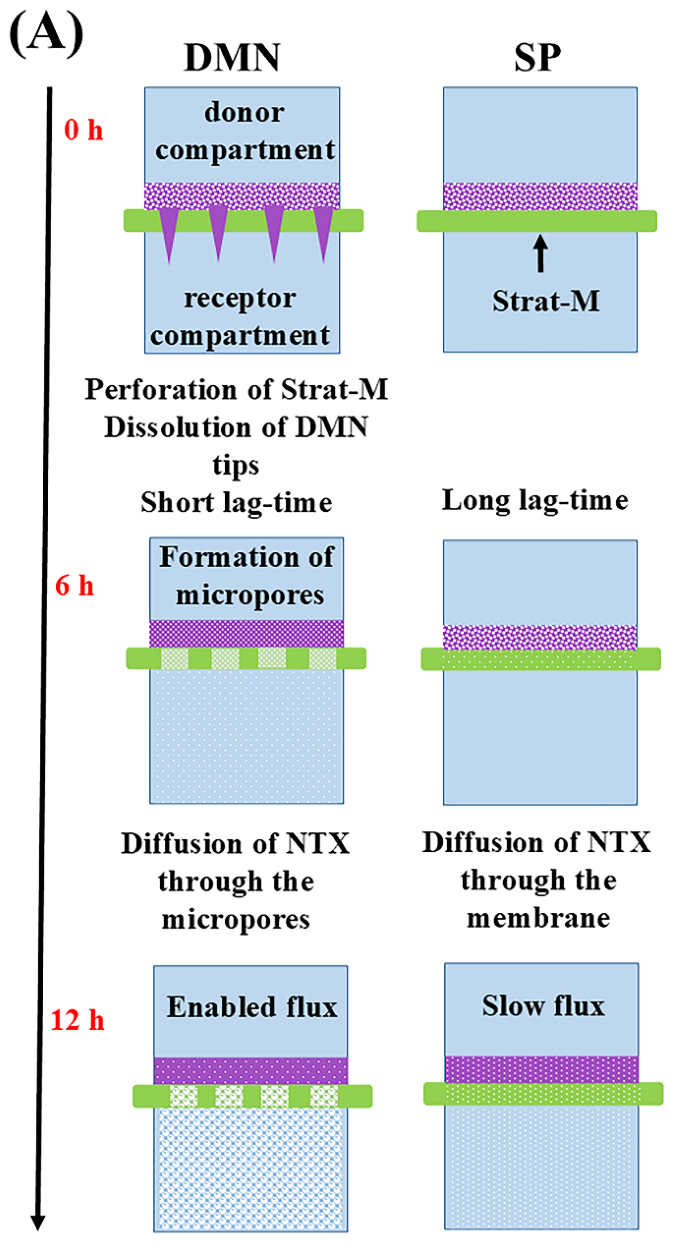
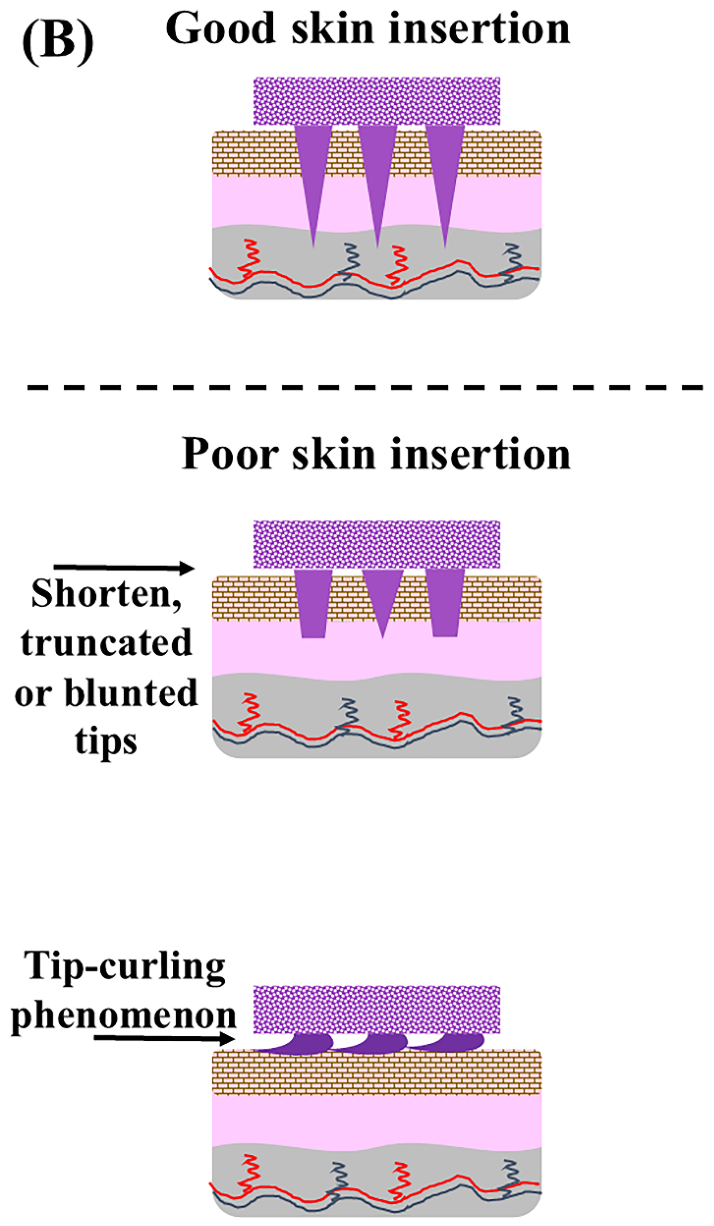
| DMN patches | SP patches | |||||
| MN-10%/2:1 | MN-20%/1:1 | MN-20%/2:1 | MN-30%/2:1 | SP-PVP:PVA | SP-PVP:P407/1:1 | SP-PVP:P407/2:1 |
| DMN tips | 30% PVP:PVA=2:1 | 30% PVP:P407=1:1 | 30% PVP:P407=2:1 |
|||
| PVP:PVA=2:1 | ||||||
| 10% | 20% | 30% | 30% | |||
| DMN backing layer | 0.1% NTX | |||||
| 0.1% NTX | ||||||
| 30% PVP:P407 | ||||||
| 2:1 | 1:1 | 2:1 | 2:1 | |||
| Formulation code | API assay, % |
| MN-10%/2:1 | 96.81 ± 3.52 |
| MN-20%/1:1 | 98.17 ± 2.97 |
| MN-20%/2:1 | 97.79 ± 4.11 |
| MN-30%/2:1 | 97.53 ± 3.91 |
| SP-PVP:PVA | 98.12 ± 2.61 |
| SP-PVP:P407/1:1 | 97.83 ± 3.24 |
| SP-PVP:P407/2:1 | 96.46 ± 2.11 |
| SP-PVP:P407/2:1 | SP-PVP:P407/1:1 | SP-PVP:P407/2:1 – API free | SP-PVP:P407/2:1 – API free |
| gelation temperature, °C | |||
| 34.21 ± 0.21 | 31.64 ± 0.32 | 33.90 ± 0.23 | 30.82 ± 0.51 |
| gelling time, s | |||
| 17.53 ± 0.58 | 9.17 ± 0.15 | 15.97 ± 0.17 | 8.32 ± 0.23 |
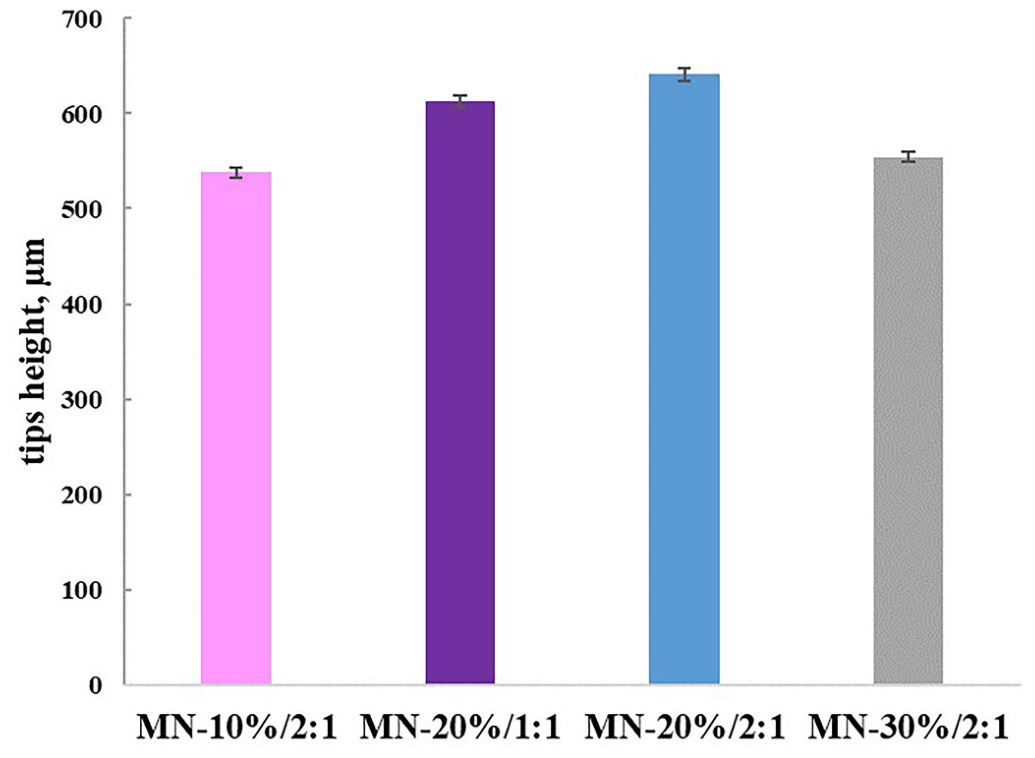
| Parameter | MN-10%/2:1 | MN-20%/1:1 | MN-20%/2:1 | MN-30%/2:1 |
| H1, µm | 538.18 ± 41.31 | 613.03 ± 57.19 | 641.15 ± 18.32 | 554.08 ± 33.12 |
| H2 , µm | 267.23 ± 34.72 | 505.06 ± 44.98 | 554.25 ± 17.36 | 469.31 ± 28.12 |
| height reduction, % | 50.37 ± 3.47 | 17.61 ± 4.71 | 13.57 ± 1.43 | 15.34 ± 3.15 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




