Submitted:
20 April 2026
Posted:
21 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.1.1. Strain
2.1.2. Main Reagents
2.1.3. Reference Genome
2.2. Methods
2.2.1. Plate Culture Assay
2.2.2. Transcriptome Sequencing
2.2.3. Gene Expression Level Analysis
2.2.4. Differential Gene Expression and Enrichment Analyses
2.2.5. Quantitative Real-Time PCR (qRT-PCR) Validation
3. Results
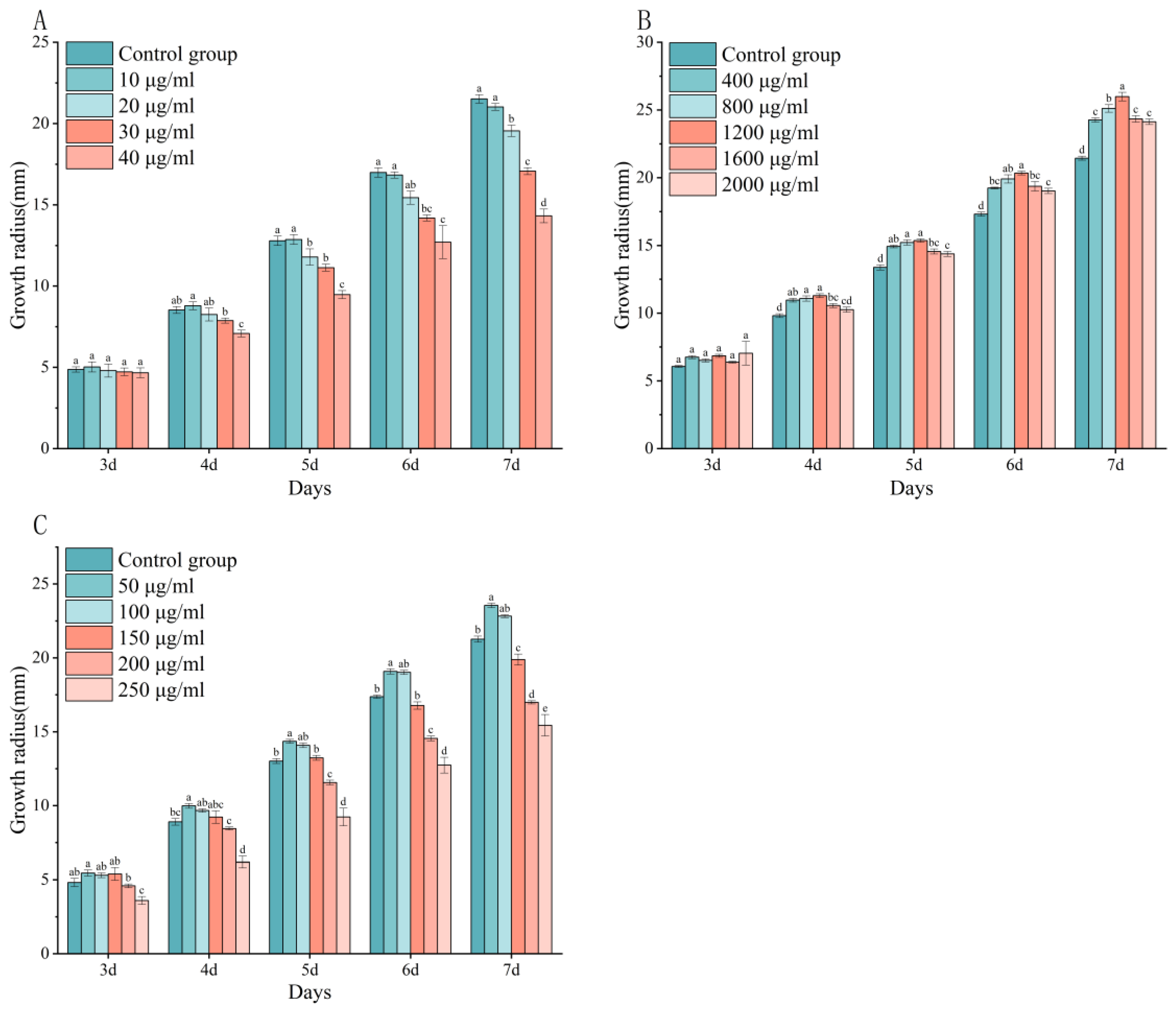
3.1. Plate Culture Assay
3.2. Transcriptome Analysis
3.2.1. Raw Data and Sequencing Quality Assessment
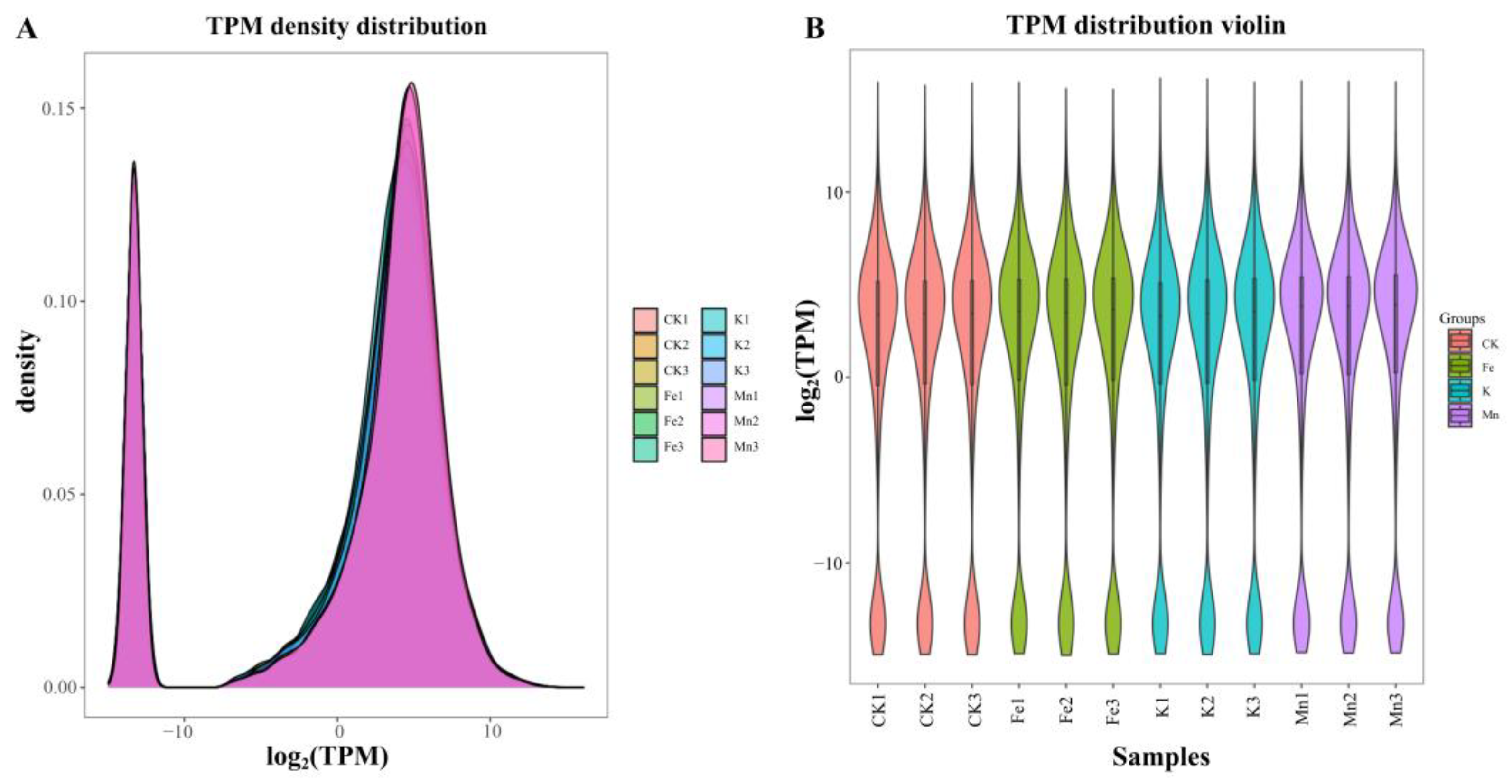
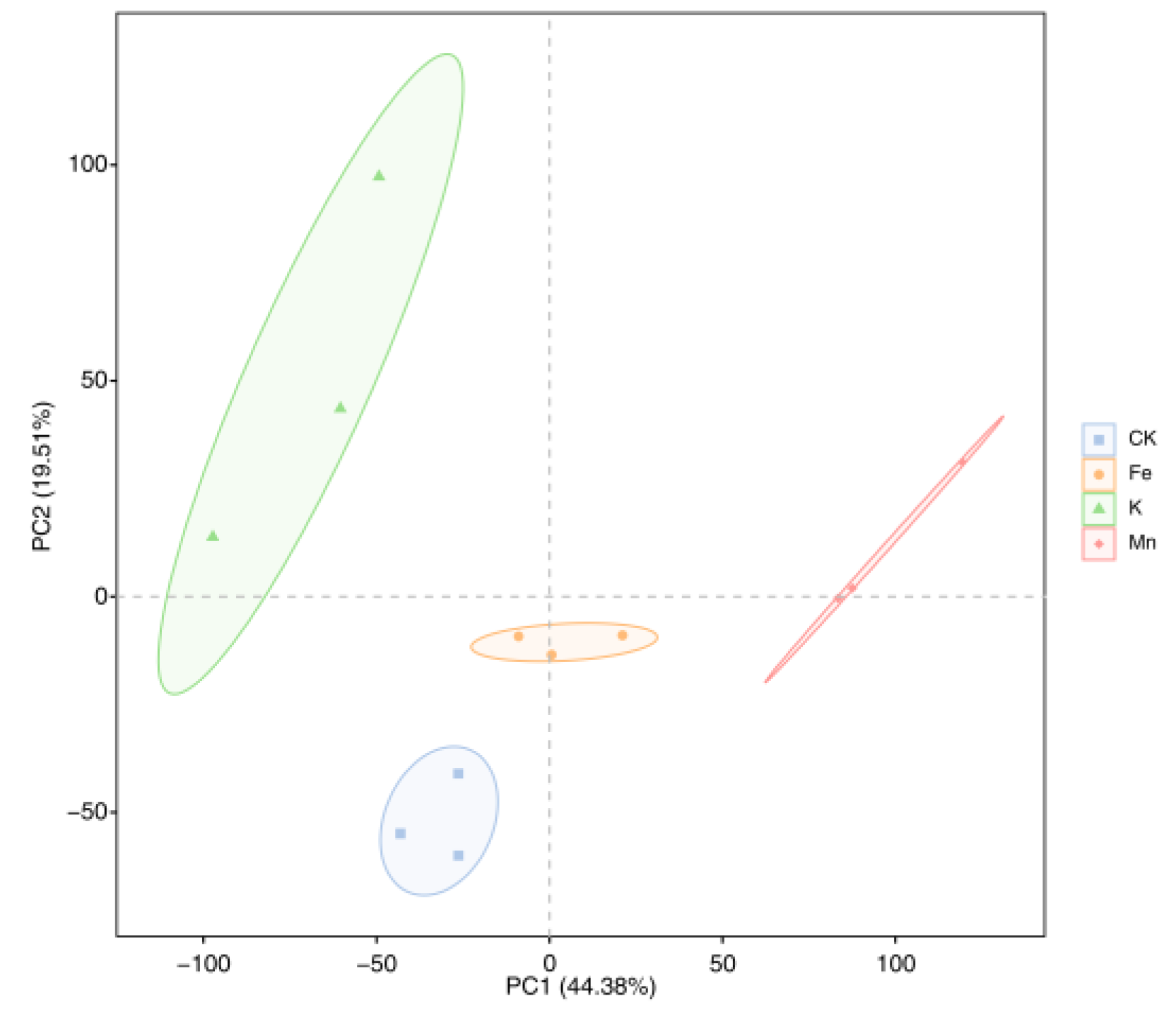
3.2.2. Expression Level Analysis
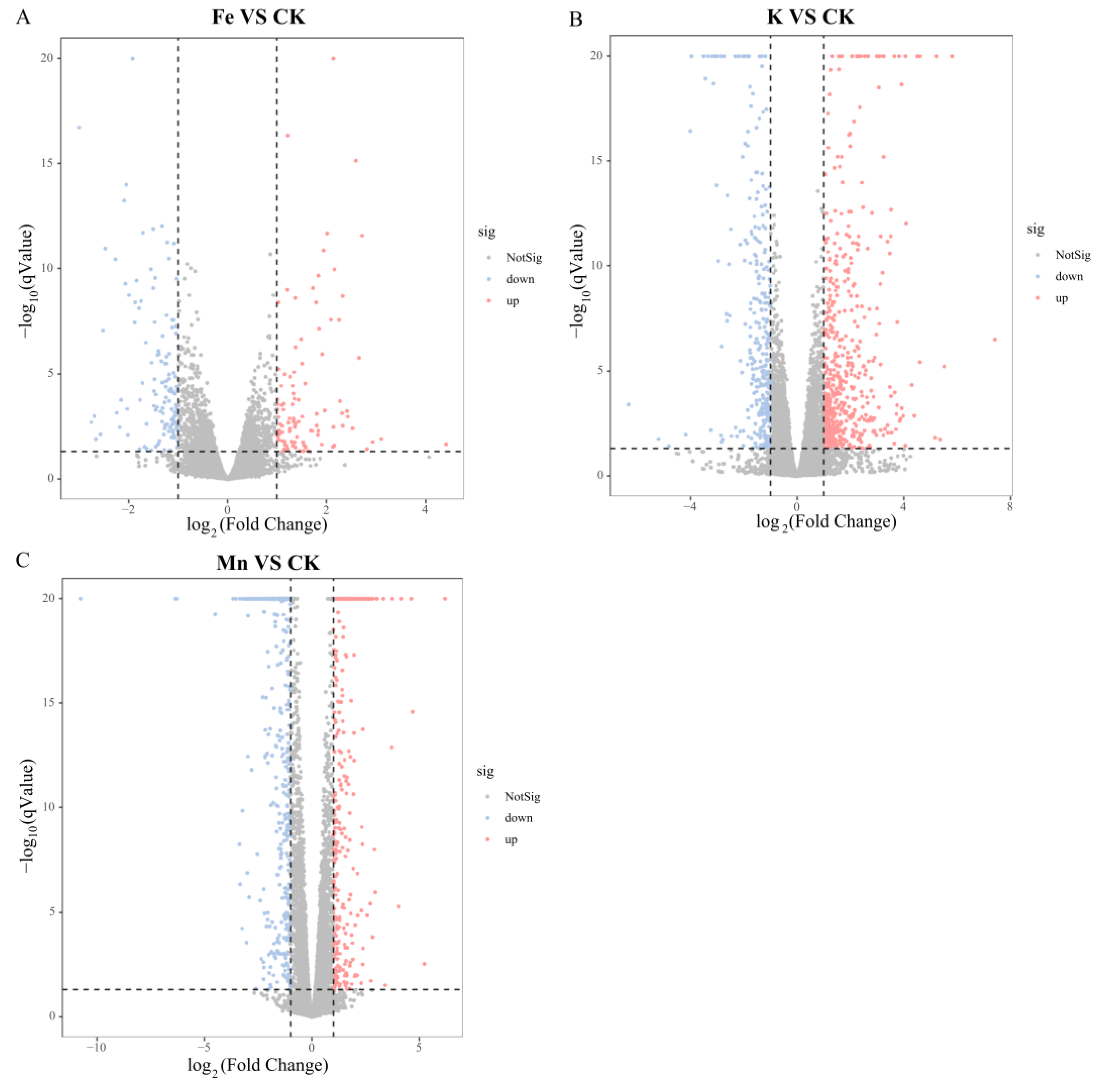
3.2.3. Differential Gene Expression Analysis
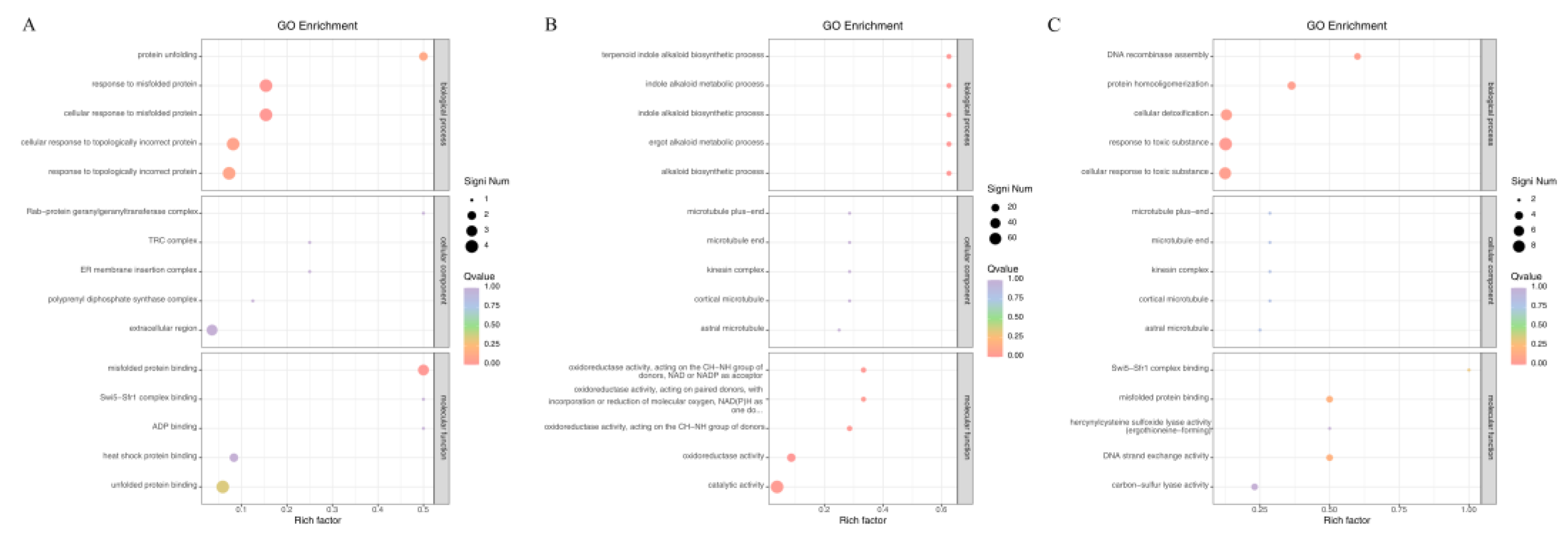
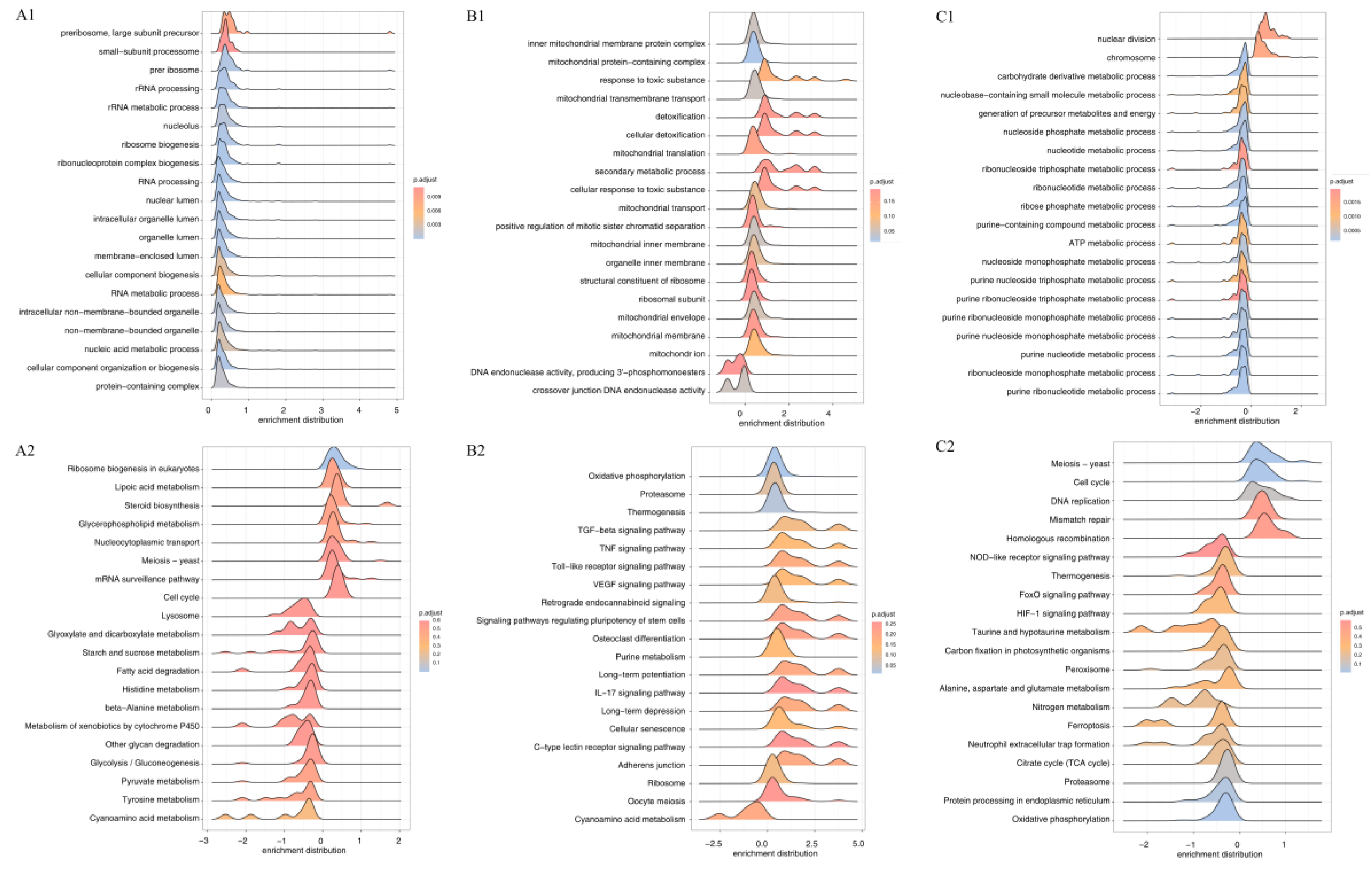
3.2.4. GO Functional Enrichment Analysis
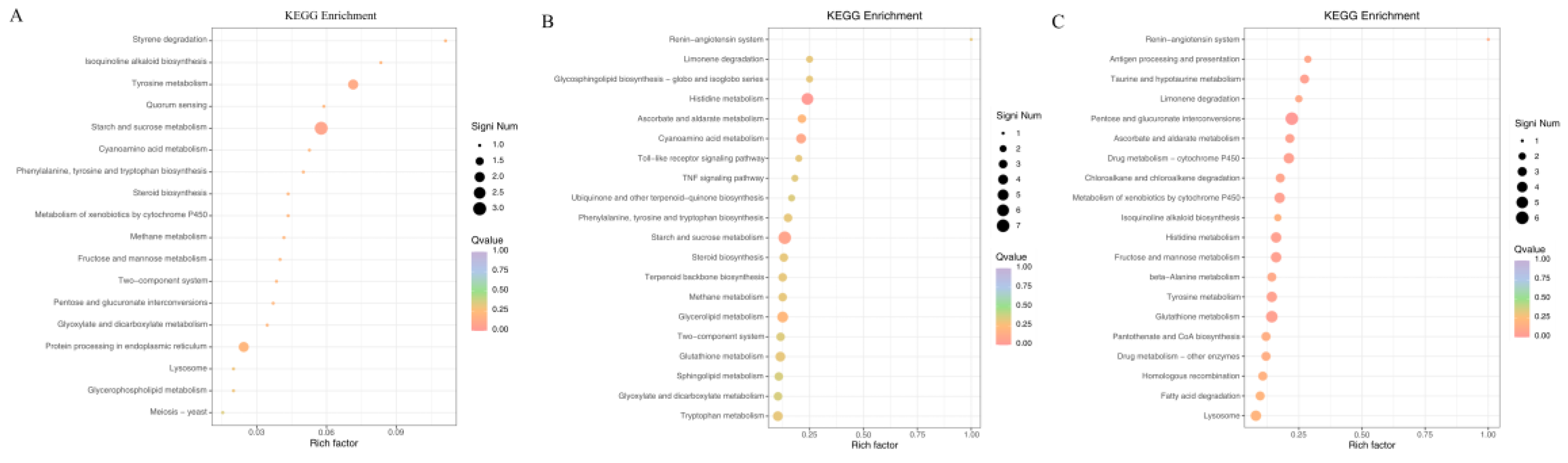
3.2.5. KEGG Pathway Enrichment Analysis
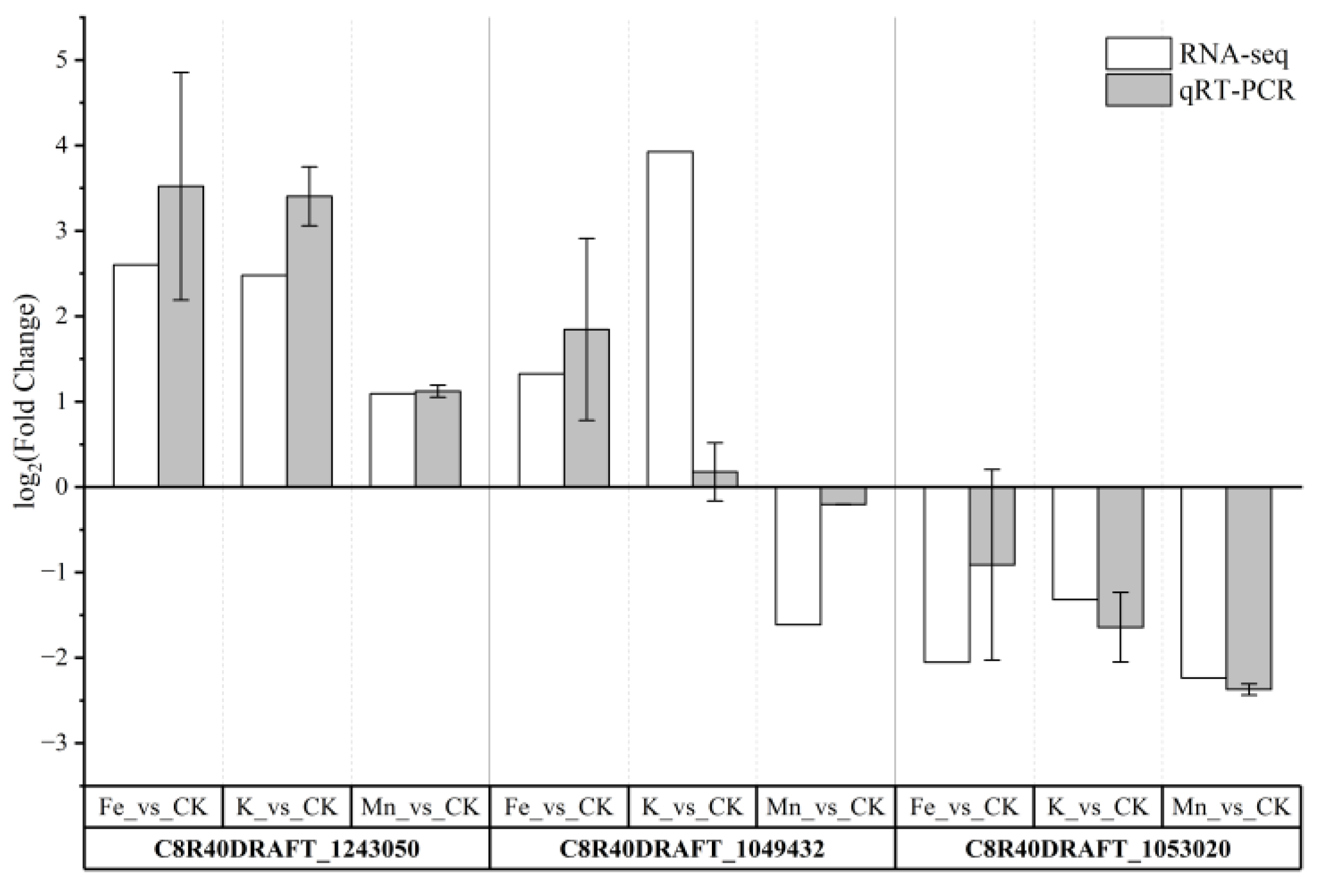
3.3. Quantitative Real-Time PCR Validation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CYP | cytochrome P450 |
| DEG | differentially expressed gene |
| GCLC | glutamate-cysteine ligase catalytic subunit |
| GCL | glutamate-cysteine ligase |
| GSH | glutathione |
| GSEA | gene set enrichment analysis |
| GO | gene ontology |
| HSP | heat shock protein |
| KEGG | Kyoto Encyclopedia of Genes and Genomes |
| MF | molecular function |
| CC | cellular component |
| BP | biological process |
| PCA | principal component analysis |
| qRT-PCR | quantitative real-time polymerase chain reaction |
| RNA-seq | RNA sequencing |
| TPM | transcripts per million |
References
- Shi, D.; Yin, C.; Feng, X.; Zhou, R.; Fan, X.; Qiao, Y.; Yao, F.; Huang, W.; Liu, Y.; Gao, H.; Cheng, W. Effect of Ultrasound and Cellulase Pre-treatment on the Water Distribution, Physical Properties, and Nutritional Components of Lentinula edodes Chips. Food and Bioprocess Technology 2020, 13, 625–636. [Google Scholar] [CrossRef]
- Adil, B.; Xiang, Q. J.; He, M. L.; Wu, Y. T.; Asghar, M. A.; Arshad, M.; Qin, P.; Gu, Y. F.; Yu, X. M.; Zhao, K.; Zhang, X. P.; Ma, M. G.; Chen, Q.; Chen, X. Q.; Yan, Y. H. Effect of sodium and calcium on polysaccharide production and the activities of enzymes involved in the polysaccharide synthesis of Lentinus edodes. Amb Express 2020, 10, 47. [Google Scholar] [CrossRef]
- Galgowska, M.; Pietrzak-Fiecko, R. Mineral Composition of Three Popular Wild Mushrooms from Poland. Molecules 2020, 25, 3588. [Google Scholar] [CrossRef]
- Gao, J.; Li, X.; Jia, S.; Zeng, H.; Zheng, B. Structural characterization and antioxidant activity of a glycoprotein isolated from shiitake mushrooms. Food Bioscience 2023, 53, 102608. [Google Scholar] [CrossRef]
- Zhang, J.; Rida, K.; Wen, J.; Yu, X.; Gu, Y.; He, M.; Chen, Q.; Xiang, Q. Screening of Lentinula edodes Strains for High Polysaccharide Production and In Vitro Antioxidant Activities. J Fungi (Basel) 2025, 11, 347. [Google Scholar] [CrossRef]
- Umeo, S. H.; Faria, M. G. I.; Dragunski, D. C.; Valle, J. S. D.; Colauto, N. B.; Linde, G. A. Iron Or Zinc Bioaccumulated In Mycelial Biomass Of Edible Basidiomycetes. An Acad Bras Cienc 2020, 92, e20191350. [Google Scholar] [CrossRef]
- Scheid, S. S.; Faria, M. G. I.; Velasquez, L. G.; do Valle, J. S.; Goncalves, A. C., Jr.; Dragunski, D. C.; Colauto, N. B.; Linde, G. A. Iron biofortification and availability in the mycelial biomass of edible and medicinal basidiomycetes cultivated in sugarcane molasses. Sci Rep 2020, 10, 12875. [Google Scholar] [CrossRef]
- Meniqueti, A. B.; Ruiz, S. P.; Faria, M. G. I.; do Valle, J. S.; Gonçalves, A. C., Jr.; Dragunski, D. C.; Colauto, N. B.; Linde, G. A. Iron-enriched mycelia of edible and medicinal basidiomycetes. Environ. Technol. 2022, 43, 1248–1254. [Google Scholar] [CrossRef]
- Ogidi, C. O.; Oyebode, K. O. Assessment of nutrient contents and bio-functional activities of edible fungus bio-fortified with copper, lithium and zinc. World J Microbiol Biotechnol 2022, 39, 56. [Google Scholar] [CrossRef] [PubMed]
- Oyetayo, V. O.; Ogidi, C. O.; Bayode, S. O.; Enikanselu, F. F. Evaluation of biological efficiency, nutrient contents and antioxidant activity of Pleurotus pulmonarius enriched with Zinc and Iron. Indian Phytopathology 2021, 74, 901–910. [Google Scholar] [CrossRef]
- Naeem, A.; Aslam, M.; Saifullah; Mühling, K. H. Lithium: Perspectives of nutritional beneficence, dietary intake, biogeochemistry, and biofortification of vegetables and mushrooms. Sci. Total Environ. 2021, 798, 149249. [Google Scholar] [CrossRef]
- Li, H.; Dai, J.; Shi, Y.; Zhu, X.; Jia, L.; Yang, Z. Molecular Regulatory Mechanism of the Iron-Ion-Promoted Asexual Sporulation of Antrodia cinnamomea in Submerged Fermentation Revealed by Comparative Transcriptomics. J Fungi (Basel) 2023, 9, 235. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Wang, W.; Wang, Y.; Yun, J.; Zhang, Y.; Zhao, F. Exogenous MnSO(4) Improves Productivity of Degenerated Volvariella volvacea by Regulating Antioxidant Activity. J Fungi (Basel) 2024, 10, 825. [Google Scholar] [CrossRef]
- Zhang, B.; Zhou, J.; Li, Q.; Gan, B. C.; Peng, W. H.; Zhang, X. P.; Tan, W.; Jiang, L.; Li, X. L. Manganese affects the growth and metabolism of Ganoderma lucidum based on LC-MS analysis. PeerJ 2019, 7, e6846. [Google Scholar] [CrossRef]
- Rodriguez Estrada, A. E.; Royse, D. J. Yield, size and bacterial blotch resistance of Pleurotus eryngii grown on cottonseed hulls/oak sawdust supplemented with manganese, copper and whole ground soybean. Bioresour Technol 2007, 98, 1898–1906. [Google Scholar] [CrossRef]
- Zieba, P.; Sekara, A.; Bernas, E.; Krakowska, A.; Sulkowska-Ziaja, K.; Kunicki, E.; Suchanek, M.; Muszynska, B. Supplementation with Magnesium Salts-A Strategy to Increase Nutraceutical Value of Pleurotus djamor Fruiting Bodies. Molecules 2021, 26, 3273. [Google Scholar] [CrossRef]
- Wlodarczyk, A.; Krakowska, A.; Sulkowska-Ziaja, K.; Suchanek, M.; Zieba, P.; Opoka, W.; Muszynska, B. Pleurotus spp. Mycelia Enriched in Magnesium and Zinc Salts as a Potential Functional Food. Molecules 2020, 26, 162. [Google Scholar] [CrossRef]
- Li, X.; Luo, L.; Wang, X.; Zhu, M. Further insights into the molecular mechanisms underlying tobacco straw cultivation of Pleurotus ostreatus by comparative transcriptome analyses. Genomics 2025, 117, 110992. [Google Scholar] [CrossRef]
- Xie, M.; Wang, F.; Zhou, X.; Zeng, J.; Zhou, Y.; Wei, T.; Chen, B.-X. Multi-omics approaches for advancing edible fungi: from genomics-enabled strain development to metabolomics-guided postharvest preservation. Food Bioscience 2025, 71, 107161. [Google Scholar] [CrossRef]
- Wang, J.; Cai, W.; Jin, Q.; Fan, L.; Guo, Z.; Feng, W. Improving the Yield and Quality of Morchella spp. Using Agricultural Waste. J Fungi (Basel) 2025, 11, 703. [Google Scholar] [CrossRef] [PubMed]
- Lu, F.; Sun, X.; Dai, X.; Zhang, P.; Ma, Y.; Xu, Y.; Wang, L.; Zhang, J. Integrated Multi-Omics Analysis to Investigate the Molecular Mechanisms Underlying the Response of Auricularia heimuer to High-Temperature Stress. J Fungi (Basel) 2025, 11, 167. [Google Scholar] [CrossRef]
- Kim, J. Y.; Kim, D. Y.; Park, Y. J.; Jang, M. J. Transcriptome analysis of the edible mushroom Lentinula edodes in response to blue light. PLoS One 2020, 15, e0230680. [Google Scholar] [CrossRef]
- Deshaware, S.; Marathe, S. J.; Bedade, D.; Deska, J.; Shamekh, S. Investigation on mycelial growth requirements of Cantharellus cibarius under laboratory conditions. Arch Microbiol 2021, 203, 1539–1545. [Google Scholar] [CrossRef]
- Shao, K.; Yao, F.; Fang, M.; Lu, L.; Ma, X.; Wang, W.; Meng, J.; Sun, X.; Cui, Y.; Sun, J. Transcriptome-Based Analysis of Mitochondrial Influence on Key Agronomic Traits and Nutritional Components in Auricularia heimuer. Agronomy 2025, 15, 2188. [Google Scholar] [CrossRef]
- Chen, S. F.; Zhou, Y. Q.; Chen, Y. R.; Gu, J. fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 2018, 34, 884–890. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Paggi, J. M.; Park, C.; Bennett, C.; Salzberg, S. L. Graph-based genome alignment and genotyping with HISAT2 and HISAT-genotype. Nat Biotechnol 2019, 37, 907–915. [Google Scholar] [CrossRef]
- Wang, L.; Wang, S.; Li, W. RSeQC: quality control of RNA-seq experiments. Bioinformatics 2012, 28, 2184–2185. [Google Scholar] [CrossRef]
- Pertea, M.; Pertea, G. M.; Antonescu, C. M.; Chang, T. C.; Mendell, J. T.; Salzberg, S. L. StringTie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat Biotechnol 2015, 33, 290–295. [Google Scholar] [CrossRef]
- Liao, Y.; Smyth, G. K.; Shi, W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 2014, 30, 923–930. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S. R.; Ye, Z.; Stanton, R. Misuse of RPKM or TPM normalization when comparing across samples and sequencing protocols. Rna 2020, 26, 903–909. [Google Scholar] [CrossRef] [PubMed]
- Love, M. I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 38. [Google Scholar] [CrossRef] [PubMed]
- Lang, B. J.; Guerrero, M. E.; Prince, T. L.; Okusha, Y.; Bonorino, C.; Calderwood, S. K. The functions and regulation of heat shock proteins; key orchestrators of proteostasis and the heat shock response. Arch Toxicol 2021, 95, 1943–1970. [Google Scholar] [CrossRef]
- Liang, Z.-Q.; Zhao, Y.; Tan, Y.-Z.; Wang, J.; Shao, Y.-L.; Zheng, Q.-W.; Zou, Y.; Lin, J.-F.; Chen, M.-T.; Ye, Z.-W. Functional characterization of the heat shock protein gene hsp78 in regulating carotenoid biosynthesis in the edible-medicinal fungus Cordyceps militaris. Food Bioscience 2025, 74, 107894. [Google Scholar] [CrossRef]
- Chen, C.; Li, Q.; Wang, Q.; Lu, D.; Zhang, H.; Wang, J.; Fu, R. Transcriptional profiling provides new insights into the role of nitric oxide in enhancing Ganoderma oregonense resistance to heat stress. Sci Rep 2017, 7, 15694. [Google Scholar] [CrossRef]
- Kumari, A.; Sutariya, J. A.; Rathore, A. P.; Rathore, M. S. The novel chaperonin 10 like protein (SbCPN10L) from Salicornia brachiata (Roxb.) augment the heat stress tolerance in transgenic tobacco. Gene 2024, 900, 148139. [Google Scholar] [CrossRef]
- Kumar, S.; Masison, D. C. Hsp70-nucleotide exchange factor (NEF) Fes1 has non-NEF roles in degradation of gluconeogenic enzymes and cell wall integrity. PLoS Genet 2019, 15, e1008219. [Google Scholar] [CrossRef]
- Zhao, W.; Dong, H.; Zhou, Z.; Wang, Y.; Hu, W. Potassium (K) application alleviates the negative effect of drought on cotton fiber strength by sustaining higher sucrose content and carbohydrates conversion rate. Plant Physiol Biochem 2020, 157, 105–113. [Google Scholar] [CrossRef]
- Gao, Z.; Chen, F.; Zheng, J.; Peng, Q.; Chen, D.; Zhang, Y.; Zhou, L.; Liu, K.; Yang, Y.; Yuan, Q. Potassium-Selective Covalent Organic Framework Membranes Enable Dynamic Monitoring of Microbial K(+) Metabolism. Small 2025, 21, e2502541. [Google Scholar] [CrossRef]
- Luo, C.; Song, Y.; Meng, L.; Cheng, Y.; Dai, H.; Qiao, Y.; Xie, X. Transcriptomic insights into the molecular mechanism of abietic acid promoting growth and branching in Armillaria gallica. Front Microbiol 2025, 16, 1632512. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Vinocur, B.; Shoseyov, O.; Altman, A. Role of plant heat-shock proteins and molecular chaperones in the abiotic stress response. Trends Plant Sci 2004, 9, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Ling, Y. Y.; Ling, Z. L.; Zhao, R. L. Construction of a heat-resistant strain of Lentinus edodes by fungal Hsp20 protein overexpression and genetic transformation. Front Microbiol 2022, 13, 1009885. [Google Scholar] [CrossRef]
- Wenhong, L.; Yang, J.; Zhao, Y.; Zhang, N.; Zhao, B.; Rongxian, L.; Shiyan, G.; Zuoshun, H. Cadmium treatment induces oxidative damage and apoptosis in vitro skeletal muscle cells. Toxicology 2025, 515, 154139. [Google Scholar] [CrossRef] [PubMed]
- Deveryshetty, J.; Mistry, A.; Pangeni, S.; Ghoneim, M.; Tokmina-Lukaszewska, M.; Gore, S. K.; Liu, J.; Kaushik, V.; Karunakaran, S.; Taddei, A.; Heyer, W. D.; Ha, T.; Bothner, B.; Antony, E. Mechanism of Rad51 filament formation by Rad52 and Rad55-Rad57 in homologous recombination. Nat Commun 2025, 16, 6685. [Google Scholar] [CrossRef]
- Wang, J.; Sun, Z.; Liu, H.; Yue, L.; Wang, F.; Liu, S.; Su, B.; Liu, B.; Kong, F.; Fang, C. Genome-Wide Identification and Characterization of the Soybean Snf2 Gene Family and Expression Response to Rhizobia. Int J Mol Sci 2023, 24, 7250. [Google Scholar] [CrossRef] [PubMed]
- Villemain, J.; Rokosky, C. Abstract 1937 Intrinsic disorder in the C-terminal Domain of the Yeast Srs2 helicase Provides Structural Diversity Essential to Implementing Multi-functional Roles in replication, Recombination and Repair. Journal of Biological Chemistry 2024, 300, 107008. [Google Scholar] [CrossRef]
- Adam, A. M. A.; Refat, M. S.; Mohamed, M. A. Synthesis and spectroscopic characterizations of noble metal complexes (gold, silver, platinum) in the presence of selenium, and their biological applications as antibacterial, antifungal, and anticancer. Research on Chemical Intermediates 2013, 41, 965–1000. [Google Scholar] [CrossRef]
- Averill-Bates, D. A. The antioxidant glutathione. In Antioxidants; Litwack, G., Ed.; Elsevier Academic Press Inc: San Diego, 2023; Volume. 121, pp. pp 109–141. [Google Scholar] [CrossRef]
- Harish, B. S.; Uppuluri, K. B. Microbial serine protease inhibitors and their therapeutic applications. Int J Biol Macromol 2018, 107, 1373–1387. [Google Scholar] [CrossRef]
- Shin, J.; Kim, J. E.; Lee, Y. W.; Son, H. Fungal Cytochrome P450s and the P450 Complement (CYPome) of Fusarium graminearum. Toxins (Basel) 2018, 10, 112. [Google Scholar] [CrossRef] [PubMed]
| Gene | Putative function | Primer sequence (5′→3′) | |
|---|---|---|---|
| Forward | Reverse | ||
| C8R40DRAFT_1124140 | β-tubulin 2 | GTTCGCGGTCCCTTAGCTT | GTAATCACCCACATCCTTTTGC |
| C8R40DRAFT_1243050 | pyridoxal phosphate-dependent transferase | CCCATTGACCACTGCCATC | CCAGCCCACATCGACTCC |
| C8R40DRAFT_1049432 | fungal peroxidase | GCTACGCTGTCGCAAGTCC | CCGTCCATGAATCCGAAATC |
| C8R40DRAFT_1053020 | uracil phosphoribosyltransferase-domain-containing protein | CTCTTGTGCTCGAGACAGGCT | TCAGTGGCATCTTTGACCGTT |
| Sample No. | Raw Reads Count | Raw Bases Count | Clean Reads Count | Clean Bases Count | Q30 Bases Ratio(%) | GC content (%) |
|---|---|---|---|---|---|---|
| CK1 | 99,954,944 | 14,993,241,600 | 94,629,298 | 13,353,955,541 | 98.91% | 48.79% |
| CK2 | 122,742,912 | 18,411,436,800 | 88,535,680 | 12,526,057,802 | 98.71% | 48.69% |
| CK3 | 100,000,000 | 15,000,000,000 | 116,115,398 | 16,309,407,246 | 98.87% | 48.48% |
| Fe1 | 100,000,000 | 15,000,000,000 | 93,037,640 | 13,060,762,548 | 98.81% | 48.84% |
| Fe2 | 100,000,000 | 15,000,000,000 | 93,798,908 | 13,141,156,027 | 98.83% | 48.96% |
| Fe3 | 100,000,000 | 15,000,000,000 | 94,475,116 | 13,426,499,466 | 98.79% | 48.85% |
| K1 | 100,000,000 | 15,000,000,000 | 93,847,592 | 13,266,192,738 | 98.86% | 48.78% |
| K2 | 100,000,000 | 15,000,000,000 | 94,226,408 | 13,230,205,439 | 98.86% | 48.94% |
| K3 | 100,000,000 | 15,000,000,000 | 94,796,364 | 13,405,157,343 | 98.87% | 48.91% |
| Mn1 | 94,438,218 | 14,165,732,700 | 94,218,924 | 13,258,424,776 | 98.88% | 48.74% |
| Mn2 | 100,000,000 | 15,000,000,000 | 95,295,406 | 13,439,817,749 | 98.77% | 48.67% |
| Mn3 | 101,680,028 | 15,252,004,200 | 94,191,118 | 13,167,217,379 | 98.82% | 48.90% |
| Treatment | Number of DEGs | Up-regulated genes | Down-regulated genes |
|---|---|---|---|
| Fe2+ | 226 | 101 | 125 |
| K+ | 858 | 536 | 322 |
| Mn2+ | 696 | 289 | 407 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




