Submitted:
19 April 2026
Posted:
21 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Current Landscape and Methods of Genetic Engineering Vero Cells
2.1. Engineering the Membrane Interface: From Viral Entry to Suspension Adaptation
2.2. The Cytoplasmic Foundry: Capacity, Stress Handling, and Survival Trade-Offs
2.3. The Nuclear Blueprint: Genomic Stability and Precision Engineering
3. Toward a Fully Programmable and Scalable Vero Cell Platform
3.1. Engineering Robustness Under Multidimensional Stress and Regulatory Constraints
3.2. From Static Optimization to Dynamic Control: Engineering Conditional Logic
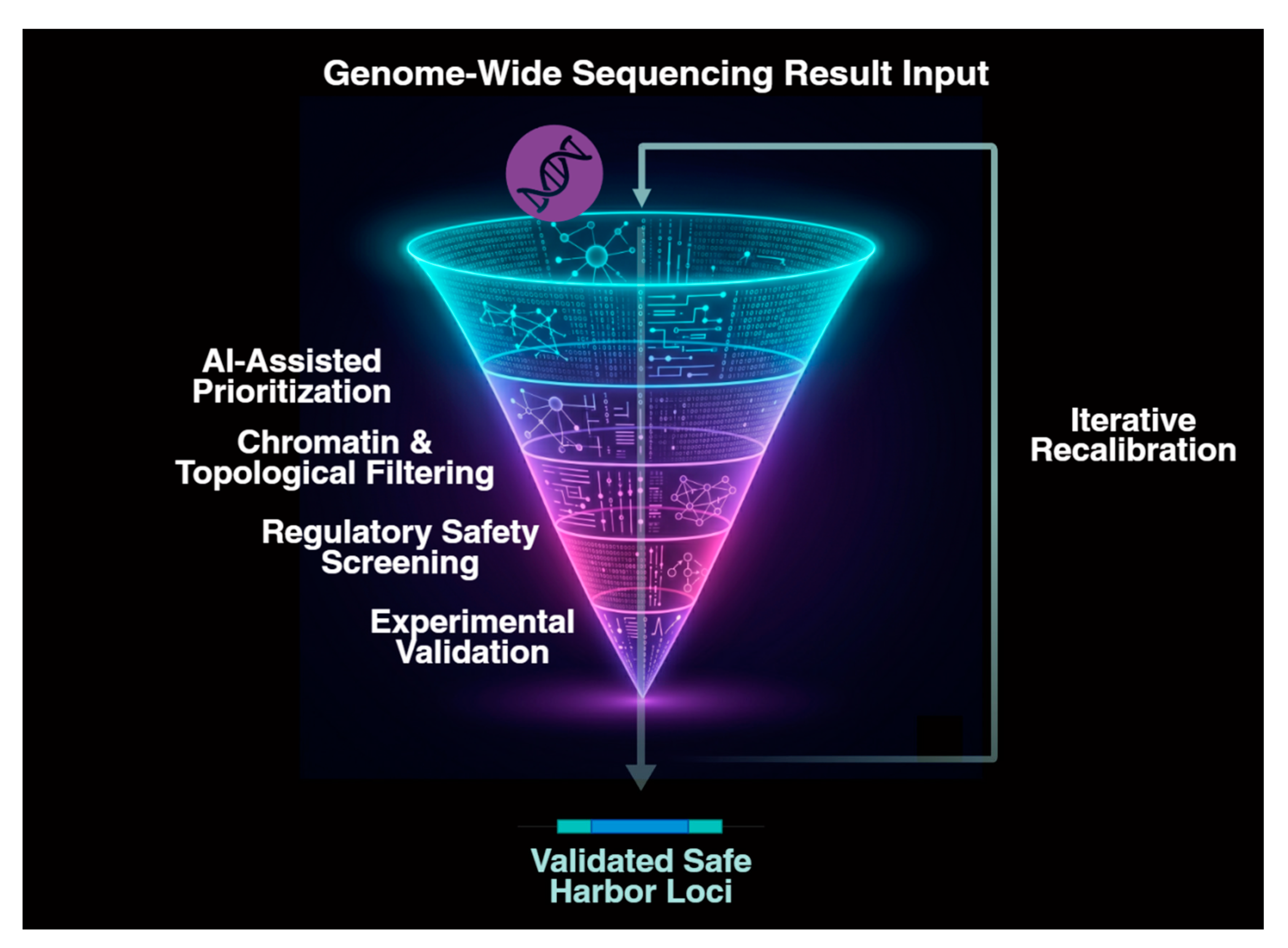
3.3. Genomic Precision as the Integrative Bottleneck
3.4. From Cell Lines to Manufacturing Platforms: The Public Health Imperative
| Engineering Layer & Component | Biological Intervention | Primary Manufacturing Benefit | Inherent Trade-off / Implementation Barrier | Reference |
| Membrane Interface | SLAM (CD150) / KREMEN1 knock-in | Circumvents host-range limitations (e.g., Measles, Coxsackievirus) to enable productive infection without relying on tumor-derived cell lines. | Receptor amplification potentially imposes additional burden on limited endoplasmic reticulum (ER) folding capacity, precipitating premature secretory stress. | [21,32] |
| Membrane Interface | SCARB2 / AXL / TIM-1 overexpression | Relieves entry kinetic bottlenecks (e.g., for EV71, filoviruses), producing order-of-magnitude yield improvements. | Yields diminishing returns for viruses with multi-factorial entry requirements or extremely low infectious inputs (e.g., SARS-CoV-2 requiring coordinated ACE2/TMPRSS2). | [22,33,34,35,36] |
| Membrane Interface | CD155 disruption | Renders cells non-permissive to poliovirus, establishing intrinsic biosafety barriers compatible with WHO GAPIII containment standards. | Narrows the broad-spectrum permissiveness characteristic of the platform, requiring separate maintenance of distinct engineered sublines. | [37] |
| Membrane Interface | CDH18 / PTEN downregulation & IGF-1 activation | Weakens intercellular adhesion and activates anoikis resistance, enabling high-density suspension cultivation. | Broad suppression of anchoring cues triggers compensatory, chronic ER stress-adaptation loops (e.g., DDIT3/CHOP upregulation), entrenching cells in persistent stress. | [38,39,40,41] |
| Cytoplasmic Foundry | XBP1s overexpression | Expands ER buffering capacity, significantly enhancing viral glycoprotein secretion under high-load conditions. | Constitutive stress-response activation globally alters host metabolic homeostasis, potentially disrupting baseline cellular functions prior to viral infection. | [49] |
| Cytoplasmic Foundry | BCL-XL overexpression | Delays apoptotic commitment, significantly extending the productive viral replication window. | Prolonged strain risks the accumulation of defective interfering particles and incomplete virions, while exacerbating tumorigenicity concerns for vaccine substrates. | [50] |
| Cytoplasmic Foundry | ISG15 deletion / IFNG–IFNGR1 knockout | Dramatically amplifies volumetric yields for highly interferon-sensitive viruses (e.g., influenza, rVSV) by removing residual host defense. | Apoptosis blockade without upstream stress mitigation may redirect death toward necrotic pathways, releasing host cell proteins/DNA and inflating downstream processing burdens. | [51] |
| Nuclear Blueprint | EMX2 knockout / Restriction factor editing | Remodels the transcriptional command layer to measurably reshape early-passage viral productivity. | Fails to ensure sustained transcriptional output; relies on physical locus disruption rather than enduring epigenetic collaboration. | [52] |
| Nuclear Blueprint | miniUCOE incorporation (Upstream of EF1α/CMV) | Reduces CpG methylation-associated silencing, sustaining transgene expression across serial passages. | Requires integration into randomly distributed loci, leaving circuits vulnerable to position effects, subline heterogeneity, and broader chromatin neighborhood remodeling. | [63] |
| Nuclear Blueprint | Static Recombinase / RMCE integration | Enables targeted cassette exchange to reduce clone-to-clone heterogeneity and preserve genomic context. | Still limited by the intrinsic karyotypic instability of Vero cells if not anchored within explicitly validated, stress-resilient Genomic Safe Harbors (GSHs). | [127] |
4. Discussion & Conclusions
References
- Yasumura, Y.; Kawakita, Y. Studies on SV40 in tissue culture: Preliminary step for cancer research in vitro. Nihon Rinsho 1963, 21, 1201–1215. [Google Scholar]
- Sweet, B.H.; Hilleman, M.R. The vacuolating virus, SV 40. Proceedings of the Society for Experimental Biology and Medicine 1960, 105, 420–427. [Google Scholar] [CrossRef] [PubMed]
- Barrett, P.N.; Mundt, W.; Kistner, O.; Howard, M.K. Vero cell platform in vaccine production: moving towards cell culture-based viral vaccines. Expert Rev Vaccines 2009, 8, 607–618. [Google Scholar] [CrossRef]
- Petricciani, J. Should continuous cell lines be used as substrates for biological products? Developments in Biological Standardization 1987, 66, 3–12. [Google Scholar]
- Desmyter, J.; Melnick, J.L.; Rawls, W.E. Defectiveness of interferon production and of rubella virus interference in a line of African green monkey kidney cells (Vero). Journal of virology 1968, 2, 955–961. [Google Scholar] [CrossRef]
- Emeny, J.M.; Morgan, M.J. Regulation of the interferon system: evidence that Vero cells have a genetic defect in interferon production. Journal of General Virology 1979, 43, 247–252. [Google Scholar] [CrossRef]
- Montagnon, B.; Fanget, B.; Vincent-Falquet, J. Industrial-scale production of inactivated poliovirus vaccine prepared by culture of Vero cells on microcarrier. Reviews of infectious diseases 1984, 6, S341–S344. [Google Scholar] [CrossRef]
- Rourou, S.; van Der Ark, A.; Majoul, S.; Trabelsi, K.; van Der Velden, T.; Kallel, H. A novel animal-component-free medium for rabies virus production in Vero cells grown on Cytodex 1 microcarriers in a stirred bioreactor. Applied microbiology and biotechnology 2009, 85, 53–63. [Google Scholar] [CrossRef]
- Orr-Burks, N.; Murray, J.; Wu, W.; Kirkwood, C.D.; Todd, K.V.; Jones, L.; Bakre, A.; Wang, H.; Jiang, B.; Tripp, R.A. Gene-edited vero cells as rotavirus vaccine substrates. Vaccine X 2019, 3, 100045. [Google Scholar] [CrossRef]
- Peden, K. Development of Quantitative Assays to Evaluate the Safety of Cell Substrates and Vaccines. Available online: https://www.fda.gov/vaccines-blood-biologics/science-research-biologics/development-quantitative-assays-evaluate-safety-cell-substrates-and-vaccines (accessed on April 12).
- Barrett, P.N.; Terpening, S.J.; Snow, D.; Cobb, R.R.; Kistner, O. Vero cell technology for rapid development of inactivated whole virus vaccines for emerging viral diseases. Expert Rev Vaccines 2017, 16, 883–894. [Google Scholar] [CrossRef]
- Murray, J.; Todd, K.V.; Bakre, A.; Orr-Burks, N.; Jones, L.; Wu, W.; Tripp, R.A. A universal mammalian vaccine cell line substrate. PLoS One 2017, 12, e0188333. [Google Scholar] [CrossRef] [PubMed]
- Sene, M.A.; Xia, Y.; Kamen, A.A. Comparative Transcriptomic Analyses of a Vero Cell Line in Suspension versus Adherent Culture Conditions. Int J Cell Biol 2023, 2023, 9364689. [Google Scholar] [CrossRef] [PubMed]
- Sene, M.A.; Kiesslich, S.; Djambazian, H.; Ragoussis, J.; Xia, Y.; Kamen, A.A. Haplotype-resolved de novo assembly of the Vero cell line genome. NPJ Vaccines 2021, 6, 106. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.K.-L.; Poh, S.L.; Dietzsch, C.; Roethl, E.; Yan, M.L.; Ng, S.K. Serum-free microcarrier based production of replication deficient Influenza vaccine candidate virus lacking NS1 using Vero cells. BMC Biotechnology 2011, 11, 81. [Google Scholar] [CrossRef]
- Kiesslich, S.; Kamen, A.A. Vero cell upstream bioprocess development for the production of viral vectors and vaccines. Biotechnol Adv 2020, 44, 107608. [Google Scholar] [CrossRef]
- Kiesslich, S.; Losa, J.P.V.-C.; Gélinas, J.-F.; Kamen, A.A. Serum-free production of rVSV-ZEBOV in Vero cells: Microcarrier bioreactor versus scale-X™ hydro fixed-bed. Journal of Biotechnology 2020, 310, 32–39. [Google Scholar] [CrossRef]
- Shen, C.F.; Guilbault, C.; Li, X.; Elahi, S.M.; Ansorge, S.; Kamen, A.; Gilbert, R. Development of suspension adapted Vero cell culture process technology for production of viral vaccines. Vaccine 2019, 37, 6996–7002. [Google Scholar] [CrossRef]
- Rourou, S.; Ben Zakkour, M.; Kallel, H. Adaptation of Vero cells to suspension growth for rabies virus production in different serum free media. Vaccine 2019, 37, 6987–6995. [Google Scholar] [CrossRef]
- Thomassen, Y.E.; Rubingh, O.; Wijffels, R.H.; van der Pol, L.A.; Bakker, W.A. Improved poliovirus D-antigen yields by application of different Vero cell cultivation methods. Vaccine 2014, 32, 2782–2788. [Google Scholar] [CrossRef]
- Hashimoto, K.; Ono, N.; Tatsuo, H.; Minagawa, H.; Takeda, M.; Takeuchi, K.; Yanagi, Y. SLAM (CD150)-independent measles virus entry as revealed by recombinant virus expressing green fluorescent protein. Journal of virology 2002, 76, 6743–6749. [Google Scholar] [CrossRef]
- Li, X.; Fan, P.; Jin, J.; Su, W.; An, D.; Xu, L.; Sun, S.; Zhang, Y.; Meng, X.; Gao, F. Establishment of cell lines with increased susceptibility to EV71/CA16 by stable overexpression of SCARB2. Virology journal 2013, 10, 250. [Google Scholar] [CrossRef] [PubMed]
- Ji, W.; Chen, Z.; Zhou, J.; Yue, X.; Qiao, Z.; Wang, J. Advances in Serum-Free Suspension Culture Technology for Animal Cells and Their Applications. Vaccines (Basel) 2025, 13. [Google Scholar] [CrossRef] [PubMed]
- Bourigault, L.; Bresson, C.; Jean, C.; Chevalard, C.; Kloutz, M.; Soulet, D.; Pelissier, F.; Richard, S.; Bassard, I.; Seve, N.; et al. Characterization of a suspension Vero cell line for viral vaccine production. NPJ Vaccines 2025, 10, 114. [Google Scholar] [CrossRef]
- Mazel-Sanchez, B.; Iwaszkiewicz, J.; Bonifacio, J.P.; Silva, F.; Niu, C.; Strohmeier, S.; Eletto, D.; Krammer, F.; Tan, G.; Zoete, V. Influenza A viruses balance ER stress with host protein synthesis shutoff. Proceedings of the National Academy of Sciences 2021, 118, e2024681118. [Google Scholar] [CrossRef] [PubMed]
- He, B. Viruses, endoplasmic reticulum stress, and interferon responses. Cell Death & Differentiation 2006, 13, 393–403. [Google Scholar] [CrossRef]
- Dupont, S.; Morsut, L.; Aragona, M.; Enzo, E.; Giulitti, S.; Cordenonsi, M.; Zanconato, F.; Le Digabel, J.; Forcato, M.; Bicciato, S. Role of YAP/TAZ in mechanotransduction. Nature 2011, 474, 179–183. [Google Scholar] [CrossRef]
- Elosegui-Artola, A.; Andreu, I.; Beedle, A.E.; Lezamiz, A.; Uroz, M.; Kosmalska, A.J.; Oria, R.; Kechagia, J.Z.; Rico-Lastres, P.; Le Roux, A.-L. Force triggers YAP nuclear entry by regulating transport across nuclear pores. Cell 2017, 171, 1397–1410. e1314. [Google Scholar] [CrossRef]
- Andreani, N.; Renzi, S.; Piovani, G.; Ajmone Marsan, P.; Bomba, L.; Villa, R.; Ferrari, M.; Dotti, S. Potential neoplastic evolution of Vero cells: in vivo and in vitro characterization. Cytotechnology 2017, 69, 741–750. [Google Scholar] [CrossRef]
- Venu, V.; Roth, C.; Adikari, S.H.; Small, E.M.; Starkenburg, S.R.; Sanbonmatsu, K.Y.; Steadman, C.R. Multi-omics analysis reveals the dynamic interplay between Vero host chromatin structure and function during vaccinia virus infection. Commun Biol 2024, 7, 721. [Google Scholar] [CrossRef]
- Feng, N.; Liu, Y.; Wang, J.; Xu, W.; Li, T.; Wang, T.; Wang, L.; Yu, Y.; Wang, H.; Zhao, Y. Canine distemper virus isolated from a monkey efficiently replicates on Vero cells expressing non-human primate SLAM receptors but not human SLAM receptor. BMC veterinary research 2016, 12, 160. [Google Scholar] [CrossRef]
- Zhang, D.; Zou, Y.; Wu, J.; Xu, L.; Ke, Z.; Wu, Y.; Zhou, Z.; Fang, M.; Chen, L.; Xu, H.; et al. Construction of a Vero cell line expression human KREMEN1 for the development of CVA6 vaccines. Virol J 2025, 22, 12. [Google Scholar] [CrossRef]
- Hunt, C.L.; Kolokoltsov, A.A.; Davey, R.A.; Maury, W. The Tyro3 receptor kinase Axl enhances macropinocytosis of Zaire ebolavirus. J Virol 2011, 85, 334–347. [Google Scholar] [CrossRef]
- Brunton, B.; Rogers, K.; Phillips, E.K.; Brouillette, R.B.; Bouls, R.; Butler, N.S.; Maury, W. TIM-1 serves as a receptor for Ebola virus in vivo, enhancing viremia and pathogenesis. PLoS Negl Trop Dis 2019, 13, e0006983. [Google Scholar] [CrossRef] [PubMed]
- van den Braak, W.J.P.; Monica, B.; Limpens, D.; Rockx-Brouwer, D.; de Boer, M.; Oosterhoff, D. Construction of a Vero Cell Line Expressing Human ICAM1 for the Development of Rhinovirus Vaccines. Viruses 2022, 14. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, H.; Yamamoto, T.; Kuroda, Y.; Inoue, Y.; Miyazaki, K.; Ohmagari, N.; Tokita, D.; Nguyen, P.H.A.; Yamada, S.; Harada, S.; et al. Improved efficacy of SARS-CoV-2 isolation from COVID-19 clinical specimens using VeroE6 cells overexpressing TMPRSS2 and human ACE2. Sci Rep 2024, 14, 24858. [Google Scholar] [CrossRef]
- Okemoto-Nakamura, Y.; Someya, K.; Yamaji, T.; Saito, K.; Takeda, M.; Hanada, K. Poliovirus-nonsusceptible Vero cell line for the World Health Organization global action plan. Sci Rep 2021, 11, 6746. [Google Scholar] [CrossRef] [PubMed]
- Luey, B.C.; May, F.E. Insulin-like growth factors are essential to prevent anoikis in oestrogen-responsive breast cancer cells: importance of the type I IGF receptor and PI3-kinase/Akt pathway. Molecular cancer 2016, 15, 8. [Google Scholar] [CrossRef]
- Pech, S.; Rehberg, M.; Janke, R.; Benndorf, D.; Genzel, Y.; Muth, T.; Sickmann, A.; Rapp, E.; Reichl, U. Tracking changes in adaptation to suspension growth for MDCK cells: cell growth correlates with levels of metabolites, enzymes and proteins. Appl Microbiol Biotechnol 2021, 105, 1861–1874. [Google Scholar] [CrossRef]
- Chu, C.; Bottaro, D.P.; Betenbaugh, M.J.; Shiloach, J. Stable Ectopic Expression of ST6GALNAC5 Induces Autocrine MET Activation and Anchorage-Independence in MDCK Cells. PLoS One 2016, 11, e0148075. [Google Scholar] [CrossRef]
- Sun, J.; Ren, H.; Wang, J.; Xiao, X.; Zhu, L.; Wang, Y.; Yang, L. CHAC1: a master regulator of oxidative stress and ferroptosis in human diseases and cancers. Frontiers in Cell and Developmental Biology 2024, 12, 1458716. [Google Scholar] [CrossRef]
- Ron, D.; Walter, P. Signal integration in the endoplasmic reticulum unfolded protein response. Nature reviews Molecular cell biology 2007, 8, 519–529. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Brodsky, J.L. Protein quality control in the secretory pathway. Journal of Cell Biology 2019, 218, 3171–3187. [Google Scholar] [CrossRef] [PubMed]
- Deng, Z.; Wang, H.; Liu, J.; Deng, Y.; Zhang, N. Comprehensive understanding of anchorage-independent survival and its implication in cancer metastasis. Cell Death & Disease 2021, 12, 629. [Google Scholar] [CrossRef]
- Zhang, B.; Li, S.; Liu, J.; Su, W.; Zhang, X.; Ren, X.; Zhao, T.; Huang, Q.; Ge, Z.; Wei, J. Phenotypes and Multi-omics Reveal Changes and Molecular Mechanism of Suspension Adaptation of HEK293 Cells: Structural Remodelling, Metabolic Reconstruction and Stress Resistance; 2026. [Google Scholar]
- Sanchez, E.L.; Lagunoff, M. Viral activation of cellular metabolism. Virology 2015, 479, 609–618. [Google Scholar] [CrossRef]
- Liu, X.; Li, F.; Zhang, J.; Wang, L.; Wang, J.; Wen, Z.; Wang, Z.; Shuai, L.; Wang, X.; Ge, J. The ATPase ATP6V1A facilitates rabies virus replication by promoting virion uncoating and interacting with the viral matrix protein. Journal of biological chemistry 2021, 296, 100096. [Google Scholar] [CrossRef]
- Clark, S.A.; Vazquez, A.; Furiya, K.; Splattstoesser, M.K.; Bashmail, A.K.; Schwartz, H.; Russell, M.; Bhark, S.-J.; Moreno, O.K.; McGovern, M. Rewiring of the host cell metabolome and lipidome during lytic gammaherpesvirus infection is essential for infectious-virus production. Journal of virology 2023, 97, e00506-00523. [Google Scholar] [CrossRef]
- Barrios, D.; Bachhav, B.; Carlos-Alcalde, W.; Llanos, C.D.; Zhou, W.; Segatori, L. Feedback-responsive cell factories for dynamic modulation of the unfolded protein response. Nature communications 2025, 16, 4106. [Google Scholar] [CrossRef]
- Zehnle, P.M.; Wu, Y.; Koleci, N.; Bohler, S.; Erlacher, M. Lentivirus-Mediated BCL-XL Overexpression Inhibits Stem Cell Apoptosis during Ex Vivo Expansion and Provides Competitive Advantage Following Xenotransplantation. International Journal of Molecular Sciences 2024, 25, 4105. [Google Scholar] [CrossRef]
- Sène, M.A.; Xia, Y.; Kamen, A.A. From functional genomics of vero cells to CRISPR-based genomic deletion for improved viral production rates. Biotechnology and Bioengineering 2022, 119, 2794–2805. [Google Scholar] [CrossRef]
- Kim, S.; Nowakowska, A.; Kim, Y.B.; Shin, H.Y. Integrated CRISPR-Cas9 system-mediated knockout of IFN-γ and IFN-γ receptor 1 in the Vero cell line promotes viral susceptibility. International Journal of Molecular Sciences 2022, 23, 8217. [Google Scholar] [CrossRef]
- Frensing, T.; Heldt, F.S.; Pflugmacher, A.; Behrendt, I.; Jordan, I.; Flockerzi, D.; Genzel, Y.; Reichl, U. Continuous influenza virus production in cell culture shows a periodic accumulation of defective interfering particles. PloS one 2013, 8, e72288. [Google Scholar] [CrossRef] [PubMed]
- Tapia, F.; Laske, T.; Wasik, M.A.; Rammhold, M.; Genzel, Y.; Reichl, U. Production of defective interfering particles of influenza A virus in parallel continuous cultures at two residence times—insights from qPCR measurements and viral dynamics modeling. Frontiers in bioengineering and biotechnology 2019, 7, 275. [Google Scholar] [CrossRef] [PubMed]
- Galluzzi, L.; Vitale, I.; Aaronson, S.A.; Abrams, J.M.; Adam, D.; Agostinis, P.; Alnemri, E.S.; Altucci, L.; Amelio, I.; Andrews, D.W. Molecular mechanisms of cell death: recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death & Differentiation 2018, 25, 486–541. [Google Scholar] [CrossRef]
- Tait, A.S.; Hogwood, C.E.; Smales, C.M.; Bracewell, D.G. Host cell protein dynamics in the supernatant of a mAb producing CHO cell line. Biotechnology and Bioengineering 2012, 109, 971–982. [Google Scholar] [CrossRef]
- Kiesslich, S. Development of a Vero cell platform for rVSV vector production; McGill University (Canada), 2021. [Google Scholar]
- Khan, O.; Tanuj, G.N.; Choravada, D.R.; Rajak, K.K.; Chandra Sekar, S.; Lingaraju, M.C.; Dhara, S.K.; Gupta, P.K.; Mishra, B.P.; Dutt, T. N6-methyladenosine RNA modification in host cells regulates peste des petits ruminants virus replication. Microbiology Spectrum 2023, 11, e02666-02622. [Google Scholar] [CrossRef]
- Mazhed, Z.; Vasilenko, V.; Siniugina, A.; Kaa, K.; Motov, A.; Pokidova, K.; Ivin, Y.; Piniaeva, A.; Khapchaev, Y.; Chernov, K. Intensification of Vero cell adherence to microcarrier particles during cultivation in a wave bioreactor. Frontiers in Bioengineering and Biotechnology 2025, 13, 1542060. [Google Scholar] [CrossRef]
- Kiesslich, S.; Kim, G.N.; Shen, C.F.; Kang, C.Y.; Kamen, A.A. Bioreactor production of rVSV-based vectors in Vero cell suspension cultures. Biotechnology and bioengineering 2021, 118, 2649–2659. [Google Scholar] [CrossRef]
- Rozelle, D.K.; Filone, C.M.; Kedersha, N.; Connor, J.H. Activation of stress response pathways promotes formation of antiviral granules and restricts virus replication. Molecular and Cellular Biology 2014, 34, 2003–2016. [Google Scholar] [CrossRef]
- Nagorska, A.; Tomás, R.M.; Tasnim, A.; Robb, N.C.; Gibson, M.I. Cryopreserved Kidney Epithelial (Vero) Cell Monolayers for Rapid Viral Quantification, Enabled by a Combination of Macromolecular Cryoprotectants. Biomacromolecules 2024, 25, 5352–5358. [Google Scholar] [CrossRef]
- Kwak, C.S.; Oflaz, F.E.; Qiu, J.; Wang, X. Human stem cell-specific epigenetic signatures control transgene expression. Biochimica et Biophysica Acta (BBA)-Gene Regulatory Mechanisms 2024, 1867, 195063. [Google Scholar] [CrossRef]
- Fedak, E.A.; Adler, F.R.; Abegglen, L.M.; Schiffman, J.D. ATM and ATR activation through crosstalk between DNA damage response pathways. Bulletin of mathematical biology 2021, 83, 38. [Google Scholar] [CrossRef] [PubMed]
- Baer, A.; Austin, D.; Narayanan, A.; Popova, T.; Kainulainen, M.; Bailey, C.; Kashanchi, F.; Weber, F.; Kehn-Hall, K. Induction of DNA damage signaling upon Rift Valley fever virus infection results in cell cycle arrest and increased viral replication. Journal of Biological Chemistry 2012, 287, 7399–7410. [Google Scholar] [CrossRef] [PubMed]
- Victor, J.; Deutsch, J.; Whitaker, A.; Lamkin, E.N.; March, A.; Zhou, P.; Botten, J.W.; Chatterjee, N. SARS-CoV-2 triggers DNA damage response in Vero E6 cells. Biochemical and biophysical research communications 2021, 579, 141–145. [Google Scholar] [CrossRef]
- Hoeksema, F.; Karpilow, J.; Luitjens, A.; Lagerwerf, F.; Havenga, M.; Groothuizen, M.; Gillissen, G.; Lemckert, A.; Jiang, B.; Tripp, R. Enhancing viral vaccine production using engineered knockout vero cell lines–A second look. Vaccine 2018, 36, 2093–2103. [Google Scholar] [CrossRef]
- Freitas, F.B.; Simões, M.; Frouco, G.; Martins, C.; Ferreira, F. Towards the generation of an ASFV-pA104R DISC mutant and a complementary cell line—a potential methodology for the production of a vaccine candidate. Vaccines 2019, 7, 68. [Google Scholar] [CrossRef]
- Rival-Gervier, S.; Lo, M.Y.; Khattak, S.; Pasceri, P.; Lorincz, M.C.; Ellis, J. Kinetics and epigenetics of retroviral silencing in mouse embryonic stem cells defined by deletion of the D4Z4 element. Molecular Therapy 2013, 21, 1536–1550. [Google Scholar] [CrossRef]
- Alhaji, S.Y.; Ngai, S.C.; Abdullah, S. Silencing of transgene expression in mammalian cells by DNA methylation and histone modifications in gene therapy perspective. Biotechnology and Genetic Engineering Reviews 2019, 35, 1–25. [Google Scholar] [CrossRef]
- McCreery, K.P.; Stubb, A.; Stephens, R.; Fursova, N.A.; Cook, A.; Kruse, K.; Michelbach, A.; Biggs, L.C.; Keikhosravi, A.; Nykänen, S. Mechano-osmotic signals control chromatin state and fate transitions in pluripotent stem cells. Nature Cell Biology 2025, 27, 1757–1770. [Google Scholar] [CrossRef]
- Feng, Y.-Q.; Desprat, R.; Fu, H.; Olivier, E.; Lin, C.M.; Lobell, A.; Gowda, S.N.; Aladjem, M.I.; Bouhassira, E.E. DNA methylation supports intrinsic epigenetic memory in mammalian cells. PLoS genetics 2006, 2, e65. [Google Scholar] [CrossRef]
- Bandyopadhyay, A.A.; O’Brien, S.A.; Zhao, L.; Fu, H.Y.; Vishwanathan, N.; Hu, W.S. Recurring genomic structural variation leads to clonal instability and loss of productivity. Biotechnology and bioengineering 2019, 116, 41–53. [Google Scholar] [CrossRef]
- Migliaccio, A.R.; Bengra, C.; Ling, J.; Pi, W.; Li, C.; Zeng, S.; Keskintepe, M.; Whitney, B.; Sanchez, M.; Migliaccio, G. Stable and unstable transgene integration sites in the human genome: extinction of the Green Fluorescent Protein transgene in K562 cells. Gene 2000, 256, 197–214. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.; Lam, C.; Bauer, N.; Auslaender, S.; Snedecor, B.; Laird, M.W.; Misaghi, S. Bax and Bak knockout apoptosis-resistant Chinese hamster ovary cell lines significantly improve culture viability and titer in intensified fed-batch culture process. Biotechnology Progress 2022, 38, e3228. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Zhang, T.; Lyu, R.; Hou, B.; Fan, T.; Yang, H.; Na, J. Effects of blocking apoptosis and lactic acid metabolism pathways on robustness and foreign protein expression of CHO cells. Sheng wu gong cheng xue bao= Chinese journal of biotechnology 2025, 41, 3098–3109. [Google Scholar]
- MacDonald, M.A.; Nöbel, M.; Martínez, V.S.; Baker, K.; Shave, E.; Gray, P.P.; Mahler, S.; Munro, T.; Nielsen, L.K.; Marcellin, E. Engineering death resistance in CHO cells for improved perfusion culture. Proceedings of MAbs; p. 2083465.
- Torres, M.; Mcconnaughie, D.; Akhtar, S.; Gaffney, C.E.; Fievet, B.; Ingham, C.; Stockdale, M.; Dickson, A.J. Engineering mammalian cell growth dynamics for biomanufacturing. Metabolic Engineering 2024, 82, 89–99. [Google Scholar] [CrossRef]
- Zustiak, M.P.; Jose, L.; Xie, Y.; Zhu, J.; Betenbaugh, M.J. Enhanced transient recombinant protein production in CHO cells through the co-transfection of the product gene with Bcl-xL. Biotechnology journal 2014, 9, 1164–1174. [Google Scholar] [CrossRef]
- Baek, E.; Noh, S.M.; Lee, G.M. Anti-apoptosis engineering for improved protein production from CHO cells. In Heterologous Protein Production in CHO Cells: Methods and Protocols; Springer, 2017; pp. 71–85. [Google Scholar]
- Rodrigues, A.F.; Soares, H.R.; Guerreiro, M.R.; Alves, P.M.; Coroadinha, A.S. Viral vaccines and their manufacturing cell substrates: New trends and designs in modern vaccinology. Biotechnology journal 2015, 10, 1329–1344. [Google Scholar] [CrossRef]
- Organization, W.H. Acceptability of cell substrates for production of biologicals: report of a WHO study group [meeting held in Geneva from 18 to 19 November 1986]. 1987. [Google Scholar]
- Organization, W.H. Requirements for continuous cell lines used for biological substances. WHO Tech Rep Ser 1987, 745, 99–115. [Google Scholar]
- Berry, J.M. Optimisation of Vero cell growth and productivity in microcarrier cultures; 1996. [Google Scholar]
- Rourou, S.; Ayed, Y.B.; Trabelsi, K.; Majoul, S.; Kallel, H. An animal component free medium that promotes the growth of various animal cell lines for the production of viral vaccines. Vaccine 2014, 32, 2767–2769. [Google Scholar] [CrossRef]
- Sousa, M.; Fenge, C.; Rupprecht, J.; Tappe, A.; Greller, G.; Alves, P.; Carrondo, M.; Roldão, A. Process intensification for Peste des Petites Ruminants Virus vaccine production. Vaccine 2019, 37, 7041–7051. [Google Scholar] [CrossRef]
- Shen, C.F.; Gilbert, R.; Burney, E.; Mehdy Elahi, S.; Lanthier, S.; Kamen, A.; Loignon, M. Suspension Vero cell culture technology for high titer production of viral vaccines. 2022. [Google Scholar]
- Mattos, D.A.; Silva, M.V.; Gaspar, L.P.; Castilho, L.R. Increasing Vero viable cell densities for yellow fever virus production in stirred-tank bioreactors using serum-free medium. Vaccine 2015, 33, 4288–4291. [Google Scholar] [CrossRef] [PubMed]
- Ursache, R.V.; Thomassen, Y.E.; van Eikenhorst, G.; Verheijen, P.J.; Bakker, W.A. Mathematical model of adherent Vero cell growth and poliovirus production in animal component free medium. Bioprocess and biosystems engineering 2015, 38, 543–555. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Orr-Burks, N.; Karpilow, J.M.; Tripp, R.A. Development of improved vaccine cell lines against rotavirus. Scientific Data 2017, 4. [Google Scholar] [CrossRef]
- Zhu, Y.; Sullender, M.E.; Campbell, D.E.; Wang, L.; Lee, S.; Kawagishi, T.; Hou, G.; Dizdarevic, A.; Jais, P.H.; Baldridge, M.T. CRISPR/Cas9 screens identify key host factors that enhance rotavirus reverse genetics efficacy and vaccine production. npj Vaccines 2024, 9, 211. [Google Scholar] [CrossRef]
- Cahn, J.K.B.; Ludwicki, H.; Shingler, J.; Gulvin, S.; Zhang, Y.; Kristopeit, A.; Ton, C.; Winters, M.A.; Wagner, J.M.; Moore, J. CRISPR-Editing of the Vero Cell Line Improves Processability of Live Virus Vaccines by Enabling Targeted Proteolysis of Fibronectin. Biotechnol Bioeng 2025, 122, 2082–2092. [Google Scholar] [CrossRef]
- Fukumoto, H.; Hishima, T.; Hasegawa, H.; Saeki, H.; Kuroda, M.; Katano, H. Evaluation of Vero-cell-derived simian endogenous retrovirus infection in humans by detection of viral genome in clinicopathological samples and commercialized vaccines and by serology of Japanese general population. Vaccine 2016, 34 24, 2700–2706. [Google Scholar] [CrossRef]
- Cheval, J.; Muth, E.; Gonzalez, G.; Coulpier, M.; Beurdeley, P.; Cruveiller, S.; Eloit, M. Adventitious Virus Detection in Cells by High-Throughput Sequencing of Newly Synthesized RNAs: Unambiguous Differentiation of Cell Infection from Carryover of Viral Nucleic Acids. mSphere 2019, 4. [Google Scholar] [CrossRef]
- Baek, E.; Noh, S.M.; Kim, S.H.; Lee, G.M. Antiapoptosis Engineering for Improved Protein Production from CHO Cells. Methods in molecular biology 2025, 2853, 103–117. [Google Scholar]
- Poulain, A.; Perret, S.; Malenfant, F.; Mullick, A.; Massie, B.; Durocher, Y. Rapid protein production from stable CHO cell pools using plasmid vector and the cumate gene-switch. Journal of biotechnology 2017, 255, 16–27. [Google Scholar] [CrossRef]
- Lîebert, M.A. Guidance for Industry: Characterization and Qualification of Cell Substrates and Other Biological Starting Materials Used in the Production of Viral Vaccines for the Prevention and Treatment of Infectious Diseases. Biotechnology Law Report 2006, 25, 697–723. [Google Scholar] [CrossRef]
- Kallunki, T.; Barišić, M.; Jäättelä, M.; Liu, B. How to Choose the Right Inducible Gene Expression System for Mammalian Studies? Cells 2019, 8. [Google Scholar] [CrossRef] [PubMed]
- Osada, N.; Kohara, A.; Yamaji, T.; Hirayama, N.; Kasai, F.; Sekizuka, T.; Kuroda, M.; Hanada, K. The Genome Landscape of the African Green Monkey Kidney-Derived Vero Cell Line. DNA Research: An International Journal for Rapid Publication of Reports on Genes and Genomes 2014, 21, 673–683. [Google Scholar] [CrossRef] [PubMed]
- Konishi, K.; Yamaji, T.; Sakuma, C.; Kasai, F.; Endo, T.; Kohara, A.; Hanada, K.; Osada, N. Whole-Genome Sequencing of Vero E6 (VERO C1008) and Comparative Analysis of Four Vero Cell Sublines. Frontiers in Genetics 2022, 13. [Google Scholar] [CrossRef]
- Zhu, I.; Liu, R.; Garcia, J.M.; Hyrenius-Wittsten, A.; Piraner, D.I.; Alavi, J.; Israni, D.V.; Liu, B.; Khalil, A.S.; Roybal, K.T. Modular design of synthetic receptors for programmed gene regulation in cell therapies. Cell 2022, 185 8, 1431–1443.e1416. [Google Scholar] [CrossRef]
- Piraner, D.I.; Abedi, M.H.; Duran Gonzalez, M.J.; Chazin-Gray, A.M.; Lin, A.; Zhu, I.; Ravindran, P.T.; Schlichthaerle, T.; Huang, B.; Bearchild, T.H.; et al. Engineered receptors for soluble cellular communication and disease sensing. Nature 2024, 638, 805–813. [Google Scholar] [CrossRef]
- Chavez, A.; Scheiman, J.; Vora, S.; Pruitt, B.W.; Tuttle, M.; Iyer, E.P.R.; Lin, S.; Kiani, S.; Guzman, C.D.; Wiegand, D.J.; et al. Highly-efficient Cas9-mediated transcriptional programming. Nature methods 2014, 12, 326–328. [Google Scholar] [CrossRef]
- Cai, R.; Lv, R.; Shi, X.e.; Yang, G.; Jin, J. CRISPR/dCas9 Tools: Epigenetic Mechanism and Application in Gene Transcriptional Regulation. International Journal of Molecular Sciences 2023, 24. [Google Scholar] [CrossRef]
- Zimak, J.; Wagoner, Z.W.; Nelson, N.; Waechtler, B.; Schlosser, H.; Kopecky, M.; Wu, J.; Zhao, W. Epigenetic silencing directs expression heterogeneity of stably integrated multi-transcript unit genetic circuits. Scientific Reports 2021, 11. [Google Scholar] [CrossRef]
- Cabera, A.; Edelstein, H.I.; Glykofrydis, F.; Love, K.S.; Palacios, S.; Tycko, J.; Zhang, M.; Lensch, S.; Shields, C.E.; Livingston, M.; et al. The sound of silence: Transgene silencing in mammalian cell engineering. Cell systems 2022, 13 12, 950–973. [Google Scholar] [CrossRef]
- Sizer, R.E.; White, R.J. Use of ubiquitous chromatin opening elements (UCOE) as tools to maintain transgene expression in biotechnology. Computational and Structural Biotechnology Journal 2022, 21, 275–283. [Google Scholar] [CrossRef]
- Lau, C.-H.; Suh, Y. In vivo epigenome editing and transcriptional modulation using CRISPR technology. Transgenic Research 2018, 27, 489–509. [Google Scholar] [CrossRef] [PubMed]
- ICH, H.U. Derivation and Characterisation of Cell Substrates Used for Production of Biotechnological/Biological Products; ICH, 2001. [Google Scholar]
- Doan, C.C.; Le, T.L.; Ho, N.Q.C.; Hoang, N.S. Effects of ubiquitous chromatin opening element (UCOE) on recombinant anti-TNFα antibody production and expression stability in CHO-DG44 cells. Cytotechnology 2021, 74, 31–49. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.H.S.; Gisonni-Lex, L.; Azizi, A. New approaches for characterization of the genetic stability of vaccine cell lines. Human Vaccines & Immunotherapeutics 2017, 13, 1669–1672. [Google Scholar] [CrossRef]
- Hilliard, W.; Lee, K.H. Systematic identification of safe harbor regions in the CHO genome through a comprehensive epigenome analysis. Biotechnol Bioeng 2021, 118, 659–675. [Google Scholar] [CrossRef]
- Hertel, O.; Neuss, A.; Busche, T.; Brandt, D.; Kalinowski, J.; Bahnemann, J.; Noll, T. Enhancing stability of recombinant CHO cells by CRISPR/Cas9-mediated site-specific integration into regions with distinct histone modifications. Frontiers in Bioengineering and Biotechnology 2022, 10. [Google Scholar] [CrossRef]
- Sadelain, M.; Papapetrou, E.P.; Bushman, F.D. Safe harbours for the integration of new DNA in the human genome. Nature Reviews Cancer 2011, 12, 51–58. [Google Scholar] [CrossRef]
- Shin, S.; Kim, S.H.; Shin, S.; Grav, L.M.; Pedersen, L.E.; Lee, J.S.; Lee, G.M. Comprehensive Analysis of Genomic Safe Harbors as Target Sites for Stable Expression of the Heterologous Gene in HEK293 Cells. ACS synthetic biology 2020. [Google Scholar] [CrossRef]
- Zeh, N.; Schmidt, M.; Schulz, P.; Fischer, S. The new frontier in CHO cell line development: From random to targeted transgene integration technologies. Biotechnology advances 2024, 108402. [Google Scholar] [CrossRef]
- Szkodny, A.C.; Lee, K.H. A Flexible Hybrid Site-Specific Integration-Based Expression System in CHO Cells for Higher-Throughput Evaluation of Monoclonal Antibody Expression Cassettes. Biotechnology Journal 2025, 20. [Google Scholar] [CrossRef]
- Campos, J.H.; Alves, G.V.; Maricato, J.T.; Braconi, C.T.; Antoneli, F.; Janini, L.M.R.; Briones, M.R.S. The epitranscriptome of Vero cells infected with SARS-CoV-2 assessed by direct RNA sequencing reveals m6A pattern changes and DRACH motif biases in viral and cellular RNAs. Frontiers in Cellular and Infection Microbiology 2022, 12. [Google Scholar] [CrossRef]
- Nyayanit, D.A.; Sarkale, P.; Baradkar, S.; Patil, S.; Yadav, P.D.; Shete-Aich, A.; Kalele, K.; Gawande, P.; Majumdar, T.; Jain, R.; et al. Transcriptome & viral growth analysis of SARS-CoV-2-infected Vero CCL-81 cells. The Indian Journal of Medical Research 2020, 152, 70–76. [Google Scholar] [PubMed]
- Roth, C.; Venu, V.; Bacot, S.; Starkenburg, S.R.; Steadman, C.R. SLUR(M)-py: a SLURM powered Pythonic pipeline for parallel processing of 3D (Epi)genomic profiles. Epigenomics 2025, 17, 1233–1247. [Google Scholar] [CrossRef] [PubMed]
- Avsec, Ž; Latysheva, N.; Cheng, J.; Novati, G.; Taylor, K.R.; Ward, T.; Bycroft, C.; Nicolaisen, L.; Arvaniti, E.; Pan, J.; et al. Advancing regulatory variant effect prediction with AlphaGenome. Nature 2026, 649, 1206–1218. [Google Scholar] [CrossRef]
- Avsec, Ž; Agarwal, V.; Visentin, D.; Ledsam, J.R.; Grabska-Barwinska, A.; Taylor, K.R.; Assael, Y.; Jumper, J.M.; Kohli, P.; Kelley, D.R. Effective gene expression prediction from sequence by integrating long-range interactions. Nature Methods 2021, 18, 1196–1203. [Google Scholar] [CrossRef]
- Linder, J.; Srivastava, D.; Yuan, H.; Agarwal, V.; Kelley, D.R. Predicting RNA-seq coverage from DNA sequence as a unifying model of gene regulation. Nat Genet 2025, 57, 949–961. [Google Scholar] [CrossRef]
- Zhou, J. Sequence-based modeling of three-dimensional genome architecture from kilobase to chromosome scale. Nature genetics 2022, 54, 725–734. [Google Scholar] [CrossRef]
- Kelley, D.R. Cross-species regulatory sequence activity prediction. PLoS Computational Biology 2019, 16. [Google Scholar] [CrossRef]
- Lee, J.S.; Kallehauge, T.B.; Pedersen, L.E.; Kildegaard, H.F. Site-specific integration in CHO cells mediated by CRISPR/Cas9 and homology-directed DNA repair pathway. Sci Rep 2015, 5, 8572. [Google Scholar] [CrossRef]
- Mayrhofer, P.; Kratzer, B.; Sommeregger, W.; Steinfellner, W.; Reinhart, D.; Mader, A.; Turan, S.; Qiao, J.; Bode, J.; Kunert, R. Accurate comparison of antibody expression levels by reproducible transgene targeting in engineered recombination-competent CHO cells. Applied Microbiology and Biotechnology 2014, 98, 9723–9733. [Google Scholar] [CrossRef]
- World Health Organization. Recommendations for the evaluation of animal cell cultures as substrates for the manufacture of biological medicinal products and for the characterization of cell banks. In WHO Technical Report Series; World Health Organization: Geneva, 2013; Volume No. 978, Annex 3. [Google Scholar]
- Bartlett, M.L.; Palese, P.; Davis, M.F.; Vermund, S.H.; Bréchot, C.; Evans, J.D.; Sauer, L.M.; Osterhaus, A.; Pekosz, A.; Nelson, M. Enhancing the response to avian influenza in the US and globally. The Lancet Regional Health–Americas 2025, 46. [Google Scholar] [CrossRef]
- Siddiquey, M.N.; Zhang, H.; Nguyen, C.C.; Domma, A.J.; Kamil, J.P. The human cytomegalovirus endoplasmic reticulum-resident glycoprotein UL148 activates the unfolded protein response. Journal of Virology 2018, 92. [Google Scholar] [CrossRef] [PubMed]
- Isler, J.A.; Skalet, A.H.; Alwine, J.C. Human cytomegalovirus infection activates and regulates the unfolded protein response. Journal of virology 2005, 79, 6890–6899. [Google Scholar] [CrossRef]
- Stahl, S.; Burkhart, J.M.; Hinte, F.; Tirosh, B.; Mohr, H.; Zahedi, R.P.; Sickmann, A.; Ruzsics, Z.; Budt, M.; Brune, W. Cytomegalovirus downregulates IRE1 to repress the unfolded protein response. PLoS pathogens 2013, 9, e1003544. [Google Scholar] [CrossRef]
- Dzau, V.; Swaminathan, S.; Baker, C.; Bright, R.A.; Castillo, J.; Chuan, T.C.; Draghia-Akli, R.; Eardley-Patel, R.; Gao, G.F.; Ishii, K. The 100 Days Mission: how a new medical-countermeasures network can deliver equity and innovation. The Lancet 2023, 402, 1507–1510. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




