Submitted:
17 April 2026
Posted:
21 April 2026
You are already at the latest version
Abstract
Keywords:
Biological Wavelength Sensitivity
Spectral Weighting Function for Biological Light Detection
Features and Applications
Concerns and Considerations
Conclusions
Acknowledgments
Conflicts of interest
References
- Linares Arroyo, H.; Abascal, A.; Degen, T.; Aube, M.; Espey, B.R.; Gyuk, G.; et al. Monitoring, trends and impacts of light pollution. Nature Reviews Earth and Environment 2024, 5, 427–30. [Google Scholar] [CrossRef]
- Gaston, K.J.; Davies, T.W.; Bennie, J.; Hopkins, J. Reducing the ecological consequences of night-time light pollution: options and developments. Journal of Applied Ecology 2012, 49(6), 1256–66. [Google Scholar] [CrossRef] [PubMed]
- Holker, F.; Bolliger, J.; Davies, T.W.; Giavi, S.; Jechow, A.; Kalinkat, G.; et al. 11 pressing research questions on how light pollution affects biodiversity. Frontiers in Ecology and Evolution 2021, 9, 767177. [Google Scholar] [CrossRef]
- Mora, C.; Tittensor, D.P.; Adl, S.; Simpson, A.G.; Worm, B. How many species are there on Earth and in the ocean? PLoS Biol. 2011, 9(8), e1001127. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Alaasam, V.J.; Kernbach, M.E.; Miller, C.R.; Ferguson, S.M. The Diversity of Photosensitivity and its Implications for Light Pollution. Integr Comp Biol. 2021, 61(3), 1170–81. [Google Scholar] [CrossRef] [PubMed]
- Bennie, J.; Davies, T.W.; Cruse, D.; Inger, R.; Gaston, K.J. Cascading effects of artificial light at night: resource-mediated control of herbivores in a grassland ecosystem. Philos Trans R Soc Lond B Biol Sci. 2015, (1667), 370. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hermas, C.I.; Litovski, I.; De Pastors, M.; Visser, M.E.; Spoelstra, K. Artificial light at night drives diel activity patterns of synanthropic pipistrelle bats and their prey. Science of the Total Environment 2024, 940, 173699. [Google Scholar] [CrossRef] [PubMed]
- Cieraad, E.; Strange, E.; Flink, M.; Schrama, M.; Spoelstra, K. Artificial light at night affects plant–herbivore interactions. Journal of Applied Ecology 2023, 60, 400–10. [Google Scholar] [CrossRef]
- Levy, O.; Fernandes de Barros Marangoni, L.; Benichou, J.I.C.; Rottier, C.; Beraud, E.; Grover, R.; et al. Artificial light at night (ALAN) alters the physiology and biochemistry of symbiotic reef building corals. Environmental Pollution 2020, 266(2), 114987. [Google Scholar] [CrossRef]
- Knop, E.; Zoller, L.; Ryser, R.; Gerpe, C.; Horler, M.; Fontaine, C. Artificial light at night as a new threat to pollination. Nature 2017, 548(7666), 206–9. [Google Scholar] [CrossRef] [PubMed]
- Giavi, S.; Fontaine, C.; Knop, E. Impact of artificial light at night on diurnal plant-pollinator interactions. Nat Commun. 2021, 12(1), 1690. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Seymoure, B.; Dell, A.; Holker, F.; Kalinkat, G. A framework for untangling the consequences of artificial light at night on species interactions. Philos Trans R Soc Lond B Biol Sci. 2023, 378(1892), 20220356. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Manfrin, A.; Singer, G.; Larsen, S.; Weiss, N.; van Grunsven, R.H.; Weiss, N.-S.; et al. Artificial Light at Night Affects Organism Flux across Ecosystem Boundaries and Drives Community Structure. Fronteirs in Envrionmental Science 2017, 5, 00061. [Google Scholar] [CrossRef]
- Holker, F.; Wurzbacher, C.; Weissenborn, C.; Monaghan, M.T.; Holzhauer, S.I.; Premke, K. Microbial diversity and community respiration in freshwater sediments influenced by artificial light at night. Philos Trans R Soc Lond B Biol Sci. 2015, (1667), 370. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Grubisic, M.; Singer, G.; Cristina Bruno, M.; van Grunsven, R.H.; Manfrin, A.; Monaghan, M.T.; et al. Artificial light at night decreases biomass and alters community composition of benthic primary producers in a sub-alpine stream. Limnology and Oceanography 2017, 62(6), 2799–810. [Google Scholar] [CrossRef]
- Sanders, D.; Kehoe, R.; Cruse, D.; van Veen, F.J.F.; Gaston, K.J. Low Levels of Artificial Light at Night Strengthen Top-Down Control in Insect Food Web. Curr Biol. 2018, 28(15), 2474–8 e3. [Google Scholar] [CrossRef] [PubMed]
- Grubisic, M.; van Grunsven, R.H.A.; Manfrin, A.; Monaghan, M.T.; Holker, F. A transition to white LED increases ecological impacts of nocturnal illumination on aquatic primary producers in a lowland agricultural drainage ditch. Environmental Pollution 2018, 240, 630–8. [Google Scholar] [CrossRef]
- Boom, M.P.; Spoelstra, K.; Biere, A.; Knop, E.; Visser, M.E. Pollination and fruit infestation under artificial light at night:light colour matters. Sci Rep. 2020, 10(1), 18389. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Parkinson, E.; Tiegs, S.D. Spectral composition of light-emitting diodes impacts aquatic and terrestrial invertebrate communities with potential implications for cross-ecosystem subsidies. Philos Trans R Soc Lond B Biol Sci. 2023, 378(1892), 20220361. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Spoelstra, K.; Teurlincx, S.; Courbois, M.; Hopkins, Z.M.; Visser, M.E.; Jones, T.M.; et al. Long-term exposure to experimental light affects the ground-dwelling invertebrate community, independent of light spectra. Philos Trans R Soc Lond B Biol Sci. 2023, 378(1892), 20220364. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Spoelstra, K.; van Grunsven, R.H.; Donners, M.; Gienapp, P.; Huigens, M.E.; Slaterus, R.; et al. Experimental illumination of natural habitat--an experimental set-up to assess the direct and indirect ecological consequences of artificial light of different spectral composition. Philos Trans R Soc Lond B Biol Sci. 2015, (1667), 370. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tao, Y.; Wolinska, J.; Holker, F.; Agha, R. Light intensity and spectral distribution affect chytrid infection of cyanobacteria via modulation of host fitness. Parasitology 2020, 147(11), 1206–15. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sanders, D.; Baker, D.J.; Cruse, D.; Bell, F.; van Veen, F.J.F.; Gaston, K.J. Spectrum of artificial light at night drives impact of a diurnal species in insect food web. Sci Total Environ. 2022, 831, 154893. [Google Scholar] [CrossRef] [PubMed]
- Longcore, T. A compendium of photopigment peak sensitivities and visual spectral response curves of terrestrial wildlife to guide design of outdoor nighttime lighting. Basic and Applied Ecology 2023, 73, 40–50. [Google Scholar] [CrossRef]
- Longcore, T.; Rodriguez, A.; Witherington, B.E.; Penniman, J.F.; Herf, L.; Herf, M. Rapid assessment of lamp spectrum to quantify ecological effects of light at night. Journal of Experimental Zoology - A 2018, 329(8-9), 511–21. [Google Scholar] [CrossRef]
- Yang, Y.; Liu, Q.; Pan, C.; Chen, J.; Xu, B.; Liu, K.; et al. Species sensitivities to artificial light at night: A phylogenetically controlled multilevel meta-analysis on melatonin suppression. Ecology Letters 2024, 27(2), e14387. [Google Scholar] [CrossRef]
- Kuhne, J.L.; van Grunsven, R.H.A.; Jechow, A.; Holker, F. Impact of Different Wavelengths of Artificial Light at Night on Phototaxis in Aquatic Insects. Integrative and Comparative Biology 2021, 61(3), 1182–90. [Google Scholar] [CrossRef]
- Seymoure, B.M.; Linares, C.; White, J. Connecting spectral radiometry of anthropogenic light.
- sources to the visual ecology of organisms. Journal of Zoology 2019, 308(2), 93–110. [CrossRef]
- Davies, T.W.; Bennie, J.; Inger, R.; de Ibarra, N.H.; Gaston, K.J. Artificial light pollution: are shifting spectral signatures changing the balance of species interactions? Glob Chang Biol. 2013, 19(5), 1417–23. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sanders, D.; Frago, E.; Kehoe, R.; Patterson, C.; Gaston, K.J. A meta-analysis of biological impacts of artificial light at night. Nat Ecol Evol. 2021, 5(1), 74–81. [Google Scholar] [CrossRef] [PubMed]
- Meah, R.J.; Sumner-Rooney, L.; Venables, S.V.; Lloyd-How, S.T.; Massy, R.; Hawkes, W.L.S.; et al. Light pollution creates multiple threats to the movement ecology of nocturnal arthropod taxa. Curr Biol. 2026, 36(2), 541–8 e4. [Google Scholar] [CrossRef] [PubMed]
- Meszaros, A.; Kriska, G.; Egri, A. Wavelength-specific negatively phototactic responses of the burrowing mayfly larvae Ephoron virgo. J Exp Biol. 2024, (10) Epub 20240521, 227. [Google Scholar] [CrossRef] [PubMed]
- Przyrembel, C.; Keller, B.; Neumeyer, C. Trichromatic color vision in the salamander (Salamandra salamandra). J Comp Physiol A 1995, 176, 575–86. [Google Scholar] [CrossRef]
- Witherington, B.E.; Bjorndal, K.A. Influences of Wavelength and Intensity on Hatchling Sea Turtle Phototaxis: Implications for Sea-Finding Behavior. Copeia 1991, 1991(4), 1060–9. [Google Scholar] [CrossRef]
- Zurl, M.; Poehn, B.; Rieger, D.; Krishnan, S.; Rokvic, D.; Veedin Rajan, V.B.; et al. Two light sensors decode moonlight versus sunlight to adjust a plastic circadian/circalunidian clock to moon phase. Proc Natl Acad Sci U S A 2022, 119(22), e2115725119. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yoshii, T.; Hermann-Luibl, C.; Helfrich-Forster, C. Circadian light-input pathways in Drosophila. Commun Integr Biol. 2016, 9(1), e1102805. [Google Scholar] [CrossRef] [PubMed Central]
- Wilby, D.; Roberts, N.W. Optical influence of oil droplets on cone photoreceptor sensitivity. J Exp Biol. 2017, 220 Pt 11, 1997–2004. [Google Scholar] [CrossRef] [PubMed Central]
- Cronin, T.W. Visual Ecology. The Senses: A Comprehensive Reference 2008, 211–45. [Google Scholar]
- Belusic, G.; Ilic, M.; Meglic, A.; Pirih, P. Red-green opponency in the long visual fibre photoreceptors of brushfoot butterflies (Nymphalidae). Proc Biol Sci. 2021, 288(1961), 20211560. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jokela-Maatta, M.; Viljanen, M.; Nevela, N.; Donner, K.; Bronmark, C. Photoreceptors and eyes of pikeperch Sander lucioperca, pike Esox lucius, perch Perca fluviatilis and roach Rutilus rutilus from a clear and a brown lake. Journal of Fish Biology 2018, 95(1), 200–13. [Google Scholar] [CrossRef]
- Stavenga, D.G.; Arikawa, K. Photoreceptor spectral sensitivities of the Small White butterfly Pieris rapae crucivora interpreted with optical modeling. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2011, 197(4), 373–85. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Palacios, A.G.; Varela, F.J.; Srivastava, R.; Goldsmith, T.H. Spectral sensitivity of cones in the goldfish, Carassius auratus. Vision Res. 1998, 38(14), 2135–46. [Google Scholar] [CrossRef] [PubMed]
- Deprato, A.; Haldar, P.; Navarro, J.F.; Harding, B.N.; Lacy, P.; Maidstone, R.; et al. Associations between light at night and mental health: A systematic review and meta-analysis. Sci Total Environ. 2025, 974, 179188. [Google Scholar] [CrossRef] [PubMed]
- Palomar-Cros, A.; Deprato, A.; Papantoniou, K.; Straif, K.; Lacy, P.; Maidstone, R.; et al. Indoor and outdoor artificial light-at-night (ALAN) and cancer risk: A systematic review and meta-analysis of multiple cancer sites and with a critical appraisal of exposure assessment. Sci Total Environ. 2024, 955, 177059. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Alsabawi, Y.; El-Serag, H.B.; Thrift, A.P. Exposure to Light at Night and Risk of Cancer: A Systematic Review, Meta-Analysis, and Data Synthesis. Cancers (Basel) 2024, (15) Epub 20240726, 16. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Palomar-Cros, A.; Espinosa, A.; Bara, S.; Sanchez, A.; Valentin, A.; Cirach, M.; et al. Outdoor artificial light-at-night and cardiometabolic disease risk: an urban perspective from the Catalan GCAT cohort study. Am J Epidemiol 2024, Epub 20240819. [Google Scholar] [CrossRef] [PubMed]
- Fan, M.; Yuan, J.; Zhang, S.; Fu, Q.; Lu, D.; Wang, Q.; et al. Association between outdoor artificial light at night and metabolic diseases in middle-aged to older adults-the CHARLS survey. Front Public Health 2025, 13, 1515597. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Obayashi, K.; Saeki, K.; Iwamoto, J.; Okamoto, N.; Tomioka, K.; Nezu, S.; et al. Exposure to light at night, nocturnal urinary melatonin excretion, and obesity/dyslipidemia in the elderly: a cross-sectional analysis of the HEIJO-KYO study. J Clin Endocrinol Metab. 2013, 98(1), 337–44. [Google Scholar] [CrossRef] [PubMed]
- Obayashi, K.; Tai, Y.; Yamagami, Y.; Saeki, K. Associations between indoor light pollution and unhealthy outcomes in 2,947 adults: Cross-sectional analysis in the HEIJO-KYO cohort. Environ Res. 2022, 215 Pt 2, 114350. [Google Scholar] [CrossRef] [PubMed]
- Obayashi, K.; Yamagami, Y.; Kurumatani, N.; Saeki, K. Bedroom lighting environment and incident diabetes mellitus: a longitudinal study of the HEIJO-KYO cohort. Sleep Med. 2020, 65, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Obayashi, K.; Yamagami, Y.; Tatsumi, S.; Kurumatani, N.; Saeki, K. Indoor light pollution and progression of carotid atherosclerosis: A longitudinal study of the HEIJO-KYO cohort. Environ Int. 2019, 133 Pt B, 105184. [Google Scholar] [CrossRef] [PubMed]
- Burns, A.C.; Windred, D.P.; Rutter, M.K.; Olivier, P.; Vetter, C.; Saxena, R.; et al. Day and night light exposure are associated with psychiatric disorders: an objective light study. Nature Mental Health 2023, 1(11), 853–62. [Google Scholar] [CrossRef]
- Sakhvidi, M.J.Z.; Mehrparvar, A.; Zare, F.; Rahmanian, M.; Yang, J.; Ebrahimi, A.S.; et al. Association between exposure to outdoor light at night and sleep habits in Shahedieh Cohort Study: A cross-sectional analysis. Environmental Pollution 2025, 385, 127144. [Google Scholar] [CrossRef]
- Windred, D.P.; Burns, A.C.; Lane, J.M.; Olivier, P.; Rutter, M.K.; Saxena, R.; et al. Brighter nights and darker days predict higher mortality risk: A prospective analysis of personal light exposure in. Proc Natl Acad Sci U S A 2024, 121(43), e2405924121. [Google Scholar] [CrossRef] [PubMed Central]
- Sun, S.; Cao, W.; Ge, Y.; Ran, J.; Sun, F.; Zeng, Q.; et al. Outdoor light at night and risk of coronary heart disease among older adults: a prospective cohort study. Eur Heart J. 2021, 42(8), 822–30. [Google Scholar] [CrossRef] [PubMed]
- Lunn, R.M.; Blask, D.E.; Coogan, A.N.; Figueiro, M.G.; Gorman, M.R.; Hall, J.E.; et al. Health consequences of electric lighting practices in the modern world: A report on the National Toxicology Program’s workshop on shift work at night, artificial light at night, and circadian disruption. Sci Total Environ. 2017, 607-608, 1073–84. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mason, I.C.; Boubekri, M.; Figueiro, M.G.; Hasler, B.P.; Hattar, S.; Hill, S.M.; et al. Circadian Health and Light: A Report on the National Heart, Lung, and Blood Institute’s Workshop. J Biol Rhythms 2018, 33(5), 451–7. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Stevens, R.G.; Brainard, G.C.; Blask, D.E.; Lockley, S.W.; Motta, M.E. Breast cancer and circadian disruption from electric lighting in the modern world. CA Cancer J Clin. 2014, 64(3), 207–18. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chellappa, S.L.; Vujovic, N.; Williams, J.S.; Scheer, F. Impact of Circadian Disruption on Cardiovascular Function and Disease. Trends Endocrinol Metab. 2019, 30(10), 767–79. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Parameswaran, G.; Ray, D.W. Sleep, circadian rhythms, and type 2 diabetes mellitus. Clin Endocrinol (Oxf) 2021. [Google Scholar] [CrossRef] [PubMed]
- Brown, T.M. Melanopic illuminance defines the magnitude of human circadian light responses under a wide range of conditions. J Pineal Res. 2020, e12655. [Google Scholar] [CrossRef] [PubMed]
- Brown, T.M.; Brainard, G.C.; Cajochen, C.; Czeisler, C.A.; Hanifin, J.P.; Lockley, S.W.; et al. Recommendations for daytime, evening, and nighttime indoor light exposure to best support physiology, sleep, and wakefulness in healthy adults. PLoS Biol. 2022, 20(3), e3001571. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Do, M.T.H. Melanopsin and the Intrinsically Photosensitive Retinal Ganglion Cells: Biophysics to Behavior. Neuron. 2019, 104(2), 205–26. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.; Brown, T. Direct effects of the light environment on daily neuroendocrine control. J Endocrinol. 2019, 243, R1–R18. [Google Scholar] [CrossRef] [PubMed]
- Lucas, R.J.; Peirson, S.N.; Berson, D.M.; Brown, T.M.; Cooper, H.M.; Czeisler, C.A.; et al. Measuring and using light in the melanopsin age. Trends in neurosciences 2014, 37(1), 1–9. [Google Scholar] [CrossRef] [PubMed]
- CIE. S 026/E CIE System for Metrology of Optical Radiation for ipRGC-Influenced Responses to Light. CIE Central Bureau: Vienna, 2018.
- Briggs, W.R. Physiology of plant responses to artifical light. In Ecological Consequences of Artificial Light at Night; Rich, C., Longcore, T., Eds.; Island Press: Washington, Covelo, London, 2006; pp. 389–412. [Google Scholar]
- Heinen, R. A spotlight on the phytobiome: plant-mediated interactions in an illuminated world. Basic and Applied Ecology 2021, 57, 146–58. [Google Scholar] [CrossRef]
- Maggi, E.; Bongiorni, L.; Fontanini, D.; Capocchi, A.; Dal Bello, M.; Giacomelli, A.; et al. Artifical light at night erases positive interactions across trophic levels. Functional Ecology 2019, 34(3), 694–706. [Google Scholar] [CrossRef]
- Bennie, J.; Davis, T.W.; Cruse, D.; Gaston, K.J. Ecological effects of artificial light at night on wild plants. Journal of Ecology 2016, 104, 611. [Google Scholar] [CrossRef]
- Bennie, J.; Cruse, T.W.; Bell, F.; Gaston, K.J. Artificial light at night alters grassland vegetation species composition and phenology. Journal of Applied Ecology 2018, 55, 442–50. [Google Scholar] [CrossRef]
- Ffrench-Constant, R.H.; Somers-Yeates, R.; Bennie, J.; Economou, T.; Hodgson, D.; Spalding, A.; et al. Light pollution is associated with earlier tree budburst across the United Kingdom. Proc Biol Sci. 2016, (1833), 283. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Alsanius, B.W.; Bergstrand, K.-J.; Hartmann, R.; Gharaie, S.; Wohanka, W.; Dorais, M.; et al. Ornamental flowers in new light: Artificial lighting shapes the microbial phyllosphere community structure of greenhouse grown sunflowers (Helianthus annus L.). Scientia Horticulturae 2017, 216, 234–47. [Google Scholar] [CrossRef]
- Costa, J.M.; Candia, A.N.; Sellaro, R. Open All Night Long: the dark side of stomatal control. Journal of Experimental Botany 2014, 167, 289–94. [Google Scholar] [CrossRef]
- Graf, A.; Smith, A.M. Starch and the clock: the dark side of plant productivity. Trends in Plant Science 2011, 16, 169–75. [Google Scholar] [CrossRef]
- Haro, S.; Bohorquez, J.; Lara, M.; Garcia-Robledo, E.; Gonzalez, C.J.; Crespo, J.M.; et al. Diel patterns of microphytobenthic primary production in intertidal sediments: the role of photoperiod on the vertical migration circadian rhythm. Sci Rep. 2019, 9(1), 13376. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jabbur, M.L.; Bratton, B.P.; Johnson, C.H. Bacteria can anticipate the seasons: Photoperiodism in cyanobacteria. Science 2024, 385(6713), 1105–11. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wang, F.; Han, T.; Jeffrey Chen, Z. Circadian and photoperiodic regulation of the vegetative to reproductive transition in plants. Commun Biol. 2024, 7(1), 579. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Paik, I.; Huq, E. Plant photoreceptors: Multi-functional sensory proteins and their signaling networks. Semin Cell Dev Biol. 2019, 92, 114–21. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yu, Z.; Fischer, R. Light sensing and responses in fungi. Nat Rev Microbiol 2019, 17(1), 25–36. [Google Scholar] [CrossRef] [PubMed]
- Fraikin, G.Y.; Strakhovskaya, M.G.; Belenikina, N.S.; Rubin, A.B. Bacterial photosensory proteins: Regulatory functions and optogenetic applications. Microbiology 2015, 84, 461–72. [Google Scholar] [CrossRef]
- Casal, J.J.; Candia, A.N.; Sellaro, R. Light perception and signalling by phytochrome. Journal of Experimental Botany 2014, 65, 2835–45. [Google Scholar] [CrossRef]
- Karniol, B.; Vierstra, R.D. The pair of bacteriophytochromes from Agrobacterium tumefaciens are histidine kinases with opposing photobiological properties. Proc Natl Acad Sci U S A 2003, 100(5), 2807–12. [Google Scholar] [CrossRef] [PubMed Central]
- Rockwell, N.C.; Duanmu, D.; Martin, S.S.; Bachy, C.; Price, D.C.; Bhattacharya, D.; et al. Eukaryotic algal phytochromes span the visible spectrum. Proc Natl Acad Sci U S A 2014, 111(10), 3871–6. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ernst, O.P.; Lodowski, D.T.; Elstner, M.; Hegemann, P.; Brown, L.S.; Kandori, H. Microbial and animal rhodopsins: structures, functions, and molecular mechanisms. Chem Rev. 2014, 114(1), 126–63. [Google Scholar] [CrossRef] [PubMed Central]
- Broser, M.; Busse, W.; Spreen, A.; Reh, M.; Bernal Sierra, Y.A.; Hwang, S.; et al. Diversity of rhodopsin cyclases in zoospore-forming fungi. Proc Natl Acad Sci U S A 2023, 120(44), e2310600120. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mobley, C.D. Light and water: radiative transfer in natural waters 1994.
- Stomp, M.; Huisman, J.; De Jongh, F.; Veraart, A.J.; Gerla, D.; Rijkeboer, M.; et al. Adaptive divergence in pigment composition promotes phytoplankton biodiversity. Nature 2004, 432(7013), 104–7. [Google Scholar] [CrossRef] [PubMed]
- Holker, F.; Jechow, A.; Schroer, S.; Tockner, K.; Gessner, M.O. Light pollution of freshwater ecosystems: principles, ecological impacts and remedies. Philos Trans R Soc Lond B Biol Sci. 2023, 378(1892), 20220360. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Davies, T.W.; Smyth, T. Darkening of the global ocean. Global Change Biology 2025, 31(5), e70227. [Google Scholar] [CrossRef]
- Loew, E.R.; Lythgoe, J.N. The ecology of colour vision. Endeavour 1985, 9(4), 170–4. [Google Scholar] [CrossRef] [PubMed]
- Wyneken, J.; Salmon, M. Linking Ecology, Morphology, and Behavior to Conservation: Lessons Learned from Studies of Sea Turtles. Integr Comp Biol. 2020, 60(2), 440–55. [Google Scholar] [CrossRef] [PubMed]
- Grubisic, M.; Haim, A.; Bhusal, P.; D’ominoni, D.M.; Gabriel, K.M.; Jechow, A.; et al. Light pollution, circadian photoreception, and melatonin in vertebrates. Sustainability 2019, 11(22), 6400. [Google Scholar] [CrossRef]
- Egri, A.; Meszaros, A.; Kriska, G. Spectral sensitivity transition in the compound eyes of a twilight-swarming mayfly and its visual ecological implications. Proc Biol Sci. 2022, 289(1973), 20220318. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chou, A.; Lin, C.; Cronin, T.W. Visual metamorphoses in insects and malacostracans: transitions between an aquatic and terrestrial life. Arthropod Structure and Development 2020, 100974. [Google Scholar] [CrossRef]
- Kutta, R.J.; Archipowa, N.; Johannissen, L.O.; Jones, A.R.; Scrutton, N.S. Vertebrate Cryptochromes are Vestigial Flavoproteins. Sci Rep. 2017, 7, 44906. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Govardovskii, V.I.; Fyhrquist, N.; Reuter, T.; Kuzmin, D.G.; Donner, K. In search of the visual pigment template. Vis Neurosci. 2000, 17(4), 509–28. [Google Scholar] [CrossRef] [PubMed]
- Christie, J.M.; Arvai, A.S.; Baxter, K.J.; Heilmann, M.; Pratt, A.J.; O’Hara, A.; et al. Plant UVR8 photoreceptor senses UV-B by tryptophan-mediated disruption of cross-dimer salt bridges. Science 2012, 335(6075), 1492–6. [Google Scholar] [CrossRef] [PubMed Central]
- Butler, W.L.; Hendricks, S.B.; Siegelman, H.W. Action spectra of phytochrome in vitro. Photochemistry and Photobiology 1964, 3, 521–8. [Google Scholar] [CrossRef]
- Banerjee, R.; Schleicher, E.; Meier, S.; Viana, R.M.; Pokorny, R.; Ahmad, M.; et al. The signaling state of Arabidopsis cryptochrome 2 contains flavin semiquinone. J Biol Chem. 2007, 282(20), 14916–22. [Google Scholar] [CrossRef] [PubMed]
- Bureau, C.C., Ed.; Colorimetry 4th Edition 2018, 015.
- Richtlijn Lichthinder [Internet]. 2020. Available online: https://www.nsvv.nl/publicaties/richtlijn-lichthinder-2020/.
- IES. 2025. Available online: https://www.ies.org/advocacy/light-at-night/.
- DarkSkyInternational. Solutions to light pollution. 2023. Available online: https://darksky.org/resources/what-is-light-pollution/light-pollution-solutions/.
- McDermott, A. Light pollution is fixable. Can researchers and policymakers work together to dim the lights? Proc Natl Acad Sci U S A 2023, 120(27), e2309539120. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Owens, A.C.; Pocock, M.J.; Seymoure, B.M. Current evidence in support of insect-friendly lighting practice. Current Opinion in Insect Science 2024, 66, 101276. [Google Scholar] [CrossRef] [PubMed]
- Czarnecka, M.; Grubisic, M.; Pilotto, F.; Jechow, A.; Holker, F. Colours of the Night: Spectrum-Specific Impacts of Light Pollution on Biota. Glob Chang Biol. 2025, 31(10), e70569. [Google Scholar] [CrossRef] [PubMed]
- Aube, M.; Roby, J.; Kocifaj, M. Evaluating potential spectral impacts of various artificial lights on melatonin suppression, photosynthesis, and star visibility. PLoS One 2013, 8(7), e67798. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jerlov, N.G. Classification of sea water in terms of quanta irradiance. ICES Journal of Marine Science 1977, 37(3), 281–7. [Google Scholar] [CrossRef]
- Aube, M. Physical behaviour of anthropogenic light propagation into the nocturnal environment. Philos Trans R Soc Lond B Biol Sci 2015, (1667), 370. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kelber, A.; Osorio, D. From spectral information to animal colour vision: experiments and concepts. Proc Biol Sci. 2010, 277(1688), 1617–25. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Charvalakis, G.A.; Stavenga, D.G.; Visser, M.E.; Spoelstra, K.; Hut, R.A. Intensity and colour of artificial light at night affect insect attraction in a taxon-dependent manner. Insect conservation and diversity 2025, 1–10. [Google Scholar] [CrossRef]
- IES. Spectral Calculator. 2025. Available online: https://www.ies.org/standards/standards-toolbox/tm-30-spectral-calculator/.
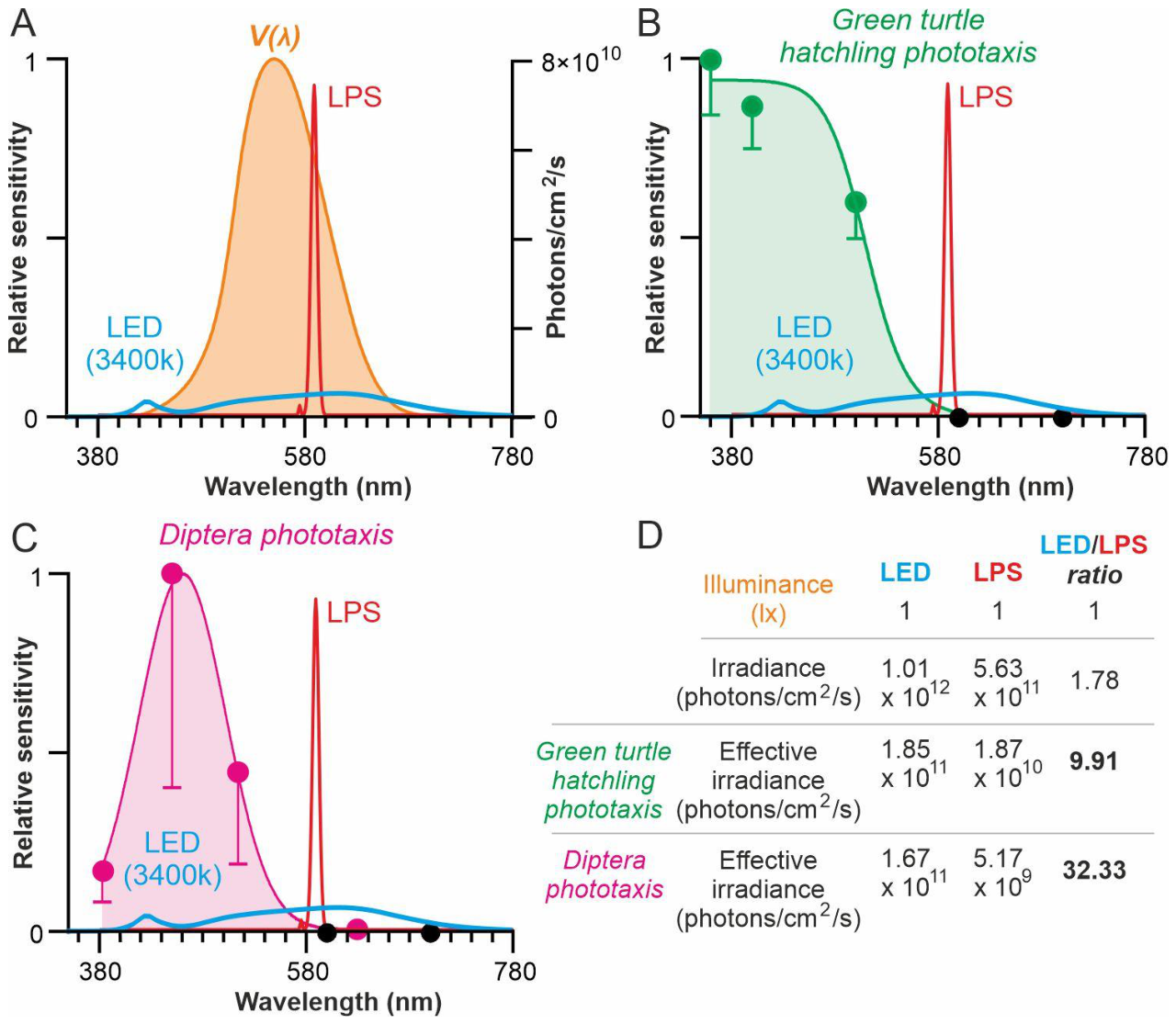
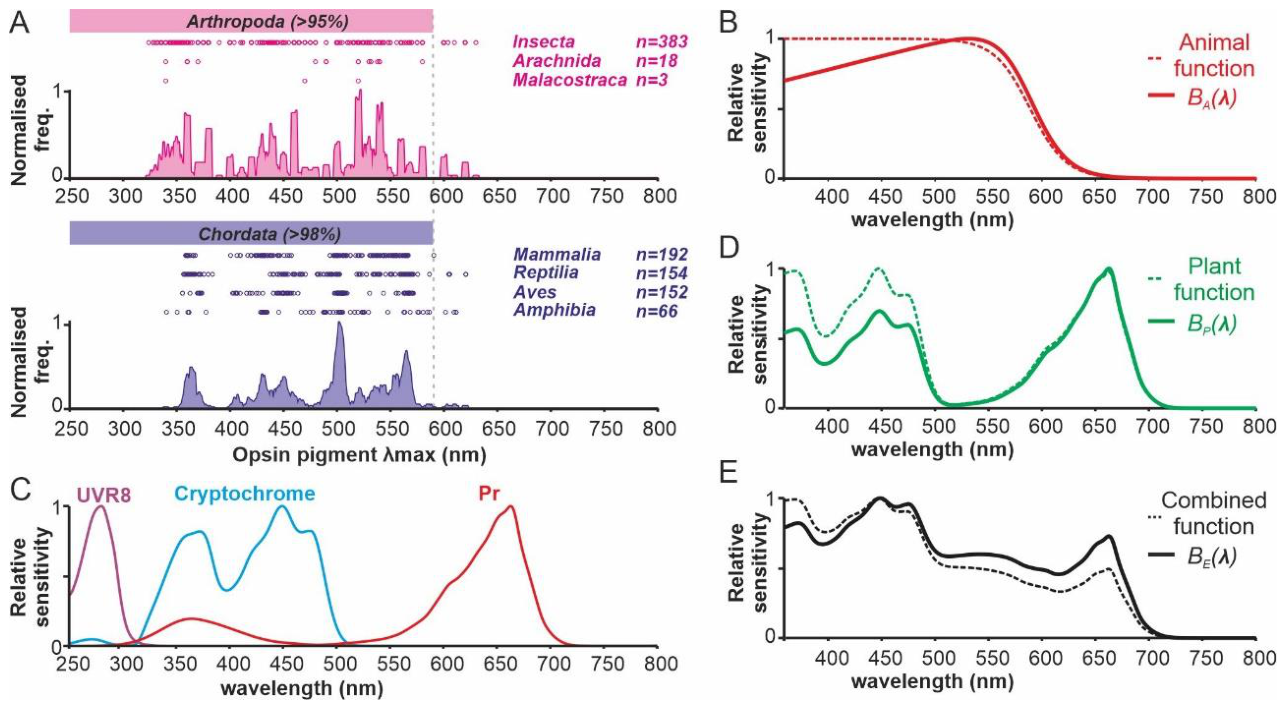
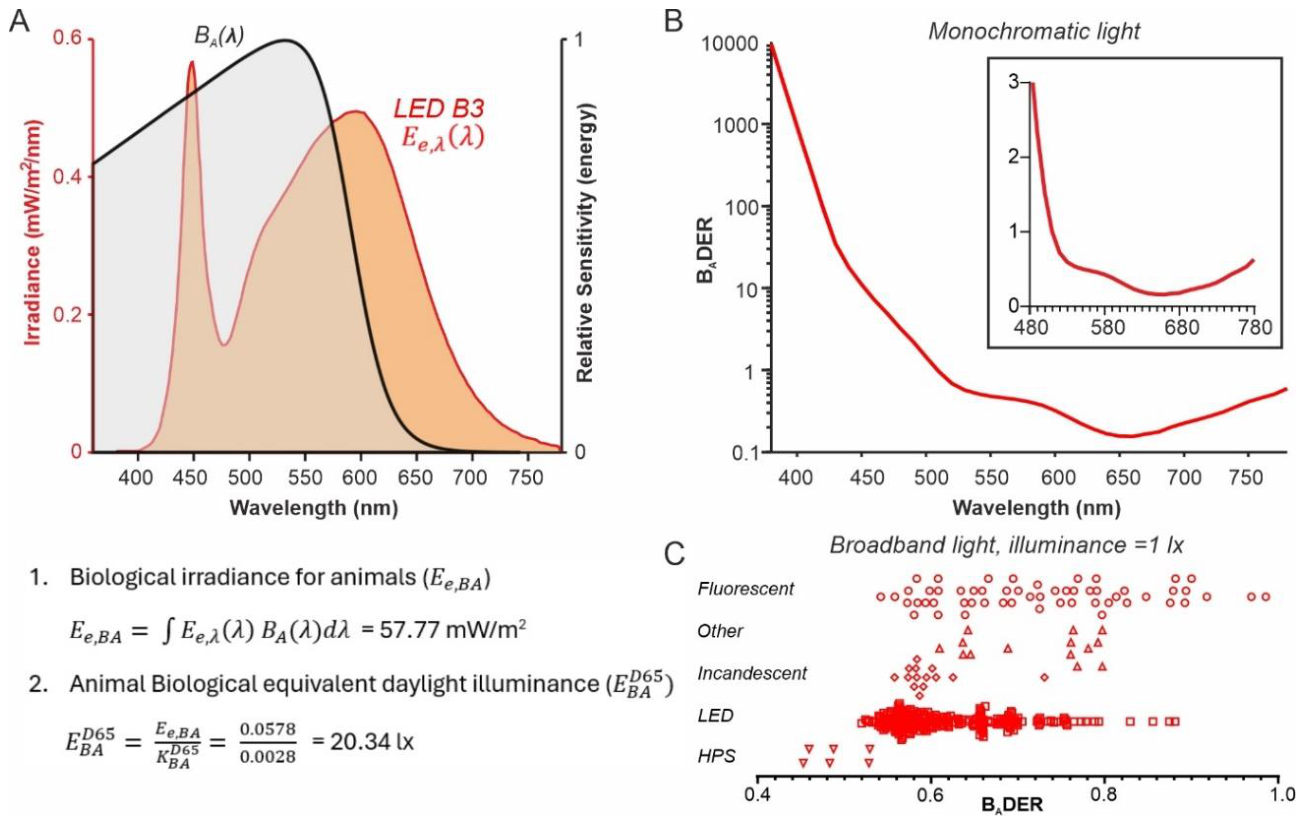
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



