Submitted:
17 April 2026
Posted:
20 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Patients’ Selection
2.2. Surgical Technique and Radiotherapy Procedure
2.3. Evaluation Parameter and Statistical Analysis
3. Results
3.1. Quantitative Analysis
4. Discussion
Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ADM | Acellular Dermal Matrix; |
| CT | Computed Tomography; |
| CTV | Clinical Target Volume; |
| DIEP | Deep Inferior Epigastric Perforator; |
| DTI | Direct-to-Implant; |
| HU | Hounsfield Unit; |
| IMRT | Intensity-Modulated Radiotherapy; |
| MRI | Magnetic Resonance Imaging; |
| PMRT | Postmastectomy Radiotherapy; |
| RFID | Radiofrequency Identification; |
| RT | Radiotherapy. |
References
- Cordeiro, P. G.; Jazayeri, L. Two-stage implant-based breast reconstruction: An evolution of the conceptual and technical approach over a two-decade period. Plast. Reconstr. Surg. 2016, vol. 138(no. 1), 1–11. [Google Scholar] [CrossRef]
- Peng, J. L.; et al. Tissue Assignment and Dosimetric Influence for Breast Radiotherapy With Metallic Port in Breast Tissue Expander Using Acurosxb Algorithms. Int. J. Radiat. Oncol. 2021, vol. 111(no. 3), e144–e145. [Google Scholar] [CrossRef]
- Akdeniz, Y.; Yegingil, I.; Yegingil, Z. Effects of metal implants and a metal artifact reduction tool on calculation accuracy of AAA and Acuros XB algorithms in small fields. Med. Phys. 2019, vol. 46(no. 11), 5326–5335. [Google Scholar] [CrossRef]
- Liang, Y.; Xu, H.; Tang, W.; Du, X. The impact of metal implants on the dose and clinical outcome of radiotherapy (Review). Mol. Clin. Oncol. 2024, vol. 21(no. 4), 1–10. [Google Scholar] [CrossRef]
- Bayasgalan, M.; Munhoz, A. M.; Shellock, F. G. Breast tissue expander with radiofrequency identification port: Assessment of MRI issues. Am. J. Roentgenol. 2020, vol. 215(no. 1), 159–164. [Google Scholar] [CrossRef]
- Schiaffino, S.; et al. MRI-Conditional Breast Tissue Expander: First In-Human Multi-Case Assessment of MRI-Related Complications and Image Quality. J. Clin. Med. 2023, vol. 12(no. 13). [Google Scholar] [CrossRef]
- Vinsensia, M. Incidence and Risk Assessment of Capsular Contracture in Breast Cancer Patients following Post-Mastectomy Radiotherapy and Implant-Based Reconstruction. Cancers (Basel) 2024, vol. 16(no. 2). [Google Scholar] [CrossRef]
- Rong, Y. Advancements in Radiation Therapy Treatment Workflows for Precision Medicine: A Review and Forward Looking. Int. J. Radiat. Oncol. 2025, vol. 122(no. 4), 1022–1034. [Google Scholar] [CrossRef]
- Yoon, J.; Xie, Y.; Heins, D.; Zhang, R. Modeling of the metallic port in breast tissue expanders for photon radiotherapy. J. Appl. Clin. Med. Phys. 2018, vol. 19(no. 3), 205–214. [Google Scholar] [CrossRef]
- Hwang, N. H.; Kim, M.; Lee, N. K.; Lee, S.; Hwang, J. Dosimetric Effect of Injection Ports in Tissue Expanders on Post-Mastectomy Volumetric Modulated Arc Therapy (VMAT) Planning for Left-Sided Breast Cancer. Appl. Sci. 2022, vol. 12(no. 13). [Google Scholar] [CrossRef]
- Lee, H. Y.; et al. Critical Factors of Dose Distribution in Breast Cancer Tomotherapy With Metallic Port Breast Tissue Expander: Image Correction, Delivery Mode, and Volume Impact. Technol. Cancer Res. Treat. 2022, vol. 21, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, N.; et al. Determination of the appropriate physical density of internal metallic ports in temporary tissue expanders for the treatment planning of post-mastectomy radiation therapy. J. Radiat. Res. 2018, vol. 59(no. 2), 190–197. [Google Scholar] [CrossRef] [PubMed]
- Tran, E. T.; Newbury, P.; Newpower, M.; Ortega, H.; Malouff, T. D.; Henson, C. Implementation of Proton Radiation Treatment Exclusion Volume Because of Inconsistent Location of Breast Expander Titanium Port. Adv. Radiat. Oncol. 2025, vol. 10(no. 12), 101876. [Google Scholar] [CrossRef] [PubMed]
- Parmeshwar, N.; Knox, J. A.; Piper, M. L. Evaluation of Pre-Pectoral Direct-to-Implant Breast Reconstruction with Post-Mastectomy Radiation: A Systematic Review and Meta-Analysis. J. Clin. Med. vol. 14(no. 14), 1–11, 2025. [CrossRef] [PubMed]
- Zhang, X.; Ning, S.; Zhang, Y. Complications After Prepectoral Versus Subpectoral Breast Reconstruction in Patients Receiving Postmastectomy Radiation Therapy: A Systematic Review and Meta-Analysis. Aesthetic Plast. Surg. 2024, vol. 48(no. 21), 4421–4429. [Google Scholar] [CrossRef] [PubMed]
- Clemens, M. W.; Kronowitz, S. J. Current perspectives on radiation therapy in autologous and prosthetic breast reconstruction. Gland Surg. 2015, vol. 4(no. 3), 222–31. [Google Scholar] [CrossRef]
- Emanuele Lisa, V.; et al. The Effect of Adjuvant Radiotherapy on One- and Two-Stage Prosthetic Breast Reconstruction and on Autologous Reconstruction: A Multicenter Italian Study among 18 Senonetwork Breast Centres. Breast J. 2023, vol. 2023. [Google Scholar] [CrossRef] [PubMed]
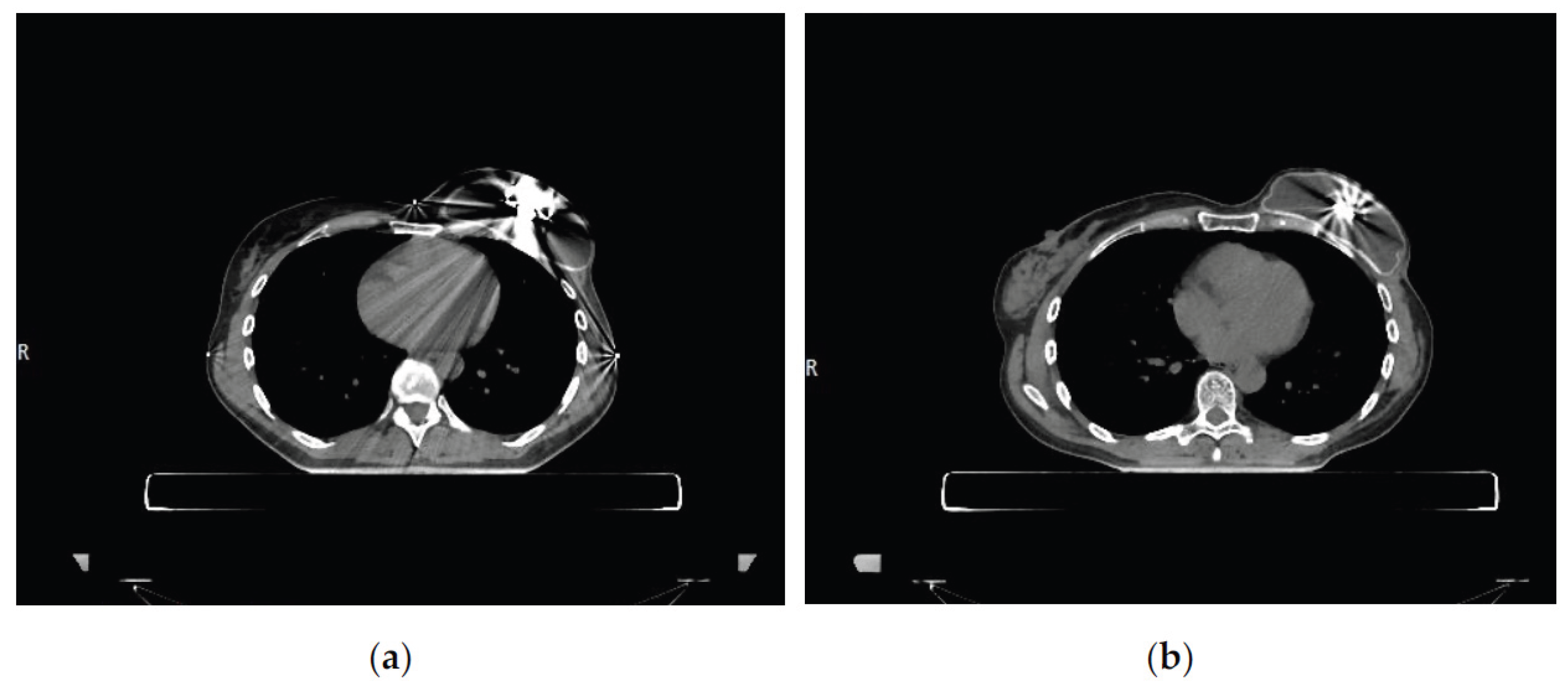
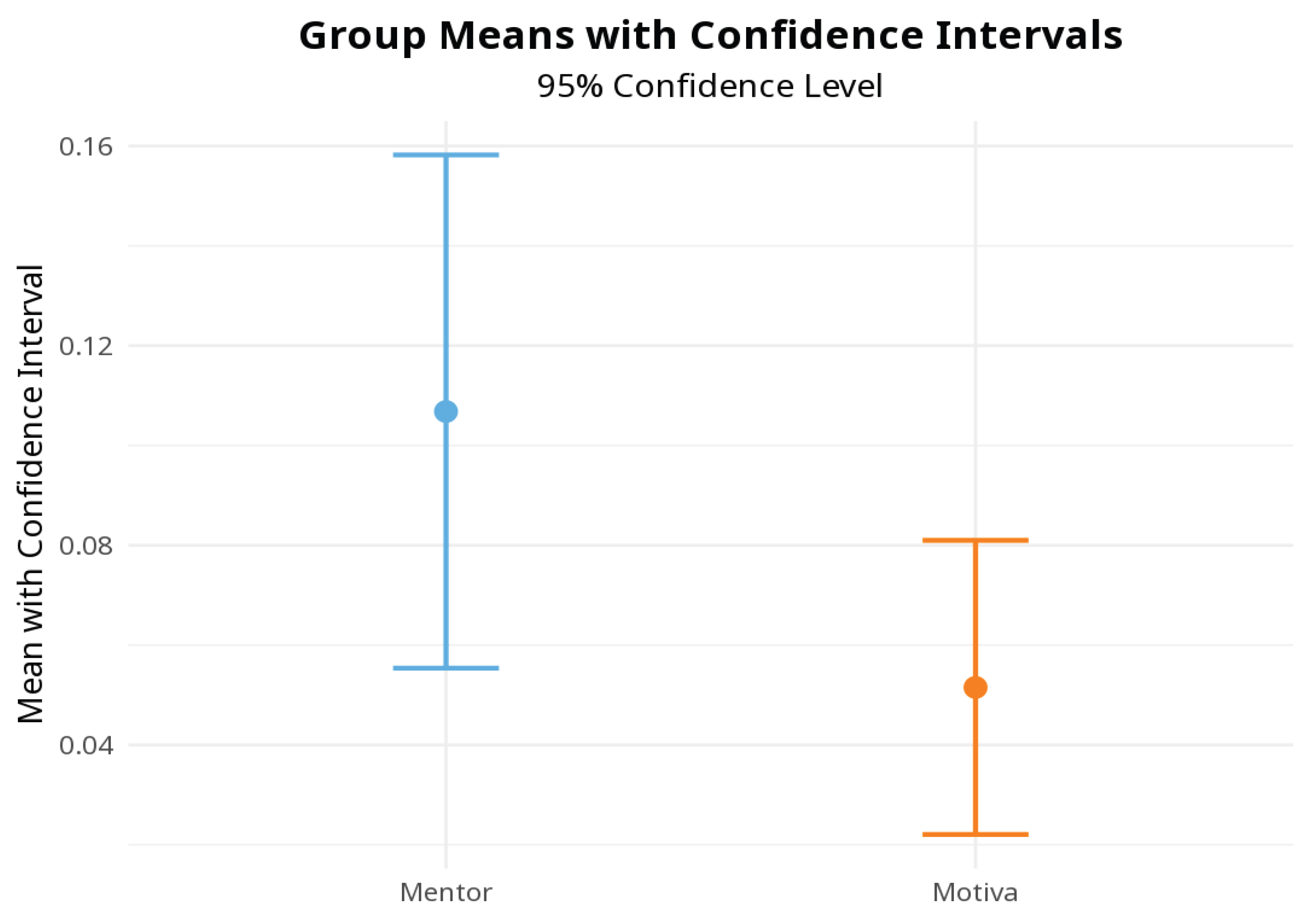
| ID pazient | Type | Artifact expander (cc) | CTV (cc) | Artifact expander / CTV |
|---|---|---|---|---|
| 1 | Motiva | 23,4 | 910,9 | 0,0257 |
| 2 | Motiva | 23,3 | 602,3 | 0,0387 |
| 3 | Motiva | 23,8 | 553,0 | 0,0430 |
| 4 | Motiva | 29,7 | 351,9 | 0,0844 |
| 5 | Motiva | 4,4 | 928,8 | 0,0047 |
| 6 | Motiva | 38,3 | 440,3 | 0,0870 |
| 7 | Motiva | 28,3 | 367,1 | 0,0771 |
| 8 | Mentor | 77,2 | 484,1 | 0,1595 |
| 9 | Mentor | 104,3 | 811,7 | 0,1285 |
| 10 | Mentor | 120,4 | 665,1 | 0,1810 |
| 11 | Mentor | 50,1 | 580,0 | 0,0864 |
| 12 | Mentor | 13,8 | 387,7 | 0,0356 |
| 13 | Mentor | 44,4 | 386,5 | 0,1149 |
| 14 | Mentor | 39,1 | 938,5 | 0,0417 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




