Submitted:
17 April 2026
Posted:
20 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction

1.1.Mass Spectrometry-Based Analysis

1.2. CAR-T Cell Therapy
2. Discussion
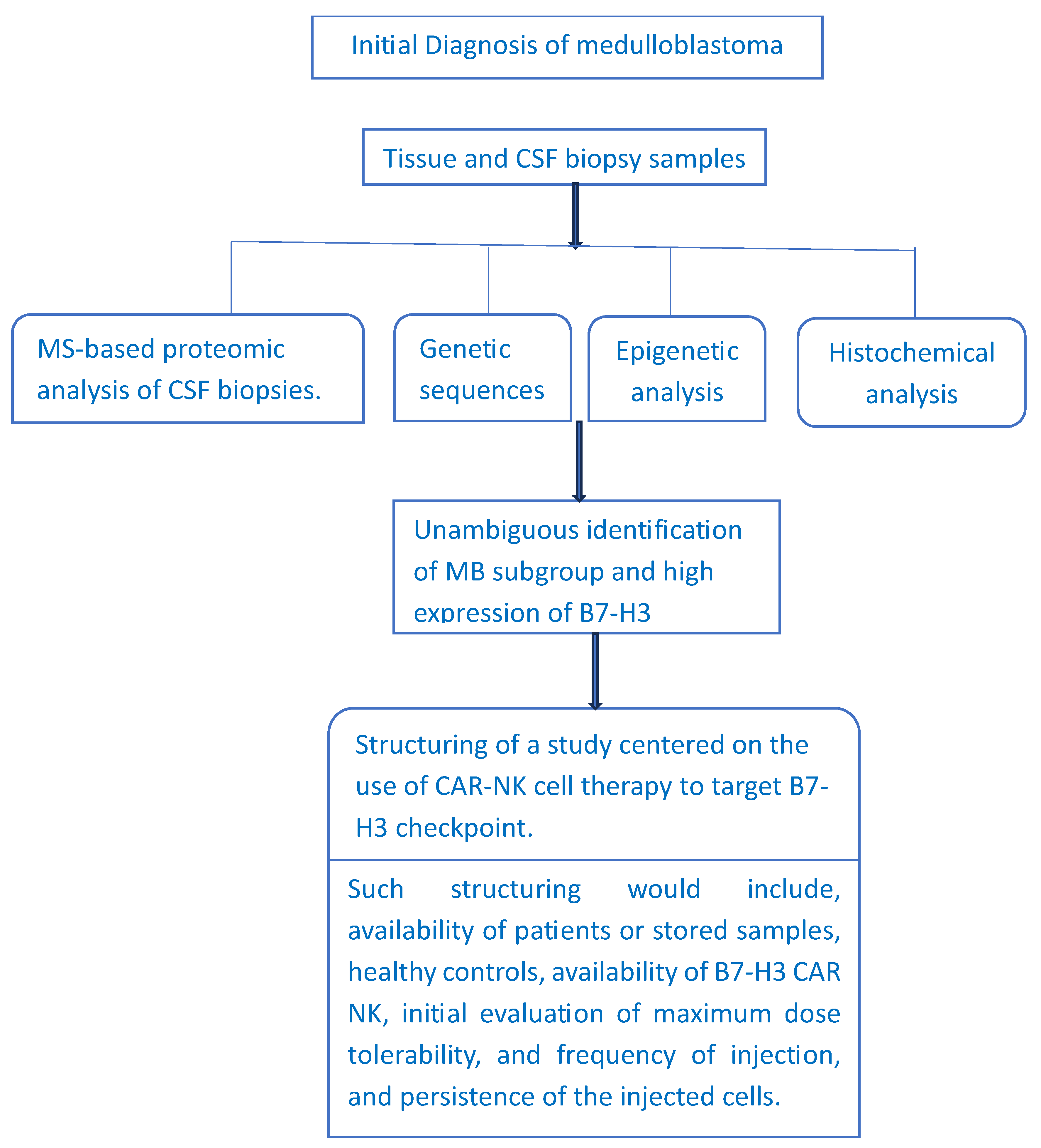
2.1. Mass Spectrometry/Liquid Biopsies Investigation of Medulloblastoma
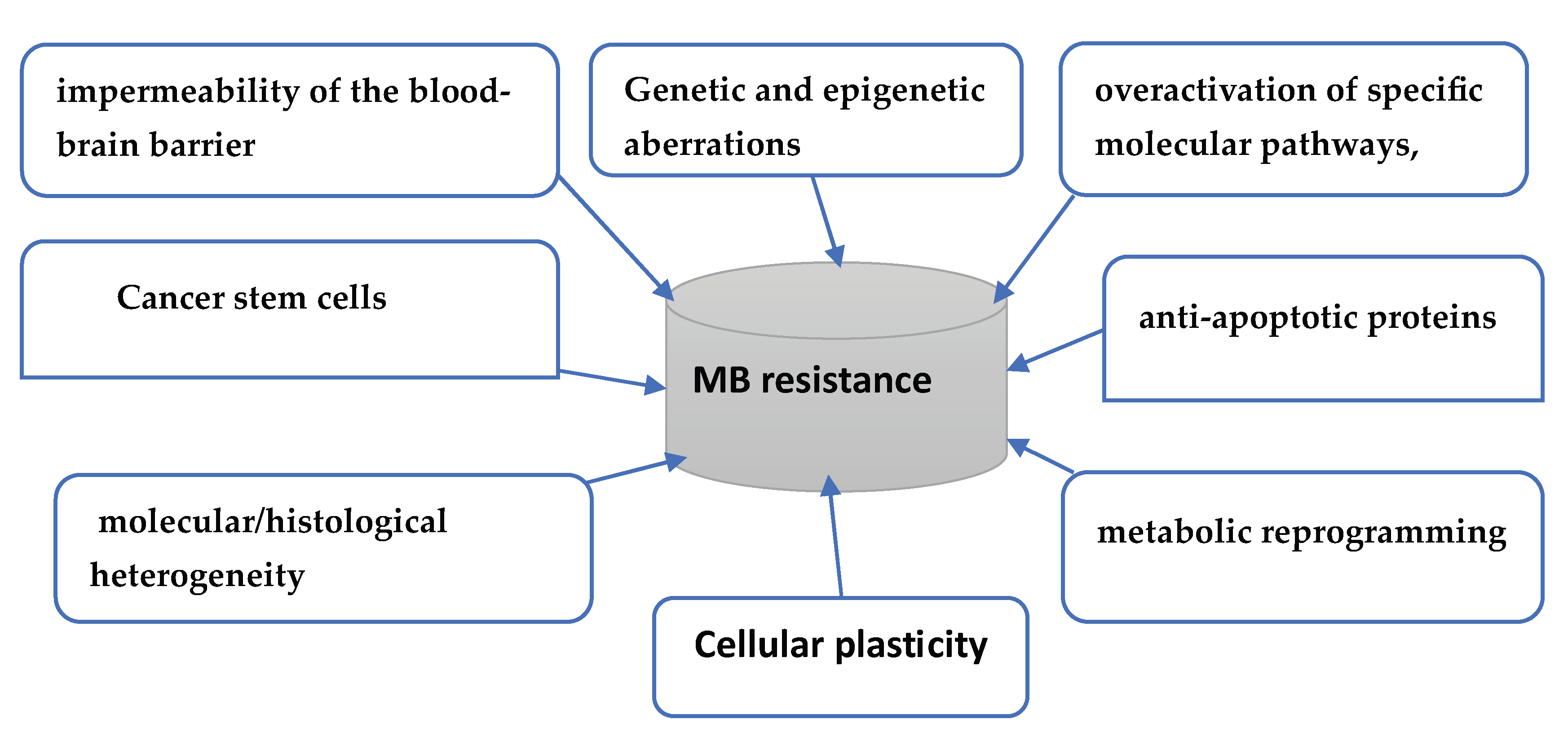
2.2. Mechanisms of Resistance in Medulloblastoma?
| drivers of MB resistance to therapy | Comments/ Refs. | |
|---|---|---|
| Permeability of blood- brain barrier (BBB). Cellular plasticity Glioblastoma stem cells Anti-apoptotic proteins Genetic aberrations Altered Molecular pathways metabolic reprogramming molecular/histological heterogeneity |
The Impermeability of BBB to a wide range of chemotherapeutic agents is considered a major driver of MB resistance to therapy. As well as the structural permeability of BBB, the expression of efflux transporter proteins on the surface of CNS endothelial cells is another factor, which contribute to the prevention of various therapeutic agents from reaching the targeted area within the brain [49,50]. Cellular plasticity is an important driver of therapy resistance. Cancer cells can alter their lineage to evade targeted therapy. To evade therapy, these cancer cells revert to a stem-like state, such phenomenon has been described as self-renewal to evade therapy. These studies also observed, that although, many genomic aberrations have been associated with lineage plasticity, few tumors exhibit such alterations, suggesting that resistance caused by lineage plasticity occurs through alternative mechanisms. [48,51]. Glioblastoma Stem cells represent one of the main challenges for attempts to develop efficacious therapies for solid CNS tumors. These tumor initiation cells are characterized with self-renewal, proliferation, and differentiation capabilities. These characteristics render this class of cells a major player in MB resistance and disease recurrence. these stem cells are also characterized by their low abundance within the tumor and low proliferative activity, which protects them from therapies targeting dividing cells [50,52,53]. Evasion of apoptosis is a hallmark of cancer cells, which is known to contribute to therapeutic resistance in various solid tumors. In the case of MB, a number of proteins have been identified as key players in cell apoptosis and associated pathways. These proteins include, BCL-2, MCL-1, BCL-XL, Bax and Bak are two nuclear-encoded proteins present in higher eukaryotes that are able to pierce the mitochondrial outer membrane to mediate cell death by apoptosis [54,55]. A small subset of subgroups 3 and 4 medulloblastoma patient’s harbor gene oncogenic drivers, including MYC and MYCN amplifications as well as PRDM6 overexpression. On the other hand, the majority of patients within the same subgroups displayed recurrent, large-scale copy number changes. Expression of genes that modulate metabolic responses and energy production (IDH1, HK2, HSPH1, GLS, and NFE2L1) are increased in recurrent tumors. [48,56,57,581]. Growth and progression of various forms of cancer, including medulloblastoma have always been linked to the activation of various pathways. For example, the IL-6/STAT3 pathway has been associated with tumorigenesis and acquired resistance in Group 3 medulloblastoma. Inhibition of STAT3 has been shown to render medulloblastoma cells sensitive to chemotherapy, leading to improved treatment outcomes [59,60]. metabolic pathways are used by medulloblastoma cells to adapt to environments, which lack the required nutrients and oxygen. Such adaptability is achieved through modulating glucose, lipids, amino acids, and nucleotide metabolism. Regarding MB, such pathways, and their impact on resistance to therapy are still understudied. Single-cell multi-omics analyses showed that In vivo modeling of radiation resistance exhibited chromatin-based metabolic reprogramming focused on wild-type isocitrate dehydrogenase (IDH1) activity. IDH1 inhibition reversed resistance-mediated chromatin changes and resulted in radiation re-sensitization [48,61]. Recent use of single-cell RNA-seq is providing much clearer picture on the role of intratumorally heterogeneity and tumor origin for the four molecular subgroups of medulloblastoma. Molecular profiling of MB at the single-cell level emphasized the central role of clonal heterogeneity in resistance to therapy. The impact of heterogeneity on therapy outcome was investigated, using quantitative profiling of global proteomes and phospho-proteomes of medulloblastoma samples, these measurements showed that post-translational modifications of MYC were associated with poor outcomes in Group 3 tumors [6,50,62]. |
2.3. Emerging Therapeutic Strategies for Treating Medulloblastoma
2.3.1. Targeting Immune Cell Checkpoints in Medulloblastoma
2.3.2. Is CAR NK Cell Therapy an Alternative to CAR T Cell Therapy?
3. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ICIs | Immune e checkpoint inhibitors. |
| MB | Medulloblastoma. |
| PTMs | post-translational modifications. |
| CAR T | Chimeric antigen receptor T cell therapy. |
| CAR NK | Chimeric antigen receptor natural killer cell therapy. |
| CTCs | Circulating tumor cells. |
| ctDNA | Circulating tumor DNA. |
| PCR | Polymerase chain reaction. |
| NGS | Next-generation sequencing. single-chain variable-fragment |
| CSF | Cerebrospinal fluid |
| MS | Mass spectrometry |
| LC | Liquid chromatography |
| LC-MS/MS | Liquid chromatography/tandem mass spectrometry |
| scFv | Single-chain variable-fragment |
| PPP | Pentose phosphate pathway. |
| TKT | Transketolase |
| ELISA | Enzyme-linked immunosorbent assay |
| BBB | Blood- brain barrier. |
| FDA | Food and Drug Administration. |
| EMA | European Medicines Agency- |
| PD-L1 | Cell death ligand-L1 |
| PD-1 | Programmed cell death-1 |
| CTLA-4 | Cytotoxic T lymphocyte antigen-4 |
| LAG-3 | Lymphocyte activation gene-3 |
| B7-H3 | B7 homolog 3 protein. |
| BTLA | B and T lymphocyte attenuator. |
| TIGIT | Cell immunoglobulin and ITIM domain. |
| ICOS | Inducible T cell costimulatory. |
| VISTA | V-domain immunoglobulin suppressor of T cell activation. |
| GvHD | Graft-versus-host disease GvHD |
| TME | Tumor microenvironment. |
References
- International Agency for Research on Cancer. WHO Classification of Tumors of the Central Nervous System. (WHO Classification of Tumours Editorial Board,5th ed.),2022 IARC.
- Gajjar, A.; Robinson, G.W.; Smith, K.S.; et al. Outcomes by clinical and molecular features in children with medulloblastoma treated with risk-adapted therapy: results of an international phase III trial (SJMB03). J Clin Oncol. 2021, 39, 822–835. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.S.; Dhanda, S.K.; Billups, C.A.; Sioson, E.; Lu, C.; Peraza, A.Z.; Gangwani, K.; Li, Y.; et al. An integrated analysis of three medulloblastoma clinical trials refines risk-stratification approaches for reducing toxicity and improving survival. Neuro-Oncology 2026, (1), 268–281. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.D.; Northcott, P.A.; Korshunov, A.; Remke, M.; Cho, Y.J.; Clifford, S.C.; et al. Molecular subgroups of medulloblastoma: The current consensus. Acta Neuropathologica 2012, 123, 465–72. [Google Scholar] [CrossRef] [PubMed]
- Cavalli, F.M.G.; Remke, M.; Rampasek, L.; Peacock, J.; Shih, D.J.H.; Luu, B.; et al. Intertumoral heterogeneity within medulloblastoma subgroups. Cancer Cell 2017, 31, 737–54 e736. [Google Scholar] [CrossRef]
- Sheng, H.; Li, H.; Zeng, H.; Zhang, B.; Lu, Y.; Liu, X.; Z.; Zhang, J., Zhang, L. Heterogeneity and tumoral origin of medulloblastoma in the single-cell era. Oncogene 2024, 43, 839 85. [CrossRef]
- Northcott, P.A.; Korshunov, A.; Witt, et al. Medulloblastoma comprises four distinct molecular variants. J. Clin. Oncol. 2011, 29, 1408–14. [CrossRef]
- Cho, Y.-J.; Tsherniak, A.; Tamayo, P.; et al. Integrative genomic analysis of medulloblastoma identifies a molecular subgroup that drives poor clinical outcome. J Clin Oncol 2011, 29, 1424–30. [Google Scholar] [CrossRef]
- Schwalbe, E.C.; Williamson, D.; Lindsey, J.C.; et al. DNA methylation profiling of medulloblastoma allows robust subclassification and improved outcome prediction using formalin-fixed biopsies. Acta Neuropathol. 2013, 125, 359–71. [Google Scholar] [CrossRef]
- Hovestadt, V.; Remke, M.; Kool, M.; et al. Robust molecular subgrouping and copy-number profiling of medulloblastoma from small amounts of archival tumor material using high-density DNA methylation arrays. Acta Neuropathol 2013, 125, 913–16. [Google Scholar] [CrossRef]
- Kim, D.T.; Uloho-Okundaye, M.; Frederico, S.C.; Guru, S.; Kim, M.J.; Chang, S.D. Advancing Medulloblastoma Therapy in Pediatrics: Integrative Molecular Classification and Emerging Treatments. Brain Sci. 2025, 15(8), 896. [Google Scholar] [CrossRef]
- Gold, M.P.; Ong, W.; Masteller, A.M.; et al. Developmental basis of SHH medulloblastoma heterogeneity. Nat. Commun. 2024, 15, 270. [Google Scholar] [CrossRef] [PubMed]
- Pan, Z.; Bao, J.; Wei, S. Advancing medulloblastoma therapy: strategies and survival insights. Clin. Exp. Med. 2025, 25(1), 119. [Google Scholar] [CrossRef] [PubMed]
- Vlachová, M.; Palinka, L.; Gregorová, J.; Moráň, L.; Růžičková, T.; Kovačovicová, P.; Almáši, M.; Pour, L.; et al. Liquid biopsy of peripheral blood using mass spectrometry detects primary extramedullary disease in multiple myeloma patients. Scientific Reports 2024, 14, 18777. [Google Scholar] [CrossRef] [PubMed]
- Satam, H.; Joshi, K.; Upasana, U.; Waghoo, S.; et al. Next-generation sequencing technology: current trends and ad-vancements biology (Basel). 2023, 12(7), 997. [Google Scholar]
- Zhang, S.; Rajadhyaksha, E.A.; Syed, F.; Canas, J.; Saxena, V.; Schwaderer, A.L.; David S. Hains, D.S. Digital droplet PCR is an accurate and precise method to measure DNA copy number. Scientific Reports 2025, 15, Article number, 36958. [Google Scholar] [CrossRef]
- Denis, J. A.; Guillerm, E.; Coulet, F.; Larsen, A. K.; Lacorte, J. M. The role of Beaming and digital PCR for multiplexed analysis in molecular oncology in the era of next-generation sequencing. Mol. Diagn.Ther 2017, 21, 587–600. [Google Scholar] [CrossRef]
- Li, S.; · Poolen, G.C.; ·van Vliet, L.C.; ·Schipper, J.G.; ·. Broekhuizen, R.; · Monnikhof, M.; Van Hecke, W.; Vermeulen J.F.; Bovenschen, N, Pediatric medulloblastoma express immune checkpoint B7-H3 Clinical and Translational Oncology 2022, 24, 1204–1208. [CrossRef]
- Majzner, R.G.; Theruvath, J.L. Nellan, A.; Heitzeneder, S., Cui, Y.; Mount, C.W.; Rietberg, S.P.; Miles H. Linde, M.H.; et al. CAR T Cells Targeting B7-H3, a Pan-Cancer Antigen, Demonstrate Potent Preclinical Activity Against Pediatric Solid Tumors and Brain Tumors. Clin. Cancer Res. 2019, 25(8), 2560–2574. [CrossRef]
- Huang, Y.; Zhong, W.Q.; Yang, X.Y.; Shan, J.L.; Zhou, L.; Li, Z.L.; Guo, Y.Q.; Zhang, K.M.; Du, T.; Zhang, H.L.; et al. Targeting site-specific N-glycosylated B7H3 induces potent antitumor immunity. Nat. Commun. 2025, 16, 3546. [Google Scholar] [CrossRef]
- Corrigan, D.T.; Ankit, T.A.; Du, M.; Martin, A.M.; Zang, X. The B7-H3 (CD276) pathway: Emerging biology and clinical therapeutics. Trends Pharmacol. Sci. 2025, 46, 975–988. [Google Scholar] [CrossRef]
- Doroshow, D.B.; Bhalla, S.; Beasley, M.B.; Sholl, L.M.; Kerr, K.M.; Gnjatic, S.; Wistuba, I.; Rimm, D.L.; Tsao, M.S.; Hirsch, F. R. PD-L1 as a biomarker of response to immune-checkpoint inhibitors. Nat. Rev. Clin. Oncol. 2021, 18, 345–362. [Google Scholar] [CrossRef] [PubMed]
- Bachtella, L.; Chunsheng, J.; Fentker, K.; Ertürk, G.R.; Safferthal, M.; Polewski, L.; Götze., M.; Graeber, S.Y.; Vos, G.M.; Struwe, W.B.; et al. Ion mobility-tandem mass spectrometry of mucin type O glycans. Nat. Commun. 2024, 15, 2611. [Google Scholar] [CrossRef] [PubMed]
- Fröhlich, F.; Fahrner, M.; Brombacher, E.; Seredynska, A.; Maldacker, M.; Schmidt1, A.; Schilling, O. Data-independent acquisition: A Milestone and prospect in clinical mass spectrometry–based proteomics. Mol. Cell Proteom. 2024, 23, 100800. [Google Scholar] [CrossRef] [PubMed]
- Wei, D.; Horton, K.L.; Chen, J.; Dong, L.; Chen, S.; Abdul-Hadi, K.; Zhang, T.T.; Casson, C.N.; Shaw, M.; Shiraishi, T.; et al. Development of a highly sensitive hybrid LC/MS assay for the quantitative measurement of CTLA-4 in human T Cells. Molecules 2023, 28, 3311. [Google Scholar] [CrossRef]
- Wenk, D.; Zuo, C.; Kislinger, T.; Sepiashvili, L. Recent developments in mass-spectrometry-based targeted proteomics of clinical cancer biomarkers. Clin. Proteom. 2024, 21, 6. [Google Scholar] [CrossRef]
- Riley, N.M.; Coon, J.J. The Role of Electron Transfer Dissociation in Modern Proteomics Analytical Chemistry. J. Am. Soc. Mass Spectrom. 2017, 36(Issue 10). [Google Scholar]
- Manly, L.S.; Roberts, A.M.; Beckman, J, S.; Roberts, B.R. Electron Capture Dissociation for Discovery Top-Down Proteomics of Peptides and Small Proteins on Chromatographic Time Scales. J. Am. Soc. Mass Spectrom 2025, 28 36(10), 2079–2093. [CrossRef]
- Cappell, K. M.; Kochenderfer, J.N. Long-term outcomes following CAR T cell therapy: what we know so far. Nat. Rev. Clin. Oncol. 2023, 20(6), 359–371. [Google Scholar] [CrossRef]
- Rivière, I.; Sadelain, M. Chimeric Antigen Receptors: A Cell and Gene Therapy Perspective. Mol. Ther. 2017, 25(5), 1117–1124. [Google Scholar] [CrossRef]
- June, C.H. Michel Sadelain, Chimeric Antigen Receptor Therapy. N. Engl. J. Med. 2018, 379(1), 64–73. [Google Scholar] [CrossRef]
- Zugasti, I.; Espinosa-Aroca, L.; Fidyt, K.; Molens-Arias, V.; Diaz-Beya, M.; Juan, M.; Urbano-Ispizua, A.; Esteve, J.; Velasco-Hernandez, T.; Menéndez, P. CAR-T cell therapy for cancer: current challenges and future directions. Signal Transduction and Targeted Therapy 2025, 10, Article number, 210. [Google Scholar] [CrossRef]
- June, C.H.; O’Connor, R.S.; Kawalekar, O.U.; Saba Ghassemi, S.; Michael C Milone, M.C. CART cell immunotherapy for human cancer. Science 2018, 359(6382), 1361–1365. [Google Scholar] [CrossRef] [PubMed]
- Marvin-Peek, J.; Savani, B.N.; Olalekan, L.O. Dholaria, B. Challenges and Advances in Chimeric Antigen Receptor Therapy for Acute Myeloid Leukemia. Cancers 2022, 14(3), 497. [CrossRef] [PubMed]
- Robert C. Sterner, R.C.; Sterner, R. M. CAR--T cell therapy: current limitations and potential strategies. Blood Cancer Journal 2021, 11, Article number, 69. [CrossRef] [PubMed]
- Li, Y.-R.; Zhu, Y.; Fang, Y.; Lyu, Z.; Yang, L. Emerging trends in clinical allogeneic CAR cell therapy. j. medj 2025, 100677. [Google Scholar] [CrossRef]
- Rosa, R.; Liu, J.; Lu, C.; Abou-el-Enein, M.; Murad, J.P.; Priceman, S.J. Current state of CAR-T cell therapies for solid tumors. medj 2026, 101028. [Google Scholar] [CrossRef]
- Jørgensen, L.V.; Christensen, E.B.; Barnkob, M.B.; Barington, T. The clinical landscape of CAR NK cells. Experimental Hematology & Oncology 2025, 14, 4634. [Google Scholar] [CrossRef]
- Kim, J.W.; Choi, S.A.H.; Dan, K.; Koh, E.J.; Ha, S.; et al. Proteomic profiling of cerebrospinal fluid reveals TKT as a potential biomarker for medulloblastoma. Scientific Reports 2024, 14, 21053. [Google Scholar] [CrossRef]
- Qin, Z.; Xiang, C.; Zhong, F.; Liu, Y.; Dong, Q.; Li, K.; Shi, W.; Ding, C.; Qin, L.; He, F. Transketolase (TKT) activity and nuclear localization promote hepatocellular carcinoma in a metabolic and a non -metabolic manner. J. Exp. Clin. Cancer Res. 2019, 38, 154. [Google Scholar] [CrossRef]
- Ricciardelli, C.; Lokman, N.A.; Cheruvu, S.; Tan, I.A.; Ween, M.P.; Pyragius, C.E.; Ruszkiewicz, A.; Hoffmann, P.; Oehler, M.K. Transketolase is up -regulated in metastatic peritoneal implants and promotes ovarian cancer cell proliferation. Clin. Exp. Metastasis 2015, 32, 441–455. [Google Scholar] [CrossRef]
- Huang, F-L.; Chang Y-M.; Lin, C-Y.; Yu, S-J.; Fu, J-T.; Chou, T-Y.; Yeh, S-W.; Liao, E-C.; Chia-Ling Li, C-L. Regulating TKT activity inhibits proliferation of human acute lymphoblastic leukemia cells. Am. J. Cancer Res. 2024, 14(2), 679–695. [CrossRef] [PubMed]
- Bruschi, M.; Petretto, A.; Cama, A.; Pavanello, M.; Bartolucci, M.; Giovanni Morana, G.; Luca Antonio, L.; et al. Potential biomarkers of childhood brain tumor identified by proteomics of cerebrospinal fluid from extraventricular drainage (EVD). Scientific Reports 2021, 11, 1818. [Google Scholar] [CrossRef] [PubMed]
- Dayon, L.; Cominetti, O.; Wojcik, J.; Galindo, A.N.; Oikonomidi, A.; Henry, H.; et al. Proteomes of Paired Human Cerebrospinal Fluid and Plasma: Relation to Blood–Brain Barrier Permeability in Older Adults. Journal of Proteome Research 2019, 18(3). [Google Scholar] [CrossRef] [PubMed]
- Archer, T.C.; Ehrenberger, T.; F Mundt, F.; Gold, M.P.; Krug, K.; Mah, C.K.; Elizabeth L. Mahone, E.L.; et al. Post-translational Modifications, and Integrative Analyses Reveal Molecular Heterogeneity within Medulloblastoma Subgroups. Cancer Cell. 2018, 34(3), 396–410.e8. [Google Scholar] [CrossRef]
- Orr, B. A. Pathology, diagnostics, and classification of medulloblastoma. Brain Pathol. 2020, 30, 664–678. [Google Scholar] [CrossRef]
- Louis, A.N.; Perry, A.; Wesseling, P.; Daniel J Brat, D.J.; Cree. I.A.; Figarella-Branger, D.; Cynthia Hawkins, et al. The 2021 WHO classification of tumors of the central nervous system: A summary. Neuro. Oncol. 2021, 23, 1231–1251. [CrossRef]
- Veo, B.; Wang, D.; DeSisto, J.; Pierce, A.; Brunt, B.; Bompada, P.C.; Donson, A.; et al. Single-cell multi-omics identifies metabolism-linked epigenetic reprogramming as a driver of therapy resistant medulloblastoma. Nature Communications 2025, 16, 10470. [Google Scholar] [CrossRef]
- Haumann, R.; Videira, J.C.; Kaspers, G.J.L.; van Vuurden, D.G.; Hulleman, E. Overview of Current Drug Delivery Methods Across the Blood-Brain Barrier for the Treatment of Primary Brain Tumors. CNS Drugs 2020, 34, 1121–1131. [Google Scholar] [CrossRef]
- Slika, H.; Shahani, A.; Wahi, R.; Miller, J.; Groves, Tyler, B. Overcoming Treatment Resistance in Medulloblastoma: Underlying Mechanisms and Potential Strategies. Cancers 2024, 16, 2249. [CrossRef]
- Bakhshinyan, D.; Ashley, A. A.; Liu, J.; William D Gwynne, W.D.; Suk, Y.; Custers, S.; et al. Temporal profiling of therapy resistance in human medulloblastoma identifies novel targetable drivers of recurrence. Sci. Adv. 2021, 7, eabi5568. [Google Scholar] [CrossRef]
- Hersh, A.M.; Gaitsch, H.; Alomari, S.; Lubelski, D.; Tyler, B.M. Molecular Pathways and Genomic Landscape of Glioblastoma Stem Cells: Opportunities for Targeted Therapy. Cancers 2022, 14, 3743. [Google Scholar] [CrossRef] [PubMed]
- Biserova, K.; Jakovlevs, A.; Uljanovs, R.; Strumfa, I. Cancer Stem Cells: Significance in Origin, Pathogenesis and Treatment of Glioblastoma. Cells 2021, 10, 621. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, M.-C.; O’Halloran, P.J.; Kerrane, S.A.; T Chonghaile, T.Ni.; Connolly, N.M.C.; Murphy, B.M. The identification of BCL-XL and MCL-1 as key anti-apoptotic proteins in medulloblastoma that mediate distinct roles in chemotherapy resistance. Cell Death and Disease 2023, 14:70. [Google Scholar] [CrossRef] [PubMed]
- Simon N. WillisS. N.; Chen, L.; Dewson, G.; Wei, A.; Naik, E.;Fletcher, J.I.; Adams, J.M.;.Huang, D.C.S. Proapoptotic Bak is sequestered by Mcl-1 and Bcl-xL, but not Bcl-2, until displaced by BH3-only proteins. Genes & Development 2005, 19, 1294–1305.
- Taylor, L.; Wade, P.K.; Johnson, J.E.C.; Aldighieri, M.; Morlando, S.; Di Leva, G.; Kerr, I.D.; Coyle, B. Drug Resistance in Medulloblastoma Is Driven by YB-1, ABCB1 and a Seven-Gene Drug Signature. Cancers 2023, 15, 1086. [Google Scholar] [CrossRef]
- Okonechnikov, K.; Joshi, P.; Körber, V.; Rademacher, A.; Bortolomeazzi, M.; Mallm, J.-P.; Vaillant, J.; da Silva, P.B.G. Oncogene aberrations drive medulloblastoma progression, not initiation. Nature 2025, 642, 1062. [Google Scholar] [CrossRef]
- Shrestha, S.; Morcavallo, A.; Gorrini, C.; Chesler, L. Biological role of MYCN in medulloblastoma: novel therapeutic opportunities and challenges ahead. Front. Oncol. 2021, 11, 694320. [Google Scholar] [CrossRef]
- Sreenivasan, L.; Wang, H.; Yap, S.Q.; Leclair, P.; Tam, A.; Lim, C.J. Autocrine IL-6/STAT3 signaling aids development of acquired drug resistance in Group 3 medulloblastoma. Cell Death Dis. 2020, 11, 1035. [Google Scholar] [CrossRef]
- Kumar, S.; Arwind, D.A.; Kumar, H.; Pandey, S.; Nayak, R.; Vithalkar, M.P.; Nitesh Kumar, N.; et al. Inhibition of STAT3: A promising approach to enhancing the efficacy of chemotherapy in medulloblastoma. Translational Oncology 2024, 46, 102023. [Google Scholar] [CrossRef]
- Manfreda, L.; Rampazzo, E.; Persano, L.; Viola, G.; Bortolozzi, R. Surviving the hunger games: Metabolic reprogramming in medulloblastoma. Biochemical Pharmacology 2023, 215, 115697. [Google Scholar] [CrossRef]
- Archer, T.C.; Ehrenberger, T.; Mundt, F.; Gold, M.P.; Krug, K.; Clarence K. Mah,C. K.; Mahoney, E.L.; Danie, C.J.; et al. Proteomics, Post-translational Modifications, and Integrative Analyses Reveal Molecular Heterogeneity within Subgroups. Cancer Cell. 2018, 34(3), 396–410.e. [CrossRef] [PubMed]
- Perachino, M.; Blondeaux, E.; Molinelli, C.; Ruelle, T.; Giannubilo, I.; Arecco, L.; Nardin, S.; Razeti, M.G.; Borea, R.; Favero, D.; et al. Adverse events and impact on quality of life of antibody drug conjugates in the treatment of metastatic breast cancer: A systematic review and meta-analysis. Eur. J. Clin. Investig. 2025, 55, e70001. [Google Scholar] [CrossRef] [PubMed]
- Markides, D.M.; Hita, A.G.; Merlin, J.; Reyes-Gibby, C.; Yeung, S.-C.J. Antibody-Drug Conjugates: The Toxicities and Adverse Effects That Emergency Physicians Must Know. Ann. Emerg. Med. 2025, 85, 214–229. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Case, R.; Wei, K.Y.; Ma, H.; Manzanillo, P.; Deng, W. Characterization and comparative analysis of multifunctional natural killer cell engagers during antitumor responses. Cell Rep. Med. 2025, 6, 102117. [Google Scholar] [CrossRef]
- Jansen, L.; Wienke, M.J.; Molkenbur, R.; Rossig, C.; Meissner, R. B7-H3 in the tumor microenvironment: Implications for CAR T cell therapy in pediatric solid tumors. Cancer and Metastasis Reviews 2025, 44:77. [Google Scholar] [CrossRef]
- Lopez, P.H.H.; Schnaar, R.L. Gangliosides in cell recognition and membrane protein regulation. Curr. Opin. Struct. Biol. 2009, 19(5), 549–57. [Google Scholar] [CrossRef]
- Fernández-Rilo, A.C.; de Billy, E.; Del Baldo, G.; De Angelis, B.; Rossi, S.; Del Bufalo, F.; et al. GD2: hopes and challenges for the treatment of pediatric patients with tumors of the central nervous system. NPJ. Precision Oncology 2025, 9, 295. [Google Scholar] [CrossRef]
- Ciccone, R.; Quintarelli, C.; Camera, A.; Pezzella, M.; S Caruso, S.; Manni, S.; Ottaviani1, A.; Guercio, M.; Francesca Del Bufalo; F.; et al. GD2-Targeting CAR T-cell Therapy for Patients with GD2þ Medulloblastoma Clin Cancer Res 2024, 30, 2545–57. [CrossRef]
- Okada, R.; Reyes-González, I.M.; Rodriguez, C.; Kondo, T.; Oh, J.; Sun, M.; Kelly, M.C.; Ling Zhang, L.; James Gulley, J. GPC2-targeted CAR T cells engineered with NFAT-inducible membrane-tethered IL-15/IL-21 exhibit enhanced activity against neuroblastoma. Cancer Immunol. Res. 2025, 13(9), 1363–1373. [Google Scholar] [CrossRef]
- Zhang, Y.; Feng, R.; Chi, X.; Na Xian, Chen, X.; Huang, N.; Zhang, Y.; Zhang, K.; et al. Jindong. Safety and efficacy of B7-H3 targeting CAR-T cell therapy for patients with recurrent GBM. J Clin Oncol 2024, 42, 2062. [CrossRef]
- Schaible, P.; Bethge, W.; Lengerke, C.; Reka Agnes Haraszti, R.A. RNA therapeutics for improving CAR T cell safety and efficacy. Cancer Res. 2023, 83(3), 354–362. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Li, H.; Lin, Y.; Wang,G.; , Xu.; Chen,Y.; XiaoK.; Rao,X.;. CircDUSP16 Contributes to Cell Development in Esophageal Squamous Cell Carcinoma by Regulating miR-497-5p/TKTL1 Axis. J Surg Res. 2021, 260, 64–75. [CrossRef] [PubMed]
- Zhang, C.; Hu, Y.; Shi, C. Targeting Natural Killer Cells for Tumor Immunotherapy. Front. Immunol. 2020, 11, 60. [Google Scholar] [CrossRef] [PubMed]
- Simonetta, F.; Lohmeyer, J.K.; Hirai, T.; Maas-Bauer, K.;.,Alvarez, M.; Wenokur, A.S.; Baker, J.; Aalipour, A. Ji, X.; Haile, S. et al. Allogeneic CAR Invariant Natural Killer T Cells Exert Potent Antitumor Effects through Host CD8 T-Cell Cross-Priming. Clin. Cancer Res. 2021, 27, 6054. [CrossRef]
- Mavers, M.; Maas-Bauer, K.; Negrin, R.S. Invariant natural killer T cells as suppressors of graft-versus-host disease in allogeneic hematopoietic stem cell transplantation. Front. Immunol. 2017, 8, 900. [Google Scholar] [CrossRef]
- Lanier, L.L. NK cell recognition. Annu. Rev. Immunol. 2005, 23(1), 225–74. [Google Scholar] [CrossRef]
- Jørgensen, L.V.; Christensen, E.B.; Barnkob, M.B.; Baringto, T. The clinical landscape of CAR NK cells. Experimental Hematology & Oncology 2025 14, 46. [Google Scholar]
- Wang, V.; Gauthier, M.; Decot, V.; Replì.; pel, L.; Bensoussan, D. Systematic review on CAR-T cell clinical trials up to 2022: academic center input. Cancers 2023, 15(4), 1003. [CrossRef]
- Barros, L.R.C.; Couto, S.C.F.; da Silva, S.D.; Paixão, E.A.; Cardoso, F.; da Silva, V.J.; et al. Systematic review of available CAR-T cell trials around the world. Cancers 2022, 14(11), 266. [Google Scholar] [CrossRef]
| Trial’s Identifier/Sponsor | Trial objective | Observations/Refs. |
|---|---|---|
| NCT05298995/ Gesù Hospital and Research Institute, ITALY NCT07087002/ Stanford University, USA NCT07390539/ Robbie MajznerDana-Farber Cancer Institute, USA. NCT05241392/ Beijing Tiantan Hospital, Cina NCT06061809/ImmunityBio, Inc. |
evaluate the safety and therapeutic efficacy of CAR.GD2 therapy in high-risk Medulloblastoma patients. Evaluate GPC2 Chimeric Antigen Receptor T (GPC2-CAR T) Cells for the treatment of Relapsed or Refractory Medulloblastoma in Children and Young Adults The purpose of this research study is to test the safety and effectiveness of a cell therapy at different doses for children and young adults with recurrent or progressive brain tumors. Evaluate the Safety/Preliminary Effectiveness and Determine the Maximal Tolerated Dose of B7-H3-targeting CAR-T Cell Therapy in Treating Recurrent Glioblastomas. Evaluate the safety and efficacy of NAI, PD-L1 t-haNK, and bevacizumab combination therapy in participants with recurrent or progressive glioblastoma (GMB) |
This a phase I clinical trial, designed to assess the safety and efficacy of 4.1BB-CD28 CAR T cell treatment targeting GD2 in pediatric or young adult patients affected by relapsed/refractory malignant central nervous system (CNS) tumors: started 2023, estimated primary completion,2027, estimated completion, 2038 Ciccone et al.2024 [69]. Single-site, open-label Phase 1 clinical trial to evaluate, safety, and efficacy of autologous GPC2-targeted chimeric antigen receptor CART cells in children and young adults with relapsed or refractory medulloblastoma or other eligible Central Nervous System (CNS) embryonal tumors. Study Start,2025-08, Primary Completion (Estimated) 2027-08 Study Completion (Estimated) 2027-08 [70] This is a single-institution, Phase 1/1b, open-label study, which uses B7-H3 CAR T cells therapy to treat some forms of recurrent or progressive brain tumors: Study start 07/2026, estimated primary completion,08/2030, estimated completion 08/2032 This is phase I open, single-arm, dose-escalation and multiple-dose study to evaluate the safety, tolerability and preliminary effectiveness of B7-H3-targeting Chimeric Antigen Receptor-T (CAR-T) cell therapy on patients with recurrent glioblastomas. Study Start 2022 Primary Completion estimated study completion [71]. This study consists of two parts: the first part is an open-label, single-arm study to evaluate the safety and efficacy of NAI, Pt-haNK, and bevacizumab combination therapy in participants with recurrent or progressive GBM. The second part consists of two experimental arms, in the firs,t participants with recurrent or progressive GBM: NAI, bevacizumab, and TTFields combination therapy, In the second arm, NAI, PD-L1 t-haNK, bevacizumab, and TTFields combination therapy is evaluated. Study start ,2024-08, primary completion (estimated) 2029-12, study completion (Estimated) 2030-12. |
| Trial’s Identifier/Sponsor | Observations |
|---|---|
| NCT05020678/ Nkarta, Inc. NCT05194709/ Wuxi People’s Hospital NCT02271711/ M.D. Anderson Cancer Center NCT05588453/Kari Kendra, (Ohio State University Comprehensive Cancer Center) NCT05962450/ Beijing YouAn Hospital |
This is a single arm, open-label, multi-center, Phase 1 study to determine the safety and tolerability of an experimental therapy called NKX019 (allogeneic CAR NK cells targeting CD19) in patients with relapsed/refractory non-Hodgkin lymphoma, chronic lymphocytic leukemia or B cell acute lymphoblastic leukemia. Study Start: 2021-08. Primary Completion, 2025-03(estimated), Study Completion 2038-12(estimated), Enrollment, 150 (Estimated). This study is an interventional, single arm, open-label, to evaluate the safety, tolerability, initial efficacy and pharmacokinetics of anti-5T4 CAR-NK cells in patients with advanced solid tumors. Study Start: Dec. 2021, Primary Completion, Dec..2022(estimated), Study Completion Dec,2022, enrollment 40(estimated). This is a phase I study to assess the safety, efficacy, side effects and maximum tolerated dose of administering autologous natural killer (NK) cells. These cells were administered directly into the ventricle in recurrent /refractory malignant posterior fossa tumors. Study Start: 2015-03, Primary Completion, 2020-08 Study Completion, 2020-08, enrollment,12. This phase I/II trial the safety and tolerability of UD TGFbetai NK cells in combination with temozolomide as a lymphodepleting agent in patients with melanoma metastatic to the brain and to determine the recommended phase 2 dose (RP2D). (Phase 1) II. To determine the intracranial response rate. (Phase 2) Study Start: 2023-03, Primary Completion, 2026-04, Study Completion 2026-04(estimated), enrollment, 30 (Estimated). The main objective of thisphase II clinical trial is to assess the efficacy and safety of autologous iNKT cells in patients with progressed hepatocellular carcinoma (HCC) after treatment with PD-1 antibody. Study start 10-.2023(estimated), Primary Completion 08-2025 (Estimated), Study Completion 08-2025 (Estimated), Enrollment (Estimated)84. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




