Submitted:
17 April 2026
Posted:
20 April 2026
You are already at the latest version
Abstract
Keywords:
Background
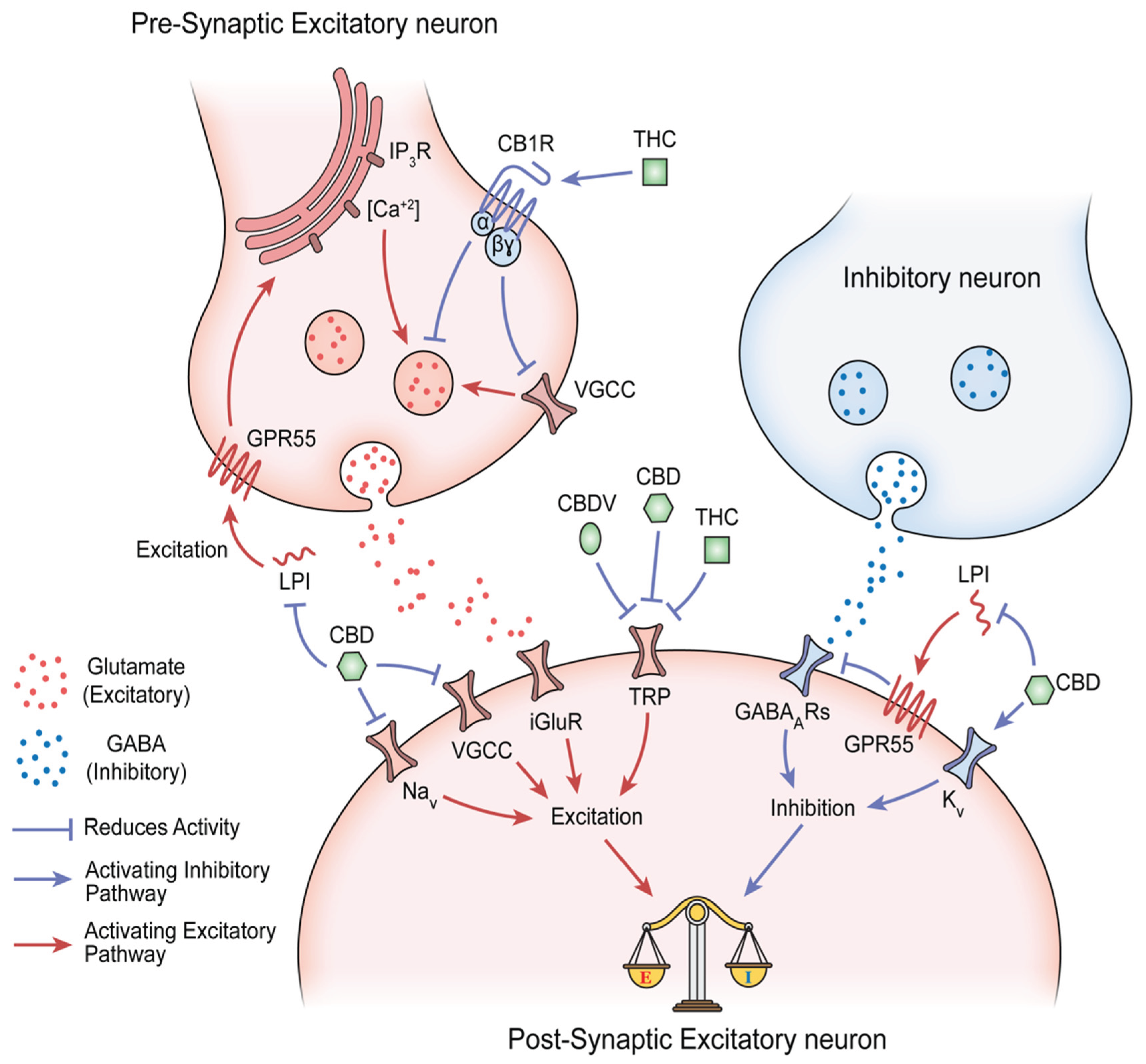
Mechanistic Basis of Multi-Compound Phytocannabinoid Signaling
Cannabinoid–Terpene Synergy
Conclusion
Acknowledgments
Abbreviations
References
- Rosenberg, E.C.; Tsien, R.W.; Whalley, B.J.; Devinsky, O. Cannabinoids and Epilepsy. Neurotherapeutics: the journal of the American Society for Experimental NeuroTherapeutics 2015, 12, 747–768. [Google Scholar] [CrossRef]
- Scharfman, H.E. The neurobiology of epilepsy. Current neurology and neuroscience reports 2007, 7, 348–354. [Google Scholar] [CrossRef]
- McCormick, D.A.; Contreras, D. On the cellular and network bases of epileptic seizures. Annual review of physiology 2001, 63, 815–846. [Google Scholar] [CrossRef]
- Mechoulam, R. Cannabis and epilepsy. Epilepsy & behavior: E&B 2017, 70, 278–279. [Google Scholar] [CrossRef]
- Rosenthal, F. The herb hashish versus medieval Muslim society. Leiden: E. J. Brill 1971. [Google Scholar]
- Chesher, G.B.; Jackson, D.M. Anticonvulsant effects of cannabinoids in mice: drug interactions within cannabinoids and cannabinoid interactions with phenytoin. Psychopharmacologia 1974, 37, 255–264. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.D.; McNeill, J.R.; Crawford, R.D.; Wilcox, W.C. Epileptiform seizures in domestic fowl. V. The anticonvulsant activity of delta9-tetrahydrocannabinol. Canadian journal of physiology and pharmacology 1975, 53, 1007–1013. [Google Scholar] [CrossRef]
- Wada, J.A.; Osawa, T.; Corcoran, M.E. Effects of tetrahydrocannabinols on kindled amygdaloid seizures and photogenic seizures in Senegalese baboons, Papio papio. Epilepsia 1975, 16, 439–448. [Google Scholar] [CrossRef]
- Ten Ham, M.; Loskota, W.J.; Lomax, P. Acute and chronic effects of beta9-tetrahydrocannabinol on seizures in the gerbil. European journal of pharmacology 1975, 31, 148–152. [Google Scholar] [CrossRef]
- Boggan, W.O.; Steele, R.A.; Freedman, D.X. 9 -Tetrahydrocannabinol effect on audiogenic seizure susceptibility. Psychopharmacologia 1973, 29, 101–106. [Google Scholar] [CrossRef]
- Corcoran, M.E.; McCaughran, J.A., Jr.; Wada, J.A. Acute antiepileptic effects of 9- tetrahydrocannabinol in rats with kindled seizures. Experimental neurology 1973, 40, 471–483. [Google Scholar] [CrossRef]
- Consroe, P.; Benedito, M.A.; Leite, J.R.; Carlini, E.A.; Mechoulam, R. Effects of cannabidiol on behavioral seizures caused by convulsant drugs or current in mice. European journal of pharmacology 1982, 83, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Huizenga, M.N.; Sepulveda-Rodriguez, A.; Forcelli, P.A. Preclinical safety and efficacy of cannabidivarin for early life seizures. Neuropharmacology 2019, 148, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Hill, T.D.; Cascio, M.G.; Romano, B.; Duncan, M.; Pertwee, R.G.; Williams, C.M.; Whalley, B.J.; Hill, A.J. Cannabidivarin-rich cannabis extracts are anticonvulsant in mouse and rat via a CB1 receptor-independent mechanism. British journal of pharmacology 2013, 170, 679–692. [Google Scholar] [CrossRef] [PubMed]
- Amada, N.; Yamasaki, Y.; Williams, C.M.; Whalley, B.J. Cannabidivarin (CBDV) suppresses pentylenetetrazole (PTZ)-induced increases in epilepsy-related gene expression. PeerJ 2013, 1, e214. [Google Scholar] [CrossRef]
- Hurley, E.N.; Ellaway, C.J.; Johnson, A.M.; Truong, L.; Gordon, R.; Galettis, P.; Martin, J.H.; Lawson, J.A. Efficacy and safety of cannabidivarin treatment of epilepsy in girls with Rett syndrome: A phase 1 clinical trial. Epilepsia 2022, 63, 1736–1747. [Google Scholar] [CrossRef]
- Hill, A.J.; Williams, C.M.; Whalley, B.J.; Stephens, G.J. Phytocannabinoids as novel therapeutic agents in CNS disorders. Pharmacology & therapeutics 2012, 133, 79–97. [Google Scholar] [CrossRef]
- Karler, R.; Cely, W.; Turkanis, S.A. The anticonvulsant activity of cannabidiol and cannabinol. Life sciences 1973, 13, 1527–1531. [Google Scholar] [CrossRef]
- Thornton, C.; Dickson, K.E.; Carty, D.R.; Ashpole, N.M.; Willett, K.L. Cannabis constituents reduce seizure behavior in chemically-induced and scn1a-mutant zebrafish. Epilepsy & behavior: E&B 2020, 110, 107152. [Google Scholar] [CrossRef]
- de Sousa, D.P.; Nóbrega, F.F.; Santos, C.C.; de Almeida, R.N. Anticonvulsant activity of the linalool enantiomers and racemate: investigation of chiral influence. Natural product communications 2010, 5, 1847–1851. [Google Scholar] [CrossRef]
- Koutroumanidou, E.; Kimbaris, A.; Kortsaris, A.; Bezirtzoglou, E.; Polissiou, M.; Charalabopoulos, K.; Pagonopoulou, O. Increased seizure latency and decreased severity of pentylenetetrazol-induced seizures in mice after essential oil administration. Epilepsy research and treatment 2013, 2013, 532657. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, C.C.; de Oliveira, C.V.; Grigoletto, J.; Ribeiro, L.R.; Funck, V.R.; Grauncke, A.C.; de Souza, T.L.; Souto, N.S.; Furian, A.F.; Menezes, I.R.; Oliveira, M.S. Anticonvulsant activity of β- caryophyllene against pentylenetetrazol-induced seizures. Epilepsy & behavior: E&B 2016, 56, 26–31. [Google Scholar] [CrossRef]
- Mallmann, M.P.; Mello, F.K.; Neuberger, B.; da Costa Sobral, K.G.; Fighera, M.R.; Royes, L.F.F.; Furian, A.F.; Oliveira, M.S. Beta-caryophyllene attenuates short-term recurrent seizure activity and blood-brain- barrier breakdown after pilocarpine-induced status epilepticus in rats. Brain research 2022, 1784, 147883. [Google Scholar] [CrossRef] [PubMed]
- Peltner, L.K.; Gluthmann, L.; Börner, F.; Pace, S.; Hoffstetter, R.K.; Kretzer, C.; Bilancia, R.; Pollastro, F.; Koeberle, A.; Appendino, G.; Rossi, A.; Newcomer, M.E.; Gilbert, N.C.; Werz, O.; Jordan, P.M. Cannabidiol acts as molecular switch in innate immune cells to promote the biosynthesis of inflammation- resolving lipid mediators. Cell chemical biology 2023, 30, 1508–1524.e7. [Google Scholar] [CrossRef]
- Shebaby, W.; Saliba, J.; Faour, W.H.; Ismail, J.; El Hage, M.; Daher, C.F.; Taleb, R.I.; Nehmeh, B.; Dagher, C.; Chrabieh, E.; Mroueh, M. In vivo and in vitro anti-inflammatory activity evaluation of Lebanese Cannabis sativa L. ssp. indica (Lam.). Journal of ethnopharmacology 2021, 270, 113743. [Google Scholar] [CrossRef]
- Hampson, A.J.; Grimaldi, M.; Lolic, M.; Wink, D.; Rosenthal, R.; Axelrod, J. Neuroprotective antioxidants from marijuana. Annals of the New York Academy of Sciences 2000, 899, 274–282. [Google Scholar] [CrossRef]
- Sánchez, A.J.; García-Merino, A. Neuroprotective agents: cannabinoids. Clinical immunology 2012, 142, 57–67. [Google Scholar] [CrossRef]
- Kovalchuk, O.; Kovalchuk, I. Cannabinoids as anticancer therapeutic agents. Cell cycle 2020, 19, 961–989. [Google Scholar] [CrossRef]
- Bogdanović, V.; Mrdjanović, J.; Borišev, I. A Review of the Therapeutic Antitumor Potential of Cannabinoids. Journal of alternative and complementary medicine 2017, 23, 831–836. [Google Scholar] [CrossRef]
- Meng, H.; Johnston, B.; Englesakis, M.; Moulin, D.E.; Bhatia, A. Selective Cannabinoids for Chronic Neuropathic Pain: A Systematic Review and Meta-analysis. Anesthesia and analgesia 2017, 125, 1638–1652. [Google Scholar] [CrossRef]
- Romero-Sandoval, E.A.; Kolano, A.L.; Alvarado-Vázquez, P.A. Cannabis and Cannabinoids for Chronic Pain. Current rheumatology reports 2017, 19, 67. [Google Scholar] [CrossRef] [PubMed]
- Safakish, R.; Ko, G.; Salimpour, V.; Hendin, B.; Sohanpal, I.; Loheswaran, G.; Yoon, S.Y.R. Medical Cannabis for the Management of Pain and Quality of Life in Chronic Pain Patients: A Prospective Observational Study. Pain medicine 2020, 21, 3073–3086. [Google Scholar] [CrossRef] [PubMed]
- Mahmud, M.S.; Hossain, M.S.; Ahmed, A.T.M.F.; Islam, M.Z.; Sarker, M.E.; Islam, M.R. Antimicrobial and Antiviral (SARS-CoV-2) Potential of Cannabinoids and Cannabis sativa: A Comprehensive Review. Molecules 2021, 26, 7216. [Google Scholar] [CrossRef] [PubMed]
- Ferber, S.G.; Namdar, D.; Hen-Shoval, D.; Eger, G.; Koltai, H.; Shoval, G.; Shbiro, L.; Weller, A. The "Entourage Effect": Terpenes Coupled with Cannabinoids for the Treatment of Mood Disorders and Anxiety Disorders. Current neuropharmacology 2020, 18, 87–96. [Google Scholar] [CrossRef]
- Iseger, T.A.; Bossong, M.G. A systematic review of the antipsychotic properties of cannabidiol in humans. Schizophrenia research 2015, 162(1-3), 153–161. [Google Scholar] [CrossRef]
- Velayudhan, L.; McGoohan, K.; Bhattacharyya, S. Safety and tolerability of natural and synthetic cannabinoids in adults aged over 50 years: A systematic review and meta-analysis. PLoS medicine 2021, 18, e1003524. [Google Scholar] [CrossRef]
- McPartland, J.; Russo, E. Cannabis and Cannabis Extracts: Greater Than the Sum of Their Parts? J Cannabis Therapeutics 2001, 1, 103–132. Available online: https://www.researchgate.net/publication/228897917_Cannabis_and_Cannabis_Extracts_Greater_Than_the_Sum_of_Their_Parts. [CrossRef]
- Russo, E.B. The Case for the Entourage Effect and Conventional Breeding of Clinical Cannabis: No "Strain," No Gain. Frontiers in plant science 2019, 9, 1969. [Google Scholar] [CrossRef]
- Pamplona, F.A.; da Silva, L.R.; Coan, A.C. Potential Clinical Benefits of CBD-Rich Cannabis Extracts Over Purified CBD in Treatment-Resistant Epilepsy: Observational Data Meta- analysis. Frontiers in neurology 2018, 9, 759. [Google Scholar] [CrossRef]
- Maayah, Z.H.; Takahara, S.; Ferdaoussi, M.; Dyck, J.R.B. The molecular mechanisms that underpin the biological benefits of full-spectrum cannabis extract in the treatment of neuropathic pain and inflammation. Biochimica et biophysica acta. Molecular basis of disease 2020, 1866, 165771. [Google Scholar] [CrossRef]
- Carlini, E.A.; Karniol, I.G.; Renault, P.F.; Schuster, C.R. Effects of marihuana in laboratory animals and in man. British journal of pharmacology 1974, 50, 299–309. [Google Scholar] [CrossRef]
- Mechoulam, R.; Ben-Shabat, S. From gan-zi-gun-nu to anandamide and 2-arachidonoylglycerol: the ongoing story of cannabis. Natural product reports 1999, 16, 131–143. [Google Scholar] [CrossRef] [PubMed]
- Russo, E.B. Taming THC: potential cannabis synergy and phytocannabinoid-terpenoid entourage effects. British journal of pharmacology 2011, 163, 1344–1364. [Google Scholar] [CrossRef] [PubMed]
- Farrelly, A.M.; Vlachou, S.; Grintzalis, K. Efficacy of Phytocannabinoids in Epilepsy Treatment: Novel Approaches and Recent Advances. International journal of environmental research and public health 2021, 18, 3993. [Google Scholar] [CrossRef] [PubMed]
- Castillo, P.E.; Younts, T.J.; Chávez, A.E.; Hashimotodani, Y. Endocannabinoid signaling and synaptic function. Neuron 2012, 76, 70–81. [Google Scholar] [CrossRef]
- Mackie, K. Cannabinoid receptors: where they are and what they do. Journal of neuroendocrinology 2008, 20 Suppl 1, 10–14. [Google Scholar] [CrossRef]
- Marsicano, G.; Lutz, B. Expression of the cannabinoid receptor CB1 in distinct neuronal subpopulations in the adult mouse forebrain. The European journal of neuroscience 1999, 11, 4213–4225. [Google Scholar] [CrossRef]
- Katona, I.; Urbán, G.M.; Wallace, M.; Ledent, C.; Jung, K.M.; Piomelli, D.; Mackie, K.; Freund, T.F. Molecular composition of the endocannabinoid system at glutamatergic synapses. The Journal of neuroscience: the official journal of the Society for Neuroscience 2006, 26, 5628–5637. [Google Scholar] [CrossRef]
- Soltesz, I.; Alger, B.E.; Kano, M.; Lee, S.H.; Lovinger, D.M.; Ohno-Shosaku, T.; Watanabe, M. Weeding out bad waves: towards selective cannabinoid circuit control in epilepsy. Nature reviews. Neuroscience 2015, 16, 264–277. [Google Scholar] [CrossRef]
- Katona, I.; Freund, T.F. Endocannabinoid signaling as a synaptic circuit breaker in neurological disease. Nature medicine 2008, 14, 923–930. [Google Scholar] [CrossRef]
- Panikashvili, D.; Simeonidou, C.; Ben-Shabat, S.; Hanus, L.; Breuer, A.; Mechoulam, R.; Shohami, E. An endogenous cannabinoid (2-AG) is neuroprotective after brain injury. Nature 2001, 413, 527–531. [Google Scholar] [CrossRef] [PubMed]
- Hansen, H.H.; Schmid, P.C.; Bittigau, P.; Lastres-Becker, I.; Berrendero, F.; Manzanares, J.; Ikonomidou, C.; Schmid, H.H.; Fernández-Ruiz, J.J.; Hansen, H.S. Anandamide, but not 2-arachidonoylglycerol, accumulates during in vivo neurodegeneration. Journal of neurochemistry 2001, 78, 1415–1427. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Ruiz, J.; Moro, M.A.; Martínez-Orgado, J. Cannabinoids in Neurodegenerative Disorders and Stroke/Brain Trauma: From Preclinical Models to Clinical Applications. Neurotherapeutics: the journal of the American Society for Experimental NeuroTherapeutics 2015, 12, 793–806. [Google Scholar] [CrossRef] [PubMed]
- Marsicano, G.; Goodenough, S.; Monory, K.; Hermann, H.; Eder, M.; Cannich, A.; Azad, S.C.; Cascio, M.G.; Gutiérrez, S.O.; van der Stelt, M.; López-Rodriguez, M.L.; Casanova, E.; Schütz, G.; Zieglgänsberger, W.; Di Marzo, V.; Behl, C.; Lutz, B. CB1 cannabinoid receptors and on-demand defense against excitotoxicity. Science 2003, 302, 84–88. [Google Scholar] [CrossRef]
- Wallace, M.J.; Blair, R.E.; Falenski, K.W.; Martin, B.R.; DeLorenzo, R.J. The endogenous cannabinoid system regulates seizure frequency and duration in a model of temporal lobe epilepsy. The Journal of pharmacology and experimental therapeutics 2003, 307, 129–137. [Google Scholar] [CrossRef]
- Ludányi, A.; Eross, L.; Czirják, S.; Vajda, J.; Halász, P.; Watanabe, M.; Palkovits, M.; Maglóczky, Z.; Freund, T.F.; Katona, I. Downregulation of the CB1 cannabinoid receptor and related molecular elements of the endocannabinoid system in epileptic human hippocampus. The Journal of neuroscience: the official journal of the Society for Neuroscience 2008, 28, 2976–2990. [Google Scholar] [CrossRef]
- Romigi, A.; Bari, M.; Placidi, F.; Marciani, M.G.; Malaponti, M.; Torelli, F.; Izzi, F.; Prosperetti, C.; Zannino, S.; Corte, F.; Chiaramonte, C.; Maccarrone, M. Cerebrospinal fluid levels of the endocannabinoid anandamide are reduced in patients with untreated newly diagnosed temporal lobe epilepsy. Epilepsia 2010, 51, 768–772. [Google Scholar] [CrossRef]
- Wallace, M.J.; Wiley, J.L.; Martin, B.R.; DeLorenzo, R.J. Assessment of the role of CB1 receptors in cannabinoid anticonvulsant effects. European journal of pharmacology 2001, 428, 51–57. [Google Scholar] [CrossRef]
- Wallace, M.J.; Martin, B.R.; DeLorenzo, R.J. Evidence for a physiological role of endocannabinoids in the modulation of seizure threshold and severity. European journal of pharmacology 2002, 452, 295–301. [Google Scholar] [CrossRef]
- Shafaroodi, H.; Samini, M.; Moezi, L.; Homayoun, H.; Sadeghipour, H.; Tavakoli, S.; Hajrasouliha, A.R.; Dehpour, A.R. The interaction of cannabinoids and opioids on pentylenetetrazole-induced seizure threshold in mice. Neuropharmacology 2004, 47, 390–400. [Google Scholar] [CrossRef]
- Blair, R.E.; Deshpande, L.S.; Sombati, S.; Falenski, K.W.; Martin, B.R.; DeLorenzo, R.J. Activation of the cannabinoid type-1 receptor mediates the anticonvulsant properties of cannabinoids in the hippocampal neuronal culture models of acquired epilepsy and status epilepticus. The Journal of pharmacology and experimental therapeutics 2006, 317, 1072–1078. [Google Scholar] [CrossRef] [PubMed]
- Jensen, K.R.; Berthoux, C.; Nasrallah, K.; Castillo, P.E. Multiple cannabinoid signaling cascades powerfully suppress recurrent excitation in the hippocampus. Proceedings of the National Academy of Sciences of the United States of America 2021, 118, e2017590118. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, R. On the application of cannabis in paediatrics and epileptology. Neuro endocrinology letters 2004, 25, 40–44. [Google Scholar] [PubMed]
- De Petrocellis, L.; Vellani, V.; Schiano-Moriello, A.; Marini, P.; Magherini, P.C.; Orlando, P.; Di Marzo, V. Plant-derived cannabinoids modulate the activity of transient receptor potential channels of ankyrin type-1 and melastatin type-8. The Journal of pharmacology and experimental therapeutics 2008, 325, 1007–1015. [Google Scholar] [CrossRef]
- Muller, C.; Morales, P.; Reggio, P.H. Cannabinoid Ligands Targeting TRP Channels. Frontiers in molecular neuroscience 2019, 11, 487. [Google Scholar] [CrossRef]
- Qin, N.; Neeper, M.P.; Liu, Y.; Hutchinson, T.L.; Lubin, M.L.; Flores, C.M. TRPV2 is activated by cannabidiol and mediates CGRP release in cultured rat dorsal root ganglion neurons. The Journal of neuroscience: the official journal of the Society for Neuroscience 2008, 28, 6231–6238. [Google Scholar] [CrossRef]
- Ross, H.R.; Napier, I.; Connor, M. Inhibition of recombinant human T-type calcium channels by Delta9-tetrahydrocannabinol and cannabidiol. The Journal of biological chemistry 2008, 283, 16124–16134. [Google Scholar] [CrossRef]
- Talley, E.M.; Cribbs, L.L.; Lee, J.H.; Daud, A.; Perez-Reyes, E.; Bayliss, D.A. Differential distribution of three members of a gene family encoding low voltage-activated (T-type) calcium channels. The Journal of neuroscience: the official journal of the Society for Neuroscience 1999, 19, 1895–1911. [Google Scholar] [CrossRef]
- Huguenard, J.R. Low-threshold calcium currents in central nervous system neurons. Annual review of physiology 1996, 58, 329–348. [Google Scholar] [CrossRef]
- Abood, M.E.; Rizvi, G.; Sallapudi, N.; McAllister, S.D. Activation of the CB1 cannabinoid receptor protects cultured mouse spinal neurons against excitotoxicity. Neuroscience letters 2001, 309, 197–201. [Google Scholar] [CrossRef]
- van der Stelt, M.; Veldhuis, W.B.; Bär, P.R.; Veldink, G.A.; Vliegenthart, J.F.; Nicolay, K. Neuroprotection by Delta9-tetrahydrocannabinol, the main active compound in marijuana, against ouabain- induced in vivo excitotoxicity. The Journal of neuroscience: the official journal of the Society for Neuroscience 2001, 21, 6475–6479. [Google Scholar] [CrossRef] [PubMed]
- El-Remessy, A.B.; Khalil, I.E.; Matragoon, S.; Abou-Mohamed, G.; Tsai, N.J.; Roon, P.; Caldwell, R.B.; Caldwell, R.W.; Green, K.; Liou, G.I. Neuroprotective effect of (-)Delta9-tetrahydrocannabinol and cannabidiol in N-methyl-D-aspartate-induced retinal neurotoxicity: involvement of peroxynitrite. The American journal of pathology 2003, 163, 1997–2008. [Google Scholar] [CrossRef] [PubMed]
- Mechoulam, R.; Panikashvili, D.; Shohami, E. Cannabinoids and brain injury: therapeutic implications. Trends in molecular medicine 2002, 8, 58–61. [Google Scholar] [CrossRef]
- Gilbert, G.L.; Kim, H.J.; Waataja, J.J.; Thayer, S.A. Delta9-tetrahydrocannabinol protects hippocampal neurons from excitotoxicity. Brain research 2007, 1128, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Nagayama, T.; Sinor, A.D.; Simon, R.P.; Chen, J.; Graham, S.H.; Jin, K.; Greenberg, D.A. Cannabinoids and neuroprotection in global and focal cerebral ischemia and in neuronal cultures. The Journal of neuroscience: the official journal of the Society for Neuroscience 1999, 19, 2987–2995. [Google Scholar] [CrossRef]
- Zani, A.; Braida, D.; Capurro, V.; Sala, M. Delta9-tetrahydrocannabinol (THC) and AM 404 protect against cerebral ischaemia in gerbils through a mechanism involving cannabinoid and opioid receptors. British journal of pharmacology 2007, 152, 1301–1311. [Google Scholar] [CrossRef]
- Hampson, A.J.; Grimaldi, M.; Axelrod, J.; Wink, D. Cannabidiol and (-)Delta9-tetrahydrocannabinol are neuroprotective antioxidants. Proceedings of the National Academy of Sciences of the United States of America 1998, 95, 8268–8273. [Google Scholar] [CrossRef]
- Steindel, F.; Lerner, R.; Häring, M.; Ruehle, S.; Marsicano, G.; Lutz, B.; Monory, K. Neuron-type specific cannabinoid-mediated G protein signalling in mouse hippocampus. Journal of neurochemistry 2013, 124, 795–807. [Google Scholar] [CrossRef]
- Busquets-Garcia, A.; Bains, J.; Marsicano, G. CB1 Receptor Signaling in the Brain: Extracting Specificity from Ubiquity. Neuropsychopharmacology: official publication of the American College of Neuropsychopharmacology 2018, 43, 4–20. [Google Scholar] [CrossRef]
- Katona, I.; Sperlágh, B.; Sík, A.; Käfalvi, A.; Vizi, E.S.; Mackie, K.; Freund, T.F. Presynaptically located CB1 cannabinoid receptors regulate GABA release from axon terminals of specific hippocampal interneurons. The Journal of neuroscience: the official journal of the Society for Neuroscience 1999, 19, 4544–4558. [Google Scholar] [CrossRef]
- Adams, R.; Pease, D.C.; Clark, J.H. Isolation of cannabinol, cannabidiol, and quebrachitrol from red oil of Minnesota wild hemp. J Am Chem Soc 1940, 62, 2194–2196. [Google Scholar]
- Crippa, J.A.; Guimarães, F.S.; Campos, A.C.; Zuardi, A.W. Translational Investigation of the Therapeutic Potential of Cannabidiol (CBD): Toward a New Age. Frontiers in immunology 2018, 9, 2009. [Google Scholar] [CrossRef] [PubMed]
- Devinsky, O.; Cross, J.H.; Laux, L.; Marsh, E.; Miller, I.; Nabbout, R.; Scheffer, I.E.; Thiele, E.A.; Wright, S. Cannabidiol in Dravet Syndrome Study Group Trial of Cannabidiol for Drug-Resistant Seizures in the Dravet Syndrome. The New England journal of medicine 2017, 376, 2011–2020. [Google Scholar] [CrossRef] [PubMed]
- Cunha, J.M.; Carlini, E.A.; Pereira, A.E.; Ramos, O.L.; Pimentel, C.; Gagliardi, R.; Sanvito, W.L.; Lander, N.; Mechoulam, R. Chronic administration of cannabidiol to healthy volunteers and epileptic patients. Pharmacology 1980, 21, 175–185. [Google Scholar] [CrossRef]
- Devinsky, O.; Marsh, E.; Friedman, D.; Thiele, E.; Laux, L.; Sullivan, J.; Miller, I.; Flamini, R.; Wilfong, A.; Filloux, F.; et al. Cannabidiol in patients with treatment-resistant epilepsy: an open-label interventional trial. The Lancet. Neurology 2016, 15, 270–278. [Google Scholar] [CrossRef]
- Tzadok, M.; Uliel-Siboni, S.; Linder, I.; Kramer, U.; Epstein, O.; Menascu, S.; Nissenkorn, A.; Yosef, O.B.; Hyman, E.; Granot, D.; Dor, M.; Lerman-Sagie, T.; Ben-Zeev, B. CBD-enriched medical cannabis for intractable pediatric epilepsy: The current Israeli experience. Seizure 2016, 35, 41–44. [Google Scholar] [CrossRef]
- Carlini, E.A.; Cunha, J.M. Hypnotic and antiepileptic effects of cannabidiol. Journal of clinical pharmacology 1981, 21, 417S–427S. [Google Scholar] [CrossRef]
- Consroe, P.; Wolkin, A. Cannabidiol--antiepileptic drug comparisons and interactions in experimentally induced seizures in rats. The Journal of pharmacology and experimental therapeutics 1977, 201, 26–32. [Google Scholar] [CrossRef]
- Jones, N.A.; Glyn, S.E.; Akiyama, S.; Hill, T.D.; Hill, A.J.; Weston, S.E.; Burnett, M.D.; Yamasaki, Y.; Stephens, G.J.; Whalley, B.J.; Williams, C.M. Cannabidiol exerts anti-convulsant effects in animal models of temporal lobe and partial seizures. Seizure 2012, 21, 344–352. [Google Scholar] [CrossRef]
- Mao, K.; You, C.; Lei, D.; Zhang, H. High dosage of cannabidiol (CBD) alleviates pentylenetetrazole- induced epilepsy in rats by exerting an anticonvulsive effect. International journal of clinical and experimental medicine 2015, 8, 8820–8827. [Google Scholar]
- Patra, P.H.; Barker-Haliski, M.; White, H.S.; Whalley, B.J.; Glyn, S.; Sandhu, H.; Jones, N.; Bazelot, M.; Williams, C.M.; McNeish, A.J. Cannabidiol reduces seizures and associated behavioral comorbidities in a range of animal seizure and epilepsy models. Epilepsia 2019, 60, 303–314. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, J.S.; Stella, N.; Catterall, W.A.; Westenbroek, R.E. Cannabidiol attenuates seizures and social deficits in a mouse model of Dravet syndrome. Proceedings of the National Academy of Sciences of the United States of America 2017, 114, 11229–11234. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.A.; Shekh-Ahmad, T.; Khalil, A.; Walker, M.C.; Ali, A.B. Cannabidiol exerts antiepileptic effects by restoring hippocampal interneuron functions in a temporal lobe epilepsy model. British journal of pharmacology 2018, 175, 2097–2115. [Google Scholar] [CrossRef] [PubMed]
- Scheffer, I.E.; Halford, J.J.; Miller, I.; Nabbout, R.; Sanchez-Carpintero, R.; Shiloh-Malawsky, Y.; Wong, M.; Zolnowska, M.; Checketts, D.; Dunayevich, E.; Devinsky, O. Add-on cannabidiol in patients with Dravet syndrome: Results of a long-term open-label extension trial. Epilepsia 2021, 62, 2505–2517. [Google Scholar] [CrossRef]
- Miller, I.; Scheffer, I.E.; Gunning, B.; Sanchez-Carpintero, R.; Gil-Nagel, A.; Perry, M.S.; Saneto, R.P.; Checketts, D.; Dunayevich, E.; Knappertz, V. GWPCARE2 Study Group Dose-Ranging Effect of Adjunctive Oral Cannabidiol vs Placebo on Convulsive Seizure Frequency in Dravet Syndrome: A Randomized Clinical Trial. JAMA neurology 2020, 77, 613–621. [Google Scholar] [CrossRef]
- Devinsky, O.; Patel, A.D.; Cross, J.H.; Villanueva, V.; Wirrell, E.C.; Privitera, M.; Greenwood, S.M.; Roberts, C.; Checketts, D.; VanLandingham, K.E.; Zuberi, S.M. GWPCARE3 Study Group Effect of Cannabidiol on Drop Seizures in the Lennox-Gastaut Syndrome. The New England journal of medicine 2018, 378, 1888–1897. [Google Scholar] [CrossRef]
- Thiele, E.A.; Bebin, E.M.; Bhathal, H.; Jansen, F.E.; Kotulska, K.; Lawson, J.A.; O'Callaghan, F.J.; Wong, M.; Sahebkar, F.; Checketts, D.; Knappertz, V. GWPCARE6 Study Group Add-on Cannabidiol Treatment for Drug-Resistant Seizures in Tuberous Sclerosis Complex: A Placebo-Controlled Randomized Clinical Trial. JAMA neurology 2021, 78, 285–292. [Google Scholar] [CrossRef]
- Rosenberg, E.C.; Chamberland, S.; Bazelot, M.; Nebet, E.R.; Wang, X.; McKenzie, S.; Jain, S.; Greenhill, S.; Wilson, M.; Marley, N.; et al. Cannabidiol modulates excitatory-inhibitory ratio to counter hippocampal hyperactivity. Neuron 2023, 111, 1282–1300.e8. [Google Scholar] [CrossRef]
- Sylantyev, S.; Jensen, T.P.; Ross, R.A.; Rusakov, D.A. Cannabinoid- and lysophosphatidylinositol- sensitive receptor GPR55 boosts neurotransmitter release at central synapses. Proceedings of the National Academy of Sciences of the United States of America 2013, 110, 5193–5198. [Google Scholar] [CrossRef]
- De Petrocellis, L.; Ligresti, A.; Moriello, A.S.; Allarà, M.; Bisogno, T.; Petrosino, S.; Stott, C.G.; Di Marzo, V. Effects of cannabinoids and cannabinoid-enriched Cannabis extracts on TRP channels and endocannabinoid metabolic enzymes. British journal of pharmacology 2011, 163, 1479–1494. [Google Scholar] [CrossRef]
- Bisogno, T.; Hanus, L.; De Petrocellis, L.; Tchilibon, S.; Ponde, D.E.; Brandi, I.; Moriello, A.S.; Davis, J.B.; Mechoulam, R.; Di Marzo, V. Molecular targets for cannabidiol and its synthetic analogues: effect on vanilloid VR1 receptors and on the cellular uptake and enzymatic hydrolysis of anandamide. British journal of pharmacology 2001, 134, 845–852. [Google Scholar] [CrossRef] [PubMed]
- Etemad, L.; Karimi, G.; Alavi, M.S.; Roohbakhsh, A. Pharmacological effects of cannabidiol by transient receptor potential channels. Life sciences 2022, 300, 120582. [Google Scholar] [CrossRef]
- Russo, E.B.; Burnett, A.; Hall, B.; Parker, K.K. Agonistic properties of cannabidiol at 5-HT1a receptors. Neurochemical research 2005, 30, 1037–1043. [Google Scholar] [CrossRef] [PubMed]
- Ahrens, J.; Demir, R.; Leuwer, M.; de la Roche, J.; Krampfl, K.; Foadi, N.; Karst, M.; Haeseler, G. The nonpsychotropic cannabinoid cannabidiol modulates and directly activates alpha-1 and alpha-1-Beta glycine receptor function. Pharmacology 2009, 83, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Ghovanloo, M.R.; Shuart, N.G.; Mezeyova, J.; Dean, R.A.; Ruben, P.C.; Goodchild, S.J. Inhibitory effects of cannabidiol on voltage-dependent sodium currents. The Journal of biological chemistry 2018, 293, 16546–16558. [Google Scholar] [CrossRef]
- Zhang, H.B.; Heckman, L.; Niday, Z.; Jo, S.; Fujita, A.; Shim, J.; Pandey, R.; Al Jandal, H.; Jayakar, S.; Barrett, L.B.; Smith, J.; Woolf, C.J.; Bean, B.P. Cannabidiol activates neuronal Kv7 channels. eLife 2022, 11, e73246. [Google Scholar] [CrossRef]
- Leweke, F.M.; Piomelli, D.; Pahlisch, F.; Muhl, D.; Gerth, C.W.; Hoyer, C.; Klosterkötter, J.; Hellmich, M.; Koethe, D. Cannabidiol enhances anandamide signaling and alleviates psychotic symptoms of schizophrenia. Translational psychiatry 2012, 2, e94. [Google Scholar] [CrossRef]
- Rakhshan, F.; Day, T.A.; Blakely, R.D.; Barker, E.L. Carrier-mediated uptake of the endogenous cannabinoid anandamide in RBL-2H3 cells. The Journal of pharmacology and experimental therapeutics 2000, 292, 960–967. [Google Scholar]
- Ben-Shabat, S.; Fride, E.; Sheskin, T.; Tamiri, T.; Rhee, M.H.; Vogel, Z.; Bisogno, T.; De Petrocellis, L.; Di Marzo, V.; Mechoulam, R. An entourage effect: inactive endogenous fatty acid glycerol esters enhance 2-arachidonoyl-glycerol cannabinoid activity. European journal of pharmacology 1998, 353, 23–31. [Google Scholar] [CrossRef]
- Wagner, H.; Ulrich-Merzenich, G. Synergy research: approaching a new generation of phytopharmaceuticals. Phytomedicine: international journal of phytotherapy and phytopharmacology 2009, 16, 97–110. [Google Scholar] [CrossRef]
- Brodie, M.J.; Czapinski, P.; Pazdera, L.; Sander, J.W.; Toledo, M.; Napoles, M.; Sahebkar, F.; Schreiber, A. GWEP1330 Study Group A Phase 2 Randomized Controlled Trial of the Efficacy and Safety of Cannabidivarin as Add-on Therapy in Participants with Inadequately Controlled Focal Seizures. Cannabis and cannabinoid research 2021, 6, 528–536. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.J.; Jones, N.A.; Smith, I.; Hill, C.L.; Williams, C.M.; Stephens, G.J.; Whalley, B.J. Voltage-gated sodium (NaV) channel blockade by plant cannabinoids does not confer anticonvulsant effects per se. Neuroscience letters 2014, 566, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Nachnani, R.; Raup-Konsavage, W.M.; Vrana, K.E. The Pharmacological Case for Cannabigerol. The Journal of pharmacology and experimental therapeutics 2021, 376, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Navarro, G.; Varani, K.; Reyes-Resina, I.; Sánchez de Medina, V.; Rivas-Santisteban, R.; Sánchez-Carnerero Callado, C.; Vincenzi, F.; Casano, S.; Ferreiro-Vera, C.; Canela, E.I.; Borea, P.A.; Nadal, X.; Franco, R. Cannabigerol Action at Cannabinoid CB1 and CB2 Receptors and at CB1-CB2 Heteroreceptor Complexes. Frontiers in pharmacology 2018, 9, 632. [Google Scholar] [CrossRef]
- Cascio, M.G.; Gauson, L.A.; Stevenson, L.A.; Ross, R.A.; Pertwee, R.G. Evidence that the plant cannabinoid cannabigerol is a highly potent alpha2-adrenoceptor agonist and moderately potent 5HT1A receptor antagonist. British journal of pharmacology 2010, 159, 129–141. [Google Scholar] [CrossRef]
- Gilsbach, R.; Hein, L. Are the pharmacology and physiology of α₂ adrenoceptors determined by α₂-heteroreceptors and autoreceptors respectively? British journal of pharmacology 2012, 165, 90–102. [Google Scholar] [CrossRef]
- Talley, E.M.; Rosin, D.L.; Lee, A.; Guyenet, P.G.; Lynch, K.R. Distribution of alpha 2A-adrenergic receptor-like immunoreactivity in the rat central nervous system. The Journal of comparative neurology 1996, 372, 111–134. [Google Scholar] [CrossRef]
- Wang, M.; Ramos, B.P.; Paspalas, C.D.; Shu, Y.; Simen, A.; Duque, A.; Vijayraghavan, S.; Brennan, A.; Dudley, A.; Nou, E.; Mazer, J.A.; McCormick, D.A.; Arnsten, A.F. Alpha2A-adrenoceptors strengthen working memory networks by inhibiting cAMP-HCN channel signaling in prefrontal cortex. Cell 2007, 129, 397–410. [Google Scholar] [CrossRef]
- Bickler, P.E.; Hansen, B.M. Alpha 2-adrenergic agonists reduce glutamate release and glutamate receptor-mediated calcium changes in hippocampal slices during hypoxia. Neuropharmacology 1996, 35, 679–687. [Google Scholar] [CrossRef]
- Riad, M.; Garcia, S.; Watkins, K.C.; Jodoin, N.; Doucet, E.; Langlois, X.; el Mestikawy, S.; Hamon, M.; Descarries, L. Somatodendritic localization of 5-HT1A and preterminal axonal localization of 5-HT1B serotonin receptors in adult rat brain. The Journal of comparative neurology 2000, 417, 181–194. [Google Scholar]
- Pertwee, R.G. The diverse CB1 and CB2 receptor pharmacology of three plant cannabinoids: delta9-tetrahydrocannabinol, cannabidiol and delta9-tetrahydrocannabivarin. British journal of pharmacology 2008, 153, 199–215. [Google Scholar] [CrossRef] [PubMed]
- Ghovanloo, M.R.; Effraim, P.R.; Tyagi, S.; Zhao, P.; Dib-Hajj, S.D.; Waxman, S.G. Functionally-selective inhibition of threshold sodium currents and excitability in dorsal root ganglion neurons by cannabinol. Communications biology 2024, 7, 120. [Google Scholar] [CrossRef] [PubMed]
- Sepulveda, D.E.; Vrana, K.E.; Kellogg, J.J.; Bisanz, J.E.; Desai, D.; Graziane, N.M.; Raup-Konsavage, W.M. The Potential of Cannabichromene (CBC) as a Therapeutic Agent. The Journal of pharmacology and experimental therapeutics 2024, 391, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Udoh, M.; Santiago, M.; Devenish, S.; McGregor, I.S.; Connor, M. Cannabichromene is a cannabinoid CB2 receptor agonist. British journal of pharmacology 2019, 176, 4537–4547. [Google Scholar] [CrossRef]
- Chávez, A.E.; Chiu, C.Q.; Castillo, P.E. TRPV1 activation by endogenous anandamide triggers postsynaptic long-term depression in dentate gyrus. Nature neuroscience 2010, 13, 1511–1518. [Google Scholar] [CrossRef]
- Anderson, L.L.; Ametovski, A.; Lin Luo, J.; Everett-Morgan, D.; McGregor, I.S.; Banister, S.D.; Arnold, J.C. Cannabichromene, Related Phytocannabinoids, and 5-Fluoro-cannabichromene Have Anticonvulsant Properties in a Mouse Model of Dravet Syndrome. ACS chemical neuroscience 2021, 12, 330–339. [Google Scholar] [CrossRef]
- Wang, Z.; Zheng, H.; Yang, H.; Song, H.; Lian, J.; Peng, C.; Wang, H.; Zhang, H.; Zheng, Y.; Wang, Q.; Lan, L.; Duan, G.; Ma, L.; Peng, X.; Huang, Z. Cannabichromene from full-spectrum hemp extract exerts acute anti-seizure effects through allosteric activation of GABAA receptors. Fundamental research 2024, 4, 1357–1364. [Google Scholar] [CrossRef]
- Guzmán-Gutiérrez, S.L.; Bonilla-Jaime, H.; Gómez-Cansino, R.; Reyes-Chilpa, R. Linalool and β-pinene exert their antidepressant-like activity through the monoaminergic pathway. Life sciences 2015, 128, 24–29. [Google Scholar] [CrossRef]
- Weston-Green, K.; Clunas, H.; Jimenez Naranjo, C. A Review of the Potential Use of Pinene and Linalool as Terpene-Based Medicines for Brain Health: Discovering Novel Therapeutics in the Flavours and Fragrances of Cannabis. Frontiers in psychiatry 2021, 12, 583211. [Google Scholar] [CrossRef]
- Kessler, A.; Sahin-Nadeem, H.; Lummis, S.C.; Weigel, I.; Pischetsrieder, M.; Buettner, A.; Villmann, C. GABA(A) receptor modulation by terpenoids from Sideritis extracts. Molecular nutrition & food research 2014, 58, 851–862. [Google Scholar] [CrossRef]
- Narusuye, K.; Kawai, F.; Matsuzaki, K.; Miyachi, E. Linalool suppresses voltage-gated currents in sensory neurons and cerebellar Purkinje cells. Journal of neural transmission 2005, 112, 193–203. [Google Scholar] [CrossRef]
- Gertsch, J.; Leonti, M.; Raduner, S.; Racz, I.; Chen, J.Z.; Xie, X.Q.; Altmann, K.H.; Karsak, M.; Zimmer, A. Beta-caryophyllene is a dietary cannabinoid. Proceedings of the National Academy of Sciences of the United States of America 2008, 105, 9099–9104. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.L.; Yang, Z.Y.; Fan, G.; Ren, J.N.; Yin, K.J.; Pan, S.Y. Antidepressant-like Effect of Citrus sinensis (L.) Osbeck Essential Oil and Its Main Component Limonene on Mice. Journal of agricultural and food chemistry 2019, 67, 13817–13828. [Google Scholar] [CrossRef] [PubMed]
- Vieira, A.J.; Beserra, F.P.; Souza, M.C.; Totti, B.M.; Rozza, A.L. Limonene: Aroma of innovation in health and disease. Chemico-biological interactions 2018, 283, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Seo, S.; Song, Y.; Gu, S.M.; Min, H.K.; Hong, J.T.; Cha, H.J.; Yun, J. D-limonene Inhibits Pentylenetetrazole-Induced Seizure via Adenosine A2A Receptor Modulation on GABAergic Neuronal Activity. International journal of molecular sciences 2020, 21, 9277. [Google Scholar] [CrossRef]
- Surendran, S.; Qassadi, F.; Surendran, G.; Lilley, D.; Heinrich, M. Myrcene-What Are the Potential Health Benefits of This Flavouring and Aroma Agent? Frontiers in nutrition 2021, 8, 699666. [Google Scholar] [CrossRef]
- Viana, G.S.; do Vale, T.G.; Silva, C.M.; Matos, F.J. Anticonvulsant activity of essential oils and active principles from chemotypes of Lippia alba (Mill.) N.E. Brown. Biological & pharmaceutical bulletin 2000, 23, 1314–1317. [Google Scholar] [CrossRef]
- Ueno, H.; Shimada, A.; Suemitsu, S.; Murakami, S.; Kitamura, N.; Wani, K.; Takahashi, Y.; Matsumoto, Y.; Okamoto, M.; Ishihara, T. Alpha-pinene and dizocilpine (MK-801) attenuate kindling development and astrocytosis in an experimental mouse model of epilepsy. IBRO reports 2020, 9, 102–114. [Google Scholar] [CrossRef]
- Felipe, C.F.B.; Albuquerque, A.M.S.; de Pontes, J.L.X.; de Melo JÍ, V.; Rodrigues, T.C.M.L.; de Sousa, A.M.P.; Monteiro, ÁB.; Ribeiro, A.E.D.S.; Lopes, J.P.; de Menezes, I.R.A.; de Almeida, R.N. Comparative study of alpha- and beta-pinene effect on PTZ-induced convulsions in mice. Fundamental & clinical pharmacology 2019, 33, 181–190. [Google Scholar] [CrossRef]
- Yang, H.; Woo, J.; Pae, A.N.; Um, M.Y.; Cho, N.C.; Park, K.D.; Yoon, M.; Kim, J.; Lee, C.J.; Cho, S. α-Pinene, a Major Constituent of Pine Tree Oils, Enhances Non-Rapid Eye Movement Sleep in Mice through GABAA-benzodiazepine Receptors. Molecular pharmacology 2016, 90, 530–539. [Google Scholar] [CrossRef]
- Miyazawa, M.; Yamafuji, C. Inhibition of acetylcholinesterase activity by bicyclic monoterpenoids. Journal of agricultural and food chemistry 2005, 53, 1765–1768. [Google Scholar] [CrossRef]
- Poorthuis, R.B.; Enke, L.; Letzkus, J.J. Cholinergic circuit modulation through differential recruitment of neocortical interneuron types during behaviour. The Journal of physiology 2014, 592, 4155–4164. [Google Scholar] [CrossRef]
- Martinello, K.; Mascia, A.; Casciato, S.; Di Gennaro, G.; Esposito, V.; Zoli, M.; Gotti, C.; Fucile, S. α4β2* nicotinic acetylcholine receptors drive human temporal glutamate/GABA balance toward inhibition. The Journal of physiology 2025, 603, 1645–1662. [Google Scholar] [CrossRef]
- Takács, V.T.; Cserép, C.; Schlingloff, D.; Pósfai, B.; Szőnyi, A.; Sos, K.E.; Környei, Z.; Dénes, Á; Gulyás, A.I.; Freund, T.F.; Nyiri, G. Co-transmission of acetylcholine and GABA regulates hippocampal states. Nature communications 2018, 9, 2848. [Google Scholar] [CrossRef]
- Zahra, A.; Wang, Y.; Wang, Q.; Wu, J. Shared Etiology in Autism Spectrum Disorder and Epilepsy with Functional Disability. Behavioural neurology 2022, 2022, 5893519. [Google Scholar] [CrossRef]
- Jensen, K.R. Supporting Cognitive Flexibility in Autism: Mechanistic Insights into Cannabinoid–Terpene Interactions. Preprints 2026. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



