Submitted:
17 April 2026
Posted:
20 April 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
Material
Collection and Authentication of Plants Material
Methods
Preparation of Polyherbal Extract
| Plant | Phytochemicals | Pharmacological properties | Reference |
|---|---|---|---|
| Withania somnifera | Isopellertierine, anferine, withanolides, withaferins. | Antihyperglycemic, Anti-inflammatory, Antioxidant |
[13] [14] |
| Gymnema sylvestre | Hentriacontane, pentatriacontane, flavones, gymnemasaponins, and gymnemic acids. | Antidiabetic Anti-inflammatory |
[15] [16] |
| Azadirachta indica | Azadirachtin, nimbin, nimbidin, gedunin, salannin, quercetin, beta-sitosterol, limonoids, flavonoids, triterpenoids, nimbolide, margolone, cyclic peptides | Anti-inflammatory, Antidiabetic, Antioxidant, | [17] [18] |
| Tinospora cordifolia | Aporphine alkaloids, tinosporides, berberine, palmatine, magnoflorine, tinosporine, tetrahydropalmatine, isocolumbine,and choline | Antidiabetic, Anti-inflammatory, Antioxidant |
[19] [20] [21] |
| Syzygium cumini | Jambosine, anthocyanins, gallic acid, ellagic acid, corilagin, quercetin, kaempferol, myricetin, oleanolic acid, betulinic acid, friedelin, β-sitosterol, triterpenoids, tannins, flavonoids. | Antidiabetic, Antioxidant, Anti-inflammatory, |
[22] |
| Emblica officinalis | Phyllaemblic acid, quercetin, kaempferol, luteolin, apigenin, tannins, gallic acid, ellagic acid, geraniin, and vitamic C | Antioxidant, Antidiabetic, Anti-inflammatory. |
[23] [24] |
| Pterocorous marsupium | Marsupsin, pterostilbene, liquirtigenin, hydroxyflavanone, P-hydroxybenzaldehyde | Antioxidant, Antidiabetic, Anti-inflammatory. |
[25] [26] |
In Vivo Studies
Experimental Animals and Study Design
Behavioral Estimations
Biochemical Parameters in Blood and Tissue Homogenate
Gastric Emptying Time
Histopathological Assessment of the Sciatic Nerve
In Vitro Studies
MIN6 Cell Culture
Cell Viability Assay
Insulin Secretion Assay
Statistical Analysis
Results
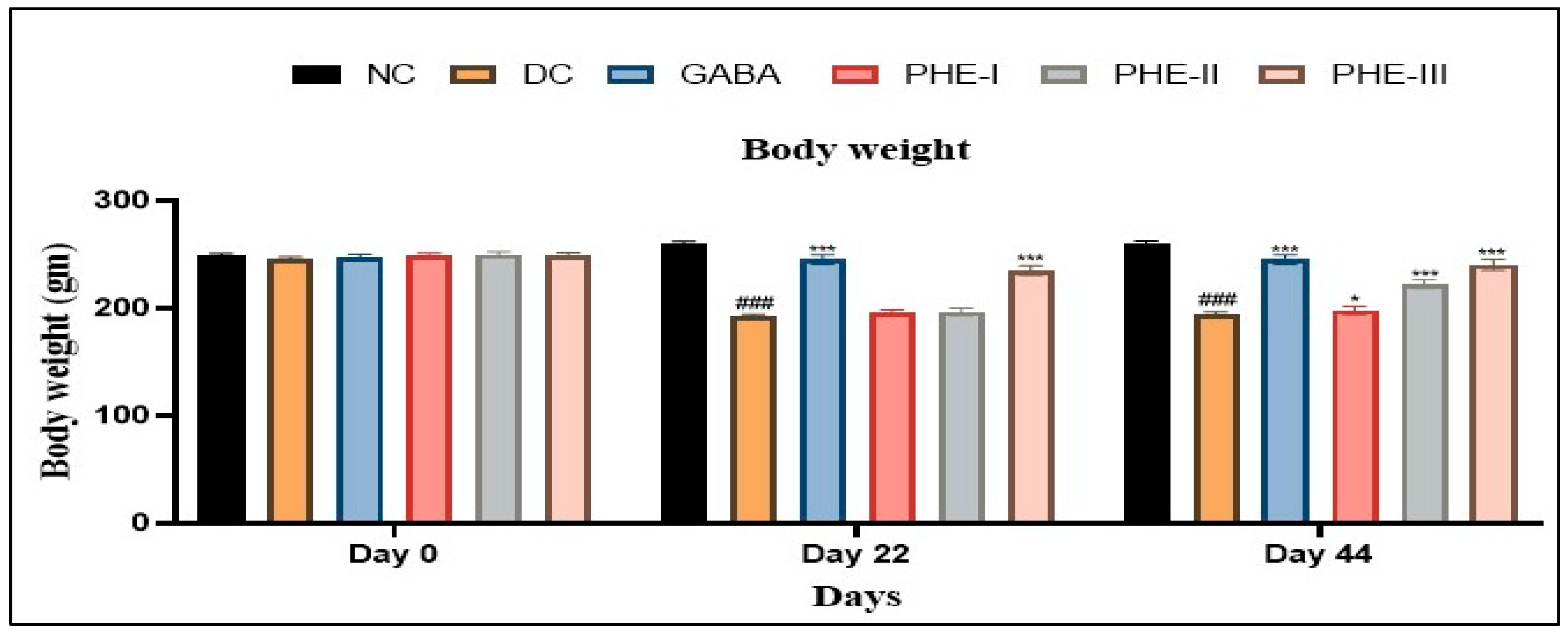
Effect of Aqueous Polyherbal Extract on Body Weight
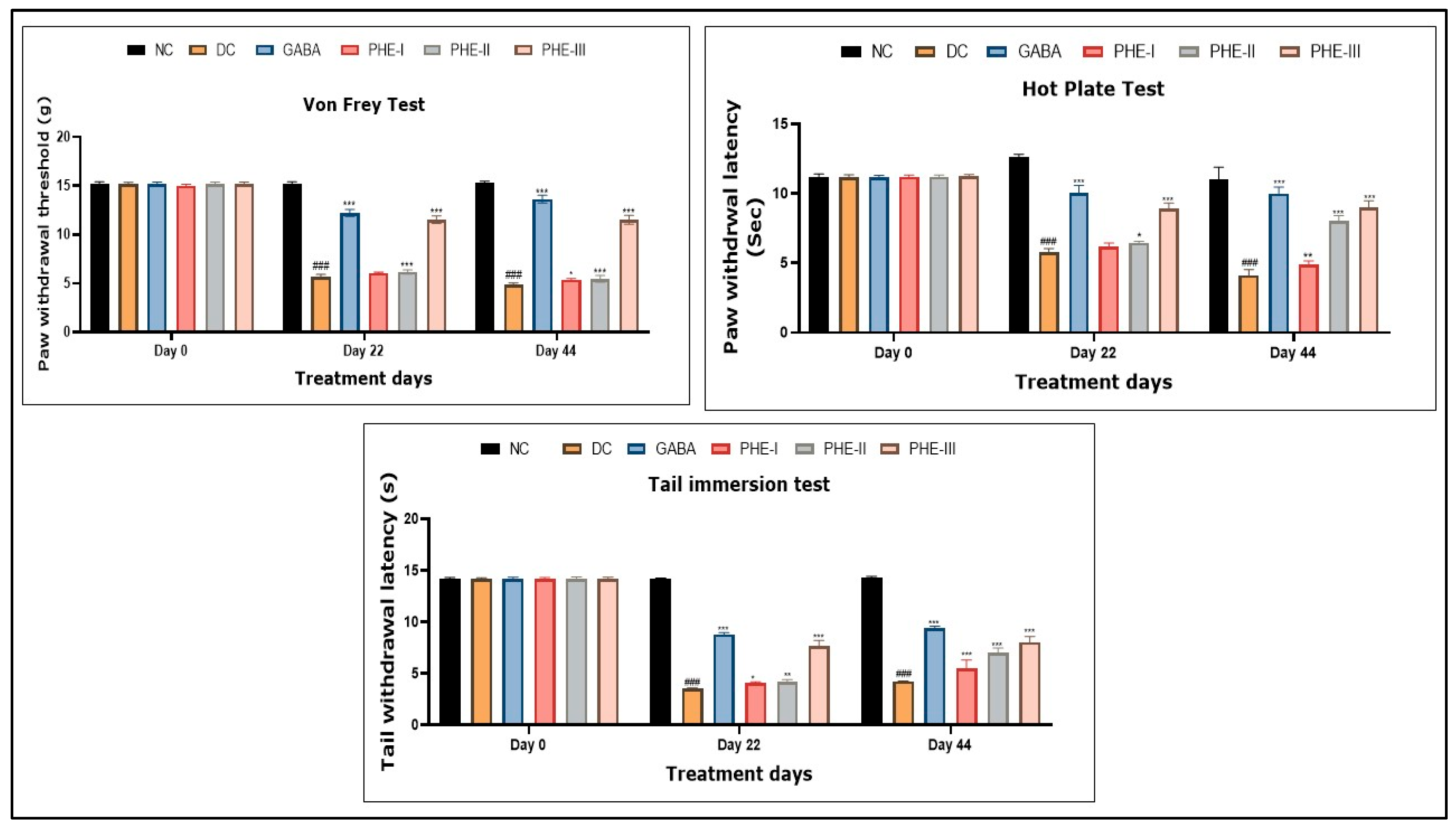
Effect of Aqueous Polyherbal Extract on Mechano-Tactile Allodynia (Von Frey Hair Test) in Diabetic Rats
Effect of Aqueous Polyherbal Extract on Thermal Hyperalgesia (Hot Plate Test and Tail Immersion Test) in Diabetic Rats
Effect of Aqueous Polyherbal Extract on Serum Biochemical Parameters in Diabetic Rats
Effect of Aqueous Polyherbal Extract on Oxidative Stress and Inflammatory Marker in Diabetic Rats
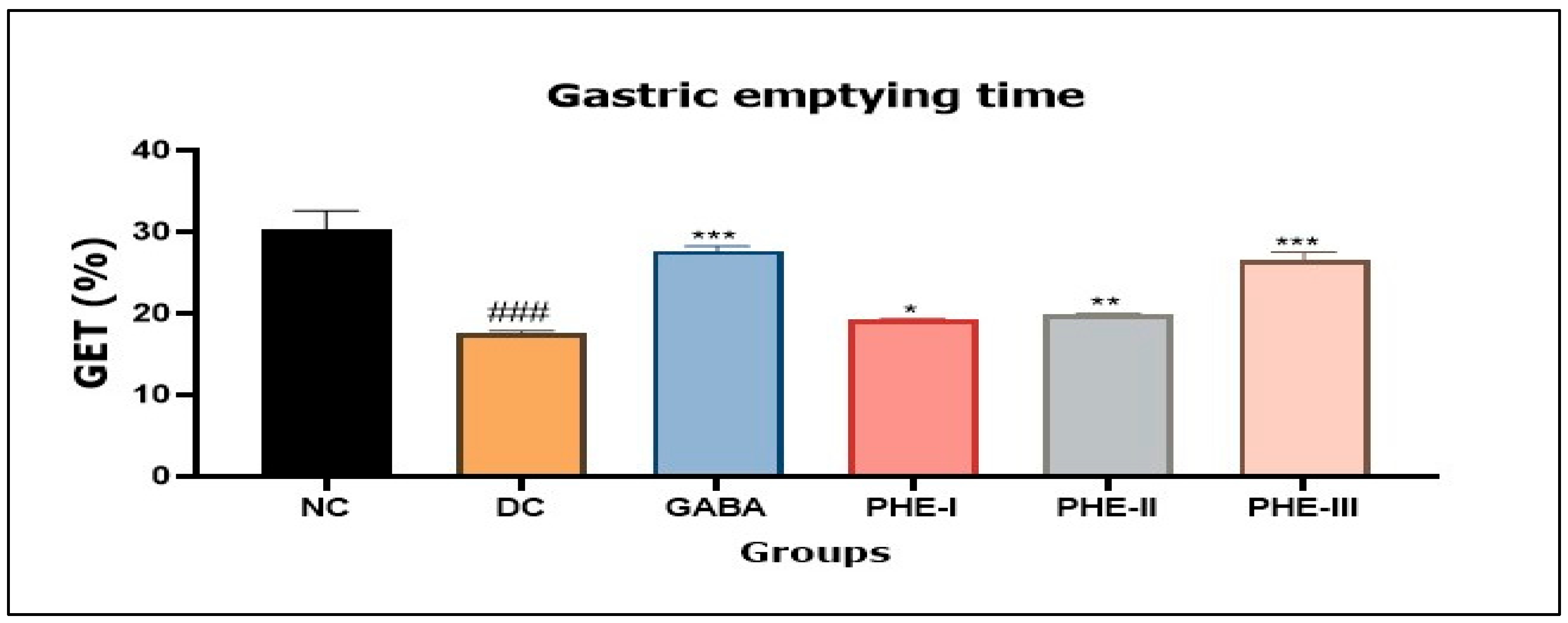
Effect of Aqueous Polyherbal Extract on Gastric Emptying Time in Diabetic Rats
Effects of Aqueous Polyherbal Extract on Cell Viability Assays in MIN6 Cell Line
Effects of Aqueous Polyherbal Extract on Insulin Secretion Assays (MIN6 Cell Line)
| Sr no. | Compounds | Concentration | C Peptide secretion (pmol/ml) | Insulin secretion (µU/ml) |
|---|---|---|---|---|
| 1 | Control | 10 μM | 01.46 | |
| 2 | Standard (Glibenclamide) |
10 μM | 9.97 | 3.32 µU/ml |
| 3 | Polyherbal extract | 1000 µg/mL | 6.47 | 2.12µU/ml |
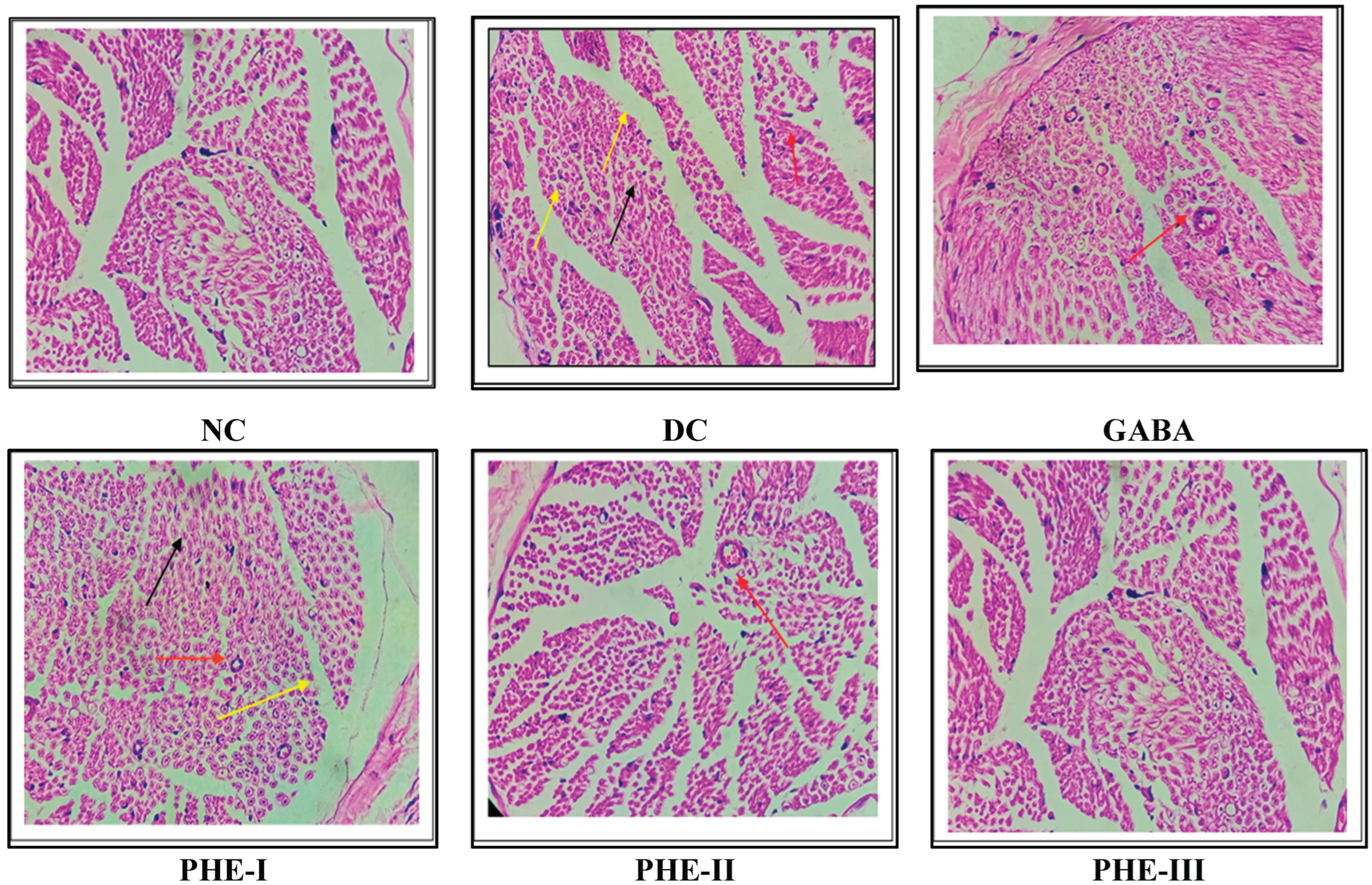
Effect of Aqueous Polyherbal Extract on Sciatic Nerve Histology in Diabetic Rats
Discussion
References
- Pop-Busui, R.; Ang, L.; Boulton, A.J.; Feldman, E.L.; Marcus, R.L.; Mizokami-Stout, K.; Singleton, J.R.; Ziegler, D. Diagnosis and treatment of painful diabetic peripheral neuropathy. 2020, 1–32. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.B.; Qiu, M.Z.; Zhang, D.Y. Apigenin ameliorates diabetic neuropathy in rats by modulating the TLR4/MyD88 signaling pathway. Asian Pacific Journal of Tropical Biomedicine 2023, 13(11), 469–78. [Google Scholar] [CrossRef]
- Smith, S.; Normahani, P.; Lane, T.; Hohenschurz-Schmidt, D.; Oliver, N.; Davies, A.H. Pathogenesis of distal symmetrical polyneuropathy in diabetes. Life 2022, 12(7), 1074. [Google Scholar] [CrossRef] [PubMed]
- Arora, K.; Tomar, P.C.; Mohan, V. Diabetic neuropathy: an insight on the transition from synthetic drugs to herbal therapies. Journal of Diabetes & Metabolic Disorders 2021, 20(2), 1773–84. [Google Scholar] [CrossRef]
- Kumar, A.; Deshpande, S.; Deshpande, V. Gas Chromatography-Mass Spectrometry (GC-MS)-Based Phytochemical Analysis and Potential Therapeutic Evaluation of Vajra Guggulu in Diabetic Neuropathy. 2025. [Google Scholar] [CrossRef]
- Mehmood, M.H.; Malik, A.; Akhtar, M.S.; Haider, G.; Gilani, A.H. Antihyperglycaemic, antihyperlipidaemic and antihypertensive effect of a polyherbal formulation in alloxan-.
- Induced diabetic rats. Farmacia ORIGINAL ARTICLE. 2020, 68(5), 882–90. [CrossRef]
- Suvarna, R.; Shenoy, R.P.; Hadapad, B.S.; Nayak, A.V. Effectiveness of polyherbal formulations for the treatment of type 2 Diabetes mellitus-A systematic review and meta-analysis. Journal of Ayurveda and integrative medicine 2021, 12(1), 213–22. [Google Scholar] [CrossRef] [PubMed]
- Başaran, N.; Paslı, D.; Başaran, A.A. Unpredictable adverse effects of herbal products. Food and Chemical Toxicology 2022, 159, 112762. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Negi, A.S.; Chauhan, A.; Semwal, R.; Kumar, R.; Semwal, R.B.; Singh, R.; Joshi, T.; Chandra, S.; Joshi, S.K.; Semwal, D.K. Formulation and evaluation of SGLT2 inhibitory effect of a polyherbal mixture inspired from Ayurvedic system of medicine. Journal of Traditional and Complementary Medicine 2022, 12(5), 477–87. [Google Scholar] [CrossRef] [PubMed]
- Anusha, R.; Akhila, N.; Nikhitha, J.; Harish, K.; Abdull, R.S.; Inamul, S.Y. Formulation and evaluation of herbal hair serum: a review. International Journal of Basic & Clinical Pharmacology 2023, 12(5), 759–66. [Google Scholar] [CrossRef]
- Reddy, K.S.; Sudheer, A.; Pradeepkumar, B.; Reddy, C.S. Effect of a polyherbal formulation in streptozotocin-induced diabetic nephropathy in wistar rats. Indian Journal of Pharmacology 2019, 51(5), 330–6. [Google Scholar] [CrossRef] [PubMed]
- GUEHAIRIA Mohamed, T.H. Effect of Withania somnifera (Ashwagandha) on biochemical and neurobehavioral disturbances induced by chronic restraint stress in an animal model . Doctoral dissertation, Université Echahid Chikh Larbi Tebessi-Tebessa, 2023. [Google Scholar]
- Hussain, A.; Aslam, B.; Muhammad, F.; Faisal, M.N. In vitro antioxidant activity and in vivo anti-inflammatory effect of Ricinus communis (L.) and Withania somnifera (L.) hydroalcoholic extracts in rats. Brazilian Archives of Biology and Technology 2021, 64, e21200783. [Google Scholar] [CrossRef]
- Hashem, H.A.; Nabil, Z.I.; El-Hak, H.N. Ashwagandha root extract’s phenolic compound counteracts alloxan’s effects on oxidative stress, inflammatory cytokines, and peripheral neuropathy in rats. Comparative Clinical Pathology 2023, 32(5), 867–80. [Google Scholar] [CrossRef]
- Gunasekaran, V.; Srinivasan, S.; Rani, S.S. Potential antioxidant and antimicrobial activity of Gymnema sylvestre related to diabetes. J. Med. Plants. 2019, 7(2), 05–11. [Google Scholar]
- Kang, M.H.; Lee, M.S.; Choi, M.K.; Min, K.S.; Shibamoto, T. Hypoglycemic activity of Gymnema sylvestre extracts on oxidative stress and antioxidant status in diabetic rats. Journal of agricultural and food chemistry 2012, 60(10), 2517–24. [Google Scholar] [CrossRef] [PubMed]
- Satyanarayana, K.; Sravanthi, K.; Shaker, I.A.; Ponnulakshmi, R. Molecular approach to identify antidiabetic potential of Azadirachta indica. Journal of Ayurveda and integrative medicine 2015, 6(3), 165. [Google Scholar] [CrossRef] [PubMed]
- Rahmani, A.; Almatroudi, A.; Alrumaihi, F.; Khan, A. Pharmacological and therapeutic potential of neem (Azadirachta indica). Pharmacognosy Reviews 2018, 12(24), 250–5. [Google Scholar] [CrossRef]
- Sharma, R.; Amin, H.; Prajapati, P.K. Antidiabetic claims of Tinospora cordifolia (Willd.) Miers: critical appraisal and role in therapy. Asian Pacific Journal of Tropical Biomedicine 2015, 5(1), 68–78. [Google Scholar] [CrossRef]
- Philip, S.; Tom, G.; Vasumathi, A.V. Evaluation of the anti-inflammatory activity of Tinospora cordifolia (Willd.) Miers chloroform extract–a preclinical study. Journal of Pharmacy and Pharmacology 2018, 70(8), 1113–25. [Google Scholar] [CrossRef] [PubMed]
- Kannadhasan, R.; Venkataraman, S. In vitro capacity and in vivo antioxidant potency of sedimental extract of Tinospora cordifolia in streptozotocin induced type 2 diabetes. Avicenna journal of phytomedicine 2013, 3(1), 7. [Google Scholar] [PubMed]
- Franco, R.R.; Zabisky, L.F.; de Lima Júnior, J.P.; Alves, V.H.; Justino, A.B.; Saraiva, A.L.; Goulart, L.R.; Espindola, F.S. Antidiabetic effects of Syzygium cumini leaves: A non-hemolytic plant with potential against process of oxidation, glycation, inflammation and digestive enzymes catalysis. Journal of Ethnopharmacology 2020, 261, 113132. [Google Scholar] [CrossRef] [PubMed]
- Nain, P.; Saini, V.; Sharma, S.; Nain, J. Antidiabetic and antioxidant potential of Emblica officinalis Gaertn. leaves extract in streptozotocin-induced type-2 diabetes mellitus (T2DM) rats. Journal of ethnopharmacology 2012, 142(1), 65–71. [Google Scholar] [CrossRef] [PubMed]
- Middha, S.K.; Goyal, A.K.; Lokesh, P.; Yardi, V.; Mojamdar, L.; Keni, D.S.; Babu, D.; Usha, T. Toxicological evaluation of Emblica officinalis fruit extract and its anti-inflammatory and free radical scavenging properties. Pharmacognosy Magazine 2015, 11 (Suppl 3), S427. [Google Scholar] [CrossRef] [PubMed]
- Pant, D.R.; Pant, N.D.; Yadav, U.N.; Khanal, D.P. Phytochemical screening and study of antioxidant, antimicrobial, antidiabetic, anti-inflammatory and analgesic activities of extracts from stem wood of Pterocarpus marsupium Roxburgh. Journal of Intercultural Ethnopharmacology 2017, 6(2), 170. [Google Scholar] [CrossRef] [PubMed]
- Perera, H. Antidiabetic effects of Pterocarpus marsupium (Gammalu). Eur. J. Med. Plants. 2016, 13(4), 1–4. [Google Scholar] [CrossRef]
- Dhanshree, S.; Aman, U.; Chandrashe, U. Neuroprotective effects of protocatechuic acid in diabetes induced neuropathic pain. American Journal of Biochemistry and Molecular Biology 2017, 7(3), 111–7. [Google Scholar] [CrossRef]
- Krishnamurthy, B.; Prashanth, A.; Sathyanarayanan, A. Comparison of the effects of zonisamide and flupirtine on paclitaxel induced peripheral neuropathy in rats. J Clin Diagno Res. 2018, 12(1). [Google Scholar] [CrossRef]
- Bakare, A.O.; Owoyele, B.V. Bromelain reduced pro-inflammatory mediators as a common pathway that mediate antinociceptive and anti-anxiety effects in sciatic nerve ligated Wistar rats. Scientific Reports 2021, 11(1), 289. [Google Scholar] [CrossRef] [PubMed]
- Slater, T.F.; Sawyer, B.C. The stimulatory effects of carbon tetrachloride and other halogenoalkanes on peroxidative reactions in rat liver fractions in vitro. General features of the systems used. Biochemical Journal 1971, 123(5), 805–14. [Google Scholar] [CrossRef] [PubMed]
- Khan, R.; Upaganlawar, A.B.; Upasani, C. Protective effects of Dioscorea alata L. in aniline exposure-induced spleen toxicity in rats: a biochemical study. Toxicology International 2014, 21(3), 294. [Google Scholar] [CrossRef] [PubMed]
- Bagheri, S.; Sarabi, M.M.; Gholami, M.; Assadollahi, V.; Khorramabadi, R.M.; Moradi, F.H.; Ahmadvand, H. D-limonene in diabetic rats. Journal of Renal Injury Prevention 2021, 10(3), e24. [Google Scholar] [CrossRef]
- Hadwan, M.H. Simple spectrophotometric assay for measuring catalase activity in biological tissues. BMC biochemistry 2018, 19, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Amira, S.; Soufane, S.; Gharzouli, K. Effect of sodium fluoride on gastric emptying and intestinal transit in mice. Experimental and Toxicologic Pathology 2005, 57(1), 59–64. [Google Scholar] [CrossRef] [PubMed]
- Lv, J.; Cao, L.; Zhang, R.; Bai, F.; Wei, P. A curcumin derivative J147 ameliorates diabetic peripheral neuropathy in streptozotocin (STZ)-induced DPN rat models through negative regulation AMPK on TRPA1. Acta cirurgica brasileira 2018, 33(06), 533–41. [Google Scholar] [CrossRef] [PubMed]
- Ishihara, H.; Asano, T.; Tsukuda, K.; Katagiri, H.; Inukai, K.; Anai, M.; Kikuchi, M.; Yazaki, Y.; Miyazaki, J.I.; Oka, Y. Pancreatic beta cell line MIN6 exhibits characteristics of glucose metabolism and glucose-stimulated insulin secretion similar to those of normal islets. Diabetologia 1993, 36, 1139–45. [Google Scholar] [CrossRef] [PubMed]
- Mehta, B.K.; Nerkar, D.; Banerjee, S. Characterization of peripheral neuropathy in rat model of type 2 diabetes. Indian J Pharm Educ Res. 2017, 51(51), 92–101. [Google Scholar] [CrossRef]
- Srinivasan, K.; Viswanad, B.; Asrat, L.; Kaul, C.L.; Ramarao, P.J. Combination of high-fat diet-fed and low-dose streptozotocin-treated rat: a model for type 2 diabetes and pharmacological screening. Pharmacological research 2005, 52(4), 313–20. [Google Scholar] [CrossRef] [PubMed]
- Dirir, A.M.; Daou, M.; Yousef, A.F.; Yousef, L.F. A review of alpha-glucosidase inhibitors from plants as potential candidates for the treatment of type-2 diabetes. Phytochemistry Reviews 2022, 21(4), 1049–79. [Google Scholar] [CrossRef] [PubMed]
- Lam, W.Y. Phytochemical profiling, antioxidant property, alpha amylase and alpha-glucosidase inhibitory activities of medicinal fern christella dentata . Doctoral dissertation, UTAR, 2014. [Google Scholar]
- Albzoor, R. Medicinal Plants as a Source of Inhibitors of the Digestive Enzymes: Alpha-glucosidase, Alpha-amylase and Pancreatic Lipase . Doctoral dissertation, Raghad Albzoor, 2019. [Google Scholar]
- Chung, M.Y.; Choi, H.K.; Hwang, J.T. AMPK activity: a primary target for diabetes prevention with therapeutic phytochemicals. Nutrients 2021, 13(11), 4050. [Google Scholar] [CrossRef] [PubMed]
- Persaud, S.J.; Al-Majed, H.; Raman, A.; Jones, P.M. Gymnema sylvestre stimulates insulin release in vitro by increased membrane permeability. Journal of endocrinology 1999, 163(2), 207–12. [Google Scholar] [CrossRef] [PubMed]
- Noshahr, Z.S.; Shahraki, M.R.; Ahmadvand, H.; Nourabadi, D.; Nakhaei, A. Protective effects of Withania somnifera root on inflammatory markers and insulin resistance in fructose-fed rats. Reports of biochemistry & molecular biology 2015, 3(2), 62. [Google Scholar] [PubMed]
- Anwer, T.; Sharma, M.; Pillai, K.K.; Iqbal, M. Effect of Withania somnifera on insulin sensitivity in non-insulin-dependent diabetes mellitus rats. Basic & clinical pharmacology & toxicology 2008, 102(6), 498–503. [Google Scholar] [CrossRef] [PubMed]
- Udayakumar, R.; Kasthurirengan, S.; Mariashibu, T.S.; Rajesh, M.; Anbazhagan, V.R.; Kim, S.C.; Ganapathi, A.; Choi, C.W. Hypoglycaemic and hypolipidaemic effects of Withania somnifera root and leaf extracts on alloxan-induced diabetic rats. International journal of molecular sciences 2009, 10(5), 2367–82. [Google Scholar] [CrossRef] [PubMed]
- Chaplan, S.R.; Bach, F.W.; Pogrel, J.W.; Chung, J.M.; Yaksh, T.L. Quantitative assessment of tactile allodynia in the rat paw. Journal of neuroscience methods 1994, 53(1), 55–63. [Google Scholar] [CrossRef] [PubMed]
- Dawane, J.S.; Pandit, V.A.; Bhosale, M.S.; Khatavkar, P.S. Evaluation of effect of nishamalaki on STZ and HFHF diet induced diabetic neuropathy in wistar rats. Journal of Clinical and Diagnostic Research: JCDR 2016, 10(10), FF01. [Google Scholar] [CrossRef] [PubMed]
- Lim, D.W.; Kim, J.G.; Lim, E.Y.; Kim, Y.T. Antihyperalgesic effects of ashwagandha (Withania somnifera root extract) in rat models of postoperative and neuropathic pain. Inflammopharmacology 2018, 26, 207–15. [Google Scholar] [CrossRef] [PubMed]
- Gesto, D.S.; Pereira, C.M.; Cerqueira, N.M.; Sousa, S.F. An atomic-level perspective of HMG-CoA-reductase: the target enzyme to treat hypercholesterolemia. Molecules 2020, 25(17), 3891. [Google Scholar] [CrossRef] [PubMed]
- Xie, P.; Luo, H.T.; Pei, W.J.; Xiao, M.Y.; Li, F.F.; Gu, Y.L.; Piao, X.L. Saponins derived from Gynostemma pentaphyllum regulate triglyceride and cholesterol metabolism and the mechanisms: A review. Journal of Ethnopharmacology 2024, 319, 117186. [Google Scholar] [CrossRef] [PubMed]
- Suganya, S.; Natarajan, S.; Chamundeeswari, D.; Anbarasu, A.; Balasubramanian, K.A.; Schneider, L.C.; Nandagopal, B. Clinical evaluation of a polyherbal nutritional supplement in dyslipidemic volunteers. Journal of Dietary Supplements 2017, 14(6), 679–90. [Google Scholar] [CrossRef] [PubMed]
- Shields, H.J.; Traa, A.; Van Raamsdonk, J.M. Beneficial and detrimental effects of reactive oxygen species on lifespan: a comprehensive review of comparative and experimental studies. Frontiers in cell and developmental biology 2021, 9, 628157. [Google Scholar] [CrossRef] [PubMed]
- Fujita, H.; Fujishima, H.; Chida, S.; Takahashi, K.; Qi, Z.; Kanetsuna, Y.; Breyer, M.D.; Harris, R.C.; Yamada, Y.; Takahashi, T. Reduction of renal superoxide dismutase in progressive diabetic nephropathy. Journal of the American Society of Nephrology 2009, 20(6), 1303–13. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.; Jin, J.; Jeon, S.; Moon, S.H.; Park, M.Y.; Yum, D.Y.; Kim, J.H.; Kang, J.E.; Park, M.H.; Kim, E.J.; Pan, J.G. SOD1 suppresses pro-inflammatory immune responses by protecting against oxidative stress in colitis. Redox Biology 2020, 37, 101760. [Google Scholar] [CrossRef] [PubMed]
- Sekhar, R.V.; McKay, S.V.; Patel, S.G.; Guthikonda, A.P.; Reddy, V.T.; Balasubramanyam, A.; Jahoor, F. Glutathione synthesis is diminished in patients with uncontrolled diabetes and restored by dietary supplementation with cysteine and glycine. Diabetes care 2011, 34(1), 162–7. [Google Scholar] [CrossRef] [PubMed]
- Uddandrao, V.S.; Parim, B.; Singaravel, S.; Ponnusamy, P.; Ponnusamy, C.; Sasikumar, V.; Saravanan, G. Polyherbal formulation ameliorates diabetic cardiomyopathy through attenuation of cardiac inflammation and oxidative stress via NF-κB/Nrf-2/HO-1 pathway in diabetic rats. Journal of Cardiovascular Pharmacology 2022, 79(1), e75-86. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.S.; Shah, R.S.; Goyal, R.K. Antihyperglycemic, antihyperlipidemic and antioxidant effects of Dihar, a polyherbal ayurvedic formulation in streptozotocin induced diabetic rats. 2009. [Google Scholar] [PubMed]
- Abdel-Kader, M.S.; Abdel-Rahman, R.F.; Soliman, G.A.; Ogaly, H.A.; Alamri, M.A.; Alharbi, A.G. Oleuropein Relieves Pancreatic Ischemia Reperfusion Injury in Rats by Suppressing Inflammation and Oxidative Stress through HMGB1/NF-κB Pathway. International Journal of Molecular Sciences 2024, 25(18), 10171. [Google Scholar] [CrossRef] [PubMed]
- Akpoveso, O.O.; Ubah, E.E.; Obasanmi, G. Antioxidant phytochemicals as potential therapy for diabetic complications. Antioxidants 2023, 12(1), 123. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Amin, H.; Prajapati, P.K. Antidiabetic claims of Tinospora cordifolia (Willd.) Miers: critical appraisal and role in therapy. Asian Pacific Journal of Tropical Biomedicine 2015, 5(1), 68–78. [Google Scholar] [CrossRef]
- Qiu, W.C.; Wang, Z.G.; Lv, R.; Wang, W.G.; Han, X.D.; Yan, J.; Wang, Y.; Zheng, Q.; Ai, K.X. Ghrelin improves delayed gastrointestinal transit in alloxan-induced diabetic mice. World Journal of Gastroenterology: WJG 2008, 14(16), 2572. [Google Scholar] [CrossRef] [PubMed]
- Lu, K.H.; Cao, J.; Oleson, S.; Ward, M.P.; Phillips, R.J.; Powley, T.L.; Liu, Z. Vagus nerve stimulation promotes gastric emptying by increasing pyloric opening measured with magnetic resonance imaging. Neurogastroenterology & Motility 2018, 30(10), e13380. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Zou, Q.; Chen, Q.; Zhang, J.; Tang, H.; Chen, J.; Qin, Y.; Yang, L.; Chen, Z.; Cao, L. Therapeutic role of Wuda granule in gastrointestinal motility disorder through promoting gastrointestinal motility and decreasing inflammatory level. Frontiers in Pharmacology 2023, 14, 1237686. [Google Scholar] [CrossRef] [PubMed]
- Bharti, S.K.; Krishnan, S.; Kumar, A.; Kumar, A. Antidiabetic phytoconstituents and their mode of action on metabolic pathways. Therapeutic Advances in Endocrinology and metabolism 2018, 9(3), 81–100. [Google Scholar] [CrossRef] [PubMed]
| Name of plant | Family | Part used | Weight (g) |
|---|---|---|---|
| Withania somnifera | Solanaceae | Root | 15 |
| Gymnema sylvestre | Asclepiadaceae | Fruit | 15 |
| Azadirachta indica | Meliaceae | Leaves | 15 |
| Tinospora cordifolia | Menispermaceae | Stem | 15 |
| Momardica charanita | Cucurbitaceae | Fruit | 15 |
| 2 parts of the above herbs | |||
| Syzygium cumini | Myrtaceae | Seeds | 10 |
| Emblica officinalis | Phyllanthaceae | Fruit | 10 |
| 2 parts of the above herbs | |||
| Pterocorous marsupium | Fabaceae | Stem | 5 |
| 1 part of the above herb | |||
| Parameters | NC | DC | GABA | PHE-I | PHE-II | PHE-III |
|---|---|---|---|---|---|---|
|
Glucose (mg/dL) |
92.67±1.36 | 257.5± 4.63### | 212.0± 2.44*** | 249.7± 1.36* | 238.0± 1.41*** | 188.5± 8.57*** |
|
TC (mg/dL) |
110.2 ± 5.94 | 197.6 ± 3.31### | 115.4 ± 6.84*** | 190.8 ± 1.47 | 182.3 ± 2.73*** | 143.5 ± 3.61*** |
|
TG (mg/dL) |
107.5 ± 2.15 | 186.5±3.27### | 98.3 ± 6.11*** | 178.5 ± 1.87** | 168.2 ± 4.46*** | 110.5 ± 2.25*** |
|
LDL-C (mg/dL) |
29.85 ± 2.60 | 65.34 ± 2.06### | 36.41 ± 3.74*** | 61.84 ± 1.72 | 58.68 ± 1.17** | 35.56 ± 3.94*** |
|
HDL-C (mg/dL) |
55.6 ± 3.4 | 27.40 ± 1.53### | 52.8 ± 3.2*** | 31.10 ± 0.88* | 34.18 ± 1.71*** | 47.31 ± 1.54*** |
| Parameters | NC | DC | GABA | PHE-I | PHE-II | PHE-III |
|---|---|---|---|---|---|---|
|
LPO (nM/mg protein) |
3.30 ± 0.88 | 10.29 ± 0.82### | 4.85 ± 1.04*** | 8.45 ± 0.92* | 7.77 ± 1.34*** | 4.93 ± 1.04*** |
|
GSH (μg/mg protein) |
2.20 ± 0.50 | 0.64 ± 0.06### | 2.07 ± 0.56*** | 0.83 ± 0.05 | 1.32 ± 0.07** | 1.82 ± 0.25*** |
|
SOD (U/mg protein) |
22.83 ± 1.49 | 8.35 ± 1.07 ### | 19.53 ± 1.08*** | 8.35 ± 0.68 | 10.67 ± 1.18** | 16.63 ± 1.03*** |
|
CAT (U/mg protein) |
51.30 ± 3.23 | 19.55 ± 1.37### | 45.63 ± 3.67*** | 23.23 ± 0.57* | 24.38 ± 1.37** | 42.15 ± 1.39*** |
|
TNF-α (Pg/ml) |
56.28 ± 2.29 | 184.3 ± 4.09### | 105.2 ± 3.77*** | 178.4 ± 1.37** | 116.2 ± 3.11*** | 84.88 ± 2.00*** |
| Sr. No | Concentration (µg/ml) | Absorbance | Cell viability | |||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | Average | |||
| 1. | Control | 1.596 | 1.598 | 1.599 | 1.597 | 85.09 |
| 2. | H2O2 induced (0.4 mM) | 0.426 | 0.441 | 0.435 | 0.434 | 27.17 |
| 3. | PHF-I (10 µg/ml) | 0.633 | 0.639 | 0.637 | 0.636 | 39.82 |
| PHF-II (40 µg/ml) | 0.955 | 0.953 | 0.951 | 0.953 | 59.64 | |
| PHF-III (100µg/ml) | 1.263 | 1.261 | 1.262 | 1.262 | 78.99 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




