Submitted:
18 April 2026
Posted:
20 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sodium Alginate Extraction via Formaldehyde
2.3. Sodium Alginate Extraction via Ethanol
2.4. Solution Preparation and Rheological Measurements
2.5. Nuclear Magnetic Resonance (NMR) Spectroscopy Analysis
2.6. Fourier Transform Infrared (FTIR) Spectroscopy Analysis
3. Results and Discussion
3.1. Sodium Alginate Extraction: Study and Yield Analysis
3.2. Rheological Studies
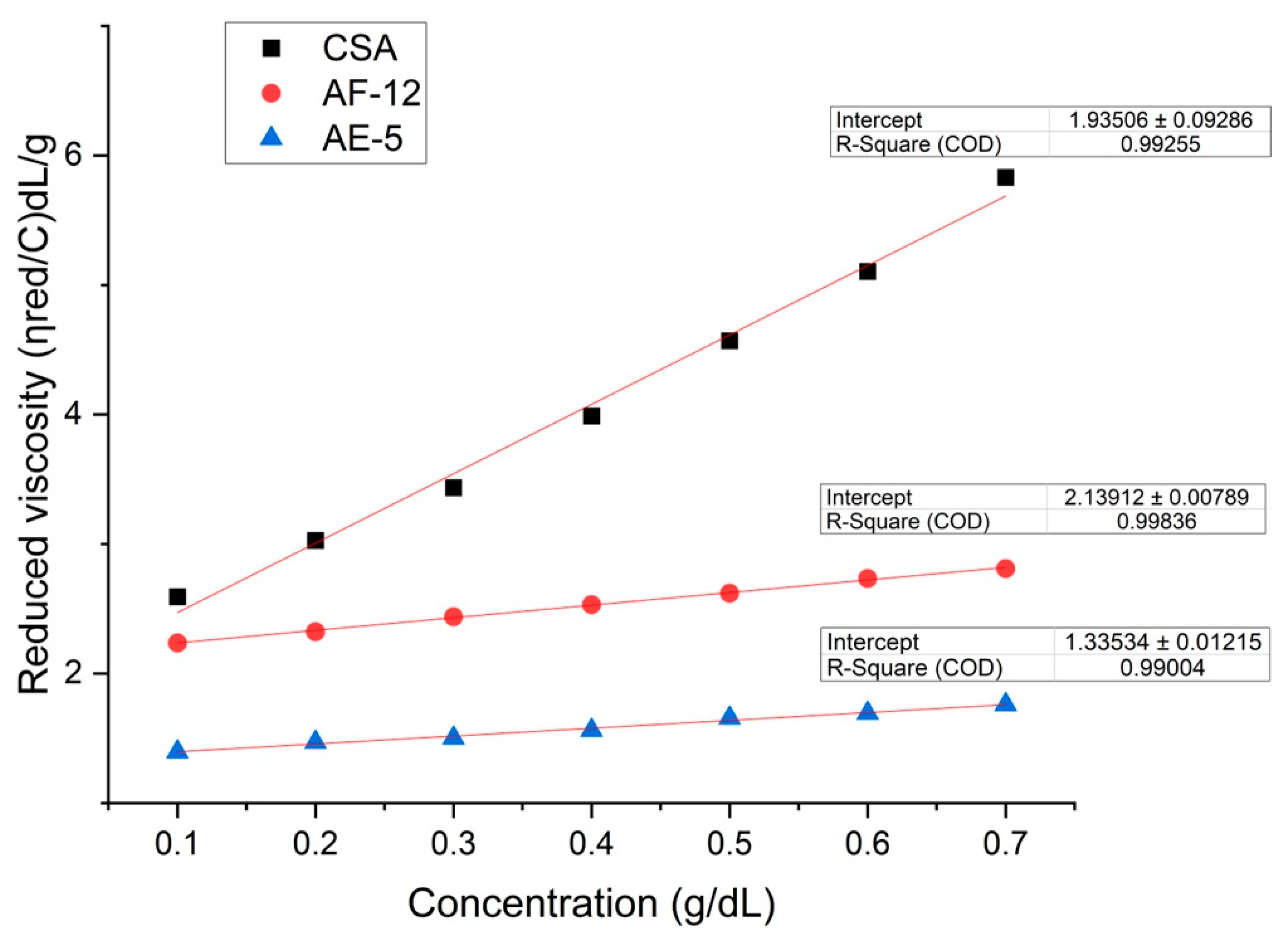
3.2.1. Intrinsic Viscosity
3.2.2. Determination of Molecular Weight
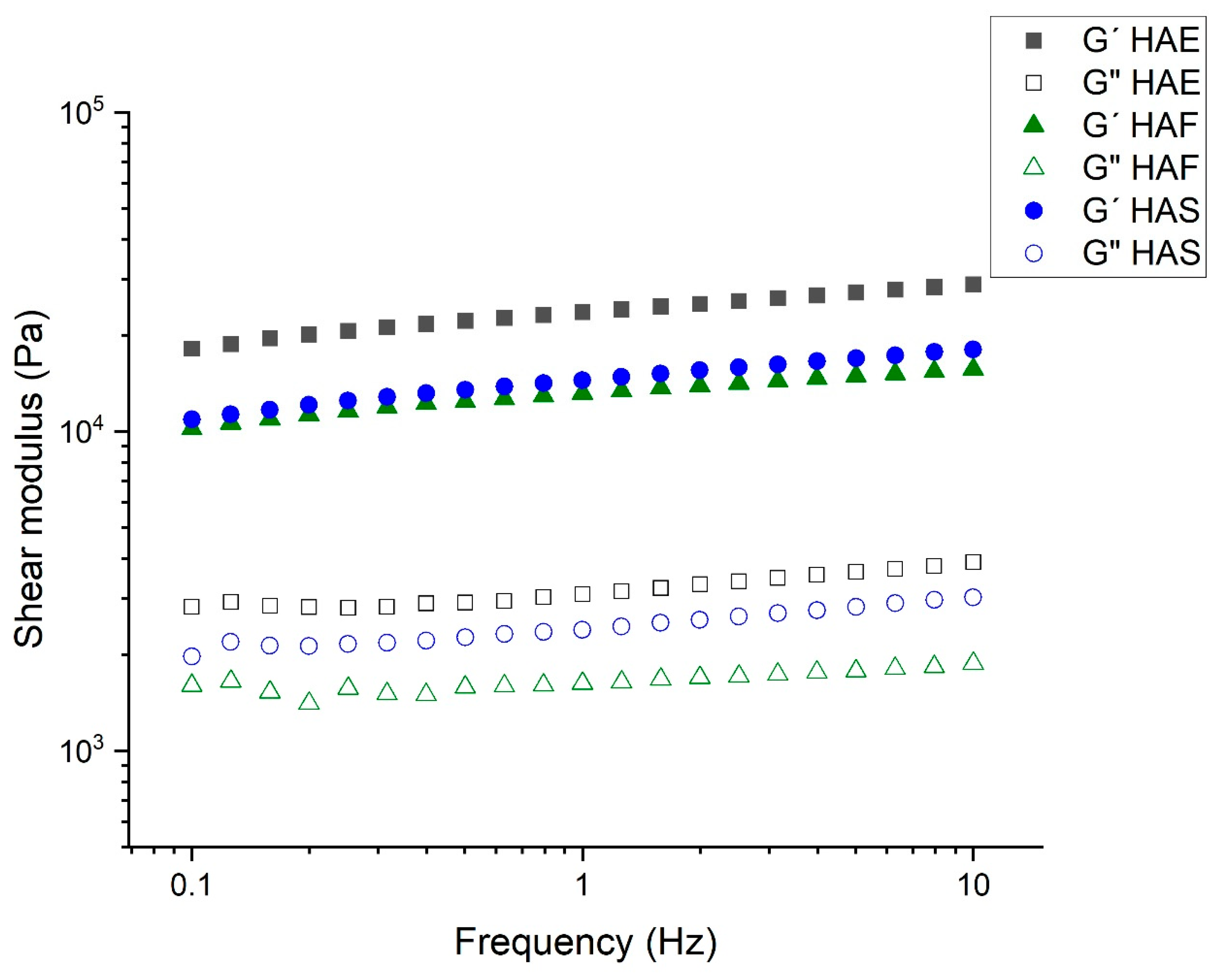
3.2.3. Viscoelastic Properties of Hydrogels
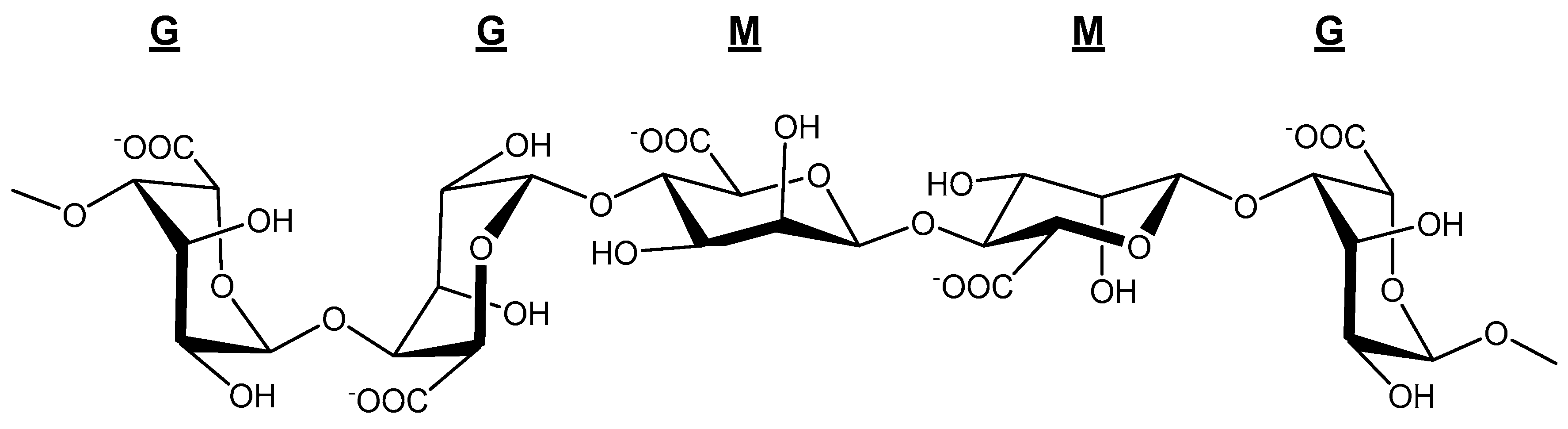
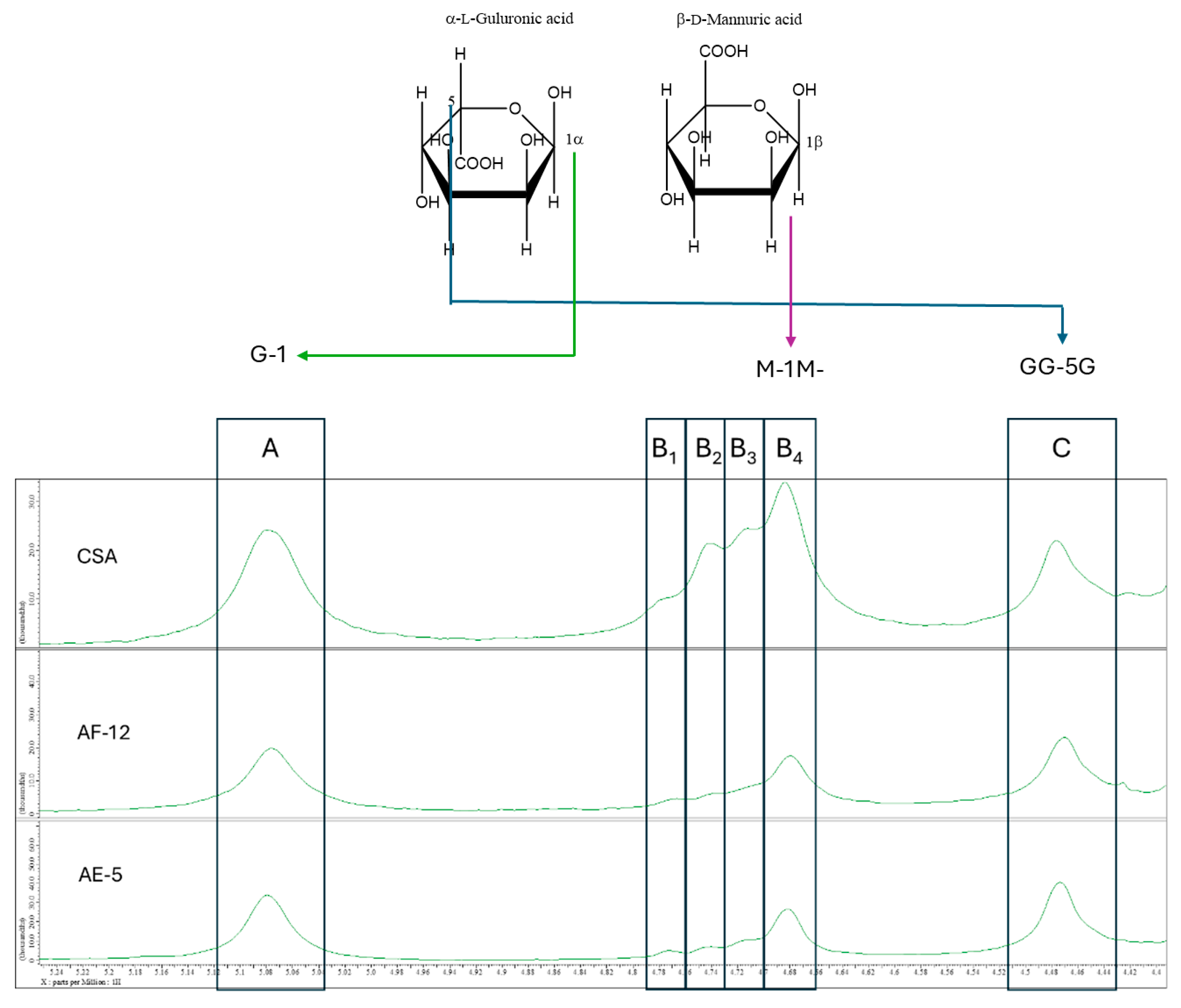
3.3. NMR Analysis of Alginate Structure
3.4. Structure-Property Relationships
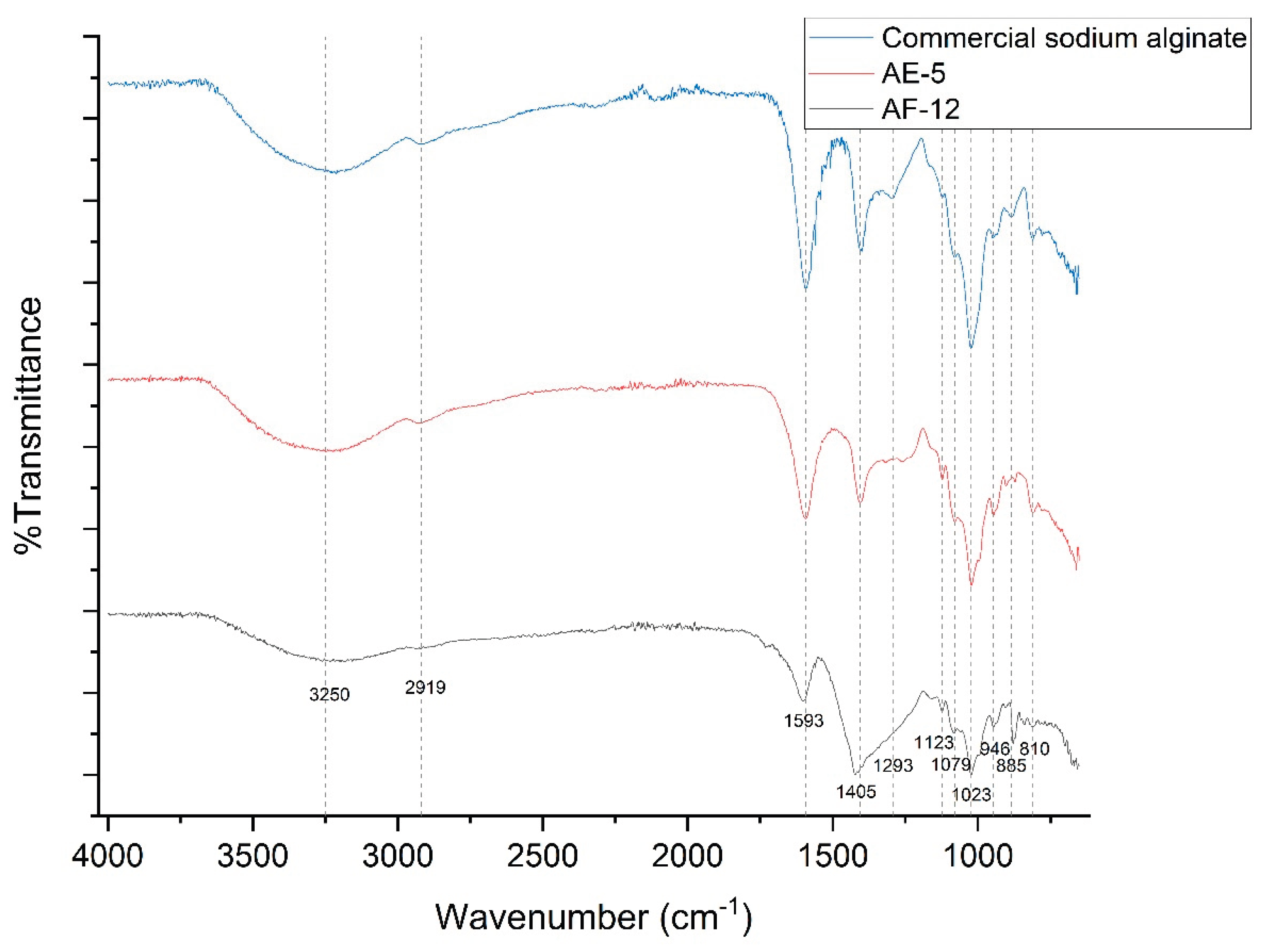
3.5. Fourier Transform Infrared (FTIR) Spectroscopy
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, M.; Hu, C.; Barnes, B.B.; Mitchum, G.; Lapointe, B.; Montoya, J.P. The Great Atlantic Sargassum Belt. Science 2019, 365, 83–87. [Google Scholar] [CrossRef]
- Chávez, V.; Uribe-Martínez, A.; Cuevas, E.; Rodríguez-Martínez, R.E.; Van Tussenbroek, B.I.; Francisco, V.; Estévez, M.; Celis, L.B.; Monroy-Velázquez, L.V.; Leal-Bautista, R.; et al. Massive Influx of Pelagic Sargassum Spp. on the Coasts of the Mexican Caribbean 2014–2020: Challenges and Opportunities. Water 2020, 12, 2908. [Google Scholar] [CrossRef]
- Debue, M.; Guinaldo, T.; Jouanno, J.; Chami, M.; Barbier, S.; Berline, L.; Chevalier, C.; Daniel, P.; Daniel, W.; Descloitres, J.; et al. Understanding the Sargassum Phenomenon in the Tropical Atlantic Ocean: From Satellite Monitoring to Stranding Forecast. Mar. Pollut. Bull. 2025, 216, 117923. [Google Scholar] [CrossRef]
- Vázquez-Delfín, E.; Freile-Pelegrín, Y.; Salazar-Garibay, A.; Serviere-Zaragoza, E.; Méndez-Rodríguez, L.C.; Robledo, D. Species Composition and Chemical Characterization of Sargassum Influx at Six Different Locations along the Mexican Caribbean Coast. Sci. Total Environ. 2021, 795, 148852. [Google Scholar] [CrossRef]
- García-Sánchez, M.; Graham, C.; Vera, E.; Escalante-Mancera, E.; Álvarez-Filip, L.; Tussenbroek, B.I. van Temporal Changes in the Composition and Biomass of Beached Pelagic Sargassum Species in the Mexican Caribbean. Aquat. Bot. 2020, 167, 103275. [Google Scholar] [CrossRef]
- Rosado-Espinosa, L.A.; Freile-Pelegrín, Y.; Hernández-Nuñez, E.; Robledo, D. A Comparative Study of Sargassum Species from the Yucatan Peninsula Coast: Morphological and Chemical Characterisation. Phycologia 2020, 59, 261–271. [Google Scholar] [CrossRef]
- Olguin-Maciel, E.; Leal-Bautista, R.M.; Alzate-Gaviria, L.; Domínguez-Maldonado, J.; Tapia-Tussell, R. Environmental Impact of Sargassum Spp. Landings: An Evaluation of Leachate Released from Natural Decomposition at Mexican Caribbean Coast. Environ. Sci. Pollut. Res. 2022, 29, 91071–91080. [Google Scholar] [CrossRef]
- Rodríguez-Martínez, R.E.; Gómez Reali, M.Á; Torres-Conde, E.G.; Bates, M.N. Temporal and Spatial Variation in Hydrogen Sulfide (H2S) Emissions during Holopelagic Sargassum Spp. Decomposition on Beaches. Environ. Res. 2024, 247, 118235. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Martínez, R.E.; Medina-Valmaseda, A.E.; Blanchon, P.; Monroy-Velázquez, L.V.; Almazán-Becerril, A.; Delgado-Pech, B.; Vásquez-Yeomans, L.; Francisco, V.; García-Rivas, M.C. Faunal Mortality Associated with Massive Beaching and Decomposition of Pelagic Sargassum. Mar. Pollut. Bull. 2019, 146, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Resiere, D.; Mehdaoui, H.; Florentin, J.; Gueye, P.; Lebrun, T.; Blateau, A.; Viguier, J.; Valentino, R.; Brouste, Y.; Kallel, H.; et al. Sargassum Seaweed Health Menace in the Caribbean: Clinical Characteristics of a Population Exposed to Hydrogen Sulfide during the 2018 Massive Stranding. Clin. Toxicol. 2021, 59, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Fu, X.; Duan, D.; Xu, J.; Gao, X. Comparison Study of Bioactive Substances and Nutritional Components of Brown Algae Sargassum Fusiforme Strains with Different Vesicle Shapes. J. Appl. Phycol. 2018, 30, 3271–3283. [Google Scholar] [CrossRef]
- Milledge, J.J.; Nielsen, B.V.; Bailey, D. High-Value Products from Macroalgae: The Potential Uses of the Invasive Brown Seaweed, Sargassum Muticum. Rev. Environ. Sci. Biotechnol. 2016, 15, 67–88. [Google Scholar] [CrossRef]
- Flores-Contreras, E.A.; Araújo, R.G.; Rodríguez-Aguayo, A.A.; Guzmán-Román, M.; García-Venegas, J.C.; Nájera-Martínez, E.F.; Sosa-Hernández, J.E.; Iqbal, H.M.N.; Melchor-Martínez, E.M.; Parra-Saldivar, R. Polysaccharides from the Sargassum and Brown Algae Genus: Extraction, Purification, and Their Potential Therapeutic Applications. Plants 2023, 12, 2445. [Google Scholar] [CrossRef] [PubMed]
- Gordillo Sierra, A.R.; Amador-Castro, L.F.; Ramírez-Partida, A.E.; García-Cayuela, T.; Carrillo-Nieves, D.; Alper, H.S. Valorization of Caribbean Sargassum Biomass as a Source of Alginate and Sugars for de Novo Biodiesel Production. J. Environ. Manage. 2022, 324, 116364. [Google Scholar] [CrossRef] [PubMed]
- Rhein-Knudsen, N.; Ale, M.T.; Ajalloueian, F.; Meyer, A.S. Characterization of Alginates from Ghanaian Brown Seaweeds: Sargassum Spp. and Padina Spp. Food Hydrocoll. 2017, 71, 236–244. [Google Scholar] [CrossRef]
- Saji, S.; Hebden, A.; Goswami, P.; Du, C. A Brief Review on the Development of Alginate Extraction Process and Its Sustainability. Sustainability 2022, 14, 5181. [Google Scholar] [CrossRef]
- Derkach, S.R.; Voron’ko, N.G.; Sokolan, N.I.; Kolotova, D.S.; Kuchina, Y.A. Interactions between Gelatin and Sodium Alginate: UV and FTIR Studies. J. Dispers. Sci. Technol. 2020, 41, 690–698. [Google Scholar] [CrossRef]
- Migonney, V. Biomaterials; John Wiley & Sons, 2014; ISBN 978-1-119-04367-6. [Google Scholar]
- Zubia, M.; Payri, C.; Deslandes, E. Alginate, Mannitol, Phenolic Compounds and Biological Activities of Two Range-Extending Brown Algae, Sargassum Mangarevense and Turbinaria Ornata (Phaeophyta: Fucales), from Tahiti (French Polynesia). J. Appl. Phycol. 2008, 20, 1033–1043. [Google Scholar] [CrossRef]
- Fenoradosoa, T.A.; Ali, G.; Delattre, C.; Laroche, C.; Petit, E.; Wadouachi, A.; Michaud, P. Extraction and Characterization of an Alginate from the Brown Seaweed Sargassum Turbinarioides Grunow. J. Appl. Phycol. 2010, 22, 131–137. [Google Scholar] [CrossRef]
- Khajouei, R.A.; Keramat, J.; Hamdami, N.; Ursu, A.-V.; Delattre, C.; Laroche, C.; Gardarin, C.; Lecerf, D.; Desbrières, J.; Djelveh, G.; et al. Extraction and Characterization of an Alginate from the Iranian Brown Seaweed Nizimuddinia Zanardini. Int. J. Biol. Macromol. 2018, 118, 1073–1081. [Google Scholar] [CrossRef]
- Urbanova, M.; Pavelkova, M.; Czernek, J.; Kubova, K.; Vyslouzil, J.; Pechova, A.; Molinkova, D.; Vyslouzil, J.; Vetchy, D.; Brus, J. Interaction Pathways and Structure–Chemical Transformations of Alginate Gels in Physiological Environments. Biomacromolecules 2019, 20, 4158–4170. [Google Scholar] [CrossRef]
- Martău, G.A.; Mihai, M.; Vodnar, D.C. The Use of Chitosan, Alginate, and Pectin in the Biomedical and Food Sector—Biocompatibility, Bioadhesiveness, and Biodegradability. Polymers 2019, 11, 1837. [Google Scholar] [CrossRef]
- Usov, A.I.; Zelinsky, N.D. Chemical Structures of Algal Polysaccharides. In Functional Ingredients from Algae for Foods and Nutraceuticals; Elsevier, 2013; pp. 23–86. ISBN 978-0-85709-512-1. [Google Scholar]
- Sanchez-Ballester, N.M.; Bataille, B.; Soulairol, I. Sodium Alginate and Alginic Acid as Pharmaceutical Excipients for Tablet Formulation: Structure-Function Relationship. Carbohydr. Polym. 2021, 270, 118399. [Google Scholar] [CrossRef]
- Draget, K.I.; Taylor, C. Chemical, Physical and Biological Properties of Alginates and Their Biomedical Implications. Food Hydrocoll. 2011, 25, 251–256. [Google Scholar] [CrossRef]
- Hecht, H.; Srebnik, S. Structural Characterization of Sodium Alginate and Calcium Alginate. Biomacromolecules 2016, 17, 2160–2167. [Google Scholar] [CrossRef]
- Grant, G.T.; Mon, E.R.; Rees, S.D.A. Biological Interactions between Polysaccharides and Divalent Cations: The Egg-Box Model. FEBS Lett. 1973, 32, 195–198. [Google Scholar] [CrossRef]
- Varela-Feijoo, A.; Djemia, P.; Narita, T.; Pignon, F.; Baeza-Squiban, A.; Sirri, V.; Ponton, A. Multiscale Investigation of Viscoelastic Properties of Aqueous Solutions of Sodium Alginate and Evaluation of Their Biocompatibility. Soft Matter 2023, 19, 5942–5955. [Google Scholar] [CrossRef] [PubMed]
- Chee, S.-Y.; Wong, P.-K.; Wong, C.-L. Extraction and Characterisation of Alginate from Brown Seaweeds (Fucales, Phaeophyceae) Collected from Port Dickson, Peninsular Malaysia. J. Appl. Phycol. 2011, 23, 191–196. [Google Scholar] [CrossRef]
- Arroyo, B.J.; Bezerra, A.C.; Oliveira, L.L.; Arroyo, S.J.; Melo, E.A.D.; Santos, A.M.P. Antimicrobial Active Edible Coating of Alginate and Chitosan Add ZnO Nanoparticles Applied in Guavas (Psidium Guajava L.). Food Chem. 2020, 309, 125566. [Google Scholar] [CrossRef]
- Makaremi, M.; Yousefi, H.; Cavallaro, G.; Lazzara, G.; Goh, C.B.S.; Lee, S.M.; Solouk, A.; Pasbakhsh, P. Safely Dissolvable and Healable Active Packaging Films Based on Alginate and Pectin. Polymers 2019, 11, 1594. [Google Scholar] [CrossRef]
- Hu, C.; Lu, W.; Mata, A.; Nishinari, K.; Fang, Y. Ions-Induced Gelation of Alginate: Mechanisms and Applications. Int. J. Biol. Macromol. 2021, 177, 578–588. [Google Scholar] [CrossRef]
- Bertagnolli, C.; Da Silva, M.G.C.; Guibal, E. Chromium Biosorption Using the Residue of Alginate Extraction from Sargassum Filipendula. Chem. Eng. J. 2014, 237, 362–371. [Google Scholar] [CrossRef]
- Yang, J.; Chen, S.; Fang, Y. Viscosity Study of Interactions between Sodium Alginate and CTAB in Dilute Solutions at Different pH Values. Carbohydr. Polym. 2009, 75, 333–337. [Google Scholar] [CrossRef]
- Hakim, M.M.; Patel, I.C. A Review on Phytoconstituents of Marine Brown Algae. Future J. Pharm. Sci. 2020, 6, 129. [Google Scholar] [CrossRef]
- Elwakeel, K.Z.; Ahmed, M.M.; Akhdhar, A.; Sulaiman, M.G.M.; Khan, Z.A. Recent Advances in Alginate-Based Adsorbents for Heavy Metal Retention from Water: A Review. Desalination Water Treat. 2022, 272, 50–74. [Google Scholar] [CrossRef]
- Davis, T.A.; Ramirez, M.; Mucci, A.; Larsen, B. Extraction, Isolation and Cadmium Binding of Alginate from Sargassum Spp. J. Appl. Phycol. 2004, 16, 275–284. [Google Scholar] [CrossRef]
- Kleinübing, S.J.; Gai, F.; Bertagnolli, C.; Silva, M.G.C.D. Extraction of Alginate Biopolymer Present in Marine Alga Sargassum Filipendula and Bioadsorption of Metallic Ions. Mater. Res. 2013, 16, 481–488. [Google Scholar] [CrossRef]
- Wang, S.; Vincent, T.; Faur, C.; Guibal, E. Alginate and Algal-Based Beads for the Sorption of Metal Cations: Cu(II) and Pb(II). Int. J. Mol. Sci. 2016, 17, 1453. [Google Scholar] [CrossRef]
- Li, L.; Zhu, B.; Yao, Z.; Jiang, J. Directed Preparation, Structure–Activity Relationship and Applications of Alginate Oligosaccharides with Specific Structures: A Systematic Review. Food Res. Int. 2023, 170, 112990. [Google Scholar] [CrossRef]
- Fertah, M.; Belfkira, A.; Dahmane, E.M.; Taourirte, M.; Brouillette, F. Extraction and Characterization of Sodium Alginate from Moroccan Laminaria Digitata Brown Seaweed. Arab. J. Chem. 2017, 10, S3707–S3714. [Google Scholar] [CrossRef]
- Xiao, Q.; Gu, X.; Tan, S. Drying Process of Sodium Alginate Films Studied by Two-Dimensional Correlation ATR-FTIR Spectroscopy. Food Chem. 2014, 164, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Johnson, F.A.; Craig, D.Q.M.; Mercer, A.D. Characterization of the Block Structure and Molecular Weight of Sodium Alginates. J. Pharm. Pharmacol. 2011, 49, 639–643. [Google Scholar] [CrossRef] [PubMed]
- Lorbeer, A.J.; Lahnstein, J.; Bulone, V.; Nguyen, T.; Zhang, W. Multiple-Response Optimization of the Acidic Treatment of the Brown Alga Ecklonia Radiata for the Sequential Extraction of Fucoidan and Alginate. Bioresour. Technol. 2015, 197, 302–309. [Google Scholar] [CrossRef]
- Smith, H.A.; Zhou, J.; Buckley, H.L. Greener Citrate-Assisted Extraction of Sodium Alginate: Process Optimization and the Mechanical Performance of Alginate-Based Films. Green Chem. 2026, 10.1039.D5GC05514H. [Google Scholar] [CrossRef]
- Klein-Marcuschamer, D.; Simmons, B.A.; Blanch, H.W. Techno-economic Analysis of a Lignocellulosic Ethanol Biorefinery with Ionic Liquid Pre-treatment. Biofuels Bioprod. Biorefining 2011, 5, 562–569. [Google Scholar] [CrossRef]
- Van Sittert, D.; Lufu, R.; Mapholi, Z.; Goosen, N.J. Ultrasound-Assisted Extraction of Alginate from Ecklonia Maxima with and without the Addition of Alkaline Cellulase – Factorial and Kinetic Analysis. J. Appl. Phycol. 2024, 36, 2781–2793. [Google Scholar] [CrossRef]
- Bojorges, H.; López-Rubio, A.; Martínez-Abad, A.; Fabra, M.J. Overview of Alginate Extraction Processes: Impact on Alginate Molecular Structure and Techno-Functional Properties. Trends Food Sci. Technol. 2023, 140, 104142. [Google Scholar] [CrossRef]
- Ortegón-Aznar, I.; Ávila-Mosqueda, S.V. Arribazón de sargazo en la península de Yucatán: ¿Problema local, regional o mundial? Bioagrociencias 2020, 13. [Google Scholar] [CrossRef]
- Standard, A. Standard Test Method for Determining the Chemical Composition and Sequence in Alginate by Proton Nuclear Magnetic Resonance (1H NMR) Spectroscopy; 2012. [Google Scholar]
- Torres, M.R.; Sousa, A.P.A.; Silva Filho, E.A.T.; Melo, D.F.; Feitosa, J.P.A.; De Paula, R.C.M.; Lima, M.G.S. Extraction and Physicochemical Characterization of Sargassum Vulgare Alginate from Brazil. Carbohydr. Res. 2007, 342, 2067–2074. [Google Scholar] [CrossRef]
- Gao, F.; Liu, X.; Chen, W.; Guo, W.; Chen, L.; Li, D. Hydroxyl Radical Pretreatment for Low-Viscosity Sodium Alginate Production from Brown Seaweed. Algal Res. 2018, 34, 191–197. [Google Scholar] [CrossRef]
- Mohammed, A.; Bissoon, R.; Bajnath, E.; Mohammed, K.; Lee, T.; Bissram, M.; John, N.; Jalsa, Nigel.K.; Lee, K.-Y.; Ward, K. Multistage Extraction and Purification of Waste Sargassum Natans to Produce Sodium Alginate: An Optimization Approach. Carbohydr. Polym. 2018, 198, 109–118. [Google Scholar] [CrossRef]
- Caballero, E.; Flores, A.; Olivares, A. Sustainable Exploitation of Macroalgae Species from Chilean Coast: Characterization and Food Applications. Algal Res. 2021, 57, 102349. [Google Scholar] [CrossRef]
- Laroche, C.; Michaud, P. A Novel Alginate from the Brown Seaweed Sargassum Turbinaroides (Sargassae). In Curr. Top. Bioprocesses Food Ind.; Larroche C Pandey Dussap, C.G., Ed.; 2009; pp. 71–92. [Google Scholar]
- Sellimi, S.; Younes, I.; Ayed, H.B.; Maalej, H.; Montero, V.; Rinaudo, M.; Dahia, M.; Mechichi, T.; Hajji, M.; Nasri, M. Structural, Physicochemical and Antioxidant Properties of Sodium Alginate Isolated from a Tunisian Brown Seaweed. Int. J. Biol. Macromol. 2015, 72, 1358–1367. [Google Scholar] [CrossRef]
- Smidsrød, O.; Haug, A. Estimation of the Relative Stiffness of the Molecular Chain in Polyelectrolytes from Measurements of Viscosity at Different Ionic Strengths. Biopolymers 1971, 10, 1213–1227. [Google Scholar] [CrossRef]
- Martinsen, A.; Skjåk-Bræk, G.; Smidsrød, O. Alginate as Immobilization Material: I. Correlation between Chemical and Physical Properties of Alginate Gel Beads. Biotechnol. Bioeng. 1989, 33, 79–89. [Google Scholar] [CrossRef]
- Szekalska, M.; Puciłowska, A.; Szymańska, E.; Ciosek, P.; Winnicka, K. Alginate: Current Use and Future Perspectives in Pharmaceutical and Biomedical Applications. Int. J. Polym. Sci. 2016, 2016, 1–17. [Google Scholar] [CrossRef]
- Storz, H.; Müller, K.J.; Ehrhart, F.; Gómez, I.; Shirley, S.G.; Gessner, P.; Zimmermann, G.; Weyand, E.; Sukhorukov, V.L.; Forst, T.; et al. Physicochemical Features of Ultra-High Viscosity Alginates. Carbohydr. Res. 2009, 344, 985–995. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and Biomedical Applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef]
- Agulhon, P.; Robitzer, M.; David, L.; Quignard, F. Structural Regime Identification in Ionotropic Alginate Gels: Influence of the Cation Nature and Alginate Structure. Biomacromolecules 2012, 13, 215–220. [Google Scholar] [CrossRef]
- Huggins, M.L. The Viscosity of Dilute Solutions of Long-Chain Molecules. IV. Dependence on Concentration. J. Am. Chem. Soc. 1942, 64, 2716–2718. [Google Scholar] [CrossRef]
- Sugiono, S.; Masruri, M.; Estiasih, T.; Widjanarko, S.B. Optimization of Extrusion-Assisted Extraction Parameters and Characterization of Alginate from Brown Algae (Sargassum Cristaefolium). J. Food Sci. Technol. 2019, 56, 3687–3696. [Google Scholar] [CrossRef]
- Trica, B.; Delattre, C.; Gros, F.; Ursu, A.V.; Dobre, T.; Djelveh, G.; Michaud, P.; Oancea, F. Extraction and Characterization of Alginate from an Edible Brown Seaweed (Cystoseira Barbata) Harvested in the Romanian Black Sea. Mar. Drugs 2019, 17, 405. [Google Scholar] [CrossRef]
- Rinaudo, M. Chitin and Chitosan: Properties and Applications. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- Clementi, F.; Mancini, M.; Moresi, M. Rheology of Alginate from Azotobacter Vinelandii in Aqueous Dispersions. J. Food Eng. 1998, 36, 51–62. [Google Scholar] [CrossRef]
- Sari-Chmayssem, N.; Taha, S.; Mawlawi, H.; Guégan, J.-P.; Jeftić, J.; Benvegnu, T. Extracted and Depolymerized Alginates from Brown Algae Sargassum Vulgare of Lebanese Origin: Chemical, Rheological, and Antioxidant Properties. J. Appl. Phycol. 2016, 28, 1915–1929. [Google Scholar] [CrossRef]
- Liparoti, S.; Speranza, V.; Marra, F. Alginate Hydrogel: The Influence of the Hardening on the Rheological Behaviour. J. Mech. Behav. Biomed. Mater. 2021, 116, 104341. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Lin, Y.; Chen, X.; Zhao, B.; Zhang, J. Flow Behavior, Thixotropy and Dynamical Viscoelasticity of Sodium Alginate Aqueous Solutions. Food Hydrocoll. 2014, 38, 119–128. [Google Scholar] [CrossRef]
- Stojkov, G.; Niyazov, Z.; Picchioni, F.; Bose, R.K. Relationship between Structure and Rheology of Hydrogels for Various Applications. Gels 2021, 7, 255. [Google Scholar] [CrossRef]
- Pfaff, N.M.; Dijksman, J.A.; Kemperman, A.J.B.; Van Loosdrecht, M.C.M.; Kleijn, J.M. Rheological Characterisation of Alginate-like Exopolymer Gels Crosslinked with Calcium. Water Res. 2021, 207, 117835. [Google Scholar] [CrossRef]
- Growney Kalaf, E.A.; Flores, R.; Bledsoe, J.G.; Sell, S.A. Characterization of Slow-Gelling Alginate Hydrogels for Intervertebral Disc Tissue-Engineering Applications. Mater. Sci. Eng. C 2016, 63, 198–210. [Google Scholar] [CrossRef]
- Rioux, L.-E.; Turgeon, S.L.; Beaulieu, M. Characterization of Polysaccharides Extracted from Brown Seaweeds. Carbohydr. Polym. 2007, 69, 530–537. [Google Scholar] [CrossRef]
- Larsen, B.; Salem, D.M.S.A.; Sallam, M.A.E.; Mishrikey, M.M.; Beltagy, A.I. Characterization of the Alginates from Algae Harvested at the Egyptian Red Sea Coast. Carbohydr. Res. 2003, 338, 2325–2336. [Google Scholar] [CrossRef]
- Bertagnolli, C.; Espindola, A.P.D.M.; Kleinübing, S.J.; Tasic, L.; Silva, M.G.C.D. Sargassum Filipendula Alginate from Brazil: Seasonal Influence and Characteristics. Carbohydr. Polym. 2014, 111, 619–623. [Google Scholar] [CrossRef]
- Grasdalen, H.; Larsen, B.; Smidsrød, O. A p.m.r. Study of the Composition and Sequence of Uronate Residues in Alginates. Carbohydr. Res. 1979, 68, 23–31. [Google Scholar] [CrossRef]
- Davis, T.A.; Llanes, F.; Volesky, B.; Mucci, A. Metal Selectivity of Sargassum Spp. and Their Alginates in Relation to Their α-l-Guluronic Acid Content and Conformation. Environ. Sci. Technol. 2003, 37, 261–267. [Google Scholar] [CrossRef]
- Zhang, H.; Cheng, J.; Ao, Q. Preparation of Alginate-Based Biomaterials and Their Applications in Biomedicine. Mar Drugs 2021. [Google Scholar]
- Rashedy, S.H.; Abd El Hafez, M.S.M.; Dar, M.A.; Cotas, J.; Pereira, L. Evaluation and Characterization of Alginate Extracted from Brown Seaweed Collected in the Red Sea. Appl. Sci. 2021, 11, 6290. [Google Scholar] [CrossRef]
- Lawrie, G.; Keen, I.; Drew, B.; Chandler-Temple, A.; Rintoul, L.; Fredericks, P.; Grøndahl, L. Interactions between Alginate and Chitosan Biopolymers Characterized Using FTIR and XPS. Biomacromolecules 2007, 8, 2533–2541. [Google Scholar] [CrossRef]
- Belattmania, Z.; Kaidi, S.; El Atouani, S.; Katif, C.; Bentiss, F.; Jama, C.; Reani, A.; Sabour, B.; Vasconcelos, V. Isolation and FTIR-ATR and 1H NMR Characterization of Alginates from the Main Alginophyte Species of the Atlantic Coast of Morocco. Molecules 2020, 25, 4335. [Google Scholar] [CrossRef]
- Park, Y.; Malgas, S.; Krause, R.W.M.; Pletschke, B.I. Extraction and Characterisation of Sodium Alginate from the Southern African Seaweed Ecklonia Maxima. Bot 2024. [Google Scholar] [CrossRef]
| Sample | 0.2 M HCl (Time, h) | 2% sodium carbonate (Time, h) | 0.2 M sodium carbonate (Temp., °C) | Yield (% w/w)* |
|---|---|---|---|---|
| AF-1 | 2 | 3 | 70 | 6.66±0.27 |
| AF-2 | 2 | 2 | 70 | 6.06±0.24 |
| AF-3 | 2 | 1 | 70 | 10.91±0.44 |
| AF-4 | 1 | 3 | 70 | 4.40±0.18 |
| AF-5 | 1 | 2 | 70 | 8.41±0.34 |
| AF-6 | 1 | 1 | 70 | 5.61±0.22 |
| AF-7 | 2 | 3 | 50 | 7.27±0.29 |
| AF-8 | 2 | 2 | 50 | 7.05±0.28 |
| AF-9 | 2 | 1 | 50 | 6.18±0.25 |
| AF-10 | 1 | 3 | 50 | 8.89±0.36 |
| AF-11 | 1 | 2 | 50 | 2.39±0.10 |
| AF-12 | 1 | 1 | 50 | 15.60±0.62 |
| Sample | 0.2 M HCl (Time, h) | 0.2 M HCl (Temp., °C) | 2% Sodium carbonate (Time, h) | 0.2 M Sodium carbonate (Temp., °C) | Yield (% w/w)* |
|---|---|---|---|---|---|
| AE-1 | 3 | 50 | 2 | 45 | 15.37±0.61 |
| AE-2 | 2.5 | 50 | 2 | 45 | 14.39±0.58 |
| AE-3 | 2 | 50 | 2 | 45 | 12.67±0.51 |
| AE-4 | 1.5 | 50 | 2 | 45 | 11.19±0.45 |
| AE-5 | 3 | 50 | 3 | 45 | 19.87±0.79 |
| AE-6 | 2.5 | 50 | 3 | 45 | 16.65±0.67 |
| AE-7 | 2 | 50 | 3 | 45 | 11.00±0.44 |
| AE-8 | 1.5 | 50 | 3 | 45 | 14.43±0.58 |
| AE-9 | 3 | 50 | 1 | 45 | 13.87±0.55 |
| AE-10 | 2.5 | 50 | 1 | 45 | 10.53±0.42 |
| AE-11 | 2 | 50 | 1 | 45 | 12.26±0.49 |
| AE-12 | 1.5 | 50 | 1 | 45 | 6.97±0.28 |
| Macroalgae Species | Extraction Conditions | Yield (%) | References |
|---|---|---|---|
| Sargassum vulgare (SVHV) | 2% formaldehyde (24 h), 0.2 M HCl (24 h), 2% Na2CO3 (5 h at 60-80 °C) | 16.90 | Torres et al. (2007)[52] |
| Sargassum vulgare (SVLV) | 2% formaldehyde (24 h), 0.2 M HCl (24 h), 2% Na2CO3 (5 h at 60-80 °C) | 16.90 | Torres et al. (2007)[52] |
| Nizimuddinia zanardini | 2% formaldehyde (24 h), 0.2 M HCl (3 h at 60 °C), 3% Na2CO3 (2.5 h at 60 °C) | 24.00 | Khajouei et al. (2018)[21] |
| Macrocystis pyrifera | 0.2% formaldehyde (overnight), 0.1 N HCl (30 min), 0.5% Na2CO3 (1 h at 60 °C) | 23.15 | Gao et al. (2018)[53] |
| Sargassum natans | 2% formaldehyde (overnight), 0.2 M HCl, 2% Na2CO3 (3 h at 99 °C) | 23.00 | Rhein-Knudsen et al. (2017)[15] |
| Sargassum vulgare | 17.00 | ||
| Padina gymnospora | 16.00 | ||
| Padina antillarum | 22.00 | ||
| Laminaria digitate | 29.00 | ||
| Macrocystis pyrifera | 26.00 | ||
| Waste Sargassum natans | 2% formaldehyde (overnight), 5% Na2CO3 (15:1 alkali:alga ratio, 2 h at 65 °C) | 15.00 | Mohammed et al. (2018)[54] |
| Durvillaea antarctica | 2% formaldehyde (24 h), 0.2 M HCl (3 h at 60 °C), 3% Na2CO3 (2.5 h at 60 °C) | 20.80 | Caballero et al. (2021)[55] |
| Ascophyllum nodosum | 0.2 M HCl (12 h at RT), 0.1 M NaHCO3 (2 h at RT) | 13.80 | Bojorgues et al. (2022)[49] |
| Saccharina latissima | 11.20 | ||
| Sargassum turbinaroides | 2% formaldehyde (24 h at 90 °C), 0.2 M HCl (24 h), 2% Na2CO3 (3 h at 100 °C) | 10.00 | Laroche et al. (2009)[56] |
| Cystoseira barbata | 0.1 M HCl (2 h at 60 °C), 3% Na2CO3 (2 h at 60 °C) | 9.90 | Sellimi et al. (2015)[57] |
| Macroalgae Species | Intrinsic Viscosity (dL/g) | Approx. Molecular Weight (x105 g/mol) |
References |
|---|---|---|---|
| Sargassum vulgare (SVHV) | 6.9 | 3.30 | Torres et al. (2007)[52] |
| Sargassum vulgare(SVLV) | 4.1 | 1.94 | Torres et al. (2007)[52] |
| Nizimuddinia zanardini | 3.42 | 1.03 | Khajouei et al. (2018)[21] |
| Sargassum natans | - | 5.69 | Rhein-Knudsen et al. (2017)[15] |
| Sargassum vulgare | - | 5.14 | |
| Padina gymnospora | - | 4.82 | |
| Laminaria digitate | - | 7.56 | |
| Macrocystis pyrifera | - | 7.19 | |
| Cystoseira barbata | 2.83 | 2.04 | Sellimi et al. (2015)[57] |
| Sargassum cristaefolium | 4.47 | 2.11 | Sugiono et al. (2019)[65] |
| Cystoseira barbata | 4.06 | 1.26 | Trica et al. (2019)[66] |
| Sargassum vulgare | - | 1.10 | Sari et al. (2016)[69] |
| Sargassum spp. (Formaldehyde) | 2.13±0.0079* | 1.00±0.004* | This study |
| Sargassum spp. (Ethanol) | 1.33±0.012* | 0.62±0.006* | |
| Commercial sodium alginate | 1.93±0.093* | 0.94±0.044* |
| Species | FG | FM | FGG | FMM | FMG | M/G | Reference |
|---|---|---|---|---|---|---|---|
| Laminaria digitata | 0.47 | 0.53 | 0.41 | 0.47 | 0.06 | 1.12 | Fertah et al. (2017)[42] |
| Sargassum natans | 0.68 | 0.32 | 0.61 | 0.25 | 0.07 | 0.47 | Rhein-Knudsen et al. (2017)[15] |
| Sargassum vulgare | 0.59 | 0.41 | 0.49 | 0.31 | 0.10 | 0.70 | |
| Padina gymnospora | 0.36 | 0.64 | 0.21 | 0.48 | 0.16 | 1.75 | |
| Padina antillarum | 0.35 | 0.65 | 0.15 | 0.45 | 0.20 | 1.85 | |
| Laminaria digitata | 0.31 | 0.69 | 0.16 | 0.53 | 0.16 | 2.19 | |
| Macrocystis pyrifera | 0.34 | 0.66 | 0.13 | 0.45 | 0.21 | 1.94 | |
| Sargassum turbinarioides Grunow | 0.52 | 0.48 | 0.39 | 0.36 | 0.25 | 0.94 | Fenoradosoa et al. (2010)[20] |
| Sargassum fluitans | 0.64 | 0.36 | 0.55 | 0.28 | 0.08 | 0.57 | Davis et al. (2004)[38] |
| Saccharina longicruris | 0.59 | 0.41 | 0.25 | 0.07 | 0.34 | 0.69 | Rioux et al. (2007)[75] |
| Ascophyllum nodosum | 0.54 | 0.46 | 0.36 | 0.28 | 0.18 | 0.85 | |
| Fucus vesiculosus | 0.41 | 0.59 | 0.22 | 0.39 | 0.19 | 1.44 | |
| Sargassum asperifolium | 0.59 | 0.41 | 0.48 | 0.30 | 0.11 | 0.69 | Larsen et al. (2003)[76] |
| Sargassum filipendula | 0.56 | 0.44 | 0.45 | 0.33 | 0.11 | 0.78 | Bertagnolli et al. (2014)[77] |
| Sargassum filipendula | 0.84 | 0.16 | 0.76 | 0.07 | 0.08 | 0.19 | Davis et al. (2003)[79] |
| Sargassum muticum | 0.76 | 0.24 | 0.59 | 0.07 | 0.17 | 0.31 | |
| Sargassum oligocystum | 0.57 | 0.43 | 0.37 | 0.24 | 0.20 | 0.77 | |
| Sargassum polycystum | 0.82 | 0.18 | 0.77 | 0.12 | 0.05 | 0.21 | |
| Sargassum thunbergii | 0.80 | 0.20 | 0.75 | 0.16 | 0.05 | 0.25 | |
| Sargassum spp. (AF-12) | 0.55 | 0.45 | 0.53 | 0.43 | 0.02 | 0.82 | This study |
| Sargassum spp. (AE-5) | 0.60 | 0.40 | 0.54 | 0.34 | 0.05 | 0.67 | |
| Commercial sodium alginate | 0.47 | 0.53 | 0.27 | 0.34 | 0.19 | 1.14 |
| Species | FGGG | FMGM | FGGM | Reference |
|---|---|---|---|---|
| Sargassum natans | 0.58 | 0.04 | 0.03 | Rhein-Knudsen et al. (2017)[15] |
| Sargassum vulgare | 0.44 | 0.05 | 0.05 | |
| Padina gymnospora | 0.17 | 0.12 | 0.03 | |
| Padina antillarum | 0.12 | 0.17 | 0.03 | |
| Laminaria digitata | 0.11 | 0.11 | 0.05 | |
| Macrocystis pyrifera | 0.10 | 0.17 | 0.03 | |
| Sargassum spp. (AF-12) | 0.52 | 0.0009 | 0.003 | This study |
| Sargassum spp. (AE-5) | 0.51 | 0.003 | 0.022 | |
| Commercial sodium alginate | 0.14 | 0.026 | 0.003 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




