Submitted:
17 April 2026
Posted:
20 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Statistical Analysis
3. Results
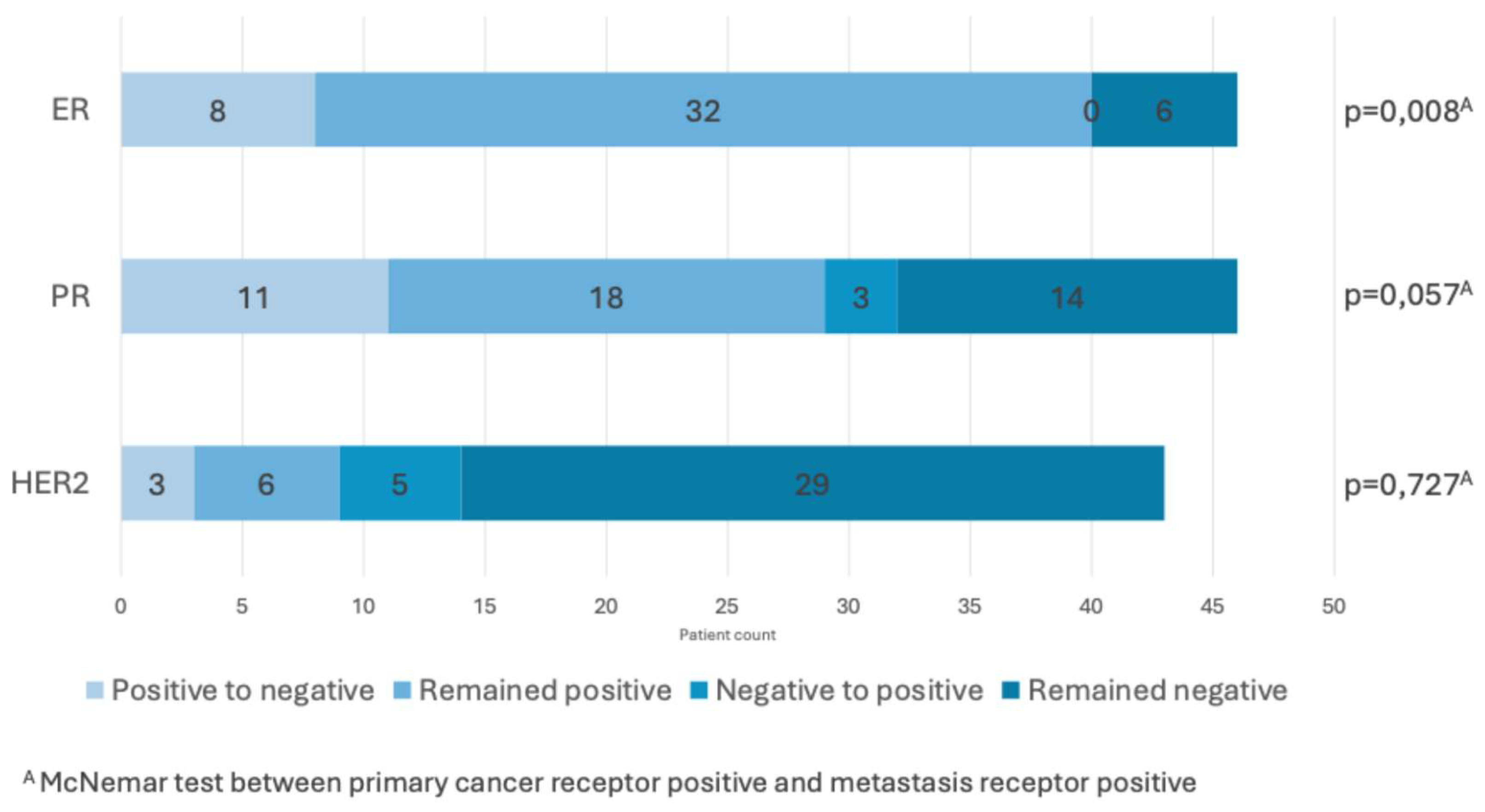
Receptor Changes in Metastatic Tissue
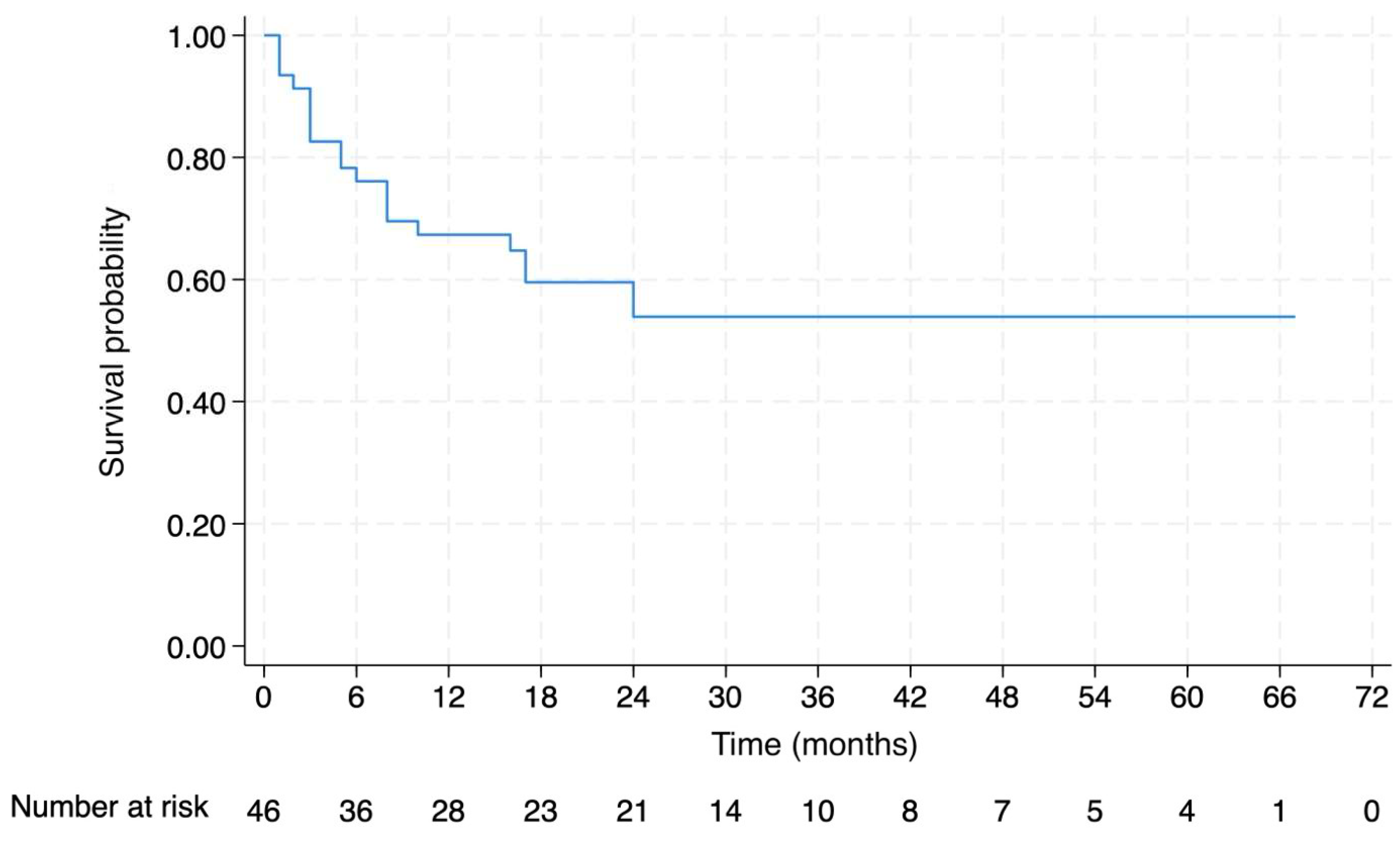
Survival Analysis According to Metastatic Site
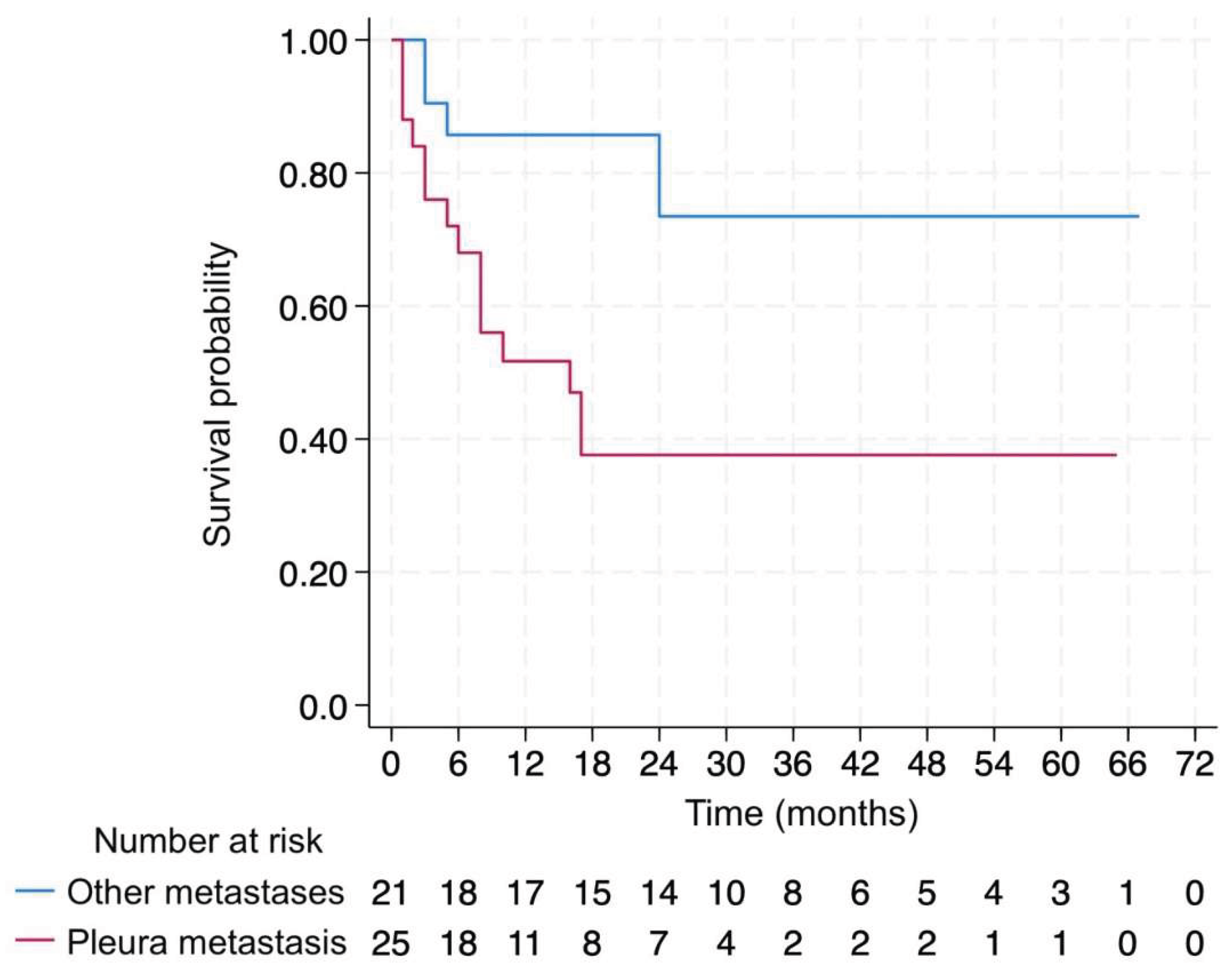
Survival Analysis After Thoracic Metastasis
Univariate Cox Regression Analysis
Multivariate Cox Regression Analysis
4. Discussion
Limitatons
5. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Yi, Z.B.; Yu, P.; Zhang, S.; Wang, W.N.; Han, Y.Q.; Ouyang, Q.C.; et al. Profile and outcome of receptor conversion in breast cancer metastases: a nation-wide multicenter epidemiological study. Int J Cancer 2021, 148[3], 692–701. [Google Scholar] [CrossRef]
- Van Poznak, C.; Somerfield, M.R.; Bast, R.C.; Cristofanilli, M.; Goetz, M.P.; Gonzalez-Angulo, A.M.; et al. Use of biomarkers to guide decisions on systemic therapy for women with metastatic breast cancer: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2015, 33[24], 2695–704. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Tong, Y.; Chen, X.; Shen, K. Association of biomarker discrepancy and treatment decision, disease outcome in recurrent/metastatic breast cancer patients. Front Oncol. 2021, 11, 638619. [Google Scholar] [CrossRef]
- McAnena, P.F.; McGuire, A.; Ramli, A.; Curran, C.; Malone, C.; McLaughlin, R.; et al. Breast cancer subtype discordance: impact on post-recurrence survival and potential treatment options. BMC Cancer 2018, 18[1], 203. [Google Scholar] [CrossRef]
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Breast Cancer. Version 1. 2026. Available online: https://www.nccn.org/professionals/physician_gls/pdf/breast.pdf.
- Cardoso, F.; Senkus, E.; Costa, A.; Papadopoulos, E.; Aapro, M.; André, F.; et al. ESO-ESMO International Consensus Guidelines for Advanced Breast Cancer [ABC 4]. Ann Oncol. 2018, 29[8], 1634–57. [Google Scholar] [CrossRef] [PubMed]
- Cazzaniga, M.E.; Pronzato, P.; Amoroso, D.; Arpino, G.; Atzori, F.; Beano, A.; et al. Survival outcomes of luminal metastatic breast cancer patients according to changes in molecular subtype at re-biopsy: insights from the GIM-13-AMBRA study. Cancers [Basel]. 2025, 17[10], 1715. [Google Scholar] [CrossRef] [PubMed]
- Lindstrom, L.S.; Karlsson, E.; Wilking, U.M.; et al. Clinically used breast cancer markers such as estrogen receptor, progesterone receptor, and human epidermal growth factor receptor 2 are unstable throughout tumor progression. J Clin Oncol. 2012, 30, 2601–2608. [Google Scholar] [CrossRef]
- Shiino, S.; Ball, G.; Syed, B.M.; Kurozumi, S.; Green, A.R.; Tsuda, H.; et al. Prognostic significance of receptor expression discordance between primary and recurrent breast cancers: a meta-analysis. Breast Cancer Res Treat 2022, 191[1], 1–14. [Google Scholar] [CrossRef]
- Duffy, M.J.; Harbeck, N.; Nap, M.; Molina, R.; Nicolini, A.; Senkus, E.; et al. Clinical use of biomarkers in breast cancer: Updated guidelines from the European Group on Tumor Markers [EGTM]. Eur J Cancer 2017, 75, 284–98. [Google Scholar] [CrossRef]
- Amir, E.; Miller, N.; Geddie, W.; et al. Prospective study evaluating the impact of tissue confirmation of metastatic disease in patients with breast cancer. J Clin Oncol. 2012, 30, 587–592. [Google Scholar] [CrossRef]
- Zhao, W.; Sun, L.; Dong, G.; Wang, X.; Jia, Y.; Tong, Z. Receptor conversion impacts outcomes of different molecular subtypes of primary breast cancer. Ther Adv Med Oncol. 2021, 13, 17588359211012982. [Google Scholar] [CrossRef]
- Shen, J.; Xu, L.; Shi, J.; Zhao, L.; Shi, S.; Feng, J.; et al. Prognostic Value and Influence of Receptor Conversion on Treatment Regimen in Metastatic Breast Cancer at the First Time of Recurrence. Oncol Res Treat 2020, 43[11], 620–7. [Google Scholar] [CrossRef] [PubMed]
- Francis, I.M.; Alath, P.; George, S.S.; Jaragh, M.; Al Jassar, A.; Kapila, K. Metastatic breast carcinoma in pleural fluid: Correlation of receptor and HER2 status with the primary carcinoma-a pilot study. Diagn Cytopathol 2016, 44[12], 980–6. [Google Scholar] [CrossRef] [PubMed]
- Niikura, N.; Liu, J.; Hayashi, N.; et al. Loss of human epidermal growth factor receptor 2 [HER2] expression in metastatic sites of HER2-overexpressing primary breast tumors. J Clin Oncol. 2012, 30, 593–599. [Google Scholar] [CrossRef]
- Schrijver, W.; Suijkerbuijk, K.P.M.; van Gils, C.H.; van der Wall, E.; Moelans, C.B.; van Diest, P.J. Receptor conversion in distant breast cancer metastases: a systematic review and meta-analysis. J Natl Cancer Inst. 2018, 110, 568–580. [Google Scholar] [CrossRef]
- Grinda, T.; Joyon, N.; Lusque, A.; Lefevre, S.; Arnould, L.; Penault-Llorca, F.; et al. Phenotypic discordance between primary and metastatic breast cancer in the large-scale real-life multicenter French ESME cohort. NPJ Breast Cancer 2021, 7[1], 41. [Google Scholar] [CrossRef]
- Sundén, M.; Norgren, S.; Lundqvist, R.; Andersson, A.; Sund, M.; Hemmingsson, O. Receptor conversion and survival in breast cancer liver metastases. Breast Cancer Res. 2023, 25[1], 105. [Google Scholar] [CrossRef] [PubMed]
- Yoshino, R.; Takahashi, K.; Hatanaka, N.; et al. Analysis of prognostic factors after pulmonary resection for metastatic breast cancer: a 23-year single-institution retrospective study. Gen Thorac Cardiovasc Surg 2025. [Google Scholar] [CrossRef]
- Connolly, J.E., Jr.; Erasmus, J.J.; Patz, E.F., Jr. Thoracic manifestations of breast carcinoma: metastatic disease and complications of treatment. Clin Radiol. 1999, 54[8], 487–94. [Google Scholar] [CrossRef]
- Butow, P.N.; Coates, A.S.; Dunn, S.M. Psychosocial predictors of survival: metastatic breast cancer. Ann Oncol. 2000, 11[4], 469–74. [Google Scholar] [CrossRef]
- Li, S.; Li, C.; Shao, W.; Liu, X.; Sun, L.; Yu, Z. Survival analysis and prognosis of patients with breast cancer with pleural metastasis. Front Oncol. 2023, 13, 1104246. [Google Scholar] [CrossRef] [PubMed]
- Light, R.W. Malignant pleural and pericardial effusions. In Sabiston & Spencer Surgery of the Chest, 10th ed.; Sellke, F.W., Rutherford, R.B., Brunicardi, F.C., Eds.; Elsevier: Philadelphia, 2024; pp. 554–66. [Google Scholar]
- Deluche, E.; Antoine, A.; Bachelot, T.; Lardy-Cleaud, A.; Dieras, V.; Brain, E.; et al. Contemporary outcomes of metastatic breast cancer among 22,000 women from the multicentre ESME cohort 2008-2016. Eur J Cancer 2020, 129, 60–70. [Google Scholar] [CrossRef]
- Aurilio, G.; Disalvatore, D.; Pruneri, G.; Bagnardi, V.; Viale, G.; Curigliano, G.; et al. A meta-analysis of oestrogen receptor, progesterone receptor and human epidermal growth factor receptor 2 discordance between primary breast cancer and metastases. Eur J Cancer 2014, 50[2], 277–89. [Google Scholar] [CrossRef] [PubMed]
- Dowling, G.P.; Keelan, S.; Cosgrove, N.S.; Daly, G.R.; Giblin, K.; Toomey, S.; et al. Receptor discordance in metastatic breast cancer: a review of clinical and genetic subtype alterations from primary to metastatic disease. Breast Cancer Res Treat. 2024, 207[3], 471–6. [Google Scholar] [CrossRef] [PubMed]
| n=46 | |
| Age at diagnosis [years] | 51.6±13.0 |
| Age range at diagnosis [years] | 26–81 |
| Specimen | |
| Pleura | 25 [54.3%] |
| Lung | 9 [19.6%] |
| Lymph node | 5 [10.9%] |
| Pericardium | 5 [10.9%] |
| Chest wall | 2 [4.3%] |
| Vital status after thoracic metastasis | |
| Alive | 26 [56.5%] |
| Deceased | 20 [43.5%] |
| Follow-up duration after thoracic metastasis [months] | 18 [1–67] |
| HR [95% CI] | Wald | p-value | |
| Metastasis | |||
| ER negative | 3.306 [1.365–8.011] | 7.014 | 0.008 |
| PR negative | 2.793 [1.065–7.324] | 4.363 | 0.037 |
| HER2 negative | 0.992 [0.322–3.053] | 0.001 | 0.988 |
| Receptor conversion [Metastatic vs primary] | |||
| ER conversion to negative | 3.095 [1.181–8.111] | 5.282 | 0.022 |
| PR conversion to negative | 1.968 [0.751–5.163] | 1.895 | 0.169 |
| HER2 conversion to negative | 0.656 [0.087–4.958] | 0.167 | 0.683 |
| HR [95% CI] | Wald | p-value | |
| Age at diagnosis | 1.020 [0.981–1.062] | 1.016 | 0.313 |
| Pleural metastasis | 3.238 [1.125–9.324] | 4.743 | 0.029 |
| ER conversion to negative | 3.669 [1.362–9.885] | 6.606 | 0.010 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




