Submitted:
16 April 2026
Posted:
17 April 2026
You are already at the latest version
Abstract
The increasing global frequency of harmful cyanobacterial blooms (CyanoHABs), driven by nutrient enrichment and climate change, poses a severe threat to aquatic ecosystems and public health. This study evaluates the effectiveness of novel clay-polymer nanocomposites—combining the charge-neutralizing capabilities of polydiallyldimethylammonium chloride (PolyDADMAC) with the high density of clay minerals (kaolinite and sepiolite) for the rapid removal of toxic cyanobacteria from water. Laboratory-scale experiments were conducted using Microcystis aeruginosa, Aphanizomenon ovalisporum, and Chlorella sp., with treatment doses determined by particle charge detector (PCD) measurements to identify the "nominal dose" required for full charge neutralization. Results demonstrate that clay-polymer nanocomposites achieve over 95% removal of turbidity and chlorophyll in M. aeruginosa at doses significantly lower (15–20%) than the calculated nominal dose, likely due to specific physical bridging interactions with the cyanobacteria’s external exopolysaccharide fibers. In contrast, A. ovalisporum and Chlorella sp. required doses closer to full charge neutralization for optimal removal. Among the materials tested, kaolinite-based nanocomposites (DKG24) showed slightly superior and more stable performance than sepiolite-based versions. Notably, application at or above the nominal dose was associated with increased soluble microcystin levels, suggesting that excessive polymer concentrations may compromise cell integrity and lead to toxin leakage. These findings suggest that engineered nanocomposites offer highly efficient, scalable technology for CyanoHAB management, provided that operational doses are carefully optimized to maximize biomass removal while minimizing toxin release.
Keywords:
1. Introduction
- Their efficiency in physically removing intact cyanobacteria and other common phytoplankton species from water samples.
- The influence of key operational parameters (e.g., contact time, nanocomposite dose) on the removal process.
2. Materials and Methods
2.1. Cyanobacteria and algae
2.2. Clays and Polymers
2.3. Particle charge detector (PCD) measurements
2.4. Experimental Set Up
2.5. Analysis of Microcystins
3. Results
3.1. Cell Surface Charge Density and Nominal Dose
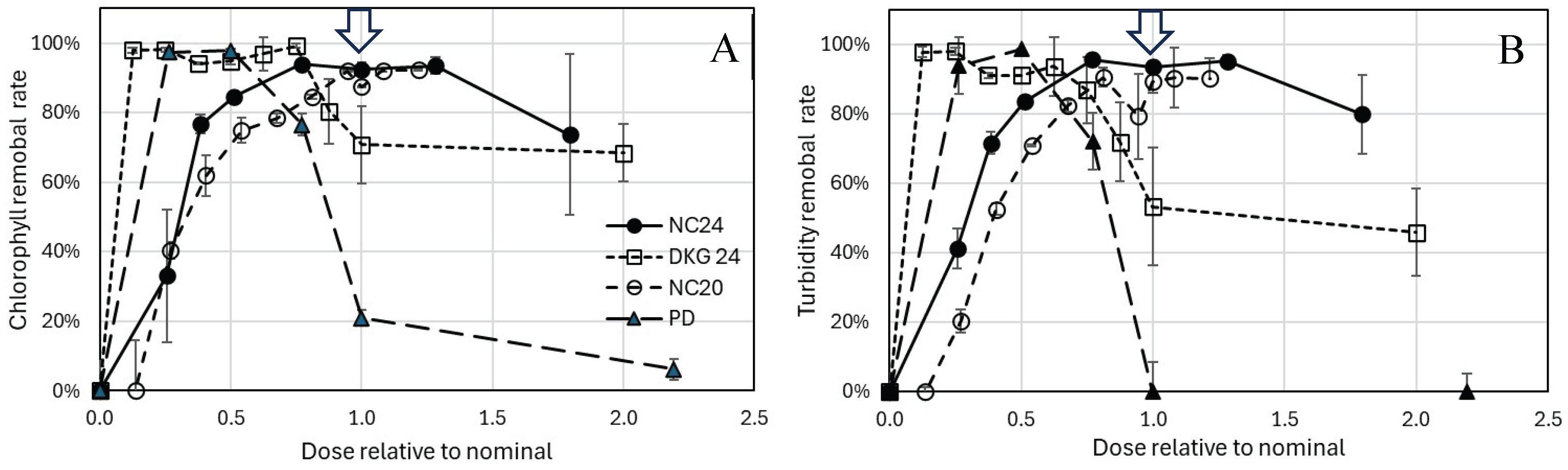
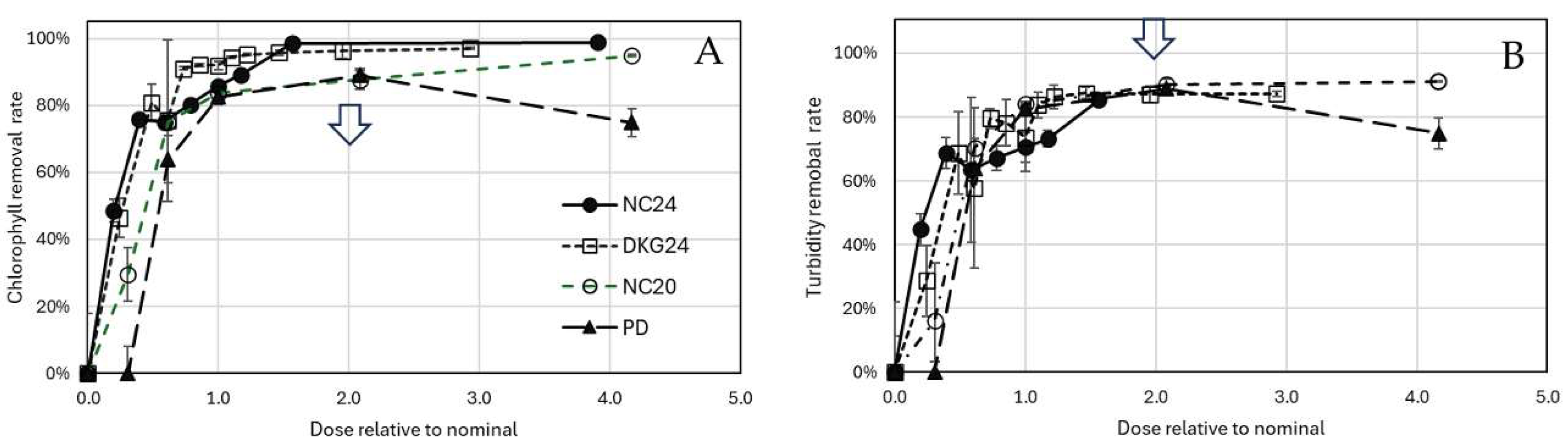
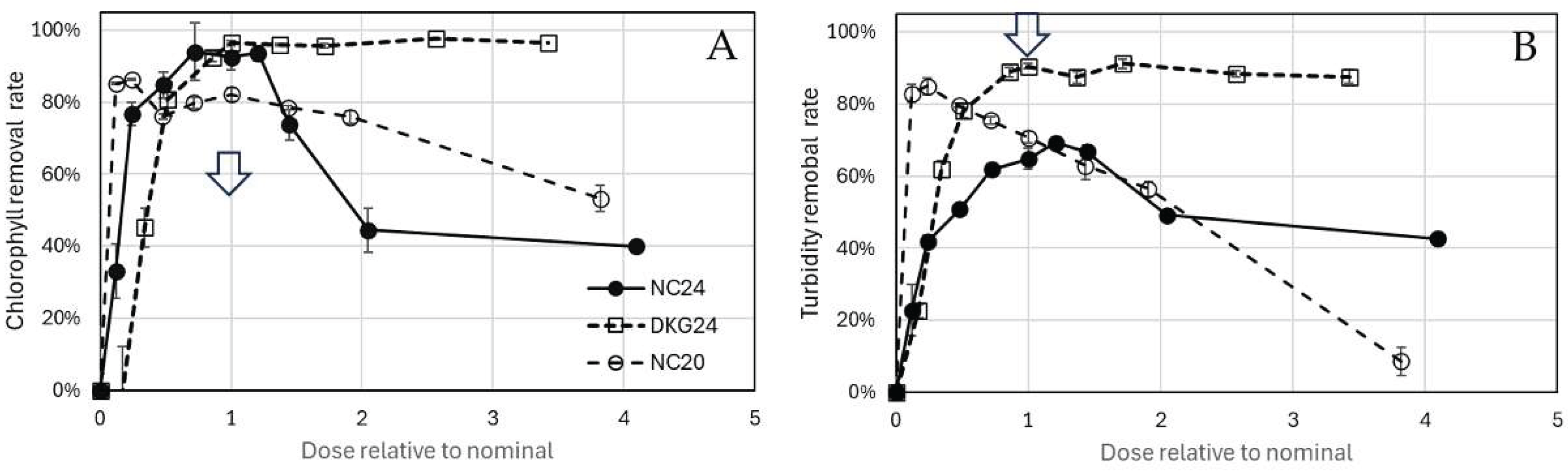
3.2. Algae and Cyanobacteria Removal by Nanocomposites
3.3. Charge neutralization and biomass aggregation
Effect of nanocomposites on the removal of microcystina
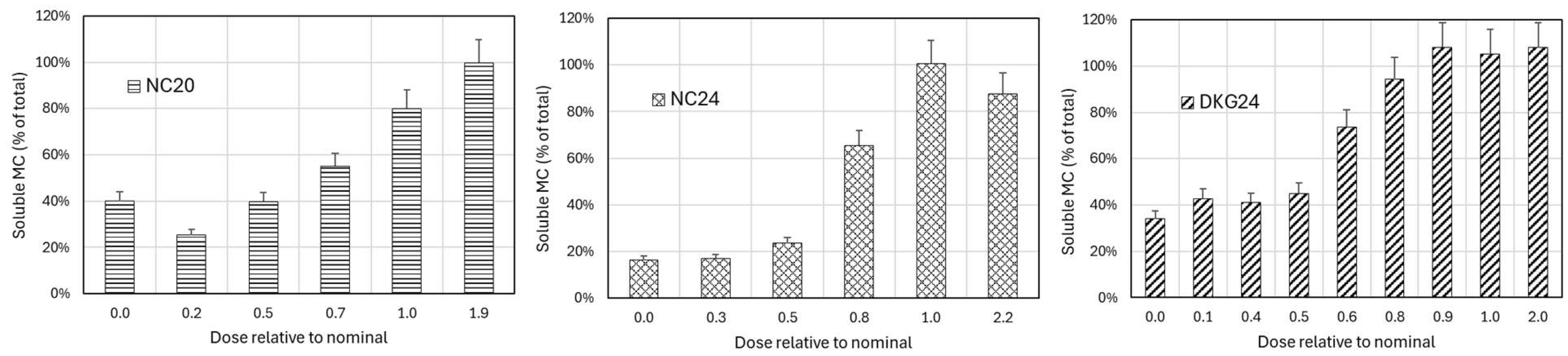
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PD | PolyDADMAC |
| PCD | Particle charge detector |
| MC | Microcystin |
| CC | Charge concentration |
| HPLC- DAD | High Performance Liquid Chromatography and Diode array detector |
| CyanoHABs | Harmful cyanobacterial blooms |
References
- Huisman, J.; Codd, G.A.; Paerl, H.W.; Ibelings, B.W.; Verspagen, J.M.H.; Visser, P.M. Cyanobacterial Blooms. Nat. Rev. Microbiol. 2018 168 2018, 16, 471–483. [Google Scholar] [CrossRef]
- Chorus, I.; Welker, M. Toxic Cyanobacteria in Water. Toxic Cyanobacteria in Water 2021. [Google Scholar] [CrossRef]
- Song, L.; Jia, Y.; Qin, B.; Li, R.; Carmichael, W.W.; Gan, N.; Xu, H.; Shan, K.; Sukenik, A. Harmful Cyanobacterial Blooms: Biological Traits, Mechanisms, Risks, and Control Strategies. Annu. Rev. Environ. Resour. 2023, 48, 123–147. [Google Scholar] [CrossRef]
- Sukenik, A.; Kaplan, A. Cyanobacterial Harmful Algal Blooms in Aquatic Ecosystems: A Comprehensive Outlook on Current and Emerging Mitigation and Control Approaches. Microorg. 2021, Vol. 9, 2021, 9. [Google Scholar] [CrossRef] [PubMed]
- Hobbs, W.; Rosewood, M. Alum Treatments Literature Review A Review on the Use and Monitoring of Alum Treatments to Control Algal Blooms; Olympia, WA, 2025. [Google Scholar]
- Tahraoui, H.; Toumi, S.; Boudoukhani, M.; Touzout, N.; Sid, A.N.E.H.; Amrane, A.; Belhadj, A.E.; Hadjadj, M.; Laichi, Y.; Aboumustapha, M.; et al. Evaluating the Effectiveness of Coagulation–Flocculation Treatment Using Aluminum Sulfate on a Polluted Surface Water Source: A Year-Long Study. Water (Switzerland) 2024, 16. [Google Scholar] [CrossRef]
- Gerchman, Y.; Vasker, B.; Tavasi, M.; Mishael, Y.; Kinel-Tahan, Y.; Yehoshua, Y. Effective Harvesting of Microalgae: Comparison of Different Polymeric Flocculants. Bioresour. Technol. 2017, 228, 141–146. [Google Scholar] [CrossRef]
- Wang, C.; Yang, Y.; Hou, J.; Wang, P.; Miao, L.; Wang, X.; Guo, L. Optimization of Cyanobacterial Harvesting and Extracellular Organic Matter Removal Utilizing Magnetic Nanoparticles and Response Surface Methodology: A Comparative Study. Algal Res. 2020, 45, 101756. [Google Scholar] [CrossRef]
- Jia, L.; Sun, N.; Wang, Z.; Tian, Y.; Fang, L.; Huang, X. Mimicking Biological Method with Inorganic and Organic Compounds Modified Clays for Continuous Controlling of Microcystis Aeruginosa. Environ. Technol. Innov. 2024, 36, 103821. [Google Scholar] [CrossRef]
- Churchman, G.J.; Gates, W.P.; Theng, B.K.G.; Yuan, G. Chapter 11.1 Clays and Clay Minerals for Pollution Control. In Developments in Clay Science; Fa00EFza Bergaya, B.K.G.T. and G.L., Ed.; Handbook of Clay Science; Elsevier, 2006; Vol. Volume 1, pp. 625–675 ISBN 1572-4352.
- Lagaly, G.; Ogawa, M.; Dékány, I. Clay Mineral–Organic Interactions. Dev. Clay Sci. 2013, 5, 435–505. [Google Scholar] [CrossRef]
- Rytwo, G. The Use of Clay-Polymer Nanocomposites in Wastewater Pretreatment. Sci. World J. 2012, 2012, 1–7. [Google Scholar] [CrossRef]
- Rytwo, G. Hybrid Clay-Polymer Nanocomposites for the Clarification of Water and Effluents. Recent Pat. Nanotechnol. 2017, 11, 181–193. [Google Scholar] [CrossRef]
- Gardi, I.; Mishael, Y.G.; Lindahl, M.; Muro-Pastor, A.M.; Undabeytia, T. Coagulation-Flocculation of Microcystis Aeruginosa by Polymer-Clay Based Composites. J. Clean. Prod. 2023, 394, 136356. [Google Scholar] [CrossRef]
- Banker, R.; Carmeli, S.; Hadas, O.; Teltsch, B.; Porat, R.; Sukenik, A. Identification of cylindrospermopsin in Aphanizomenon ovalisporum (cyanophyceae) isolated from Lake Kinneret, Israel. J. Phycol. 1997, 33, 613–616. [Google Scholar] [CrossRef]
- Kaplan-Levy, R.N.; Alster, A.; Shlichter, M.; Zohary, T. The Israel National Culture Collection of Algae (INCCA) for Biodiversity Conservation. Nov. Hedwigia 2020, 111, 231–243. [Google Scholar] [CrossRef]
- Stanier, R.Y.; Kunisawa, R.; Mandel, M.; Cohen-Bazire, G. Purification and Properties of Unicellular Blue-Green Algae (Order Chroococcales). Bacteriol. Rev. 1971, 35, 171–205. [Google Scholar] [CrossRef]
- Holm-Hansen, O.; Lorenzen, C.J.; Holmes, R.W.; Strickland, J.D.H. Fluorometric Determination of Chlorophyll. J. du Cons. 1965, 30, 3–15. [Google Scholar] [CrossRef]
- Rytwo, G.; Lavi, R.; Rytwo, Y.; Monchase, H.; Dultz, S.; König, T.N. Clarification of Olive Mill and Winery Wastewater by Means of Clay-Polymer Nanocomposites. Sci. Total Environ. 2013, 442, 134–142. [Google Scholar] [CrossRef]
- Rytwo, G.; Lavi, R.; König, T.N.; Avidan, L. Direct Relationship Between Electrokinetic Surface-Charge Measurement of Effluents and Coagulant Type and Dose. Colloids Interface Sci. Commun. 2014, 1, 27–30. [Google Scholar] [CrossRef]
- Rytwo, G. US20190152813 Method for Production of Potable Water 2019.
- Gerdes, W.F. A New Instrument – The Streaming Current Detector. 12th Natl. ISA Anal. Instrum. Symp. 1966, 181–198. [Google Scholar]
- Delgado, A. V.; González-Caballero, F.; Hunter, R.J.; Koopal, L.K.; Lyklema, J. Measurement and Interpretation of Electrokinetic Phenomena. J. Colloid Interface Sci. 2007, 309, 194–224. [Google Scholar] [CrossRef]
- Fairhurst, D. An Overview of the Zeta Potential - Part 1: The Concept. Available online: http://www.americanpharmaceuticalreview.com/Featured-Articles/133232-An-Overview-of-the-Zeta-Potential-Part-1-The-Concept/ (accessed on 14 July 2016).
- Fairhurst, D.; Lee, R.W. The Zeta Potential & Its Use in Pharmaceutical Applications – Part 1: Charged Interfaces in Polar & Non-Polar Media & the Concept of the Zeta Potential. Drug Dev. Deliv. 2011, 11, 60. [Google Scholar]
- Sun, F.; Zhang, H.; Qian, A.; Yu, H.; Xu, C.; Pan, R.; Shi, Y. The Influence of Extracellular Polymeric Substances on the Coagulation Process of Cyanobacteria. Sci. Total Environ. 2020, 720, 137573. [Google Scholar] [CrossRef]
- Braun, M.; Bernard, T.; Ung, H.; Piller, O.; Gilbert, D. Computational Fluid Dynamics Modeling of Contaminant Mixing at Junctions for an Online Security Management Toolkit in Water Distribution Networks. J. Water Supply Res. Technol. 2015, 64, 504–515. [Google Scholar] [CrossRef]
- Harel, M.; Weiss, G.; Daniel, E.; Wilenz, A.; Hadas, O.; Sukenik, A.; Sedmak, B.; Dittmann, E.; Braun, S.; Kaplan, A. Casting a Net: Fibres Produced by Microcystis Sp. in Field and Laboratory Populations. Environ. Microbiol. Rep. 2012, 4, 342–349. [Google Scholar] [CrossRef] [PubMed]
- Sukenik, A.; Viner-Mozzini, Y.; Mizrahi, D.; Tamam, I.; Benitez, A.R.; Nir, S. Removal of Cyanotoxins–Microcystins from Water by Filtration through Granulated Composites of Bentonite with Micelles of the Cation Octadecyltrimethyl Ammonium (ODTMA). Appl. Nano 2021, 2, 67–81. [Google Scholar] [CrossRef]





| Name | properties | Clay type | Polymer type |
Polymer to clay mass ratio |
Reference |
| NC20 | nanocomposite | Sepiolite | PolyDADMAC | 0.6:1 | [20] |
| NC24 | nanocomposite | Sepiolite | PolyDADMAC | 1.8:1 | [19] |
| DKG24 | nanocomposite | Kaolinite | PolyDADMAC | 1.8:1 | [21] |
| PolyDADMAC | polymer | N/A2 | N/A2 |
| Organism | Turbidity [NTU] |
Chlorophyll [μmol/L] | Charge density1 [molc /mol Chl] |
Nominal dose2 [mg/L] |
|---|---|---|---|---|
| Microcystis | 54 | 0.9 | 41.9 | 5.6 (DKG24) |
| 100 | 1.6 | 69.1 | 10.8 (PD) | |
| 157 | 1.8 | 21.8 | 14.8 (NC20) | |
| 264 | 2.9 | 13.0 | 8.2 (NC24) | |
| 379 | 6.1 | 18.0 | 15.4 PD) | |
| Aphanizomenon | 121 | 1.5 | 24.6 | 5.7 (GKG24) |
| 275 | 3.2 | 15.5 | 10.8 (NC24) | |
| 1224 | 2972 | 36.3 | 12.1 (PD) | |
| 3055 | 6193 | 31.8 | 31.1 (PD) | |
| Chlorella | 55 | 3347 | 12.0 | 29.3 (NC20) |
| 99 | 3420 | 13.8 | 8.2 (DKG24) | |
| 104 | 4424 | 16.1 | NC24) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




