Submitted:
16 April 2026
Posted:
20 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
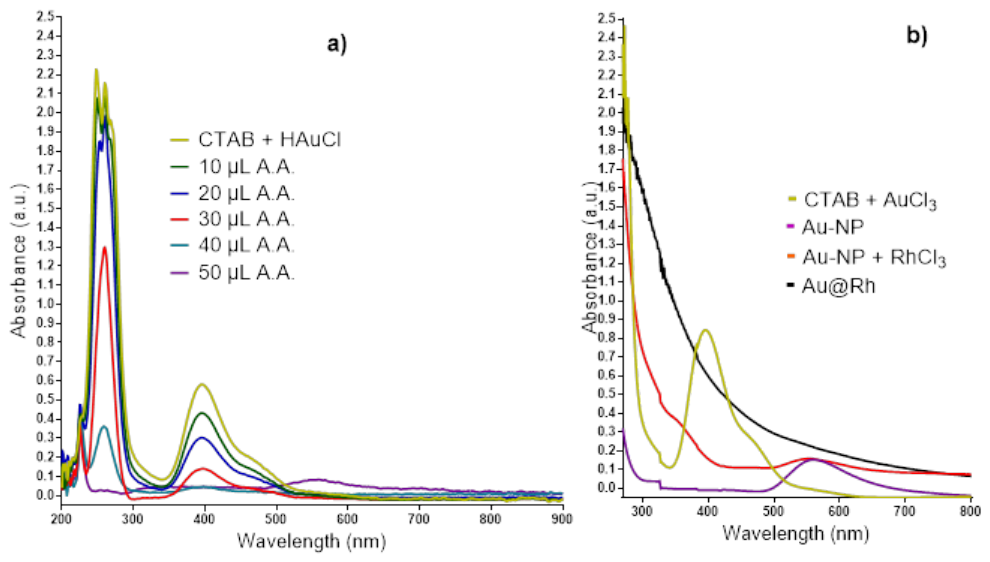
2.1. Optical Properties of Au@Rh Nanoparticles
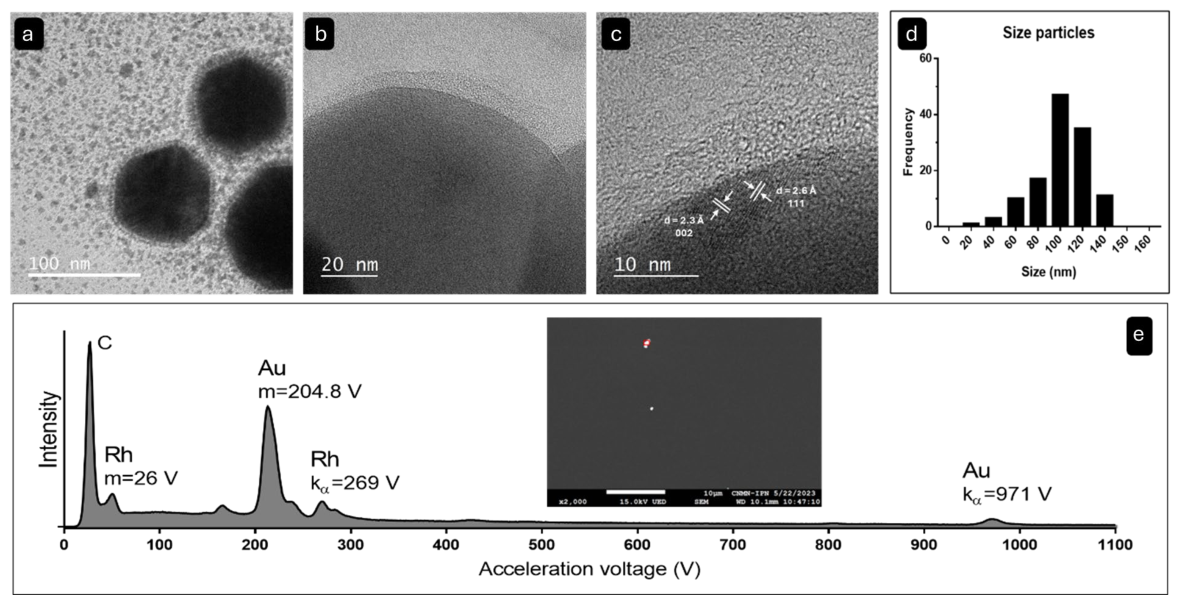
2.2. Structural Characterization of Au@Rh NPs
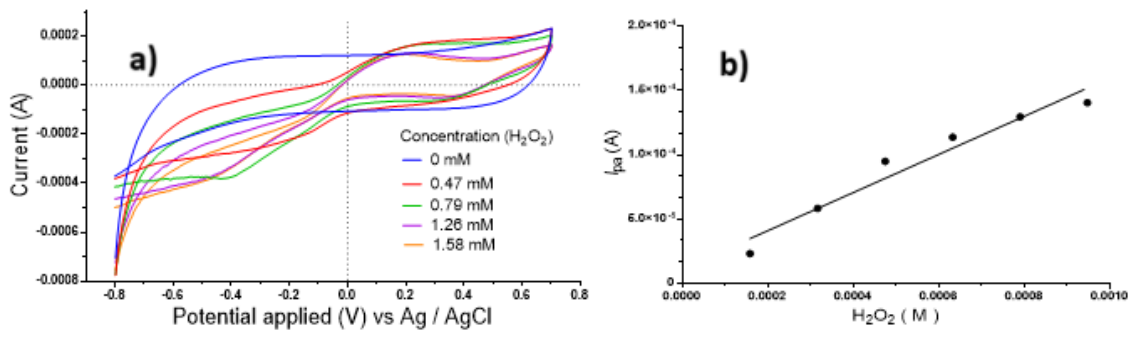
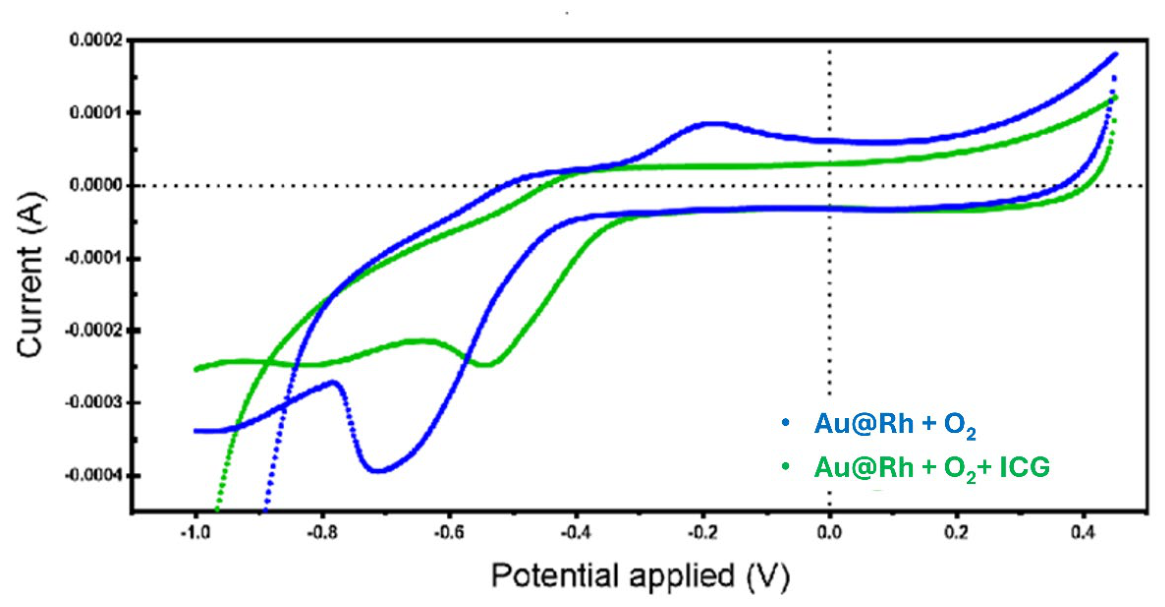
2.3. Evaluation of Electrocatalytical Activity of AuRh NPs
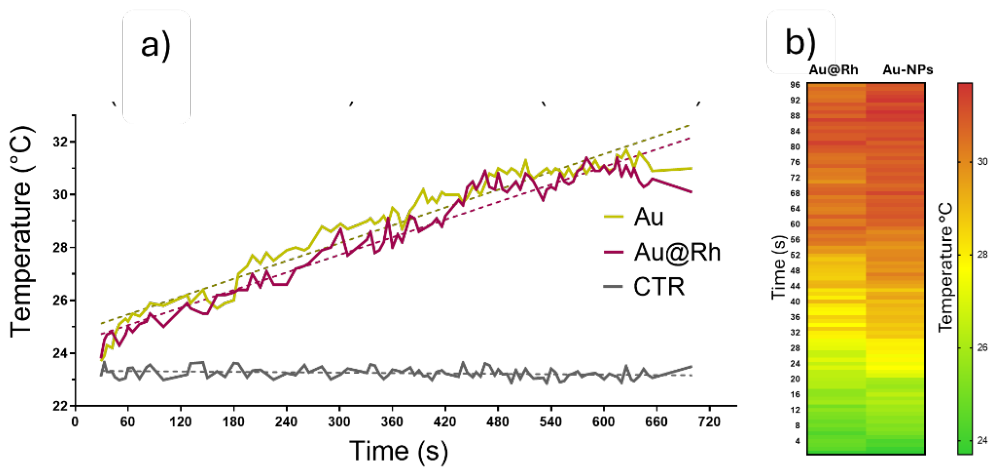
2.4. Evaluation of Photothermal Conversion Efficiency
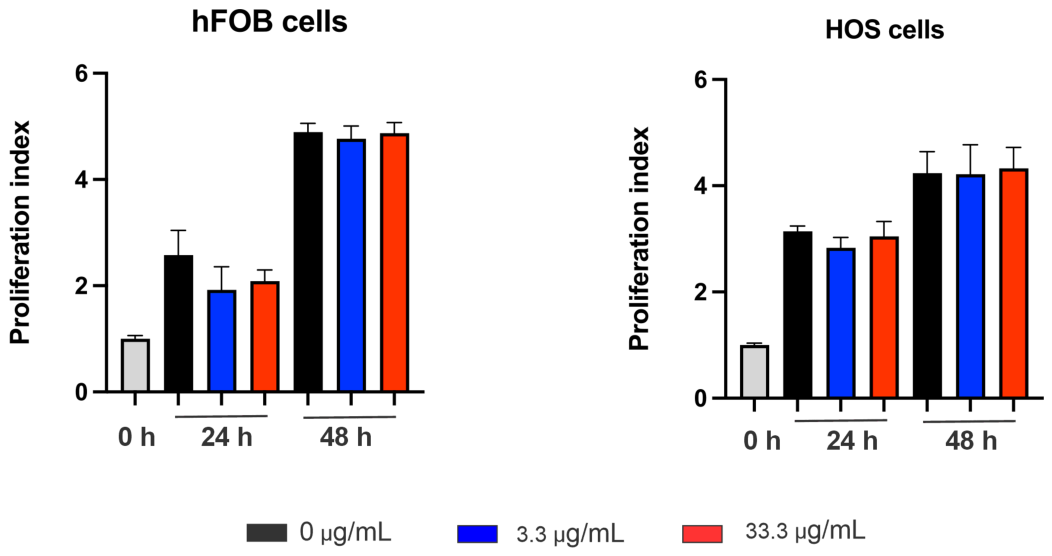
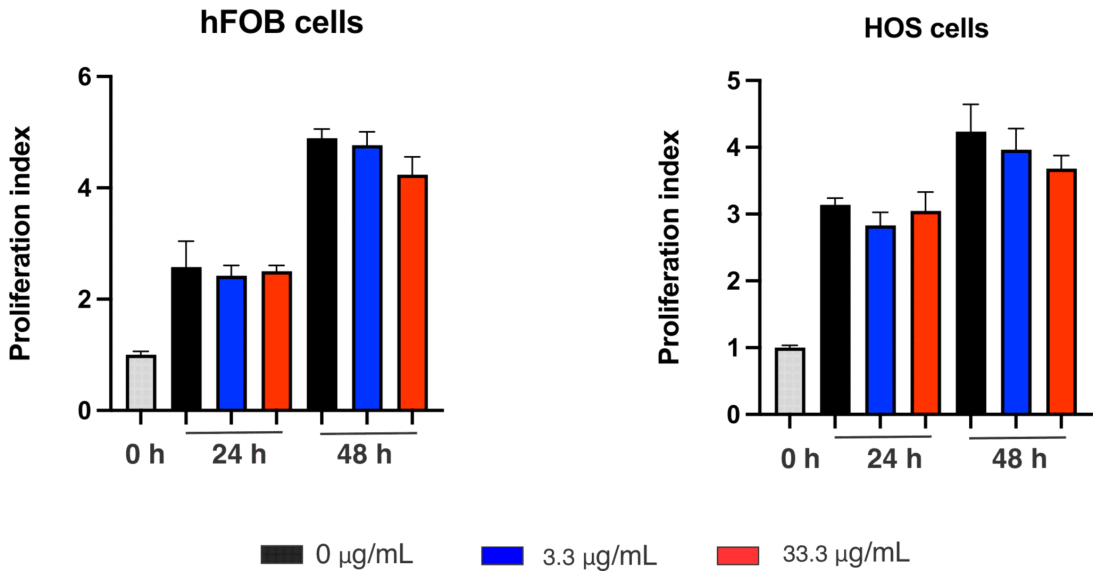
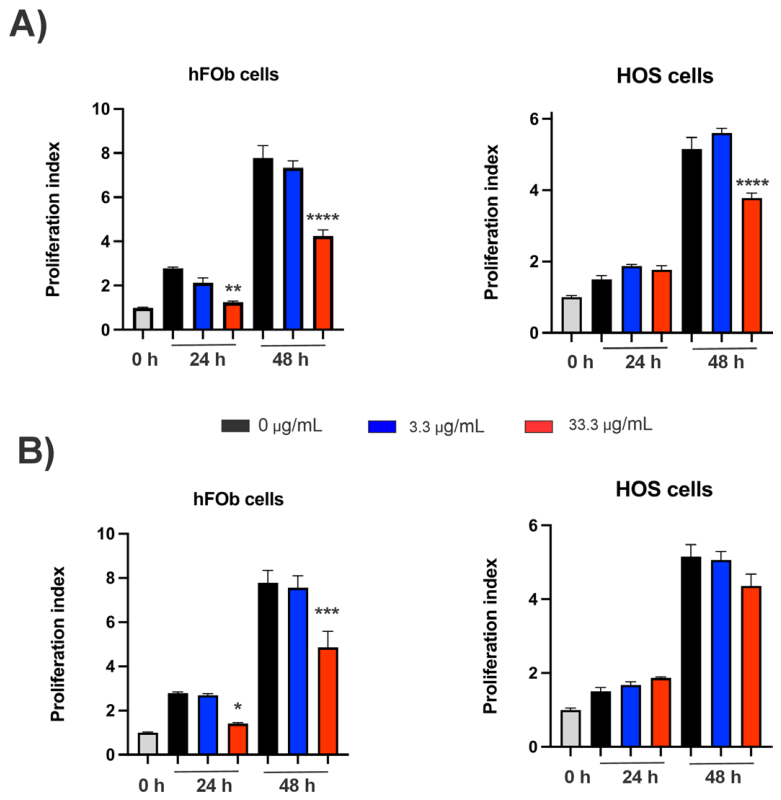
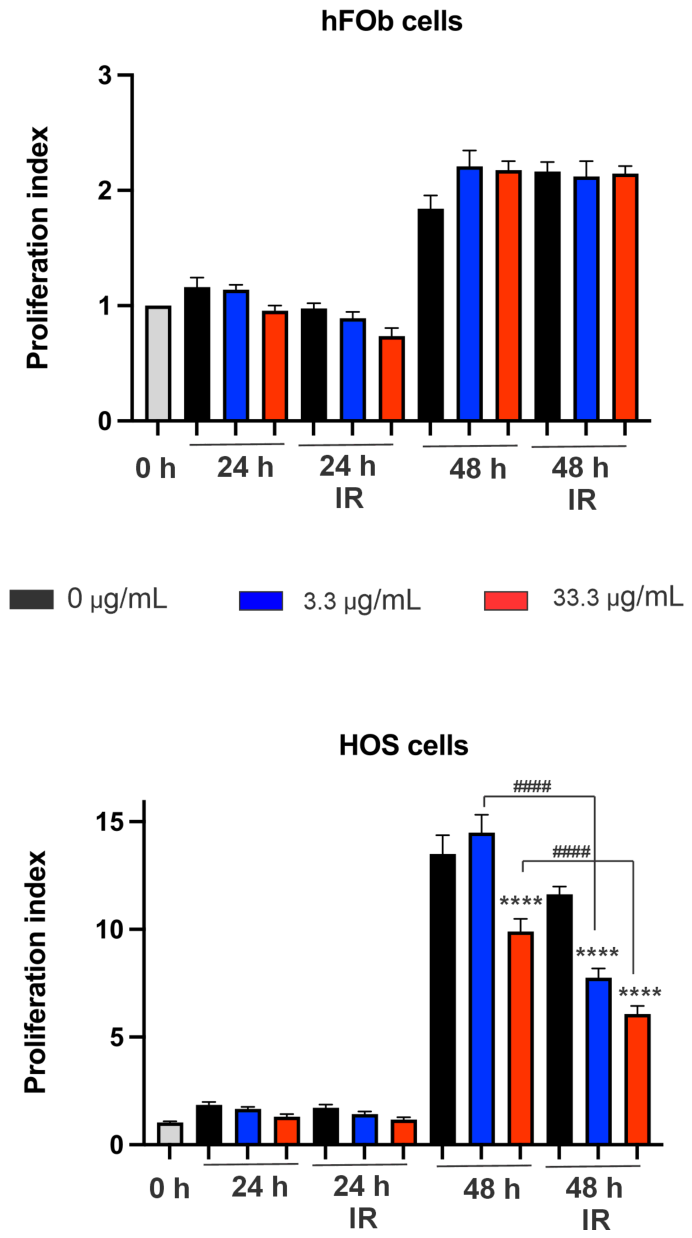
3.5. Cytotoxicity Assessment in Osteosarcoma (HOS) and Osteoblast (hFOB) Cells
3. Discussion
3.1. Electrocatalytic Properties
3.2. Cytocompatibility and Therapeutic Implications
4. Materials and Methods
4.1. Synthesis of Au@Rh Core-Shell Nanoparticles
4.2. Spectroscopic Characterization of Au@Rh Core-Shell Nanoparticles
4.3. Photothermal Conversion: Experimental Setup
4.4. Electrochemical Characterization
4.5. Formation of the Nanostructured System Au@Rh-ICG
4.7. Cytotoxicity Assessment in hFOB and HOS Cells
4.8. Photosensitiser Activation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Beird, H.C.; Bielack, S.S.; Flanagan, A.M.; Gill, J.; Heymann, D.; Janeway, K.A.; Livingston, J.A.; Roberts, R.D.; Strauss, S.J.; Gorlick, R. Osteosarcoma. Nat Rev Dis Primers 2022, 8, 77. [Google Scholar] [CrossRef]
- Nyeko, R.; Geriga, F.; Angom, R.; Kambugu, J.B.; van Heerden, J. The management of osteosarcoma in children and adolescents in a resource-limited setting: quality improvement considerations to improve treatment outcomes. BMC Cancer 2024, 24, 1061. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, Y.; Tsoi, K.; Parry, M.C.; Stevenson, J.D.; Fujiwara, T.; Sumathi, V.; Jeys, L.M. Impact of chemotherapy-induced necrosis on event-free and overall survival after preoperative MAP chemotherapy in patients with primary high-grade localized osteosarcoma. Bone Joint J. 2020, 102, 795–803. [Google Scholar] [CrossRef] [PubMed]
- Jeys, L.M.; Thorne, C.J.; Parry, M.; Gaston, C.L.; Sumathi, V.P.; Grimer, J.R. A Novel System for the Surgical Staging of Primary High-grade Osteosarcoma: The Birmingham Classification. Clin Orthop Relat Res. 2017, 475, 842–850. [Google Scholar] [CrossRef]
- Xu, J.; Xie, L.; Guo, W. Neoadjuvant Chemotherapy Followed by Delayed Surgery: Is it Necessary for All Patients With Nonmetastatic High-Grade Pelvic Osteosarcoma? Clin Orthop Relat Res. 2018, 476, 2177–2186. [Google Scholar] [CrossRef] [PubMed]
- Bajpai, J.; et al. Outcomes in non-metastatic treatment naive extremity osteosarcoma patients treated with a novel non-high dosemethotrexate-based, dose-dense combination chemotherapy regimen ‘OGS-12’. Eur J Cancer 2017, 85, 49–58. [Google Scholar] [CrossRef]
- Chen, X.; Bahrami, A.; Pappo, A.; et al. Recurrent Somatic Structural Variations Contribute to Tumorigenesis in Pediatric Osteosarcoma. Cell Reports 2014, 7, 104–112. [Google Scholar] [CrossRef]
- Jaworska, M.; et al. The Warburg effect: a score for many instruments in the concert of cancer and cancer niche cells. Pharmacol Rep. 2023, 75, 876–890. [Google Scholar] [CrossRef]
- Potter, M.; Newport, E.; Morten, K.J. The Warburg effect: 80 years on. Biochem Soc Trans. 2016, 44, 1499–1505. [Google Scholar] [CrossRef]
- Lu, J.; Tan, M.; Cai, Q. The Warburg effect in tumor progression: mitochondrial oxidative metabolism as an anti-metastasis mechanism. Cancer Lett. 2015, 356, 156–164. [Google Scholar] [CrossRef]
- Vaupel, P.; Schmidberger, H.; Mayer, A. The Warburg effect: essential part of metabolic reprogramming and central contributor to cancer progression. Int J Radiat Biol. 2019, 95, 912–919. [Google Scholar] [CrossRef]
- Kobayashi, Y.; et al. Warburg effect in Gynecologic cancers. J Obstet Gynaecol Res. 2019, 45, 542–548. [Google Scholar] [CrossRef]
- Czarnecka, A.M.; et al. Molecular Biology of Osteosarcoma. Cancers 2020, 12, 2130. [Google Scholar] [CrossRef]
- Li, G.; Wang, C.; Jin, B.; et al. Advances in smart nanotechnology-supported photodynamic therapy for cancer. Cell Death Discov. 2024, 10, 466. [Google Scholar] [CrossRef]
- Xin, Yujia. Nanodynamic therapies for osteosarcoma: Recent advances and future perspectives. Chemical Engineering Journal 2026, 527, 171582. [Google Scholar] [CrossRef]
- Yang, J. Progress of phototherapy for osteosarcoma and application prospect of blue light photobiomodulation therapy. Front. Oncol. 2022, 12, 1022973. [Google Scholar] [CrossRef] [PubMed]
- Al Jarrah, Khaled. ANovel Approach for Enhanced Osteosarcoma Photodynamic Therapy Using Encapsulated Methylene Blue in Silica Nanoparticles. J. Compos. Sci. 2023, 7, 137. [Google Scholar] [CrossRef]
- Volsi, A. L.; Scialabba, C.; Vetri, V.; Cavallaro, G.; Licciardi, M.; Giammona, G. Near-Infrared Light Responsive Folate Targeted Gold Nanorods for Combined Photothermal-Chemotherapy of Osteosarcoma. ACS Appl Mater Interfaces 2017, 9, 14453–14469. [Google Scholar] [CrossRef] [PubMed]
- Hong, Liang. Recent Advances in Strategies for Addressing Hypoxia in Tumor Photodynamic Therapy. Biomolecules 2022, 12, 81. [Google Scholar] [CrossRef] [PubMed]
- Birhan, Yihenew Simegniew. Oxygen tension regulating nanoformulation for the improved photodynamic therapy of hypoxic tumors. Materials Today Bio 2025, 31, 101587. [Google Scholar] [CrossRef]
- Khojastehnezhad, M. A.; Seyedi, S. M. R.; Raoufi, F.; Asoodeh, A. Association of hypoxia-inducible factor 1 expressions with prognosis role as a survival prognostic biomarker in the patients with osteosarcoma: a meta-analysis. Expert Rev Mol Diagn 2022, 22, 1099–1106. [Google Scholar] [CrossRef] [PubMed]
- Luo, D.; Ren, H.; Zhang, W.; Xian, H.; Lian, K.; Liu, H. Clinicopathological and prognostic value of hypoxia-inducible factor-1α in patients with bone tumor: a systematic review and meta-analysis. J Orthop Surg Res 2019, 14, 56. [Google Scholar] [CrossRef] [PubMed]
- Clever, D.; Roychoudhuri, R.; Constantinides, M. G.; et al. Oxygen Sensing by T Cells Establishes an Immunologically Tolerant Metastatic Niche. Cell 2016, 166, 1117–1131.e14. [Google Scholar] [CrossRef]
- Shirata, C.; Kaneko, J.; Inagaki, Y.; et al. Near-infrared photothermal/photodynamic therapy with indocyanine green induces apoptosis of hepatocellular carcinoma cells through oxidative stress. Sci Rep 2017, 7, 13958. [Google Scholar] [CrossRef]
- Li, W.; Guo, X.; Kong, F.; et al. Overcoming photodynamic resistance and tumor targeting dual-therapy mediated by indocyanine green conjugated gold nanospheres. J Control Release 2017, 258, 171–181. [Google Scholar] [CrossRef]
- Wang, J.; et al. A Porous Au@Rh Bimetallic Core-Shell Nanostructure as an H₂O₂-Driven Oxygenerator to Alleviate Tumor Hypoxia for Simultaneous Bimodal Imaging and Enhanced Photodynamic Therapy. Adv Mater 2020, 32, e2001862. [Google Scholar] [CrossRef]
- Kang, Y.; Xue, Q.; Peng, R.; Jin, P.; Zeng, J.; Jiang, J.; Chen, Y. Bimetallic AuRh nanodendrites consisting of Au icosahedron cores and atomically ultrathin Rh nanoplate shells: synthesis and light-enhanced catalytic activity. NPG Asia Mater 2017, 9, e407. [Google Scholar] [CrossRef]
- Machuca, A.; et al. Rhodium Nanoparticles as a Novel Photosensitizing Agent in Photodynamic Therapy against Cancer. Chemistry 2020, 26, 7685–7691. [Google Scholar] [CrossRef]
- Kang, S.; et al. Morphology-Controlled Synthesis of Rhodium Nanoparticles for Cancer Phototherapy. ACS Nano 2018, 12, 6997–7008. [Google Scholar] [CrossRef]
- Khan, Z.; Singh, T.; Hussain, J. I.; Hashmi, A. A. Au(III)–CTAB reduction by ascorbic acid: Preparation and characterization of gold nanoparticles. Colloids Surf B Biointerfaces 2012, 104, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Liao, C.; Guo, X.; et al. RhRu Alloy-Anchored MXene Nanozyme for Synergistic Osteosarcoma Therapy. Small 2023, 19, e2205511. [Google Scholar] [CrossRef]
- García Calavia, P.; et al. Photosensitiser-gold nanoparticle conjugates for photodynamic therapy of cancer. Photochem Photobiol Sci 2018, 17, 1534–1552. [Google Scholar] [CrossRef]
- Fan, D.; et al. Nanomedicine in cancer therapy. Sig Transduct Target Ther 2023, 8, 293. [Google Scholar] [CrossRef]
- Gomerdinger, V.F.; Nabar, N.; Hammond, P.T. Advancing engineering design strategies for targeted cancer nanomedicine. Nat Rev Cancer 2025, 25, 657––683. [Google Scholar] [CrossRef]
- Compton, R.G.; Banks, C.E. Understanding Voltammetry, 3rd ed.; World Scientific: New Jersey, 2018; p. 312. [Google Scholar]
- PSuchomel; Stulajterova, K.P.; Sedenkova, E. B. Impact of gold nanoparticles shape on their cytotoxicity against human osteoblast and osteosarcoma in in vitro model. Evaluation of the safety of use and anti-cancer potential. Journal of Materials Science: Materials in Medicine 2019, 30, 22. [Google Scholar] [CrossRef]
- Solarte, D.L.G.; Rau, S.J.; Hellwig, E.; Vach, K.; Al-Ahmad, A. Antimicrobial Behavior and Cytotoxicity of Indocyanine Green in Combination with Visible Light and Water-Filtered Infrared A Radiation against Periodontal Bacteria and Subgingival Biofilm. Biomedicines 2022, 10, 956. [Google Scholar] [CrossRef]
- Aghayan, S.; et al. Evaluation of Indocyanine-Mediated Photodynamic Therapy Cytotoxicity in Human Osteoblast-Like Cells: an In Vitro Study. Folia Medica 2022, 64, 932–938. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, L.; Zhao, G.; et al. Homologous targeting nanoparticles for enhanced PDT against osteosarcoma HOS cells and the related molecular mechanisms. J Nanobiotechnol 2022, 20, 83. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



