Submitted:
15 April 2026
Posted:
17 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
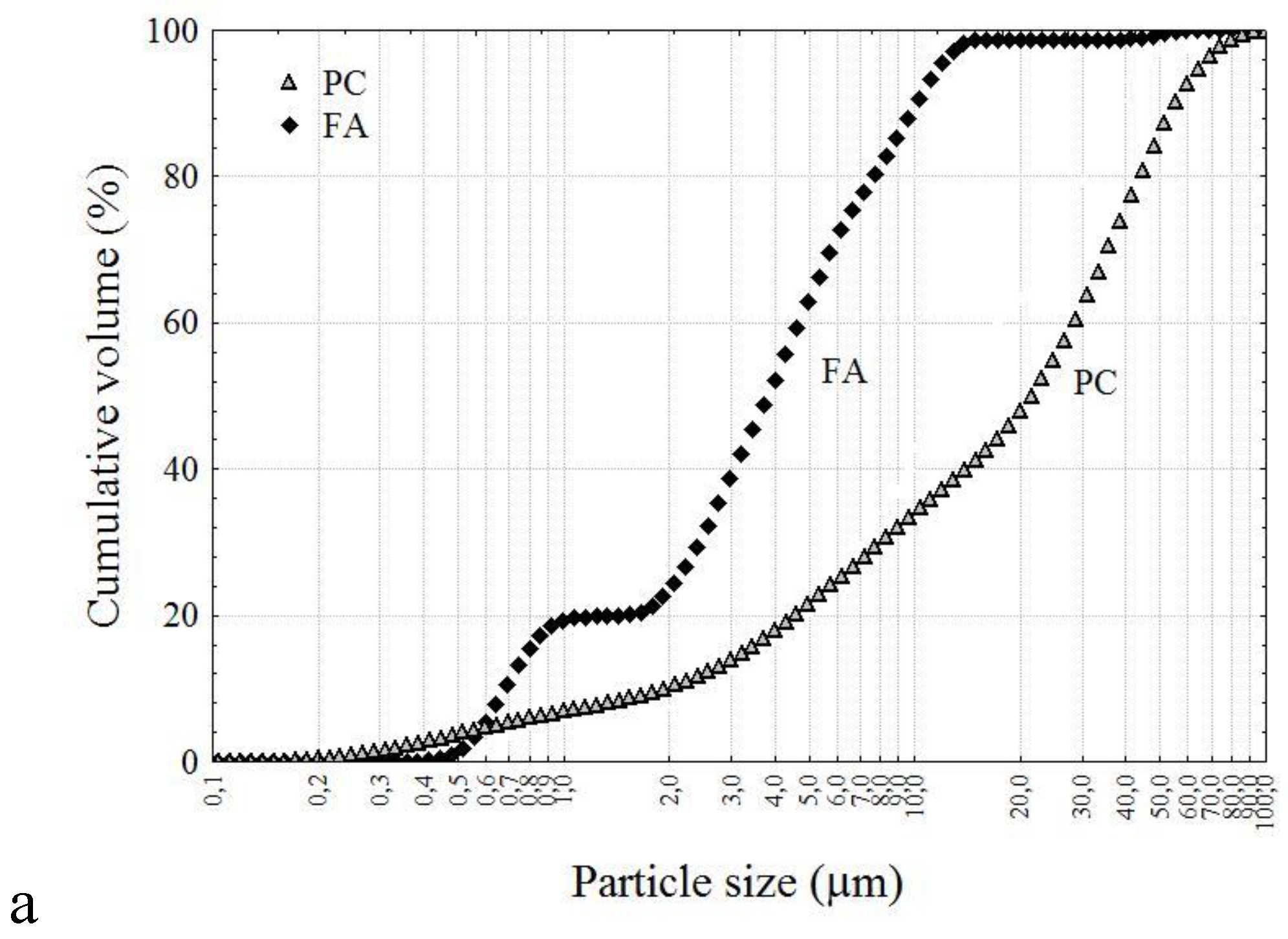
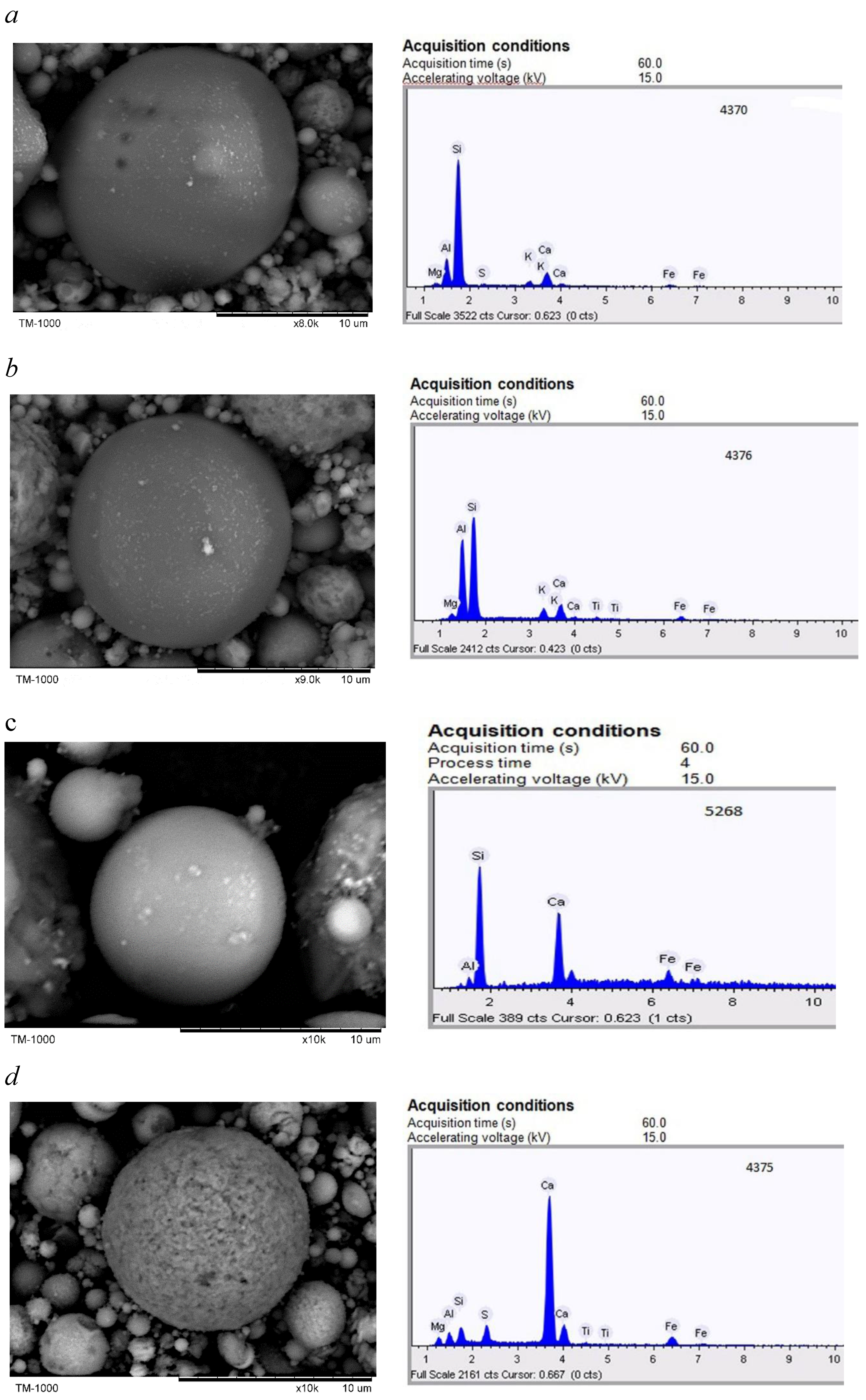
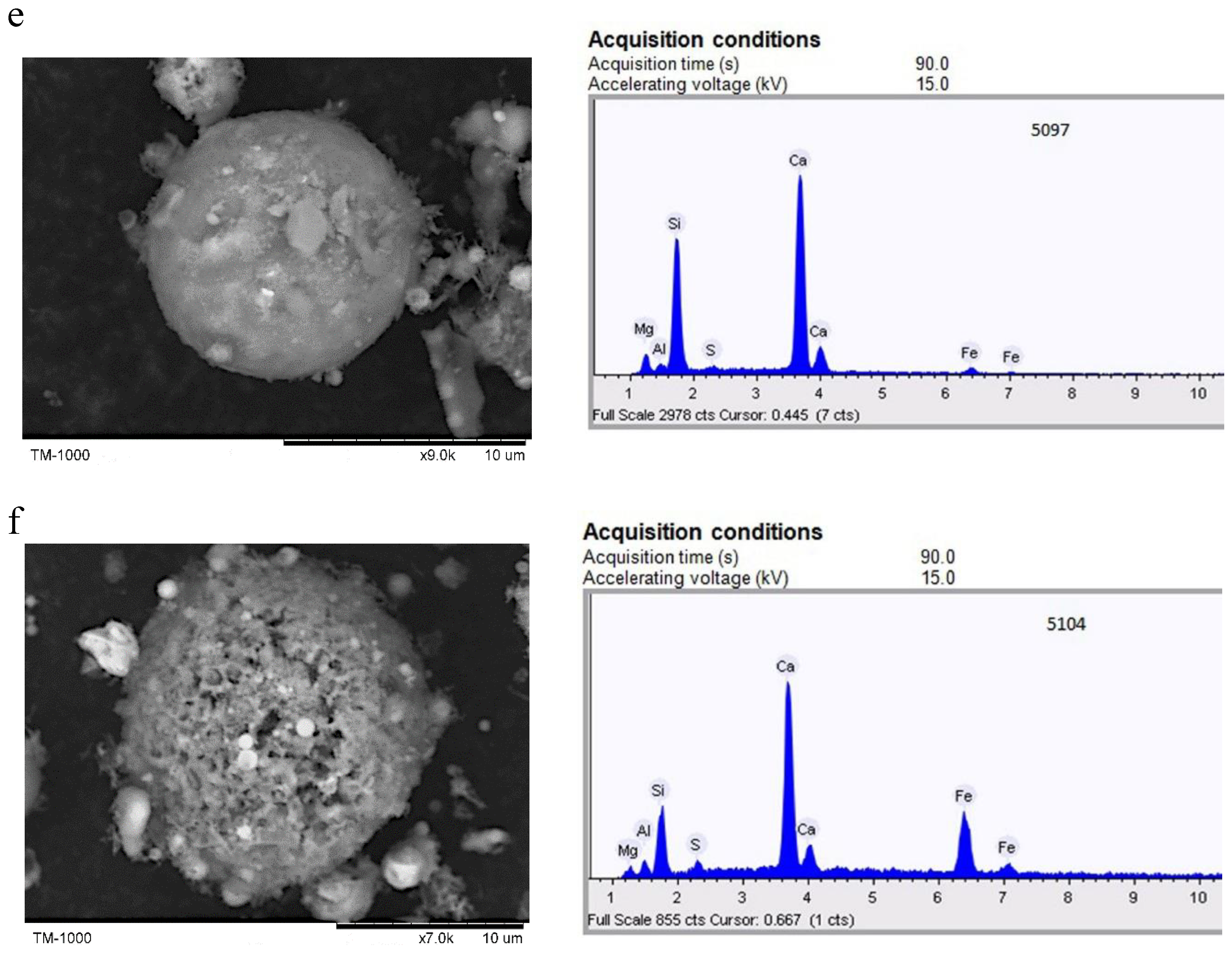
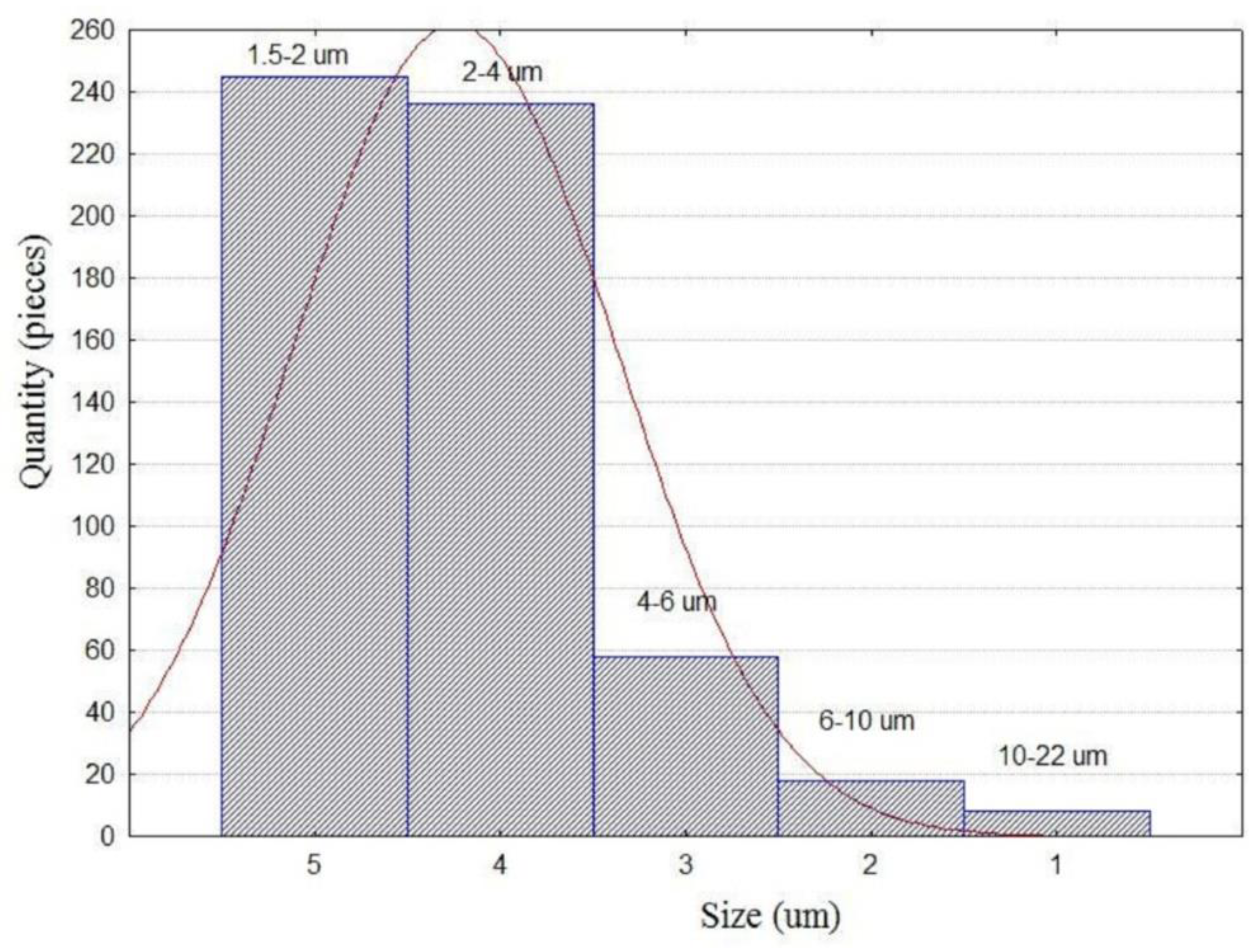
2.1. Individual Microspheres of the Original HCFA
2.2. Specimens of Binders at Different Curing Times
3. Conclusions
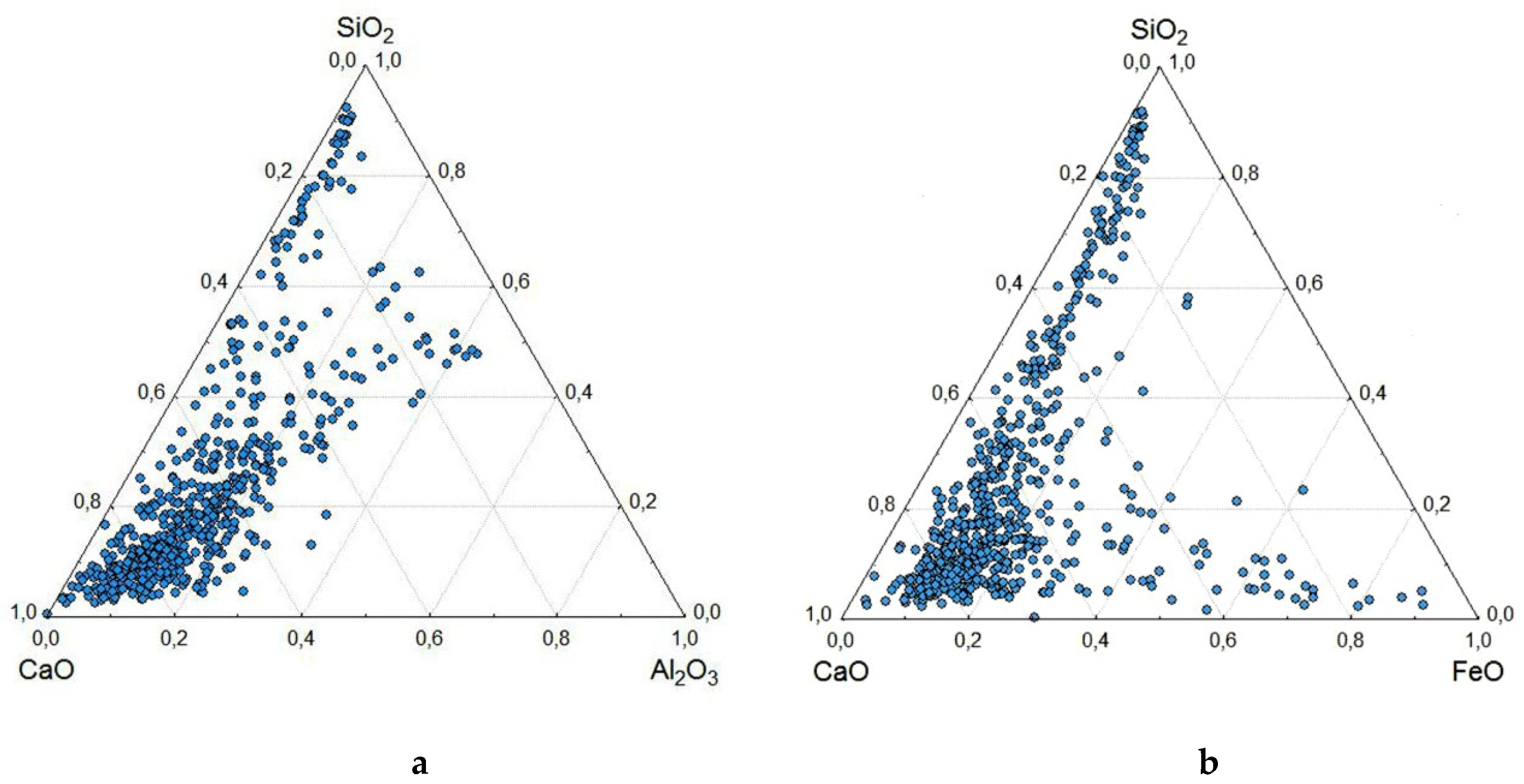
- SEM-EDS was used to study 568 individual microspheres in the original fine high-calcium fly ash with a particle size of d90–10 μm. Analysis of the composition of individual globules using ternary diagrams of CaO-Al2O3-SiO2 and CaO-FeO-SiO2 reveals a wide range of compositions. However, most microsphere compositions are in the array with a content of CaO 55–90%, SiO2 5–30%, Al2O3 0–20% and FeO 2–25%. When the CaO content is less than 50%, there are three trends in the composition of microspheres: (1) calcium silicates, (2) calcium aluminosilicates, and (3) calcium-iron-silicates and calcium ferrites.
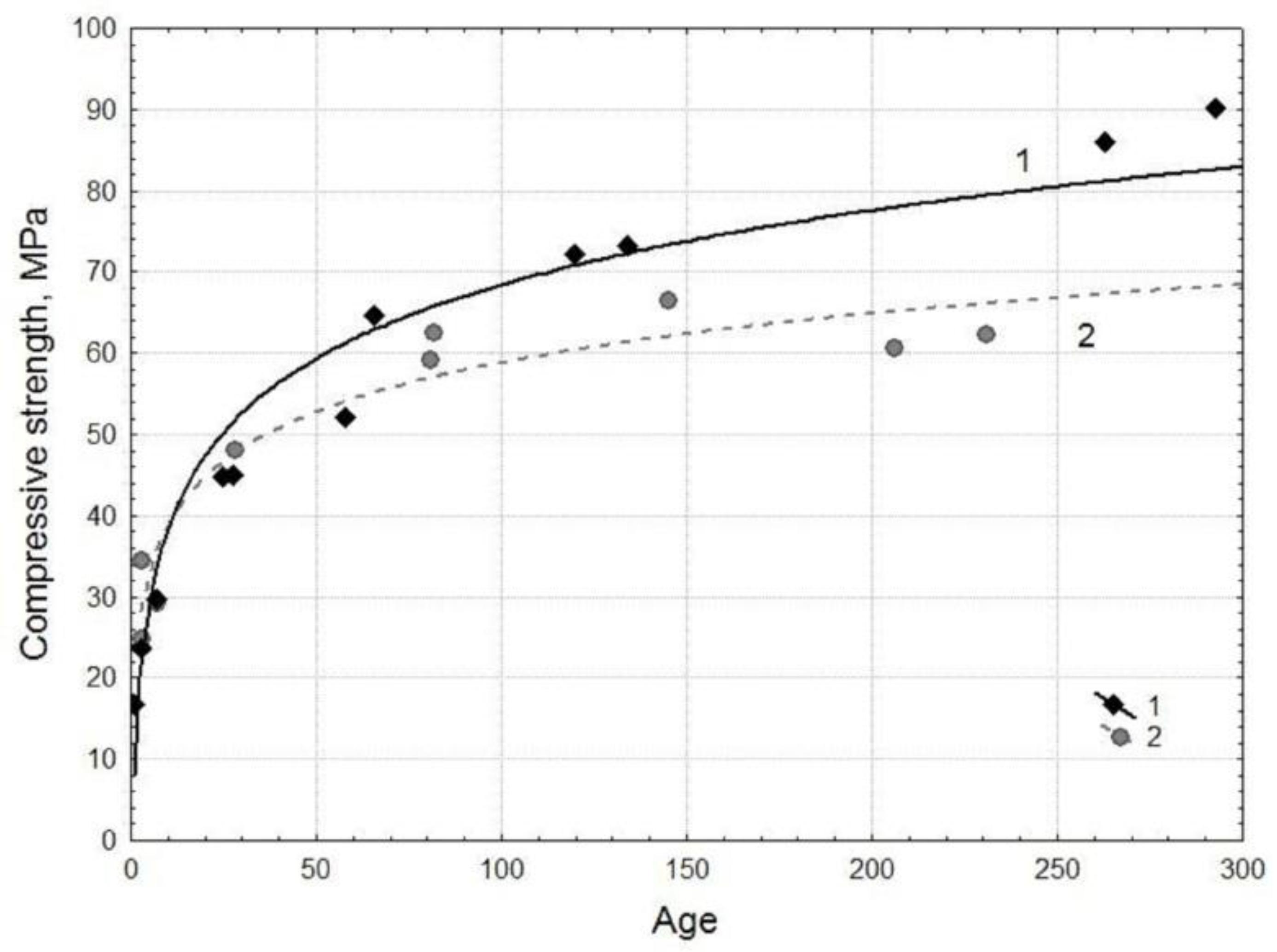
- Based on fine HCFA, the specimens were obtained (at W/B=0.25; superplasticizer 0.12% Melflux 5581F) with high compressive strength, which increases from 24 to 90 MPa during curing from 3 to 300 days. Comparison with specimens based on CEM I 42.5N cement (at W/C=0.4) showed that during curing up to 28 days their strength is practically the same and amounts to about 50 MPa. With further curing up to 300 days, the strength of the cement samples (60-66 MPa) is inferior to the specimens based on HCFA.
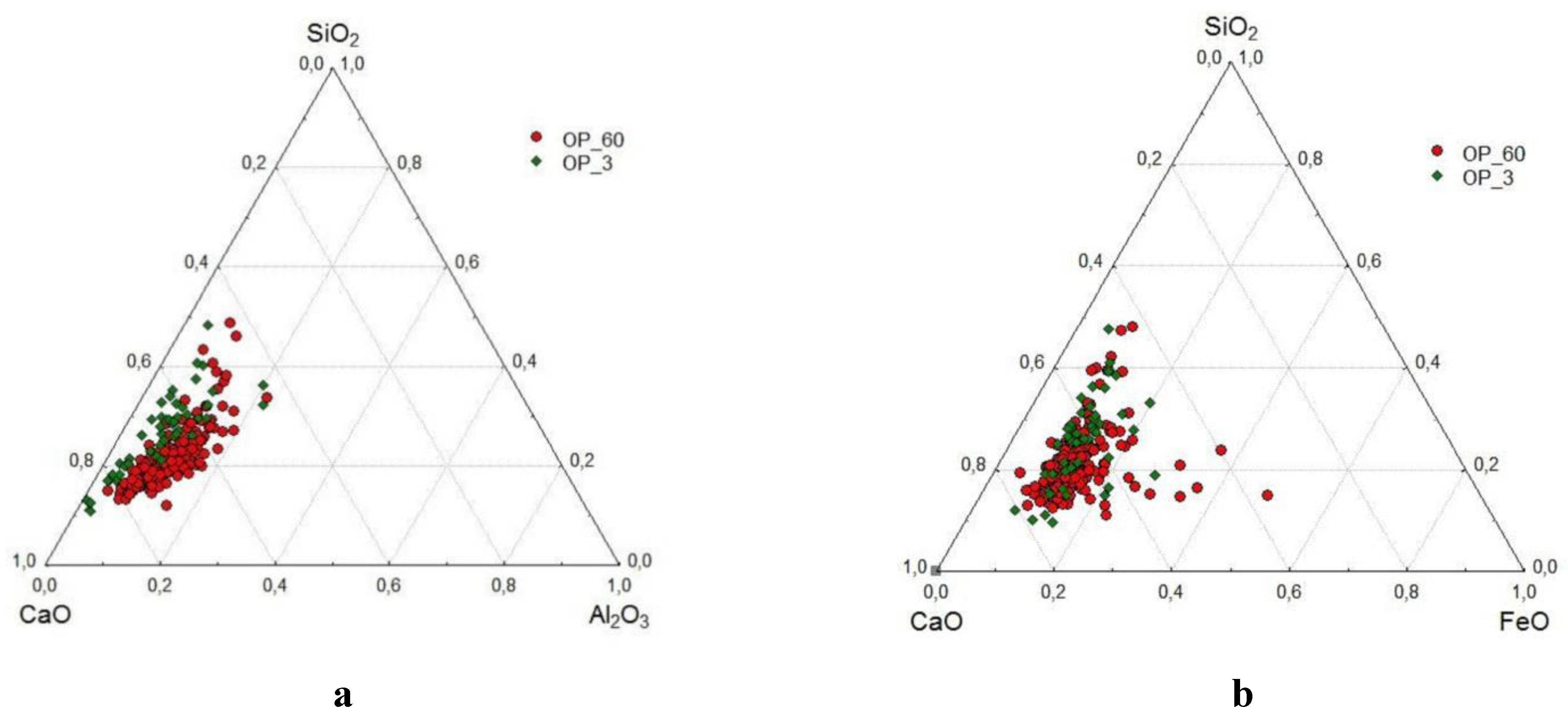
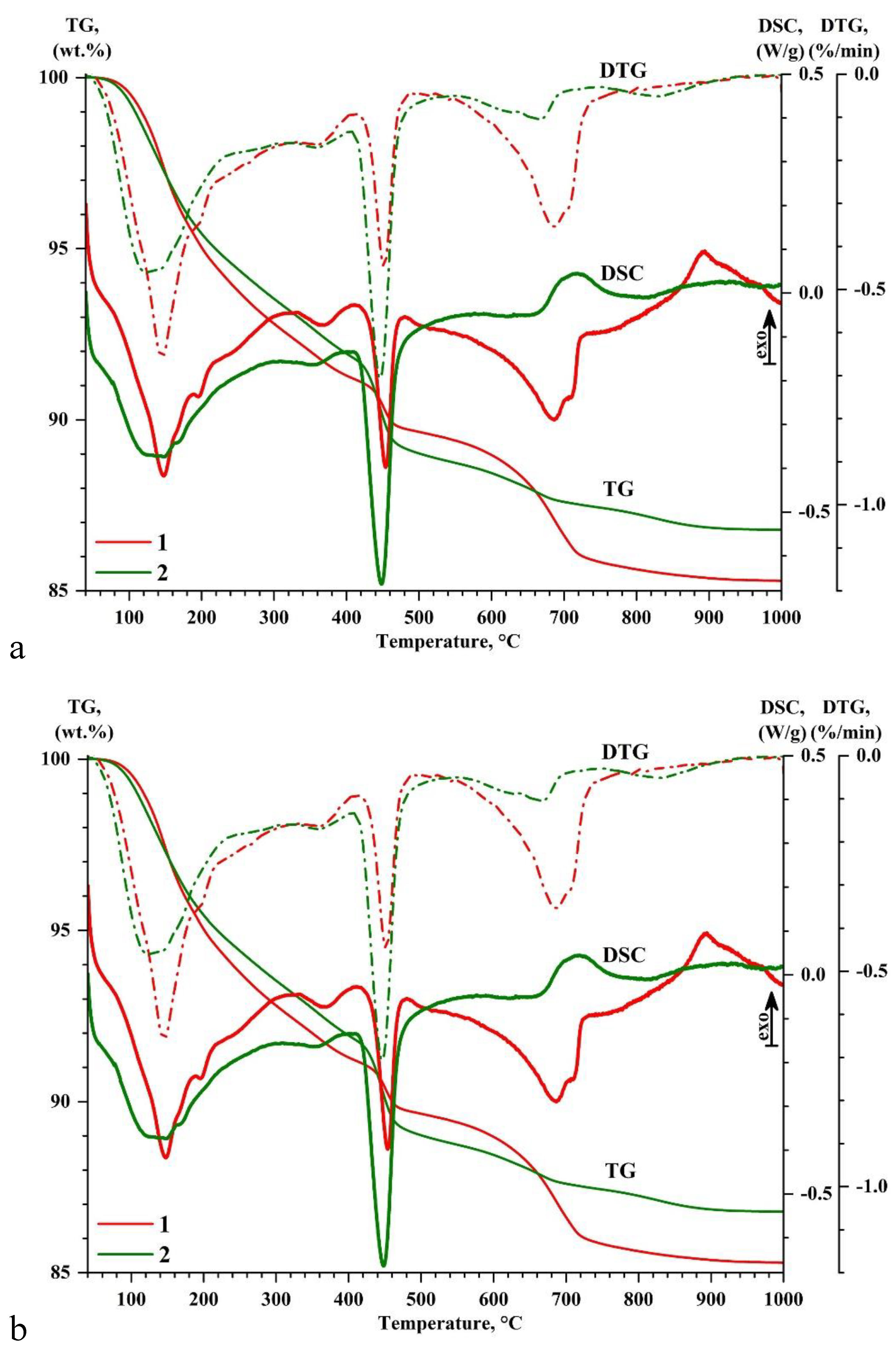
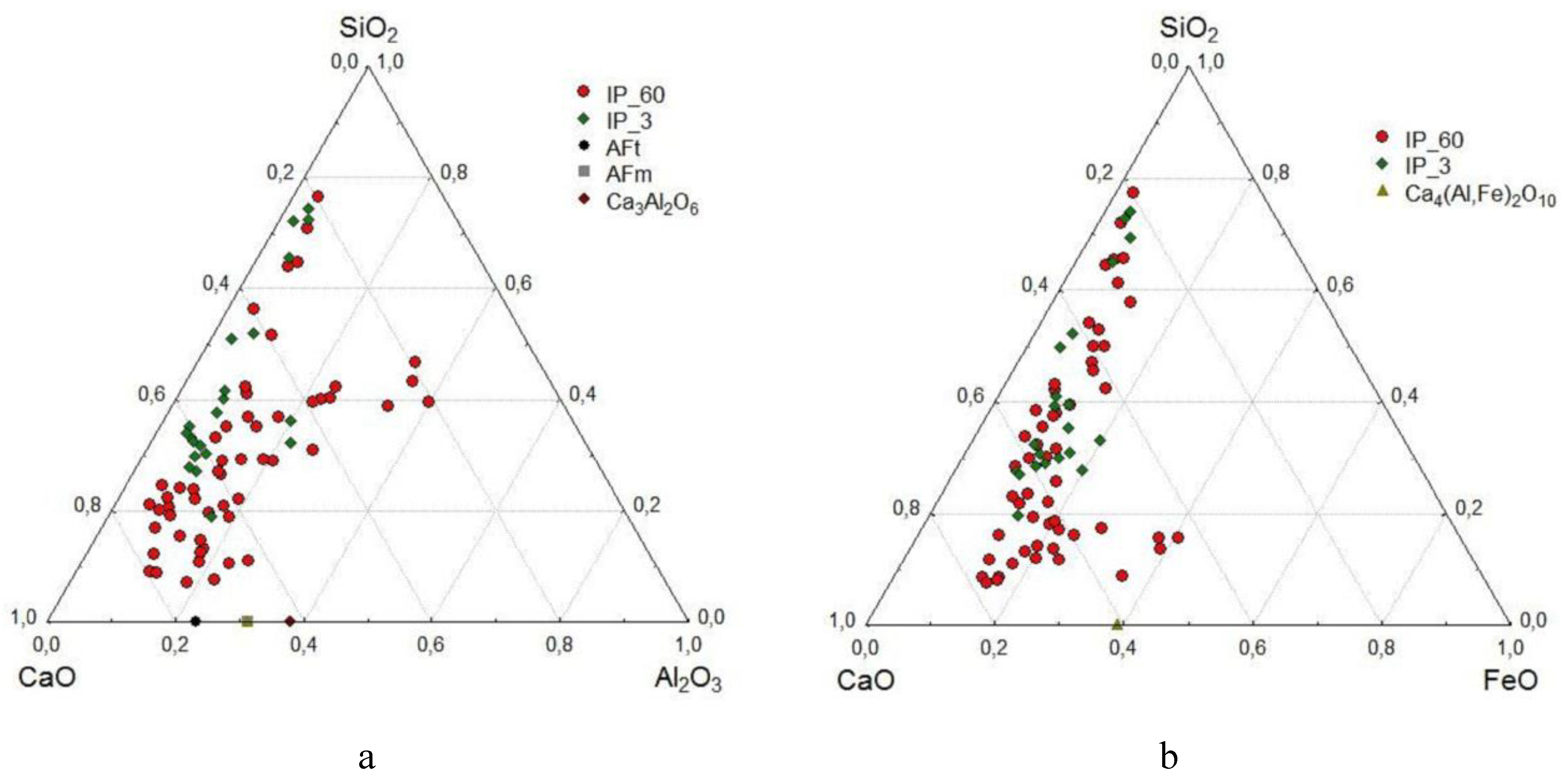
- Using the SEM-EDS method, the compositions of 410 areas of newly formed hydrate products in a space initially filled with water, which forms the strength matrix, were studied. It was found that the overwhelming majority of the compositions are concentrated in the high-calcium region of the CaO–Al2O3–SiO2 diagram, with a total CaO content of 60-80%. The SiO2 content in them is 15-30%, and they can contain from 1 to 15% Al2O3 and 5-14% FeO. In terms of composition and CaO/SiO2 ratio (from 1.7 to 4.2), they are close to the composition of the areas of hardened cement in the corresponding composition diagram. The SEM-EDS method confirmed a more active role of calcium aluminoferrite in the formation of hydrate products compared to calcium aluminate.
4. Materials and Methods
4.1. Starting Materials and Binder Specimens.
4.2. Methods
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| HCFA SEM-EDS | High-calcium fly ash Scanning electron microscopy and Energy-dispersive X-ray spectroscopy |
| XRF | X-ray fluorescence analysis |
| XRD | X-ray diffraction |
| OP | Outer products |
| IP | Inner products |
| SPMs | Supplementary cementitious materials |
| WDS | Wavelength-dispersive electron microscopy |
| ASEM-EDS | Automated scanning electron microscopy with Energy dispersive X-ray spectrometry |
| STA | Synchronous thermal analysis |
| TG | Thermogravimetry |
| DTG | Derivative Thermogravimetry |
| DSC | Differential Scanning Calorimetry |
| W/B | Water/Binder |
| W/C | Water/Cement |
References
- Gollakota, A.R.K.; Volli, V.; ShuCh-M. Progressive utilisation prospects of coal fly ash: A review. Sci Total Environ. 2019, 672, 951–989. [Google Scholar] [CrossRef]
- Singh, N.; Bhardwaj, S.A. Reviewing the role of coal bottom ash as an alternative of cement. Constr. Build. Mater. 2020, 233, 117276. [Google Scholar] [CrossRef]
- Feduik, R.S.; Smoliakov, A. K.; Timokhin, R. A.; Batarshin, V. O.; Yevdokimova, Yu. G. Using thermal power plants waste for building materials. IOP Publishing IOP Conf. Series: Earth and Environmental Science 2017, 87, 092010. [Google Scholar] [CrossRef]
- Yu, J.; Lu, C.; Leung, C.K.Y.; Li, G. Mechanical properties of green structural concrete with ultrahigh-volume fly ash. Constr. Build. Mater. 2017, 147, 510–518. [Google Scholar] [CrossRef]
- Teixeira, E.R.; Mateus, R.; Camoes, A.F.; Bragança, L.; Branco, F.G. Comparative environmental life-cycle analysis of concretes using biomass and coal fly ashes as partial cement replacement material. J. Clean. Prod. 2016, 112, 2221–2230. [Google Scholar] [CrossRef]
- ASTM С430-25; Standard Test Method for Fineness of Hydraulic Cement by the 45-μm (No. 325) Sieve. 2025; 3 p. Available online: https://clck.su/ogqcg.
- EN 197-1. Cement - Part 1: Composition, specifications and conformity criteria for common cements. 2000. 29 p. Available online: http://www.rucem.ru/yabbfiles/Attachments/EN-197-1.pdf?ysclid=mnr4zv8n5b285610497.
- GOST 31108-2020. Common cements. Specifications. Moscow, Standartinform. 2020. 20 p. Available online: https://www.status-grunt.ru/upload/bases/24.pdf?yclid=11130730052221140991.
- Durdziński, P.T.; Dunant, C.F.; Haha, M.B.; Scrivener, K.L. A new quantification method based on SEM-EDS to assess fly ash composition and study the reaction of its individual components in hydrating cement paste. Cem. Concr. Res. 2015, 73, 111–122. [Google Scholar] [CrossRef]
- Wu, R.D.; Dai, S.B.; Jian, S.W.; Huang, J.; Yang, L.; Li, B.D.; Nurmirzayev, A. Utilization of the circulating fluidized bed combustion ash in autoclaved aerated concrete: Effect of superplasticizer. Constr. Build. Mater. 2020, 237, 117644. [Google Scholar] [CrossRef]
- Zhang, N.; Yu, H.; Gong, W.; Liu, T.; Wang, N.; Tan, Yo.; Wu, C. Effects of low- and high-calcium fly ash on the water resistance of magnesium oxysulfate cement. Constr. Build. Mater. 2020, 230, 116951. [Google Scholar] [CrossRef]
- Deschner, F.; Winnefeld, F.; Lothenbach, B.; Seufert, S.; Schwesig, P.; Dittrich, S.; Goetz-Neunhoeffer, F.; Neubauer, Ju. Hydration of Portland cement with high replacement by siliceous fly ash. Cem. Concr. Res. 2012, 42, 1389–1400. [Google Scholar] [CrossRef]
- Sharonova, O.M.; Solovyov, L.A.; Oreshrina, N.A.; Yumashev, V.V.; Anshits, A.G. Composition of high-calcium fly ash middlings selectively sampled from ash collection facility and prospect of their utilization as component of cementing materials. Fuel Process. Technol. 2010, 91, 573–581. [Google Scholar] [CrossRef]
- Ilic, M.; Cheeseman, C.; Sollars, C.; Knight, J. Mineralogy and mi-crostructure of sintered lignite coal fly ash. Fuel 2003, 82, 331–336. [Google Scholar] [CrossRef]
- Tishmack, J.K.; Olek, J.; Diamond, S.; Sahu, S. Characterization of pore solutions expressed from high-calcium fly-ash-water pastes. Fuel 2001, 80, 815–819. [Google Scholar] [CrossRef]
- Escalante-Garcia, J.-I.; Sharp, J.H. The chemical composition and microstructure of hydration products in blended cements. Cem. Concr. Compos. 2004, 26, 967–976. [Google Scholar] [CrossRef]
- Supit, S.V.M.; Shaikh, F.U.A.; Sarker, P.K. Effect of ultrafine fly ash on mechanical properties of high volume fly ash mortar. Constr. Build. Mater. 2014, 51, 278–286. [Google Scholar] [CrossRef]
- Lothenbach, B.; Scrivener, K.; Hooton, R.D. Supplementary cementitious materials. Cem. Concr. Res. 2011, 41, 1244–1256. [Google Scholar] [CrossRef]
- Aughenbaugh, K.L.; Chancey, R.T.; Stutzman, P.; Juenger, M.C.; Fowler, D.W. An examination of the reactivity of fly ash in cementitious pore solutions. Mater. Struct. 2013, 46, 869–880. [Google Scholar] [CrossRef]
- Pratiwi, W.D.; Triwulan; Ekaputri, J.J.; Fansuri, H. Combination of precipitated-calcium carbonate substitution and dilute-alkali fly ash treatment in a very high-volume fly ash cement paste. Constr.Build. Mater. 2020, 234, 117273. [Google Scholar] [CrossRef]
- Johnson, A.; Catalan, L.J.J.; Kinrade, S.D. Characterization and evaluation of fly-ash from co-combustion of lignite and wood pellets for use as cement admixture. Fuel 2010, 89, 3042–3050. [Google Scholar] [CrossRef]
- Kutchko, B.G.; Kim, A.G. Fly ash characterization by SEM–EDS. Fuel 2006, 85, 2537–2544. [Google Scholar] [CrossRef]
- Kim, T.; Davis, J.M.; Ley, M.T.; Kang, S.; Amrollahi, P. Fly ash particle characterization for predicting concrete compressive strength. Constr. Build. Mater. 2018, 165, 560–571. [Google Scholar] [CrossRef]
- Kang, S.; Lloyd, Z.; Kim, T.; Ley, M.T. Predicting the compressive strength of fly ash concrete with the Particle Model. Cem. Concr. Res. 2020, 137, 106218. [Google Scholar] [CrossRef]
- Sharonova, O.M.; Yumashev, V.V.; Solovyov, L.A.; Anshits, A.G. The fine high-calcium fly ash as the basis of composite cementing material. Mag. Civ. Eng. 2019, 60–70. [Google Scholar] [CrossRef]
- GOST 5382-2019; Cements and materials for cement production. Chemical analysis methods. Standartinform: Moscow, 2019; 66 p. Available online: https://rosgosts.ru/file/gost/77/150/gost_5382-2019.pdf?ysclid=mnr4ur7shu915779500.
- Sharonova, O.M.; Solovyov, L.A.; Anshits, A.G. The influencing factors for the strength enhancement of composite materials made up of fine high-calcium fly ash. Adv. Concrete Constr. 2023, 16, 169–176. [Google Scholar] [CrossRef]
- Han, H. X.; Yang, J.; Feng, J.; Zhou, C.; Wang, X. Research on hydration mechanism of ultrafine fly ash and cement composite. Constr. Build. Mater. 2019, 227, 116697. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



