Submitted:
15 April 2026
Posted:
16 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
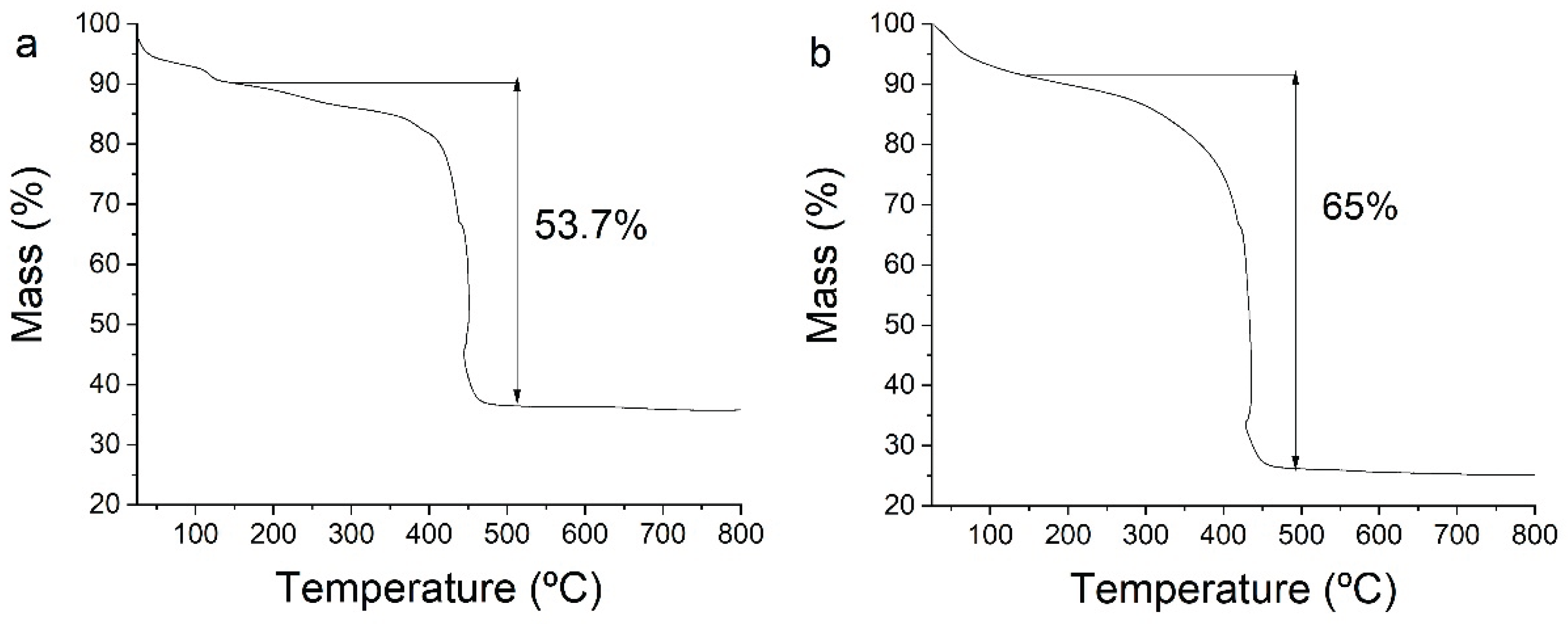
2.1. Synthesis and Characterization of NU-1000
2.2. Mitoxantrone Loading into NU-1000
2.3. PEGylation and Stabilization Under Physiological Conditions
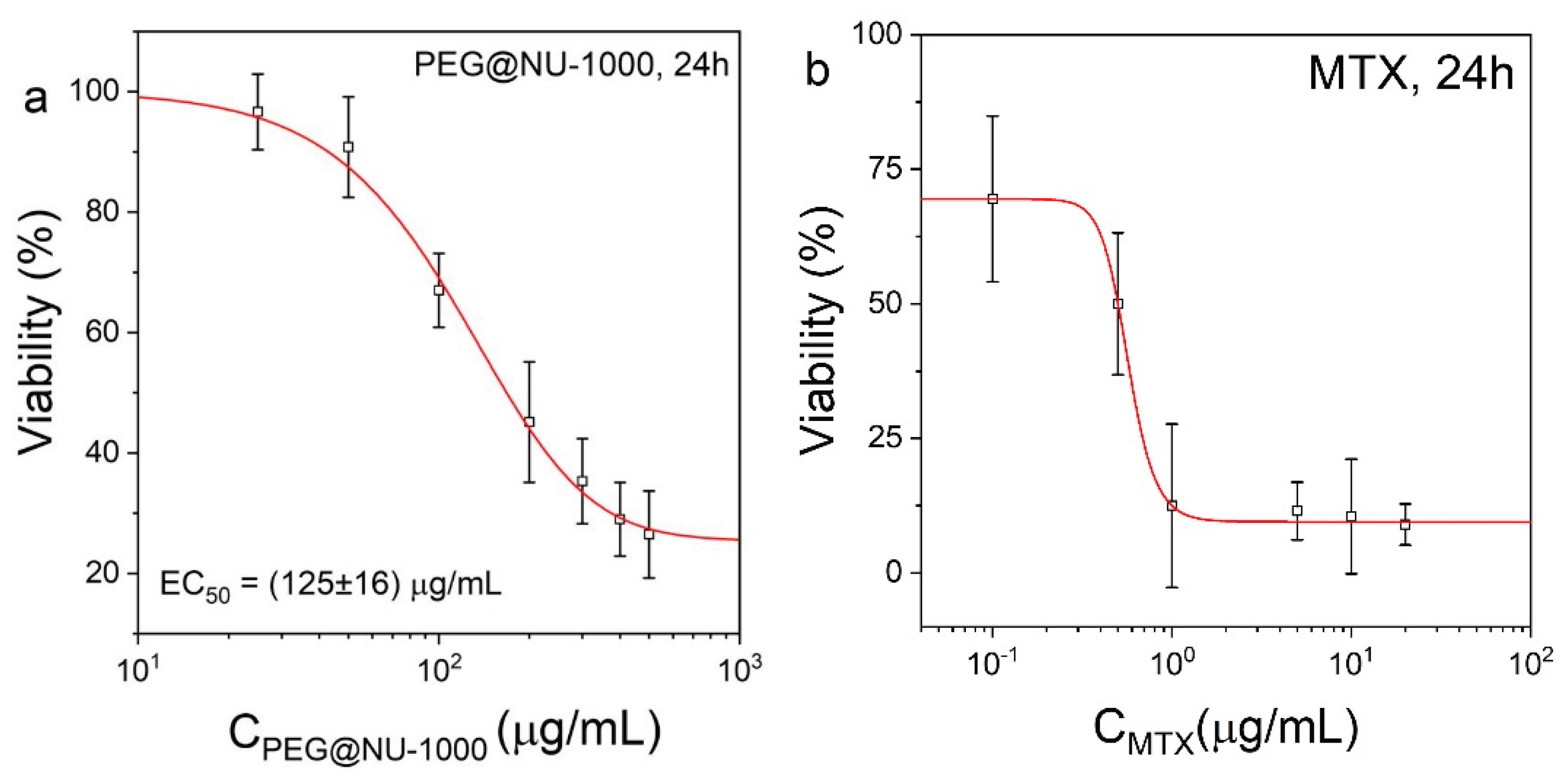
2.4. Cytotoxicity of PEG@NU-1000 and Free Mitoxantrone
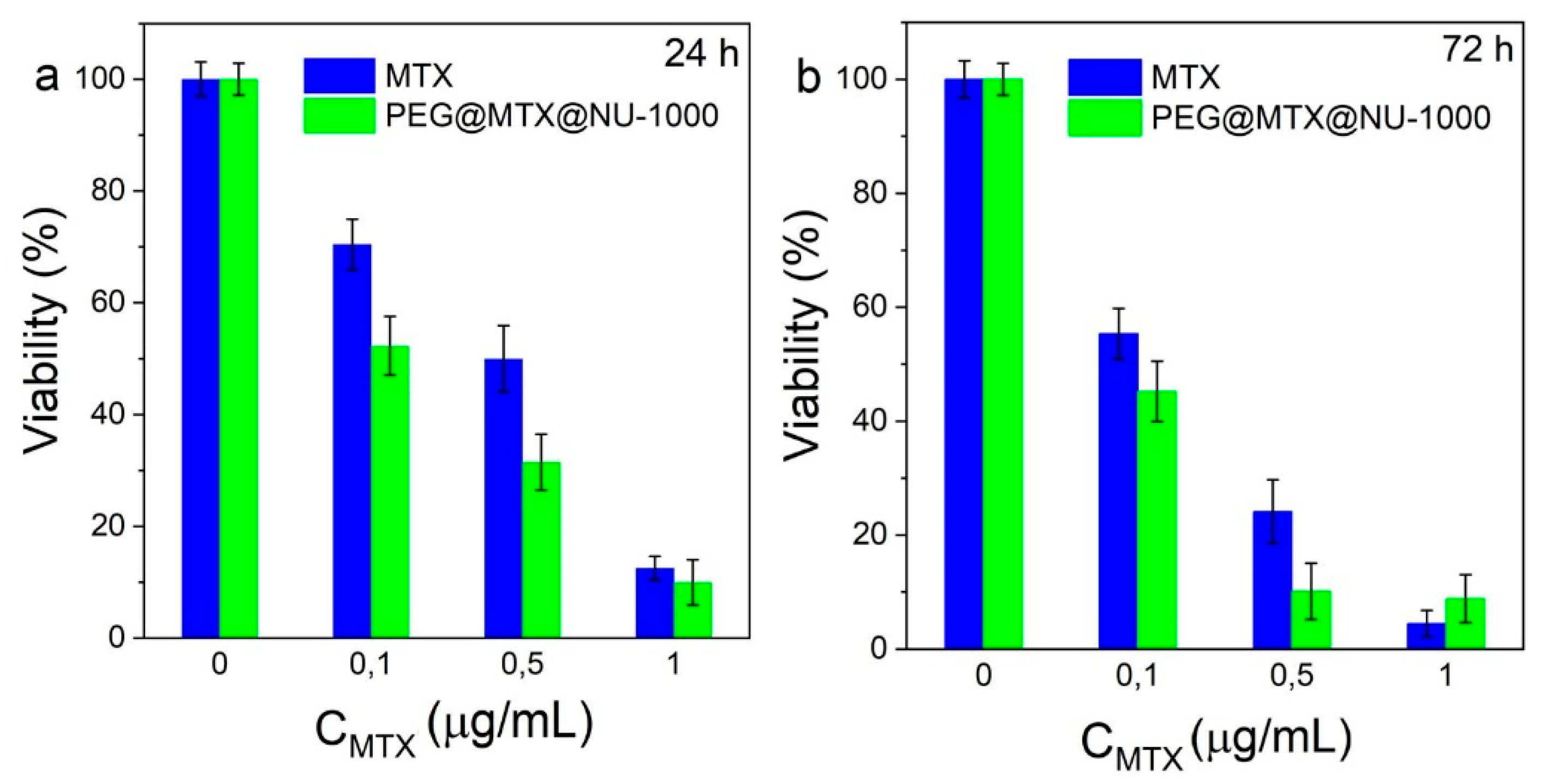
2.5. Antiproliferative Activity of PEG@MTX@NU-1000
3. Materials and Methods
3.1. Materials and Characterization Methods
3.2. NU-1000 Synthesis
3.3. MTX@NU-1000 Inclusion Assays
3.4. PEG@MTX@NU-1000 Particles Preparation
3.5. Cytotoxicity Assays
- PEG@NU-1000: 0, 25, 50, 100, 200, 300, 400, 500, and 600 µg/mL.
- MTX: 0, 0.1, 0.5, 1, 5, 10, and 20 µg/mL.
- PEG@MTX@NU-1000: concentrations equivalent to 0, 0.1, 0.5, 1, 5, 10, and 20 µg/mL of MTX.
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| MOFs | Metal–organic frameworks |
| MTX | Mitoxantrone |
| PEG | Polyethylene glycol |
| PXRD | Powder X-ray diffraction |
| SEM | Scanning electron microscopy |
| DLS | Dynamic light scattering |
| UV–Vis | Ultraviolet–visible spectroscopy |
| TGA | Thermogravimetric analysis |
| PBS | Phosphate-buffered saline |
| PDI | Polydispersity index |
| LC | Loading capacity |
References
- World Health Organization. Cancer. Available online: https://www.who.int/news-room/fact-sheets/detail/cancer (accessed on 10 April 2026).
- Chabner, B.A.; Roberts, T.G. Chemotherapy and the war on cancer. Nat. Rev. Cancer 2005, 5, 65–72. [CrossRef]
- Vasan, N.; Baselga, J.; Hyman, D.M. A view on drug resistance in cancer. Nature 2019, 575, 299–309. [CrossRef]
- Park, K. Controlled drug delivery systems: Past forward and future back. J. Control. Release 2014, 190, 3–8. [CrossRef]
- Peer, D.; Karp, J.M.; Hong, S.; Farokhzad, O.C.; Margalit, R.; Langer, R. Nanocarriers as an emerging platform for cancer therapy. Nat. Nanotechnol. 2007, 2, 751–760. [CrossRef]
- Min, Y.; Caster, J.M.; Eblan, M.J.; Wang, A.Z. Clinical translation of nanomedicine. Chem. Rev. 2015, 115, 11147–11190. [CrossRef]
- Farokhzad, O.C.; Langer, R. Impact of nanotechnology on drug delivery. ACS Nano 2009, 3, 16–20. [CrossRef]
- Zhao, Z.; Ukidve, A.; Kim, J.; Mitragotri, S. Targeting strategies for tissue-specific drug delivery. Cell 2020, 181, 151–167. [CrossRef]
- Akerman, M.E.; Chan, W.C.W.; Laakkonen, P.; Bhatia, S.N.; Ruoslahti, E. Nanocrystal targeting in vivo. Proc. Natl. Acad. Sci. U.S.A. 2002, 99, 12617–12621. [CrossRef]
- Gref, R.; Minamitake, Y.; Peracchia, M.T.; Trubetskoy, V.; Torchilin, V.; Langer, R. Biodegradable long-circulating polymeric nanospheres. Science 1994, 263, 1600–1603. [CrossRef]
- Suk, J.S.; Xu, Q.; Kim, N.; Hanes, J.; Ensign, L.M. PEGylation as a strategy for improving nanoparticle-based drug and gene delivery. Adv. Drug Deliv. Rev. 2016, 99, 28–51. [CrossRef]
- Gu, F.; Zhang, L.; Teply, B.A.; Mann, N.; Wang, A.; Radovic-Moreno, A.F.; Langer, R.; Farokhzad, O.C. Precise engineering of targeted nanoparticles by using self-assembled biointegrated block copolymers. Proc. Natl. Acad. Sci. U.S.A. 2008, 105, 2586–2591. [CrossRef]
- Torchilin, V.P. Multifunctional nanocarriers. Adv. Drug Deliv. Rev. 2012, 64, 302–315.
- Wu, M.-X.; Yang, Y.-W. Metal-organic framework (MOF)-based drug/cargo delivery and cancer therapy. Adv. Mater. 2017, 29, 1606134. [CrossRef]
- Sun, Y.; Zheng, L.; Yang, Y.; Qian, X.; Fu, T.; Li, X.; Yang, Z.; Yan, H.; Cui, C.; Tan, W. Metal-organic framework nanocarriers for drug delivery in biomedical applications. Nano-Micro Lett. 2020, 12, 103. [CrossRef]
- Lebrón, J.A.; Ostos, F.J.; Martínez-Santa, M.; García-Moscoso, F.; López-López, M.; Moyá, M.L.; Bernal, E.; Bachiller, S.; González-Ulloa, G.; Rodríguez-Lucena, D.; Lopes-Costa, T.; Fernández-Torres, R.; Ruiz-Mateos, E.; Pedrosa, J.M.; Rafii-El-Idrissi Benhnia, M.; López-Cornejo, P. Biocompatible metal–organic frameworks as promising platforms to eradicate HIV reservoirs ex vivo in people living with HIV. J. Mater. Chem. B 2024, 12, 5220–5237. [CrossRef]
- Horcajada, P.; Chalati, T.; Serre, C.; Gillet, B.; Sebrie, C.; Baati, T.; Eubank, J.F.; Heurtaux, D.; Clayette, P.; Kreuz, C.; Chang, J.-S.; Hwang, Y.K.; Marsaud, V.; Bories, P.-N.; Cynober, L.; Gil, S.; Férey, G.; Couvreur, P.; Gref, R. Porous metal-organic-framework nanoscale carriers as a potential platform for drug delivery and imaging. Nat. Mater. 2010, 9, 172–178. [CrossRef]
- McKinlay, A.C.; Morris, R.E.; Horcajada, P.; Férey, G.; Gref, R.; Couvreur, P.; Serre, C. BioMOFs: Metal-organic frameworks for biological and medical applications. Angew. Chem. Int. Ed. 2010, 49, 6260–6266. [CrossRef]
- He, S.; Wu, L.; Li, X.; Sun, H.; Xiong, T.; Liu, J.; Huang, C.; Xu, H.; Sun, H.; Chen, W.; Zhang, J. Metal-organic frameworks for advanced drug delivery. Acta Pharm. Sin. B 2021, 11, 2362–2395. [CrossRef]
- He, L.; Liu, Y.; Lau, J.; Fan, W.; Li, Q.; Zhang, C.; Huang, P.; Chen, X. Recent progress in nanoscale metal-organic frameworks for drug release and cancer therapy. Nanomedicine 2019, 14, 1343–1365. [CrossRef]
- Wang, Y.; Yan, J.; Wen, N.; Xiong, H.; Cai, S.; He, Q.; Hu, Y.; Peng, D.; Liu, Z.; Liu, Y. Metal-organic frameworks for stimuli-responsive drug delivery. Biomaterials 2020, 230, 119619. [CrossRef]
- di Nunzio, M.R.; Agostoni, V.; Cohen, B.; Gref, R.; Douhal, A. A “ship in a bottle” strategy to load a hydrophilic anticancer drug in porous metal organic framework nanoparticles: Efficient encapsulation, matrix stabilization, and photodelivery. J. Med. Chem. 2014, 57, 411–420. [CrossRef]
- Evison, B.J.; Sleebs, B.E.; Watson, K.G.; Phillips, D.R.; Cutts, S.M. Mitoxantrone, more than just another topoisomerase II poison. Med. Res. Rev. 2016, 36, 248–299. [CrossRef]
- Damiani, R.M.; Moura, D.J.; Viau, C.M.; Brito, V.; Morás, A.M.; Henriques, J.A.P.; Saffi, J. Influence of PARP-1 inhibition in the cardiotoxicity of the topoisomerase 2 inhibitors doxorubicin and mitoxantrone. Toxicol. In Vitro 2018, 52, 203–213. [CrossRef]
- Huang, S.; Yuan, J.; Xie, Y.; et al. Targeting nano-regulator based on metal-organic frameworks for enhanced immunotherapy of bone metastatic prostate cancer. Cancer Nanotechnol. 2023, 14, 43. [CrossRef]
- Singhal, M.; Riches-Suman, K.; Pors, K.; Addicoat, M.A.; Ruiz, A.; Nayak, S.; Elies, J. Encapsulation and delivery of mitoxantrone using zirconium-based metal-organic frameworks (MOFs) and their cytotoxic potential in breast cancer cells. Appl. Sci. 2024, 14, 1902. [CrossRef]
- Xu, X.; Wu, Y.; Huang, L.; Wu, P.; Huang, L. A metal-organic framework nanocarrier system for precise targeted delivery and reduced systemic toxicity of mitoxantrone in breast cancer therapy. ACS Appl. Mater. Interfaces 2025, 17, 60241–60254. [CrossRef]
- Ji, M.; Liu, H.; Liang, X.; Wei, M.; Shi, D.; Gou, J.; Yin, T.; He, H.; Tang, X.; Zhang, Y. Tumor cells are under siege from all sides: Tumor cell-mimic metal-organic framework nanoparticles triggering cuproptosis/ferroptosis/apoptosis for chemo-chemodynamic-photothermal-immunological synergistic antitumor therapy. Chem. Eng. J. 2024, 485, 149640. [CrossRef]
- Wang, T.C.; Vermeulen, N.A.; Kim, I.S.; Martinson, A.B.F.; Stoddart, J.F.; Hupp, J.T.; Farha, O.K. Scalable synthesis and post-modification of a mesoporous metal-organic framework called NU-1000. Nat. Protoc. 2016, 11, 149–162. [CrossRef]
- Vargas, E.L.; Snurr, R.Q. Heterogeneous diffusion of alkanes in the hierarchical metal-organic framework NU-1000. Langmuir 2015, 31, 10056–10065. [CrossRef]
- Chen, Y.; Li, P.; Modica, J.A.; Drout, R.J.; Farha, O.K. Acid-resistant mesoporous metal-organic framework toward oral insulin delivery: Protein encapsulation, protection, and release. J. Am. Chem. Soc. 2018, 140, 5678–5681. [CrossRef]
- Zhao, X.; Liu, S.; Hu, C.; Liu, Y.; Pang, M.; Lin, J. Controllable synthesis of monodispersed NU-1000 drug carrier for chemotherapy. ACS Appl. Bio Mater. 2019, 2, 4436–4441. [CrossRef]
- Lee, B.S.; Dutta, P.K. Optical spectroscopic studies of the antitumor drug 1,4-dihydroxy-5,8-bis[[2-[(2-hydroxyethyl)amino]ethyl]amino]-9,10-anthracenedione (mitoxantrone). J. Phys. Chem. 1989, 93, 5665–5672. [CrossRef]
- Enache, M.; Volanschi, E. Spectral characterization of self-association of antitumor drug mitoxantrone. Rev. Roum. Chim. 2010, 55, 255–262.
- Castillero, P.; Sánchez-Valencia, J.R.; Cano, M.; Pedrosa, J.M.; Roales, J.; Barranco, A.; González-Elipe, A.R. Active and optically transparent tetracationic porphyrin/TiO2 composite thin films. ACS Appl. Mater. Interfaces 2010, 2, 712–721. [CrossRef]
- Yabushita, M.; Li, P.; Durkin, K.A.; Kobayashi, H.; Fukuoka, A.; Farha, O.K.; Katz, A. Insights into supramolecular sites responsible for complete separation of biomass-derived phenolics and glucose in metal-organic framework NU-1000. Langmuir 2017, 33, 4129–4137. [CrossRef]
- Bhunia, S.; Saha, P.; Moitra, P.; Addicoat, M.A.; Bhattacharya, S. Efficacious and sustained release of an anticancer drug mitoxantrone from new covalent organic frameworks using protein corona. Chem. Sci. 2022, 13, 7920–7932. [CrossRef]
- Guerrero, F.; Moscoso, F.G.; Silvestre-Albero, J.; Martin-Malo, A.; Carrillo-Carrión, C. Fluorinated zirconium-based metal-organic frameworks as novel sorbents to improve the efficacy of hemodialysis treatment. Small Sci. 2025, 5, 2500054. [CrossRef]
- Kato, S.; Otake, K.; Chen, H.; Akpinar, I.; Buru, C.T.; Islamoglu, T.; Snurr, R.Q.; Farha, O.K. Zirconium-based metal-organic frameworks for the removal of protein-bound uremic toxin from human serum albumin. J. Am. Chem. Soc. 2019, 141, 2568–2576. [CrossRef]
- Li, S.; Yang, S.; Liang, G.; Yan, M.; Wei, C.; Lu, Y. Regulation and photocatalytic degradation mechanism of a hydroxyl modified UiO-66 type metal organic framework. RSC Adv. 2023, 13, 5273–5282. [CrossRef]
- Trushina, D.; Sapach, A.Y.; Burachevskaia, O.A.; Medvedev, P.V.; Khmelenin, D.N.; Borodina, T.N.; Soldatov, M.; Butova, V.V. Doxorubicin-loaded core-shell UiO-66@SiO2 metal-organic frameworks for targeted cellular uptake and cancer treatment. Pharmaceutics 2022, 14, 1325. [CrossRef]
- Nel, A.; Xia, T.; Mädler, L.; Li, N. Toxic potential of materials at the nanolevel. Science 2006, 311, 622–627. [CrossRef]
- Li, P.; Klet, R.C.; Moon, S.-Y.; Wang, T.C.; Deria, P.; Peters, A.W.; Klahr, B.M.; Park, H.-J.; Al-Juaid, S.S.; Hupp, J.T.; Farha, O.K. Synthesis of nanocrystals of Zr-based metal-organic frameworks with csq-net: Significant enhancement in the degradation of a nerve agent simulant. Chem. Commun. 2015, 51, 10925–10928. [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



