Submitted:
15 April 2026
Posted:
16 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Patient Recruitment, Inclusion and Exclusion Criteria
2.3. Epidemiological, Clinical and Histopathological Characteristics of Patients
2.4. Primary and Secondary Outcomes
2.5. Specimen Processing and Section Preparation
2.6. Immunohistochemical Staining Protocol
2.7. Interpretation of MMR Status
2.8. Statistical Analysis
3. Results
3.1. Cohort Characteristics
3.2. MMRd Rates
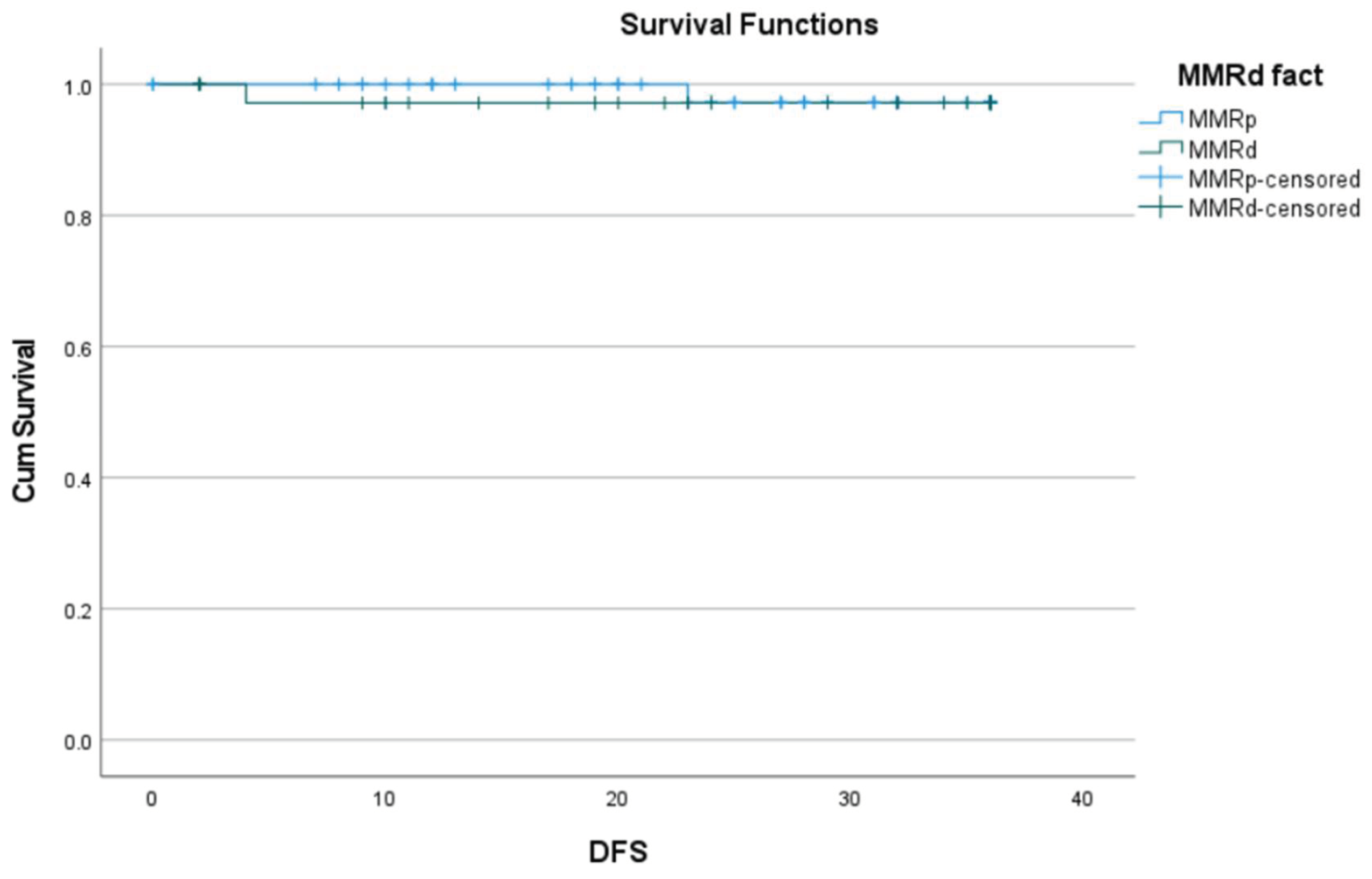
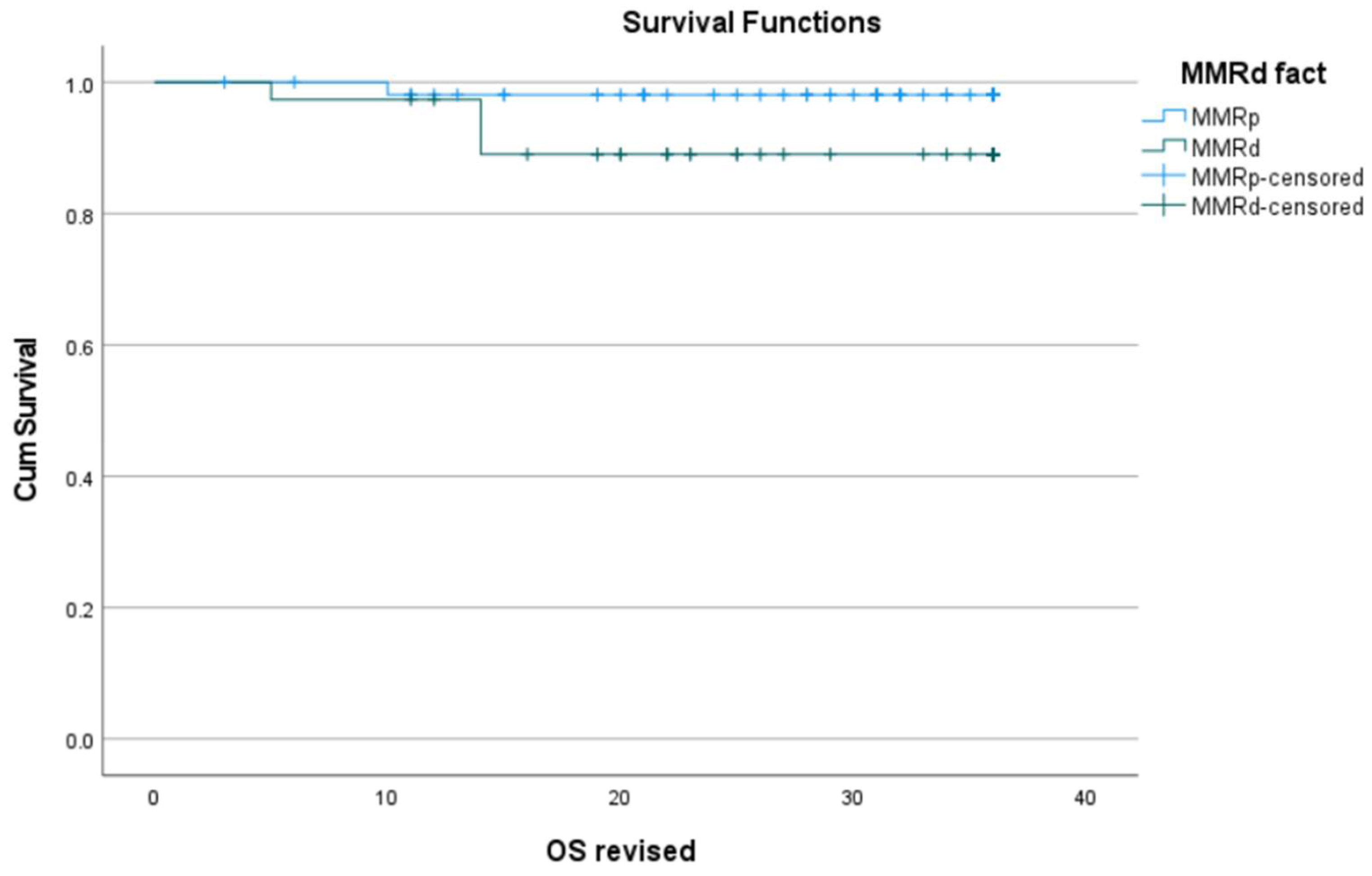
3.3. Correlation of MMRd with Histopathological Characteristics
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Siegel, R.L.; Kratzer, T.B.; Giaquinto, A.N.; Sung, H.; Jemal, A. Cancer Statistics, 2025. CA. Cancer J. Clin. 2025, 75, 10–45. [Google Scholar] [CrossRef] [PubMed]
- Kandoth, C.; Schultz, N.; Cherniack, A.D.; Akbani, R.; Liu, Y.; Shen, H.; Robertson, A.G.; Pashtan, I.; Shen, R.; et al.; Cancer Genome Atlas Research Network Integrated Genomic Characterization of Endometrial Carcinoma. Nature 2013, 497, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Concin, N.; Matias-Guiu, X.; Vergote, I.; Cibula, D.; Mirza, M.R.; Marnitz, S.; Ledermann, J.; Bosse, T.; Chargari, C.; Fagotti, A.; et al. ESGO/ESTRO/ESP Guidelines for the Management of Patients with Endometrial Carcinoma. Int. J. Gynecol. Cancer Off. J. Int. Gynecol. Cancer Soc. 2021, 31, 12–39. [Google Scholar] [CrossRef] [PubMed]
- Jumaah, A.S.; Al-Haddad, H.S.; Salem, M.M.; McAllister, K.A.; Yasseen, A.A. Mismatch Repair Deficiency and Clinicopathological Characteristics in Endometrial Carcinoma: A Systematic Review and Meta-Analysis. J. Pathol. Transl. Med. 2021, 55, 202–211. [Google Scholar] [CrossRef]
- Santoro, A.; Angelico, G.; Travaglino, A.; Inzani, F.; Arciuolo, D.; Valente, M.; D’Alessandris, N.; Scaglione, G.; Fiorentino, V.; Raffone, A.; et al. New Pathological and Clinical Insights in Endometrial Cancer in View of the Updated ESGO/ESTRO/ESP Guidelines. Cancers 2021, 13, 2623. [Google Scholar] [CrossRef]
- Herman, J.G.; Umar, A.; Polyak, K.; Graff, J.R.; Ahuja, N.; Issa, J.P.; Markowitz, S.; Willson, J.K.; Hamilton, S.R.; Kinzler, K.W.; et al. Incidence and Functional Consequences of hMLH1 Promoter Hypermethylation in Colorectal Carcinoma. Proc. Natl. Acad. Sci. U. S. A. 1998, 95, 6870–6875. [Google Scholar] [CrossRef]
- Boland, C.R.; Koi, M.; Chang, D.K.; Carethers, J.M. The Biochemical Basis of Microsatellite Instability and Abnormal Immunohistochemistry and Clinical Behavior in Lynch Syndrome: From Bench to Bedside. Fam. Cancer 2008, 7, 41–52. [Google Scholar] [CrossRef]
- Ryan, N. a. J.; Glaire, M.A.; Blake, D.; Cabrera-Dandy, M.; Evans, D.G.; Crosbie, E.J. The Proportion of Endometrial Cancers Associated with Lynch Syndrome: A Systematic Review of the Literature and Meta-Analysis. Genet. Med. Off. J. Am. Coll. Med. Genet. 2019, 21, 2167–2180. [Google Scholar] [CrossRef]
- Ligtenberg, M.J.L.; Kuiper, R.P.; Chan, T.L.; Goossens, M.; Hebeda, K.M.; Voorendt, M.; Lee, T.Y.H.; Bodmer, D.; Hoenselaar, E.; Hendriks-Cornelissen, S.J.B.; et al. Heritable Somatic Methylation and Inactivation of MSH2 in Families with Lynch Syndrome Due to Deletion of the 3’ Exons of TACSTD1. Nat. Genet. 2009, 41, 112–117. [Google Scholar] [CrossRef]
- Luchini, C.; Bibeau, F.; Ligtenberg, M.J.L.; Singh, N.; Nottegar, A.; Bosse, T.; Miller, R.; Riaz, N.; Douillard, J.-Y.; Andre, F.; et al. ESMO Recommendations on Microsatellite Instability Testing for Immunotherapy in Cancer, and Its Relationship with PD-1/PD-L1 Expression and Tumour Mutational Burden: A Systematic Review-Based Approach. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2019, 30, 1232–1243. [Google Scholar] [CrossRef]
- Gordhandas, S.; Kahn, R.M.; Gamble, C.; Talukdar, N.; Maddy, B.; Baltich Nelson, B.; Askin, G.; Christos, P.J.; Holcomb, K.; Caputo, T.A.; et al. Clinicopathologic Features of Endometrial Cancer with Mismatch Repair Deficiency. Ecancermedicalscience 2020, 14, 1061. [Google Scholar] [CrossRef]
- Stelloo, E.; Jansen, A.M.L.; Osse, E.M.; Nout, R.A.; Creutzberg, C.L.; Ruano, D.; Church, D.N.; Morreau, H.; Smit, V.T.H.B.M.; van Wezel, T.; et al. Practical Guidance for Mismatch Repair-Deficiency Testing in Endometrial Cancer. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2017, 28, 96–102. [Google Scholar] [CrossRef]
- Wing-Cheuk Wong, R.; Palicelli, A.; Hoang, L.; Singh, N. Interpretation of P16, P53 and Mismatch Repair Protein Immunohistochemistry in Gynaecological Neoplasia. Diagn. Histopathol. 2020, 26, 257–277. [Google Scholar] [CrossRef]
- Li, Z.; Pearlman, A.H.; Hsieh, P. DNA Mismatch Repair and the DNA Damage Response. DNA Repair 2016, 38, 94–101. [Google Scholar] [CrossRef]
- Kim, M.-K.; So, K.A.; Chun, Y.-K.; Kim, Y.H.; Lim, K.T.; Lee, K.H.; Kim, T.J. Clinicopathologic Significance of Mismatch Repair Protein Expression in Endometrioid Endometrial Cancer. Taiwan. J. Obstet. Gynecol. 2023, 62, 724–728. [Google Scholar] [CrossRef]
- Aytekin, O.; Cesur, N.; Gozel, S.; Karsli, S.E.; Tokalioglu, A.A.; Kilic, F.; Cuylan, Z.F.; Selcuk, I.; Comert, G.K.; Erdogan, F.; et al. Mismatch Repair Protein Deficiency and Its Relationship with Clinicopathological Factors in Endometrial Cancer: A Retrospective Study. J. Cancer 2025, 16, 2778–2786. [Google Scholar] [CrossRef]
- Wang, C.; Kuang, W.; Zeng, J.; Ren, Y.; Liu, Q.; Sun, H.; Feng, M.; Liang, D. A Retrospective Study of Consistency between Immunohistochemistry and Polymerase Chain Reaction of Microsatellite Instability in Endometrial Cancer. PeerJ 2023, 11, e15920. [Google Scholar] [CrossRef]
- Kato, M.; Takano, M.; Miyamoto, M.; Sasaki, N.; Goto, T.; Tsuda, H.; Furuya, K. DNA Mismatch Repair-Related Protein Loss as a Prognostic Factor in Endometrial Cancers. J. Gynecol. Oncol. 2015, 26, 40–45. [Google Scholar] [CrossRef]
- Raffone, A.; Travaglino, A.; Mascolo, M.; Carbone, L.; Guida, M.; Insabato, L.; Zullo, F. TCGA Molecular Groups of Endometrial Cancer: Pooled Data about Prognosis. Gynecol. Oncol. 2019, 155, 374–383. [Google Scholar] [CrossRef]
- Pasanen, A.; Loukovaara, M.; Bützow, R. Clinicopathological Significance of Deficient DNA Mismatch Repair and MLH1 Promoter Methylation in Endometrioid Endometrial Carcinoma. Mod. Pathol. Off. J. U. S. Can. Acad. Pathol. Inc 2020, 33, 1443–1452. [Google Scholar] [CrossRef]
- Stelloo, E.; Nout, R.A.; Osse, E.M.; Jürgenliemk-Schulz, I.J.; Jobsen, J.J.; Lutgens, L.C.; van der Steen-Banasik, E.M.; Nijman, H.W.; Putter, H.; Bosse, T.; et al. Improved Risk Assessment by Integrating Molecular and Clinicopathological Factors in Early-Stage Endometrial Cancer-Combined Analysis of the PORTEC Cohorts. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2016, 22, 4215–4224. [Google Scholar] [CrossRef]
- Segura, S.E.; Pedra Nobre, S.; Hussein, Y.R.; Abu-Rustum, N.R.; Weigelt, B.; Soslow, R.A.; DeLair, D.F. DNA Mismatch Repair-Deficient Endometrial Carcinosarcomas Portend Distinct Clinical, Morphologic, and Molecular Features Compared With Traditional Carcinosarcomas. Am. J. Surg. Pathol. 2020, 44, 1573–1579. [Google Scholar] [CrossRef]
- de Freitas, D.; Aguiar, F.N.; Anton, C.; de Almeida, D.C.; Bacchi, C.E.; Carvalho, J.P.; Carvalho, F.M. Clinicopathological Characteristics of Endometrial Carcinomas According to DNA Mismatch Repair Protein Status. Heliyon 2023, 9, e17495. [Google Scholar] [CrossRef]
- Raffone, A.; Travaglino, A.; Cerbone, M.; Gencarelli, A.; Mollo, A.; Insabato, L.; Zullo, F. Diagnostic Accuracy of Immunohistochemistry for Mismatch Repair Proteins as Surrogate of Microsatellite Instability Molecular Testing in Endometrial Cancer. Pathol. Oncol. Res. POR 2020, 26, 1417–1427. [Google Scholar] [CrossRef]
- Singh, N.; Wong, R.; Tchrakian, N.; Allen, S.-G.; Clarke, B.; Gilks, C.B. Interpretation of Mismatch Repair Protein Expression Using Obsolete Criteria Results in Discrepancies with Microsatellite Instability and Mutational Testing Results. Comment on Hechtman et al. Mod Pathol 2020. Mod. Pathol. Off. J. U. S. Can. Acad. Pathol. Inc 2021, 33 34, 871-879 1031–1032. [Google Scholar] [CrossRef]
- Travaglino, A.; Raffone, A.; Mascolo, M.; Guida, M.; Insabato, L.; Zannoni, G.F.; Zullo, F. TCGA Molecular Subgroups in Endometrial Undifferentiated/Dedifferentiated Carcinoma. Pathol. Oncol. Res. POR 2020, 26, 1411–1416. [Google Scholar] [CrossRef]
- Köbel, M.; Tessier-Cloutier, B.; Leo, J.; Hoang, L.N.; Gilks, C.B.; Soslow, R.A.; Delair, D.; Stewart, C.J.R.; Lee, C.-H. Frequent Mismatch Repair Protein Deficiency in Mixed Endometrioid and Clear Cell Carcinoma of the Endometrium. Int. J. Gynecol. Pathol. Off. J. Int. Soc. Gynecol. Pathol. 2017, 36, 555–561. [Google Scholar] [CrossRef]
- Travaglino, A.; Raffone, A.; Mascolo, M.; Guida, M.; Insabato, L.; Zannoni, G.F.; Zullo, F. Clear Cell Endometrial Carcinoma and the TCGA Classification. Histopathology 2020, 76, 336–338. [Google Scholar] [CrossRef]
- Travaglino, A.; Raffone, A.; Gencarelli, A.; Mollo, A.; Guida, M.; Insabato, L.; Santoro, A.; Zannoni, G.F.; Zullo, F. TCGA Classification of Endometrial Cancer: The Place of Carcinosarcoma. Pathol. Oncol. Res. POR 2020, 26, 2067–2073. [Google Scholar] [CrossRef]
- Conlon, N.; Da Cruz Paula, A.; Ashley, C.W.; Segura, S.; De Brot, L.; da Silva, E.M.; Soslow, R.A.; Weigelt, B.; DeLair, D.F. Endometrial Carcinomas with a “Serous” Component in Young Women Are Enriched for DNA Mismatch Repair Deficiency, Lynch Syndrome, and POLE Exonuclease Domain Mutations. Am. J. Surg. Pathol. 2020, 44, 641–648. [Google Scholar] [CrossRef]
- Soslow, R.A.; Tornos, C.; Park, K.J.; Malpica, A.; Matias-Guiu, X.; Oliva, E.; Parkash, V.; Carlson, J.; McCluggage, W.G.; Gilks, C.B. Endometrial Carcinoma Diagnosis: Use of FIGO Grading and Genomic Subcategories in Clinical Practice: Recommendations of the International Society of Gynecological Pathologists. Int. J. Gynecol. Pathol. Off. J. Int. Soc. Gynecol. Pathol. 2019, 38 Suppl 1, S64–S74. [Google Scholar] [CrossRef]
- Murali, R.; Davidson, B.; Fadare, O.; Carlson, J.A.; Crum, C.P.; Gilks, C.B.; Irving, J.A.; Malpica, A.; Matias-Guiu, X.; McCluggage, W.G.; et al. High-Grade Endometrial Carcinomas: Morphologic and Immunohistochemical Features, Diagnostic Challenges and Recommendations. Int. J. Gynecol. Pathol. Off. J. Int. Soc. Gynecol. Pathol. 2019, 38 Suppl 1, S40–S63. [Google Scholar] [CrossRef]
| Characteristic | Value |
|---|---|
| Age(mean ± SD) | 65.84 ± 11.32 |
| Histopathological Type, n(%) | |
| Endometrioid | 80 (86.0%) |
| Serous | 3 (3.2%) |
| MMMT | 5 (5.4%) |
| Clear cell | 1 (1.1%) |
| Mixed | 4 (4.3%) |
| Myometrial Invasion, n(%) | |
| >50% | 42 (45.2%) |
| <50% | 51 (54.8%) |
| LVSI, n(%) | |
| Yes | 22 (23.7%) |
| No | 71 (76.3%) |
| Grade, n(%) | |
| Low | 66 (71.0%) |
| High | 27 (29.0%) |
| Stage, n(%) | |
| I | 46 (49.5%) |
| II | 34 (36.6%) |
| III | 8 (8.6%) |
| IV | 5 (5.4%) |
| MMRstatus | |
| MMRp ΜΜRd |
54 (58.0%) 39 (42.0%) |
|
Nodal status No Yes |
89 (95.7%) 4 (4.3%) |
|
Recurrence No Yes |
2(2.2%) 91(97.8%) |
|
Death No Yes |
5(5.4%) 88(94.6%) |
| Variables | p value |
|---|---|
| Histologic Type | 0.009 |
| Myometrial Invasion | 0.005 |
| Grade | 0.907 |
| LVSI | 0.301 |
| Stage | 0.820 |
| Death | 0.329 |
| Any Nodal Invasion | 0.126 |
| Recurrence | 0.500 |
| Death | 0.15 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




