Submitted:
14 April 2026
Posted:
16 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
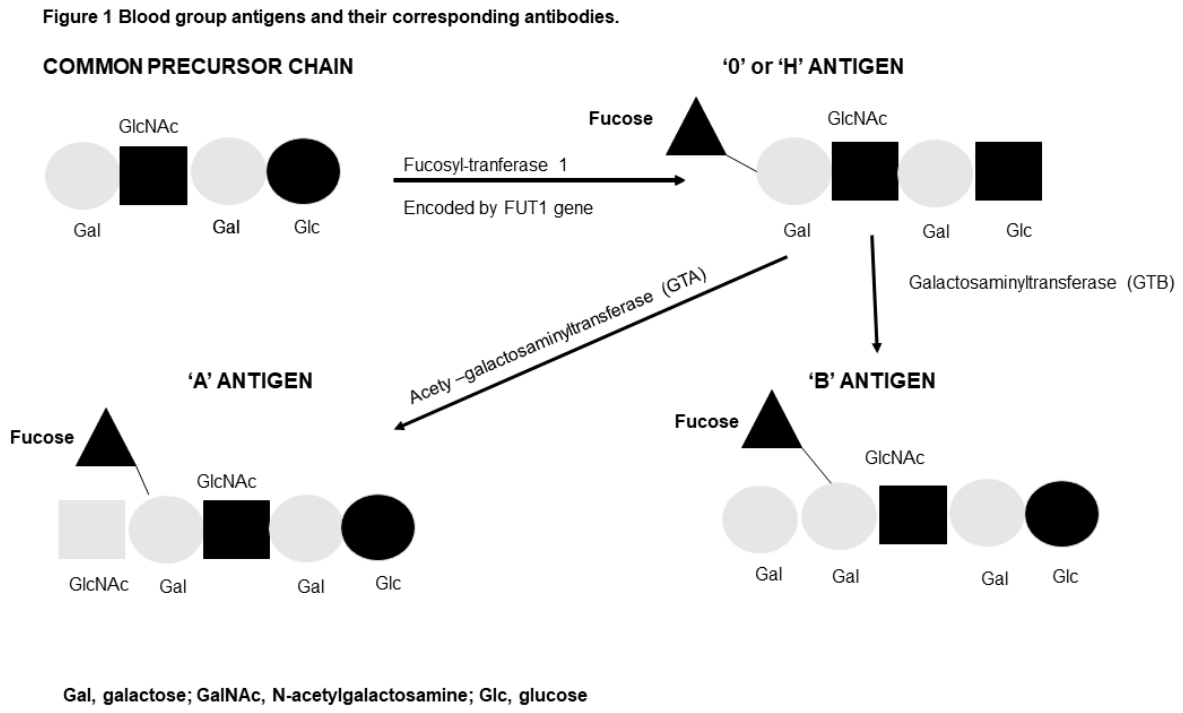
2. Immunological Aspects
3. Development Of The Different Strategies To Overcome The Problem Of A, B, 0 Incompatibility
- a)
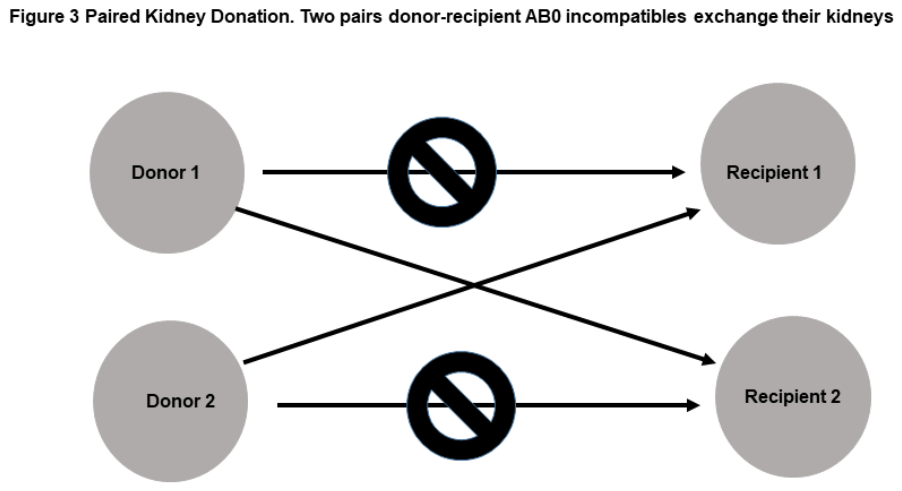
- The kidney paired donation (KPD). It is the simpler way to perform kidney transplantation in AB0i. This way concerns LDKT involving different families with a kidney exchange among them. Each family is unable to make LDKT, but the problem may be overcome exchanging kidneys. This avoid immunological problems, but may implies legal ones.
- b)
- The desensitization technique is the most utilized. There are several techniques that may be adopted for desensitization and these techniques has evolved by time as will described later.
- c)
- The use of enzymatic conversion of the blood group is the most recent and promising technique for the future. Anyway, also the enzymatic conversion is part of desensitization treatment.
4. Kidney Paired Donation
5. Desensitization
6. Principal Techniques Used to Obtain Desensitization, Results Obtained and Controversies
7. Clinical Outcomes After Kidney Transplantation Involving AB0i Kidneys
8. Conclusions
- a)
- Removal of circulating AB0 antibodies by PE or IA;
- b)
- Immunomodulation by the administration of IVIG before and even after transplantation;
- c)
- B cell depletion by the use of immunosuppressant, among which monoclonal antibody anti CD20 seems to be one of the more effective. Indeed, B cells produce isoagglutinins, and their reduction is essential to obtain an effective desensitization.
- d)
- Enzymatic elimination of the donor kidney of group A or group B. To date this technique is only experimental, but it seems to be effective. The A or B elimination could be reached adding the appropriate enzyme to the kidney during the machine perfusion of the explanted kidneys.
- e)
- It is thought that the enzymatic elimination in the future could be added to the desensitization strategy.
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Stewart, D.E.; Klassen, D.K. Kidney Transplants from HLA-Incompatible Live Donors and Survival. N Engl J Med. 2016, 375, 287–8. [Google Scholar]
- Starzl, T.E.; Marchioro, T.L.; Holmes, J.H.; Hermann, G.; Brittain, R.S.; Stonington, O.H.; Talmage, D.W.; Waddel, W.R. RENAL HOMOGRAFTS IN PATIENTS WITH MAJOR DONOR-RECIPIENT BLOOD GROUP INCOMPATIBILITIES. Surgery. 1964, 55, 159–200. [Google Scholar]
- Oriol, R. ABH and related tissue antigens. Biochem Soc Trans. 1987, 15, 596–599. [Google Scholar] [CrossRef]
- Watkins, W.M.; Morgan, W.T.J. Possible genetical pathways for the biosynthesis of blood group mucopolysaccharides. Vox 1959, 4, 27–119. [Google Scholar]
- Larsen, R.D.; Ernst, L.K.; Nair, R.P.; Lowe, J.B. Molecular cloning, sequence, and expression of a human GDP-L-fucose:beta-D-galactoside 2-alpha-L-fucosyltransferase cDNA that can form the H blood group antigen. Proc Natl Acad Sci U S A 1990, 87, 6674–6678. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, F.; Clausen, H.; White, T.; Marken, J.; Hakomori, S. Molecular genetic basis of the histo-blood group ABO system. Nature. 1990, 17(345), 229–233. [Google Scholar] [CrossRef] [PubMed]
- Rydberg, L.; Breimer, M.E.; Brynger, H.; Samuelsson, B.E. ABO-incompatible kidney transplantation (A2 to O). Qualitative and semiquantitative studies of the humoral immune response against different blood group A antigens. Transplantation 1990, 49, 954–960. [Google Scholar] [CrossRef] [PubMed]
- Rydberg, L.; Breimer, M.E.; Samuelsson, B.E.; Brynger, H. Blood group ABO-incompatible (A2 to O) kidney transplantation in human subjects: a clinical, serologic, and biochemical approach. Transplant Proc. 1987, 19, 4528–4537. [Google Scholar]
- Böhmig, G.A.; Farkas, M.A.; Eskandary, F.; Wekerle, T. Strategies to overcome the ABO barrier in kidney transplantation. Nat Rev Nephrol. 2015, 11, 732–747. [Google Scholar] [CrossRef]
- Matsui, T.; Fujimura, Y.; Nishida, S.; Titani, K. Human plasma alpha 2-macroglobulin and von Willebrand factor possess covalently linked ABO(H) blood group antigens in subjects with corresponding ABO phenotype. Blood 1993, 82, 663–668. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.A.; Bowen, D.J.; Hallett, M.B.; Giddings, J.C.; Collins, P.W. Factor VIIa induced release of von Willebrand factor from human umbilical vein endothelial cells by a tyrosine kinase dependent pathway. Thromb Haemost. 2002, 87, 1057–1061. [Google Scholar]
- Brynger, H.; Rydberg, L.; Samuelsson, B.; Blohmé, I.; Lindholm, A.; Sandberg, L. Renal transplantation across a blood group barrier--‘A2’ kidneys to ‘O’ recipients. Proc Eur Dial Transplant Assoc. 1983, 19, 427–431. [Google Scholar]
- Rapaport, F.T. The case for a living emotionally related international kidney donor exchange registry. Transplant Proc. 1986, 18, 5–9. [Google Scholar]
- Delmonico, F.L.; Morrissey, P.E.; Lipkowitz, G.S.; Stoff, J.S.; Himmelfarb, J.; Harmon, W.; Pavlakis, M.; Mah, H.; Goguen, J.; Luskin, R.; Milford, E.; Basadonna, G.; Chobanian, M.; Bouthot, B.; Lorber, M.; Rohrer, R.J. Donor kidney exchanges. Am J Transplant. 2004, 4, 1628–1634. [Google Scholar] [CrossRef]
- Segev, D.L.; Gentry, S.E.; Warren, D.S.; Reeb, B.; Montgomery, R.A. Kidney paired donation and optimizing the use of live donor organs. JAMA. 2005, 293, 1883–1890. [Google Scholar] [CrossRef] [PubMed]
- de Klerk, M.; Keizer, K.M.; Claas, F.H.; Witvliet, M.; Haase-Kromwijk, B.J.; Weimar, W. The Dutch national living donor kidney exchange program. Am J Transplant. 2005, 5, 2302–2305. [Google Scholar]
- de Klerk, M.; Witvliet, M.D.; Haase-Kromwijk, B.J.; Weimar, W.; Claas, F.H. A flexible national living donor kidney exchange program taking advantage of a central histocompatibility laboratory: the Dutch model. Clin Transpl. 2008, 69–73. [Google Scholar]
- Bingaman, A.W.; Wright, F.H.; Murphey, C.L. Kidney paired donation in live-donor kidney transplantation. N Engl J Med. 2010, 363, 1091–1092. [Google Scholar] [CrossRef]
- Toews, M.; Giancaspro, M.; Richards, B.; Ferrari, P. Kidney Paired Donation and the “Valuable Consideration” Problem: The Experiences of Australia, Canada, and the United States. Transplantation. 2017, 101, 1996–2002. [Google Scholar] [CrossRef] [PubMed]
- D’Alessandro, T.; Veale, J.L. Innovations in kidney paired donation transplantation. Curr Opin Organ Transplant. 2019, 24, 429–433. [Google Scholar] [CrossRef]
- Pins, M.R.; Saidman, S.L.; Cosimi, A.B.; Jennings, L.D.; Stowell, C.P. Accelerated acute rejection of an apparent A2 renal allograft in an O recipient: report of a case with flow cytometric analysis. Transplantation. 1997, 63, 984–988. [Google Scholar] [CrossRef]
- Park, W.D.; Grande, J.P.; Ninova, D.; Nath, K.A.; Platt, J.L.; Gloor, J.M.; Stegall, M.D. Accommodation in ABO-incompatible kidney allografts, a novel mechanism of self-protection against antibody-mediated injury. Am J Transplant. 2003, 3, 952–960. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, K.; Miwa, Y.; Ogawa, H.; Yazaki, S.; Iwamoto, M.; Furusawa, T.; Onishi, A.; Kuzuya, T.; Haneda, M.; Watarai, Y.; Uchida, K.; Kobayashi, T. Comparative study on signal transduction in endothelial cells after anti-a/b and human leukocyte antigen antibody reaction: implication of accommodation. Transplantation 2012, 93, 390–397. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Saito, K.; Takahara, S.; Okuyama, A.; Tanabe, K.; Toma, H.; Uchida, K.; Hasegawa, A.; Yoshimura, N.; Kamiryo, Y. Japanese ABO-Incompatible Kidney Transplantation Committee. Excellent long-term outcome of ABO-incompatible living donor kidney transplantation in Japan. Am J Transplant. 2004, 4, 1089–1096. [Google Scholar] [CrossRef]
- Gloor, J.M.; Lager, D.J.; Fidler, M.E.; Grande, J.P.; Moore, S.B.; Winters, J.L.; Kremers, W.K.; Stegall, M.D. A Comparison of splenectomy versus intensive posttransplant antidonor blood group antibody monitoring without splenectomy in ABO-incompatible kidney transplantation. Transplantation. 2005, 80, 1572–1577. [Google Scholar] [CrossRef]
- Sonnenday, C.J.; Warren, D.S.; Cooper, M.; Samaniego, M.; Haas, M.; King, K.E.; Shirey, R.S.; Simpkins, C.E.; Montgomery, R.A. Plasmapheresis, CMV hyperimmune globulin, and anti-CD20 allow ABO-incompatible renal transplantation without splenectomy. Am J Transplant. 2004, 4, 1315–1322. [Google Scholar] [CrossRef]
- Genberg, H.; Kumlien, G.; Wennberg, L.; Berg, U.; Tydén, G. ABO-incompatible kidney transplantation using antigen-specific immunoadsorption and rituximab: a 3-year follow-up. Transplantation. 2008, 85, 1745–1754. [Google Scholar] [CrossRef]
- Naciri Bennani, H.; Bobo Barry, K.M.; Noble, J.; Malvezzi, P.; Jouve, T.; Rostaing, L. Outcomes of ABO-incompatible kidney transplants with very high isoagglutinin titers: a single-center experience and literature review. Front Immunol. 2024, 15, 1504495. [Google Scholar] [CrossRef]
- Eum, S.H.; Lee, H.; Ko, E.J.; Min, J.W.; Ban, T.H.; Yoon, H.E.; Shin, S.J.; Chung, B.H. Long-Term Clinical Outcomes of ABO-Incompatible Kidney Transplantation in Patients With High Baseline Anti-A/B Antibody Titers. J Korean Med Sci. 2025, 40, e308. [Google Scholar] [CrossRef] [PubMed]
- Genberg, H.; Kumlien, G.; Wennberg, L.; Berg, U.; Tydén, G. Isoagglutinin adsorption in ABO-incompatible transplantation. Transfus Apher Sci. 2010, 43, 231–235. [Google Scholar] [CrossRef] [PubMed]
- Cen, M.; Wang, R.; Kong, W.; Deng, H.; Lei, W.; Chen, J. ABO-incompatible living kidney transplantation. Clin Transplant. 2020, 34, e14050. [Google Scholar] [CrossRef]
- Biglarnia, A.R.; Nilsson, B.; Nilsson Ekdahl, K.; Tufveson, G.; Nilsson, T.; Larsson, E.; Wadström, J. Desensitization with antigen-specific immunoadsorption interferes with complement in ABO-incompatible kidney transplantation. Transplantation. 2012, 93, 87–92. [Google Scholar] [CrossRef]
- Tobian, A.A.; Shirey, R.S.; Montgomery, R.A.; Ness, P.M.; King, K.E. The critical role of plasmapheresis in ABO-incompatible renal transplantation. Transfusion. 2008, 48, 2453–2460. [Google Scholar] [CrossRef]
- Tobian, A.A.; Shirey, R.S.; Montgomery, R.A.; Tisch, D.J.; Ness, P.M.; King, K.E. plasma exchange reduces ABO titers to permit ABO-incompatible renal transplantation. Transfusion. 2009, 49, 1248–1254. [Google Scholar] [CrossRef]
- Tydén, G.; Donauer, J.; Wadström, J.; Kumlien, G.; Wilpert, J.; Nilsson, T.; Genberg, H.; Pisarski, P.; Tufveson, G. Implementation of a Protocol for ABO-,incompatible kidney transplantation--a three-center experience with 60 consecutive transplantations. Transplantation. 2007, 83, 1153–1155. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, Y.; Kato, Y.; Kaneko, M.K.; Ogasawara, S.; Shimazu, M.; Tanabe, M.; Kawachi, S.; Obara, H.; Shinoda, M.; Kitagawa, Y.; Narimatsu, H.; Kitajima, M. Neutralization of blood group A-antigen by a novel anti-A antibody: overcoming ABO-incompatible solid-organ transplantation. Transplantation. 2008, 85, 378–385. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.; Niekrasz, M.; Kehoe, M.; Rolf, L.L., Jr.; Martin, M.; Baker, J.; Kosanke, S.; Romano, E.; Zuhdi, N.; Cooper, D.K. Cardiac allotransplantation across the ABO-blood group barrier by the neutralization of preformed antibodies: the baboon as a model for the human. Lab Anim Sci. 1994, 44, 121–124. [Google Scholar] [PubMed]
- Wongsaroj, P.; Kahwaji, J.; Vo, A.; Jordan, S.C. Modern approaches to incompatible kidney transplantation. World J Nephrol. 2015, 4, 354–362. [Google Scholar] [CrossRef] [PubMed]
- Heo, G.Y.; Jung, M.; Piao, H.; Kim, H.J.; Kim, H.W.; Lee, J.; Huh, K.H.; Kim, B.S.; Yang, J. Successful eculizumab treatment as an adjunctive therapy to desensitization in ABO-incompatible living donor kidney transplantation and its molecular phenotypes. Front Immunol. 2024, 15, 1465851. [Google Scholar] [CrossRef]
- Wan, Z.; He, X.; Zhou, J.; Tan, Z.; Zhong, Q.; Zhao, F. The effect of eculizaumab combined with double filtration plasmapheresis in improving recovery outcomes in ABOi-KT patients. Transpl Immunol. 2026, 95, 102362. [Google Scholar] [CrossRef]
- Mohamed, M.; Sweeney, T.; Alkhader, D.; Nassar, M.; Alqassieh, A.; Lakhdar, S.; Nso, N.; Fülöp, T.; Daoud, A.; Soliman, K.M. ABO incompatibility in renal transplantation. World J Transplant. 2021, 11, 388–399. [Google Scholar] [CrossRef]
- Scurt, F.G.; Ewert, L.; Mertens, P.R.; Haller, H.; Schmidt, B.M.W.; Chatzikyrkou, C. Clinical outcomes after ABO-incompatible renal transplantation: a systematic review and meta-analysis. Lancet. 2019, 393, 2059–2072. [Google Scholar] [CrossRef]
- Lo, P.; Sharma, A.; Craig, J.C.; Wyburn, K.; Lim, W.; Chapman, J.R.; Palmer, S.C.; Strippoli, G.F.; Wong, G. Preconditioning Therapy in ABO-Incompatible Living Kidney Transplantation: A Systematic Review and Meta-Analysis. Transplantation. 2016, 100, 933–942. [Google Scholar] [CrossRef]
- Okumi, M.; Toki, D.; Nozaki, T.; Shimizu, T.; Shirakawa, H.; Omoto, K.; Inui, M.; Ishida, H.; Tanabe, K. ABO-Incompatible Living Kidney Transplants: Evolution of Outcomes and Immunosuppressive Management. Am J Transplant. 2016, 16, 886–896. [Google Scholar] [CrossRef] [PubMed]
- Kimura, F.; Sato, K.; Kobayashi, S.; Ikeda, T.; Sao, H.; Okamoto, S.; Miyamura, K.; Mori, S.; Akiyama, H.; Hirokawa, M.; Ohto, H.; Ashida, H.; Motoyoshi, K. Japan Marrow Donor Program. Impact of AB0-blood group incompatibility on the outcome of recipients of bone marrow transplants from unrelated donors in the Japan Marrow Donor Program. Haematologica. 2008, 93, 1686–1693. [Google Scholar] [CrossRef]
- Okada, M.; Watarai, Y.; Iwasaki, K.; Murotani, K.; Futamura, K.; Yamamoto, T.; Hiramitsu, T.; Tsujita, M.; Goto, N.; Narumi, S.; Takeda, A.; Morozumi, K.; Uchida, K.; Kobayashi, T. Favorable results in ABO-incompatible renal transplantation without B cell-targeted therapy: Advantages and disadvantages of rituximab pretreatment. Clin Transplant. 2017, 31. [Google Scholar] [CrossRef]
- Xu, P.; Zhao, N.; Wang, J. Success rate and safety of living donor kidney transplantation in ABO blood group incompatible relatives: A systematic review and meta-analysis. Transpl Immunol. 2023, 81, 101921. [Google Scholar] [CrossRef] [PubMed]
- Obeid, D.A.; Broering, D.C.; AlMeshari, K.A.; Shah, Y.Z.; Aleid, H.A.; AlManea, H.M.; AlAbassi, A.M.; AlMozain, N.; Marquez, K.; Alsaadi, E.A.; Ali, T.Z. Impact of different blood group incompatibilities in kidney transplantation: a 15-year outcomes analysis from a large kidney transplant center. Front Transplant. 2025, 18, 1690999. [Google Scholar] [CrossRef] [PubMed]
- Taylan, C.; Mückenhausen, S.I.; Weber, L.T.; Stippel, D.L.; Thumfart, J. Midterm Outcome of AB0 Incompatible Kidney Transplantation in Children and Adolescents-A Single Center Experience. Pediatr Transplant. 2026, 30, e70248. [Google Scholar] [CrossRef] [PubMed]
- Cozzi, M.; Donato, P.; Ugolini, G.; Nguefouet Momo, R.E.; Nacchia, F.; Ballarini, Z.; Piccoli, P.; Cantini, M.; Caletti, C.; Andreola, S.; Gandini, G.; Gambaro, G.; Boschiero, L. Outcomes in AB0 Incompatible Living Donor Kidney Transplantation: A Case - Control Study. Front Med (Lausanne). 2022, 22, 932171. [Google Scholar] [CrossRef]
- Bachmann, F.; Lachmann, N.; Budde, K.; Liefeldt, L.; Halleck, F.; Naik, M.; Friedersdorff, F.; Rudolph, B.; Wu, K.; Meyer, O.; Slowinski, T.; Waiser, J. Late Steroid Withdrawal Following AB0-Incompatible Renal Transplantation. Transplant Proc. 2018, 50, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Barnett, A.N.; Manook, M.; Nagendran, M.; Kenchayikoppad, S.; Vaughan, R.; Dorling, A.; Hadjianastassiou, V.G.; Mamode, N. Tailored desensitization strategies in ABO blood group antibody incompatible renal transplantation. Transpl Int. 2014, 27, 187–196. [Google Scholar] [CrossRef]
- Speer, C.; Kälble, F.; Nusshag, C.; Pego da Silva, L.; Schaier, M.; Becker, L.E.; Klein, K.; Sommerer, C.; Beimler, J.; Leo, A.; Waldherr, R.; Mehrabi, A.; Süsal, C.; Zeier, M.; Morath, C. Outcomes and complications following ABO- incompatible kidney transplantation performed after desensitization by semi-selective immunoadsorption - a retrospective study. Transpl Int. 2019, 32, 1286–1296. [Google Scholar] [CrossRef] [PubMed]
- Becker, L.E.; Siebert, D.; Süsal, C.; Opelz, G.; Leo, A.; Waldherr, R.; Macher-Goeppinger, S.; Schemmer, P.; Schaefer, S.M.; Klein, K.; Beimler, J.; Zeier, M.; Schwenger, V.; Morath, C. Outcomes Following ABO-Incompatible Kidney Transplantation Performed After Desensitization by Nonantigen-Specific Immunoadsorption. Transplantation 2015, 99, 2364–2371. [Google Scholar] [CrossRef] [PubMed]
- Kohei, N.; Hirai, T.; Omoto, K.; Ishida, H.; Tanabe, K. Chronic antibody-mediated rejection is reduced by targeting B-cell immunity during an introductory period. Am J Transplant. 2012, 12, 469–476. [Google Scholar] [CrossRef]
- Subramanian, V.; Gunasekaran, M.; Gaut, J.P.; Phelan, D.; Vachharajani, N.; Santos, R.D.; Wellen, J.; Shenoy, S.; Mohanakumar, T. ABO incompatible renal transplants and decreased likelihood for developing immune responses to HLA and kidney self-antigens. Hum Immunol. 2016, 77, 76–83. [Google Scholar] [CrossRef]
- Langhorst, C.; Ganner, A.; Schneider, J.; Prager, E.P.; Walz, G.; Pisarski, P.; Jänigen, B.; Zschiedrich, S. Long-term Follow-up of ABO-Incompatible Kidney Transplantation in Freiburg, Germany: A Single-Center Outcome Report. Transplant Proc. 2021, 53, 848–855. [Google Scholar] [CrossRef]
- de Weerd, A.E.; Betjes, M.G.H. ABO-Incompatible Kidney Transplant Outcomes: A Meta-Analysis. Clin J Am Soc Nephrol. 2018, 13, 1234–1243. [Google Scholar] [CrossRef]
- Koo, T.Y.; Lee, J.; Lee, Y.; Kim, H.W.; Kim, B.S.; Huh, K.H.; Yang, J. Outcomes of ABO-Incompatible Living Donor Kidney Transplantation Compared to Waiting or Deceased Donor Kidney Transplantation. Am J Nephrol. 2024, 55, 235–244. [Google Scholar] [CrossRef]
- Massie, A.B.; Orandi, B.J.; Waldram, M.M.; Luo, X.; Nguyen, A.Q.; Montgomery, R.A.; Lentine, K.L.; Segev, D.L. Impact of ABO-Incompatible Living Donor Kidney Transplantation on Patient Survival. Am J Kidney Dis. 2020, 76, 616–623. [Google Scholar] [CrossRef]
- de Weerd, A.E.; van den Brand, J.A.J.G.; Bouwsma, H.; de Vries, A.P.J.; Dooper, I.P.M.M.; Sanders, J.F.; Christiaans, M.H.L.; van Reekum, F.E.; van Zuilen, A.D.; Bemelman, F.J.; Nurmohamed, A.S.; van Agteren, M.; Betjes, M.G.H.; de Jong, M.F.C.; Baas, M.C. ABO-incompatible kidney transplantation in perspective of deceased donor transplantation and induction strategies: a propensity-matched analysis. Transpl Int. 2021, 34, 2706–2719. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Pan, J.; Li, D.; Wang, K.; Ding, H.; Zhang, F.; Wang, W.; Li, P.; Zhong, J.; Liao, G. The viability of ABO-incompatible kidney transplants: a single-center cohort in China. Front Immunol. 2026, 17, 1747411. [Google Scholar] [CrossRef]
- Zeng, J.; Ma, M.; Tao, Z.; Rao, Z.; Wu, C.; Yin, S.; Jiang, X.; Chen, G.; Wang, Z.; Huang, D.; Zhu, M.; Liu, L.; Huo, W.; Yang, H.; Guo, H.; Chen, G.; Li, F.; Zheng, C.; Huang, D.; Rahfeld, P.; Kizhakkedathu, J.N.; Withers, S.G.; Lu, X.; Zhang, K.; Lin, T.; Song, T. Enzyme-converted O kidneys allow ABO-incompatible transplantation without hyperacute rejection in a human decedent model. Nat Biomed Eng. Online ahead of print. 2025.
- Rahfeld, P.; Sim, L.; Moon, H.; Constantinescu, I.; Morgan-Lang, C.; Hallam, S.J.; Kizhakkedathu, J.N.; Withers, S.G. An enzymatic pathway in the human gut microbiome that converts A to universal O type blood. Nat Microbiol. 2019, 4, 1475–1485. [Google Scholar] [CrossRef]
- MacMillan, S.; Hosgood, S.A.; Walker-Panse, L.; Rahfeld, P.; Macdonald, S.S.; Kizhakkedathu, J.N.; Withers, S.G.; Nicholson, M.L. Enzymatic conversion of human blood group A kidneys to universal blood group O. Nat Commun. 2024, 15, 2795. [Google Scholar] [CrossRef] [PubMed]
- MacMillan, S.; Hosgood, S.A.; Nicholson, M.L. Enzymatic blood group conversion of human kidneys during ex vivo normothermic machine perfusion. Br J Surg. 2023, 110, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.; Ma, M.; Jiang, X.; Rao, Z.; Huang, D.; Zhang, H.; Yin, S.; Bao, R.; Zhang, H.; Wang, Z.; Gao, H.; Gong, F.; Lin, T.; Zhang, K.; Song, T. Enzymatic conversion of blood group B kidney prevents hyperacute antibody-mediated injuries in ABO-incompatible transplantation. Nat Commun. 2025, 16, 1506. [Google Scholar] [CrossRef]

| Clinical Outcomes | AB0i-LDKT | AB0c-LDKT | p |
|---|---|---|---|
| Number | 41 | 132 | |
| Preoperative Hospital Stay (Day) | 22.27±7.10 | 22.20±9.30 | 0.955 |
| In-patient Care Spending | 127.13±41.21 | 91.34±38.74 | <0.001 |
| Delayed Graft Function | 2.44% | 3.03% | 1.000 |
| Pulmonary Infection | 34.15% | 20.45% | 0.092 |
| Urinary Tract Infection | 4.88% | 3.79% | 0.670 |
| Surgical Complications | 14.63% | 8.33% | 0.240 |
| Graft Survival 1st year | 95.12% | 94.70% | 1.000 |
| Graft Survival 3st year | 92.68% | 92.42% | 1.000 |
| Patient Survival 1st year | 97.56% | 96.21% | 1.000 |
| Patient Survival 3st year | 97.56% | 93.94% | 0.692 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




