Submitted:
15 April 2026
Posted:
16 April 2026
You are already at the latest version
Abstract
Background: Overlapping endemic infections often present with non-specific systemic features, which could initially lead to delayed recognition and inappropriate treatment. Strongyloides stercoralis and Coccidioides spp. are rarely encountered together, yet both may cause pulmonary disease, constitutional symptoms, and eosinophilia, complicating diagnosis. Corticosteroid exposure in particular can unmask severe strongyloidiasis, highlighting the importance of early detection. Case Presentation: We present the case of a 30-year-old man from the Dominican Republic with recent travel to Brazil and Mexico, who presented with a 3-week history of fever, cough, myalgias, rash, and 13-pound weight loss. Initial treatment for presumed asthma exacerbation and bacterial pneumonia with corticosteroids and multiple antibiotics failed to relieve symptoms. Laboratory evaluation revealed marked eosinophilia (absolute eosinophil count 3,400/µL) and elevated inflammatory markers. Chest CT demonstrated diffuse bilateral tree-in-bud and micronodular opacities. Bronchoalveolar lavage contained 44% eosinophils. Serologic testing was positive for Strongyloides IgG, Coccidioides IgM/IgG, and β-D-glucan. The patient improved with ivermectin and fluconazole but experienced a relapse of coccidioidomycosis after antifungal discontinuation, requiring reinitiation of long-term azole therapy. Discussion: Coinfection with Strongyloides stercoralis and Coccidioides spp. poses a difficult diagnosis due to overlapping respiratory and systemic manifestations that could mimic common bacterial, fungal or allergic processes. Corticosteroid exposure can precipitate Strongyloides hyperinfection while promoting fungal proliferation, worsening disease severity. Recognition of eosinophilia in patients with a compatible travel history should prompt evaluation for parasitic and fungal etiologies. This case emphasizes the need for early serologic testing and targeted therapy while providing close follow-up to prevent relapses and complications in overlapping endemic infections. Conclusion: This case shows the difficulty of diagnosing overlapping infections like Strongyloides stercoralis and Coccidioides, which can easily be mistaken for bacterial pneumonia. It highlights the risk of giving corticosteroids before ruling out parasitic diseases and stresses the value of screening those at risk. The patient’s relapse after stopping treatment reflects the chronic nature of coccidioidomycosis and the need for close follow-up. Clinicians should keep an open, exposure-based approach when evaluating unexplained pulmonary symptoms, especially in people from endemic areas. This case underscores the importance of broad differentials, timely diagnosis, and long-term monitoring in patients with complex overlapping infections.
Keywords:
Introduction
Case Presentation
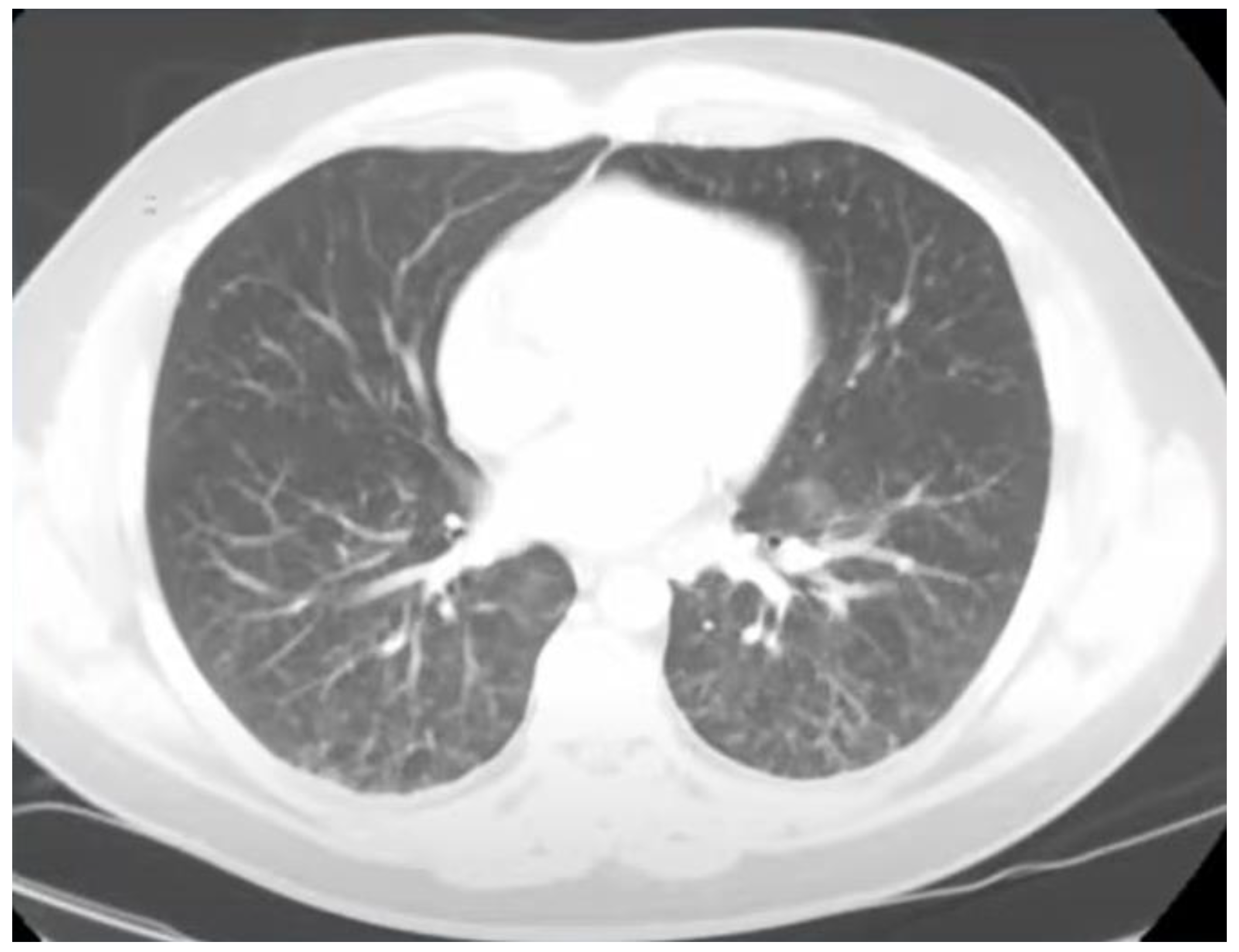
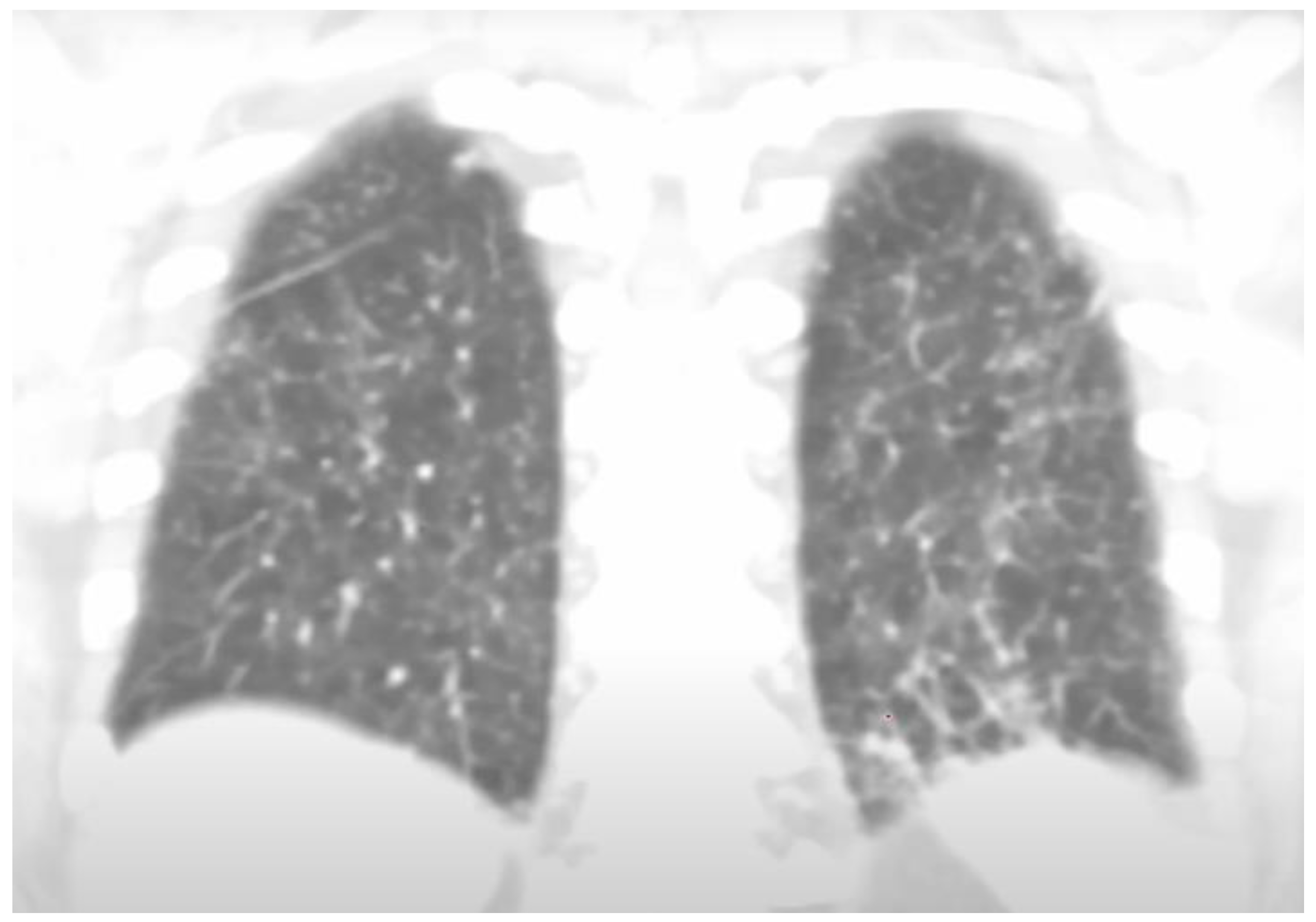
Discussion
Conclusion
Informed Consent Statement
References
- Domínguez VR, Pérez-López C, Sánchez CV, Contreras CU, Guerrero AI, Abenza Abildúa MJ. Strongyloides hyperinfection syndrome due to corticosteroid therapy after resection of meningioma: illustrative case. Journal of neurosurgery Case lessons [Internet]. 2022 Nov;4(2):CASE21667. Available from: https://pubmed.ncbi.nlm.nih.gov/35855010/.
- Fardet L, Généreau T, Poirot JL, Guidet B, Kettaneh A, Cabane J. Severe strongyloidiasis in corticosteroid-treated patients: Case series and literature review. Journal of Infection. 2007 Jan;54(1):18–27.
- Herrick KR, Trondle ME, Febles TT. Coccidioidomycosis (Valley Fever) in Primary Care. American Family Physician [Internet]. 2020 Feb 15;101(4):221–8. Available from: https://pubmed.ncbi.nlm.nih.gov/32053327/.
- Malo J, Luraschi-Monjagatta C, Wolk DM, Thompson R, Hage CA, Knox KS. Update on the Diagnosis of Pulmonary Coccidioidomycosis. Annals of the American Thoracic Society. 2014 Feb;11(2):243–53.
- Marcos LA, Terashima A, DuPont HL, Gotuzzo E. Strongyloides hyperinfection syndrome: an emerging global infectious disease. Transactions of the Royal Society of Tropical Medicine and Hygiene. 2008 Apr;102(4):314–8.
- NUTMAN TB. Human infection with Strongyloides stercoralis and other related Strongyloides species. Parasitology. 2016 May 16;144(3):263–73.
- Williams SL, Chiller T. Update on the Epidemiology, Diagnosis, and Treatment of Coccidioidomycosis. Journal of Fungi. 2022 Jun 25;8(7):666.
- Nutman TB. Human infection with Strongyloides stercoralis and other related Strongyloides species. Parasitology. 2017;144(3):263-273. [CrossRef]
- Keiser PB, Nutman TB. Strongyloides stercoralis in the Immunocompromised Population. Clin Microbiol Rev. 2004;17(1):208-217. [CrossRef]
- Ramanathan R, Nutman T. Strongyloides stercoralis infection in the immunocompromised host. Curr Infect Dis Rep. 2008;10(2):105-110. [CrossRef]
- John N. Galgiani, Neil M. Ampel, Janis E. Blair, Antonino Catanzaro, Francesca Geertsma, Susan E. Hoover, Royce H. Johnson, Shimon Kusne, Jeffrey Lisse, Joel D. MacDonald, Shari L. Meyerson, Patricia B. Raksin, John Siever, David A. Stevens, Rebecca Sunenshine, Nicholas Theodore, 2016 Infectious Diseases Society of America (IDSA) Clinical Practice Guideline for the Treatment of Coccidioidomycosis, Clinical Infectious Diseases, Volume 63, Issue 6, 15 September 2016, Pages e112–e146,. [CrossRef]
- Stockamp NW, Thompson GR 3rd. Coccidioidomycosis. Infect Dis Clin North Am. 2016;30(1):229-246. [CrossRef]
- Maizels RM, McSorley HJ. Regulation of the host immune system by helminth parasites. J Allergy Clin Immunol. 2016;138(3):666-675. [CrossRef]
- Theel ES, Doern CD. β-D-glucan testing is important for diagnosis of invasive fungal infections. J Clin Microbiol. 2013;51(11):3478-3483. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



