Submitted:
14 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Patient Population
2.2. Development of the Scoring System
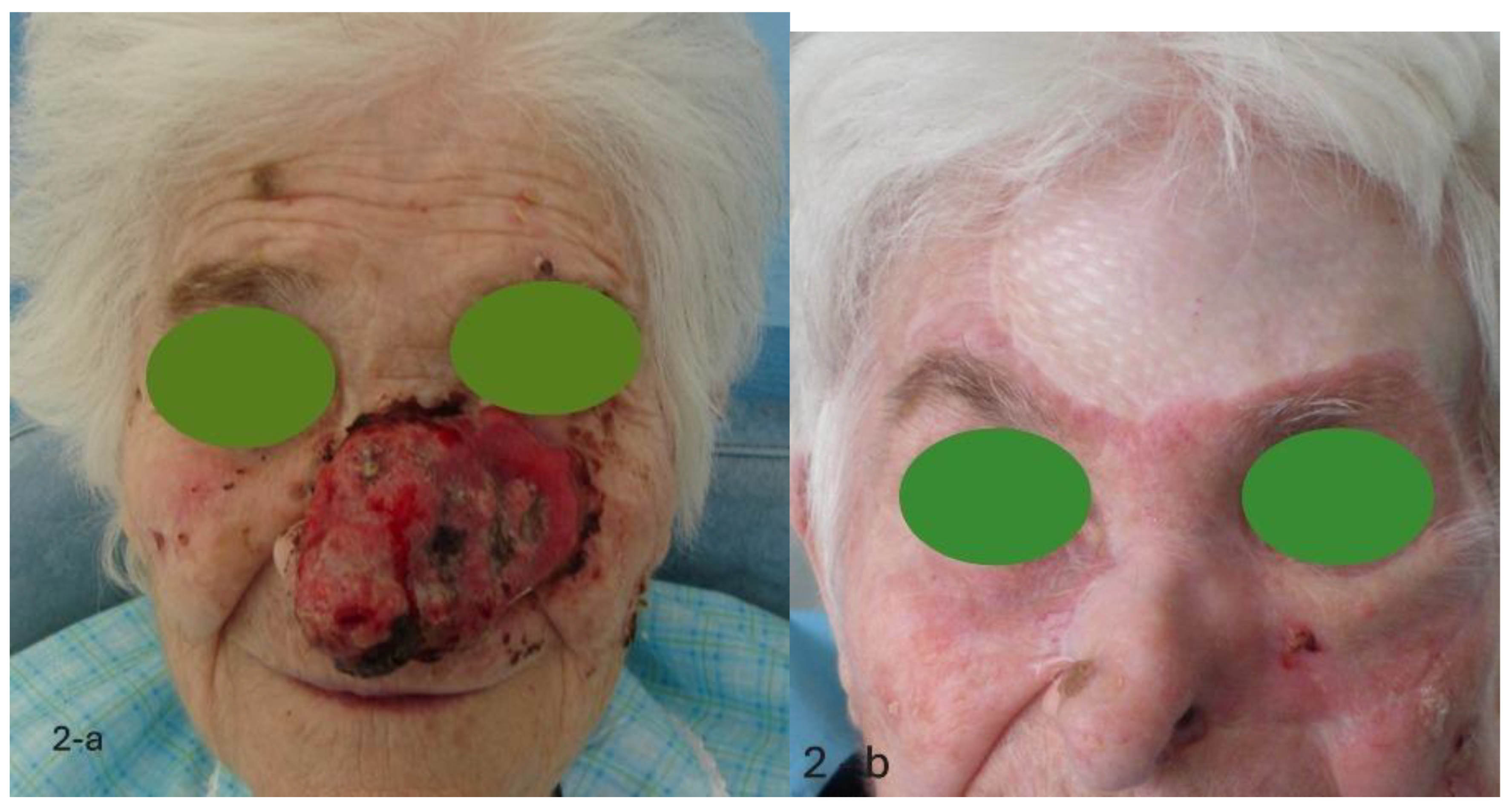
- Anatomical site of the neoplasm: a tumor can be considered locally advanced in relation to its location. Localization on the face, genitals, perineum and extremities must be considered high risk. At medium risk is localization on the scalp and the remaining parts of the upper or lower limbs. Low risk, locations in anatomical regions that are easier to manage such as the trunk.
- TNM classification in relation to size and diffusion in depth. High risk. T4. Medium risk: T2 – T3. Low risk: Tis, T1.
- Histological variant: The basal-squamous variant must be considered high risk. At medium risk, the micro-nodular or infiltrative variant, or morpheiform with neural invasion. At low risk, the variants, nodular, cystic nodule, adenoid, keratotic, with adnexal differentiation, superficial, pleomorphic, clear cell or signet ring clear cell.
- The presence of a surgical center authorized to treat large skin tumors: to be considered high risk if the patient lives in a location without a surgical department. At medium risk, presence of a peripheral hospital with a surgical department with suitable reconstructive experience. Low Risk, in case of comprehensive cancer center or academic cancer center with on-site presence of a reconstructive unit with recognized experience.
- Patient frailty: calculated according to the Hospital Frailty Risk, high risk, medium risk, low risk conditions.
- Possibility of instrumental evaluation and staging: at high risk if this evaluation is impossible, at medium risk if it is limited due to the absence of advanced radio diagnostics, or absence of nuclear medicine, or due to difficulty in reaching the dedicated site, at low risk if the staging is possible.
- Possibility of carrying out radiotherapy: high risk in case of negative opinion from the patient; at medium risk, even with a positive opinion from the patient, RT cannot be performed, because it has already been performed, or cannot be performed due to the shape and size of the field to be treated, radiodermatitis, objective difficulty in surgical repair of the radio-treated area; low risk, if RT is possible.
- Evaluation of the patient's place of residence: high risk, in case of negative evaluation or impossible patient compliance; medium risk, difficult compliance; low risk, positive compliance.
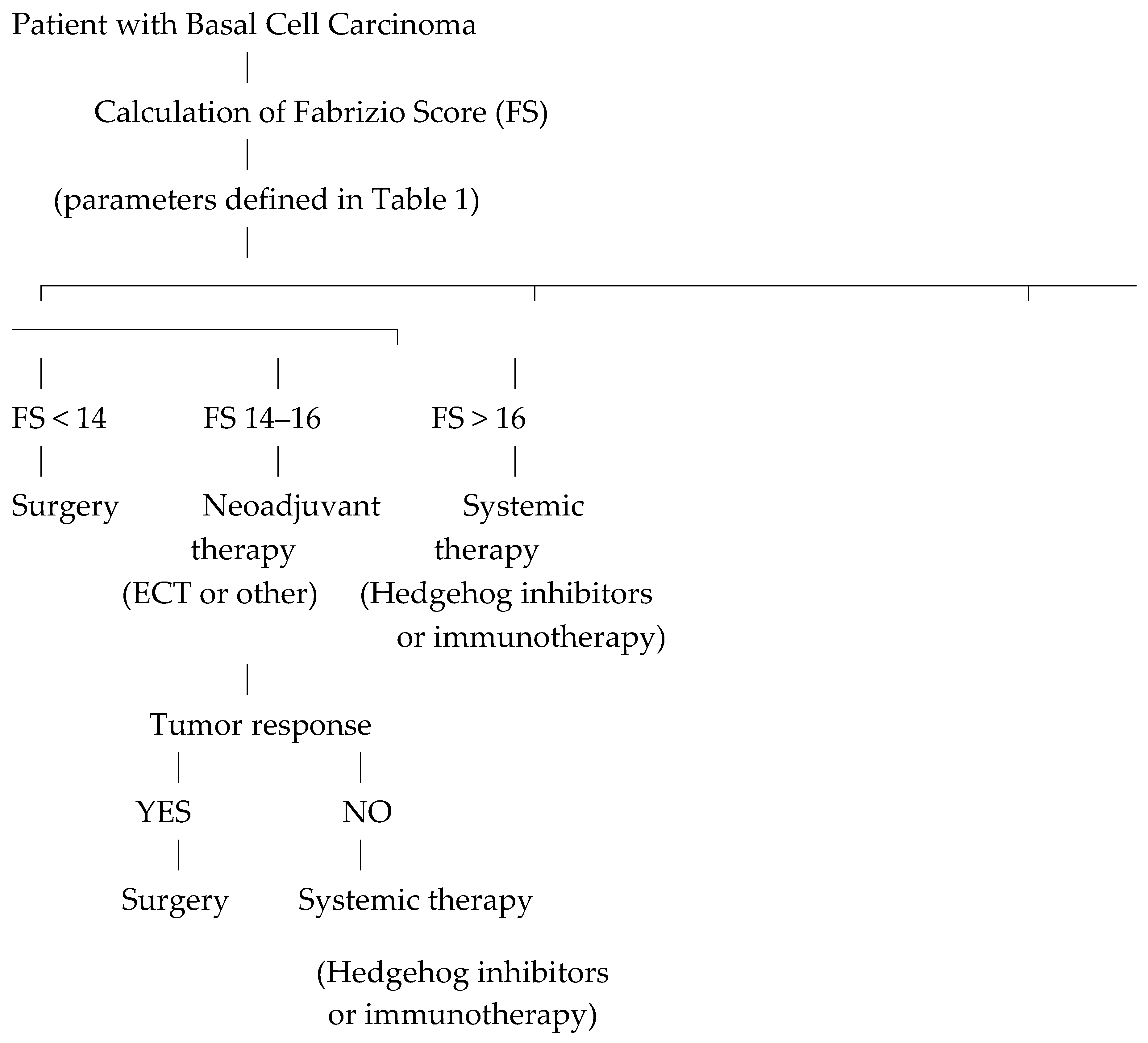
- Evaluation of the possibility of performing local or systemic neoadjuvant treatment: high risk, negative evaluation; medium risk, difficult but possible assessment; low risk, positive rating. Each of these parameters will be given a score of 3 for the high-risk rating, a score of 2 for the medium risk rating and a score of 1 for the low-risk rating. By adding the various scores, if the result is lower than 14, the neoplasm cannot be objectively indicated as locally advanced, and the patient must be sent for surgical treatment. For a score between 14 and 16, the patient is sent to neoadjuvant treatment; after which a new evaluation is carried out. If it determines a score lower than 14, surgery is performed, otherwise, the patient is sent for medical treatment with a target therapy (TKI, Tyrosine Kinase Inhibitors). Only for a score above 16, we must speak of a locally advanced neoplastic lesion, and the patient is started on treatment with target therapy (TKI, Tyrosine Kinase Inhibitors).
2.3. Scoring System
- Anatomical location of the tumor
- tumor stage (TNM classification)
- Histological Subtype
- availability of specialized surgical centers
- patient frailty (Hospital Frailty Risk Score)
- availability of staging procedures
- feasibility of radiotherapy
- geographic accessibility to treatment centers
- feasibility of electrochemotherapy or other neoadjuvant treatments
- 1 point – low risk
- 2 points – intermediate risk
- 3 points – high risk
| Score | Interpretation | Recommended management |
| <14 | Not locally advanced | Surgical treatment |
| 14–16 | Borderline | Neoadjuvant therapy |
| >16 | Locally advanced | Systemic therapy |
2.4. Interpretation of the Score
3. Results
- 1,627 cases (87.9%) had FS <14 and were treated surgically
- 218 cases (11.8%) had FS between 14 and 16 and received neoadjuvant therapy
- 6 cases (0.3%) had FS >16 and were directly referred for systemic targeted therapy
4. Discussion
5. Conclusion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rogers, H.W.; Weinstock, M.A.; Harris, A.R.; et al. Incidence estimate of nonmelanoma skin cancer in the United States, 2006. Arch Dermatol. 2010, 146, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Peris, K.; Fargnoli, M.C.; Kaufmann, R.; et al. European consensus-based interdisciplinary guideline for diagnosis and treatment of basal cell carcinoma – update 2023. Eur J Cancer 2023, 192, 113254. [Google Scholar] [CrossRef]
- Alam, M.; Goldberg, L.H.; Silapunt, S.; et al. Delayed treatment and continued growth of non-melanoma skin cancer. J Am Acad Dermatol. 2011, 64, 839–848. [Google Scholar] [CrossRef] [PubMed]
- Chren, M.M.; Linos, E.; Torres, J.S.; et al. Tumor recurrences after treatment of basal cell carcinoma. J Invest Dermatol. 2013, 133, 1188–1196. [Google Scholar] [CrossRef]
- Mohan, S.V.; Chang, A.L. Advanced basal cell carcinoma: epidemiology and therapeutic innovations. Curr Dermatol Rep. 2014, 3, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Goppner, D.; Leverkus, M. Basal cell carcinoma: pathogenesis and targeted therapy. J Skin Cancer 2011, 2011, 650258. [Google Scholar] [CrossRef] [PubMed]
- Basset-Seguin, N.; Hauschild, A.; Kunstfeld, R.; et al. Vismodegib in patients with advanced basal cell carcinoma (STEVIE): a pre-planned interim analysis. Eur J Cancer 2017, 86, 334–348. [Google Scholar] [CrossRef]
- Weinstock, M.A.; Still, J.M. Treatment options for severe basal cell carcinoma. Semin Cutan Med Surg. 2011, 30, S10–S13. [Google Scholar] [CrossRef]
- Fecher, L.A. Systemic therapy for metastatic basal cell carcinoma. Curr Treat Options Oncol. 2013, 14, 237–248. [Google Scholar] [CrossRef]
- Ganti, A.K.; Kessinger, A. Systemic therapy for disseminated basal cell carcinoma. Cancer Treat Rev. 2011, 37, 440–443. [Google Scholar] [CrossRef] [PubMed]
- Gorlin, R.J.; Goltz, R.W. Multiple nevoid basal cell epithelioma syndrome. N Engl J Med. 1960, 262, 908–912. [Google Scholar] [CrossRef]
- Stratigos, A.J.; Sekulic, A.; Peris, K.; et al. Cemiplimab in locally advanced basal cell carcinoma after hedgehog inhibitor therapy. Lancet Oncol. 2021, 22, 848–857. [Google Scholar] [CrossRef]
- Peris, K.; Licitra, L.; Ascierto, P.A.; et al. Identifying locally advanced basal cell carcinoma eligible for treatment with vismodegib: an expert panel consensus. Future Oncol. 2015, 11, 703–712. [Google Scholar] [CrossRef]
- Ally, M.S.; Aasi, S.; Wysong, A.; et al. An investigator-initiated open-label clinical trial of vismodegib as neoadjuvant therapy for high-risk basal cell carcinoma. J Am Acad Dermatol. 2014, 71, 904–911. [Google Scholar] [CrossRef] [PubMed]
- Maciel, P.C.; Veiga-Filho, J.; Carvalho, M.P.; et al. Quality of life after surgical treatment of skin carcinomas. Ann Bras Dermatol. 2014, 89, 594–598. [Google Scholar] [CrossRef]
- Bhutani, T.; Abrouk, M.; Sima, C.S.; et al. Risk of squamous cell carcinoma after vismodegib therapy for basal cell carcinoma. J Am Acad Dermatol. 2017, 77, 713–718. [Google Scholar] [CrossRef]
- Sekulic, A.; Mangold, A.R.; Northfelt, D.W. Targeting the Hedgehog signaling pathway in advanced basal cell carcinoma. Curr Opin Oncol. 2013, 25, 218–223. [Google Scholar] [CrossRef]
- Sekulic, A.; Migden, M.R.; Basset-Seguin, N.; et al. Long-term safety and efficacy of vismodegib in advanced basal cell carcinoma (ERIVANCE BCC). BMC Cancer 2017, 17, 332. [Google Scholar] [CrossRef] [PubMed]
- Sekulic, A.; Migden, M.R.; Oro, A.E.; et al. Efficacy and safety of vismodegib in advanced basal-cell carcinoma. N Engl J Med. 2012, 366, 2171–2179. [Google Scholar] [CrossRef] [PubMed]
- Sekulic, A.; Yoo, S.; Kudchadkar, R.; et al. Real-world treatment patterns in advanced basal cell carcinoma: RegiSONIC registry. PLoS One 2022, 17, e0262151. [Google Scholar] [CrossRef] [PubMed]
- Migden, M.R.; Guminski, A.; Gutzmer, R.; et al. Treatment with two different doses of sonidegib in locally advanced or metastatic basal cell carcinoma (BOLT): a multicentre, randomised, double-blind phase 2 trial. Lancet Oncol. 2015, 16, 716–728. [Google Scholar] [CrossRef] [PubMed]
- Dummer, R.; Guminski, A.; Gutzmer, R.; et al. Long-term efficacy and safety of sonidegib in advanced basal cell carcinoma: 42-month analysis of the BOLT study. Br J Dermatol. 2020, 182, 1369–1378. [Google Scholar] [CrossRef] [PubMed]
- Lewin, J.M.; Carucci, J.A. Advances in the management of basal cell carcinoma. F1000Prime Rep. 2015, 7, 53. [Google Scholar] [CrossRef]
- Mohan, S.V.; Chang, J.; Li, S.; et al. Increased risk of cutaneous squamous cell carcinoma after vismodegib therapy. JAMA Dermatol. 2016, 152, 527–532. [Google Scholar] [CrossRef] [PubMed]
- Bertozzi, N.; Simonacci, F.; Grieco, M.P.; Grignaffini, E.; Raposio, E. Single center evidence for the treatment of basal cell carcinoma of the head and neck. Acta Biomed. 2019, 90(1), 77–82. [Google Scholar]
- Simonacci, F.; Bertozzi, N.; Grieco, M.P.; Grignaffini, E.; Raposio, E. Surgical therapy of cutaneous squamous cell carcinoma: our experience. Acta Biomed. 2018, 89(2), 242–248. [Google Scholar]
- Chen, C.; Chen, Z.J. Reconstruction of the concha of the ear using a postauricular island flap. Plast Reconstr Surg 1990, 86, 569–72. [Google Scholar] [CrossRef]
- Talmi, Y.P.; Horowitz, Z.; Bedrin, L.; et al. Auricular reconstruction with a postauricular myocutaneous island flap: flip–flop flap. Plast Reconstr Surg 1996, 98, 1191–9. [Google Scholar] [CrossRef] [PubMed]
- Cordova, A.; D’Arpa, S.; Pirrello, R.; et al. Retroauricular skin: a flaps bank for ear reconstruction. J Plast Reconstr Aesthet Surg 2008, 61 (suppl 1), S44–51. [Google Scholar] [CrossRef] [PubMed]
- Stiller, M.B.; Gerressen, M.; Modabber, A.; et al. Anteriorly pedicled retroauricular flap for repair of auricular defects. Aesthetic Plast Surg 2012, 36, 623–7. [Google Scholar] [CrossRef] [PubMed]
| Outcome Measure | Locally Advanced BCC (laBCC) N= 63 | Metastatic BCC (mBCC) N=33 |
|---|---|---|
| Objective Response Rate (ORR) (95%CI) | 60.3% (47.2-71.7) | 48.5% (30.8-66.2) |
| Complete Response (CR) | 31.7% | 0% |
| Partial Response (PR) | 28.6% | 48.5% |
| Stable Disease (SD) | 14.3% | 45.5% |
| Median Duration of Response (DOR) (95%CI) | 26.2 months (9.0-37.6) | 14.8 months (5.6-17-0) |
| Median Progression-Free Survival (PFS) (95%CI) | 12.9 months (10.2-28.0) | 9.3 months(7.4-16.6) |
| Median Overall Survival (OS) (95%CI) | Not Reached (NE) | 33.4 months (18.1-NE) |
| Outcome Measure | Locally Advanced BCC (laBCC) N=1132 | Metastatic BCC (mBCC)N=83 |
|---|---|---|
| Objective Response Rate (ORR) (95%CI) | 68.5% (65.66-71.29) | 36.9% (26.63-48.13) |
| Complete Response (CR) | 33.4% | 2.4% |
| Partial Response (PR) | 35.1% | 34.5% |
| Stable Disease (SD) | 21.6% | 51.2% |
| Disease Control Rate (DCR) | 90.1% | 88.1% |
| Median duration of response (DOR) (95%CI) | 23 months (20.4-26.7) | 13.9 months (9.2-NE) |
| Median Progression-Free Survival (PFS) (95%CI) | 23.2 months (21.4-26) | 13.1 months (12.0-17.7) |
| laBCC | mBCC | |||
|---|---|---|---|---|
| 200 mg (n = 66) | 800 mg (n = 128) | 200 mg (n = 13) | 800 mg (n = 23) | |
| Objective Response Rate (ORR) (95%CI) | 56% (43–68) | 46·1% (37·2–55·1) | 8 (0·2–36) | 17% (5–39) |
| Complete Response (CR) | 5% (0·9–13) | 1·6 %(0·2–5·5) | 0% (0–25) | 0% (0–15) |
| Disease Control Rate (DCR) | 91% | 82·0% | 92% | 91% |
| Median duration of response (DOR) (95%CI) | 26·1months (NE) | 23·3months (12·2–29·6) | 24·0 (NE) | NE (NE) |
| Median Progression-Free Survival (PFS) ( 95%CI) | 22·1months (NE) | 24·9months (19·2–33·4) | 13·1months (5·6–33·1) | 11·1months (7·3–16·6) |
| TTR, time to tumor response. (95% CI) | 4·0months (3·8–5·6) | 3·8months (3·7–5·5) | 9·2months (NE) | 1·0months (1·0–2·1) |
| Parameter | Low Risk (1 point) | Medium Risk (2 points) | High Risk (3 points) |
|---|---|---|---|
| Anatomical location of the neoplasm | Easily manageable anatomical areas (trunk) | Anatomical regions with intermediate management difficulty (scalp, upper and lower limbs) | Critical anatomical regions (face, genital area, perineum, extremities) |
| Tumor stage (TNM / EADO classification) | Stage I–IIA (Tis, T1) | Stage IIB (T2–T3) | Stage IIIA–IV (T4) |
| Histological variant | Low-risk variants (nodular, nodulocystic, adenoid, keratotic, superficial, pleomorphic, clear cell, clear cell signet ring, adnexal differentiation) | Variants with high risk of recurrence (micronodular, infiltrative, morphea form, morphea form with perineural invasion) | Basosquamous variant |
| Availability of a surgical center authorized to treat extensive skin neoplasms | University Hospital Unit / Regional Oncology Center / Unit with recognized experience in demolitive and reconstructive surgery | Peripheral hospital with limited reconstructive experience or surgical department not specialized in reconstructive treatment | Absence of a general surgical department |
| Patient frailty (Hospital Frailty Risk Score) | Low frailty conditions | Moderate frailty conditions | Severe frailty conditions |
| Instrumental assessment and staging | Complete staging feasible | Limited staging availability (absence of advanced radiology or nuclear medicine, difficulty accessing referral centers) | Staging not feasible |
| Possibility of performing radiotherapy | Radiotherapy feasible | Radiotherapy not feasible (previous radiotherapy, technical limitations, large treatment field, radiodermatitis, difficulty in reconstruction after irradiation) | Patient refusal |
| Geographic accessibility to treatment center | Positive evaluation | Difficult evaluation | Negative evaluation |
| Feasibility of electrochemotherapy (ECT) or other neoadjuvant treatments | Positive evaluation | Difficult evaluation | Negative evaluation |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




