Figure 1.
Baseline properties of SWRs and associated neuronal activity in dorsal and ventral hippocampus. (A) Representative extracellular field recordings from the CA1 stratum pyramidale illustrating spontaneous SWRs in dorsal (top) and ventral (bottom) hippocampal slices under control conditions. (B) Example of an individual SWR event. Top trace: raw signal (SWR). Middle traces: low-pass filtered signal (35 Hz) showing the sharp wave component and band-pass filtered signal (90–250 Hz) showing ripple oscillations. Bottom trace: band-pass filtered signal ()0.4—1.5 kHz) revealing multiunit activity (MUA). Calibration bars: 50 μV, 10 ms. (C) Representative spectral and temporal characteristics of SWRs. Top: ripple power spectrum. Bottom: peri-event time histogram of MUA aligned to the SWR peak. (D) Quantification of SWR and MUA parameters in dorsal (blue) and ventral (red) hippocampus, including inter-event interval (IEI), probability of clustered events, sharp wave amplitude, ripple frequency, ripple power, baseline firing rate (MUA-Base), SWR-associated firing rate (MUA-SWR), and MUA delay relative to SWR peak (MUA-Delay). Significant differences between dorsal and ventral hippocampus are indicated (independent t-test, *p < 0.05, **p < 0.01).
Figure 1.
Baseline properties of SWRs and associated neuronal activity in dorsal and ventral hippocampus. (A) Representative extracellular field recordings from the CA1 stratum pyramidale illustrating spontaneous SWRs in dorsal (top) and ventral (bottom) hippocampal slices under control conditions. (B) Example of an individual SWR event. Top trace: raw signal (SWR). Middle traces: low-pass filtered signal (35 Hz) showing the sharp wave component and band-pass filtered signal (90–250 Hz) showing ripple oscillations. Bottom trace: band-pass filtered signal ()0.4—1.5 kHz) revealing multiunit activity (MUA). Calibration bars: 50 μV, 10 ms. (C) Representative spectral and temporal characteristics of SWRs. Top: ripple power spectrum. Bottom: peri-event time histogram of MUA aligned to the SWR peak. (D) Quantification of SWR and MUA parameters in dorsal (blue) and ventral (red) hippocampus, including inter-event interval (IEI), probability of clustered events, sharp wave amplitude, ripple frequency, ripple power, baseline firing rate (MUA-Base), SWR-associated firing rate (MUA-SWR), and MUA delay relative to SWR peak (MUA-Delay). Significant differences between dorsal and ventral hippocampus are indicated (independent t-test, *p < 0.05, **p < 0.01).
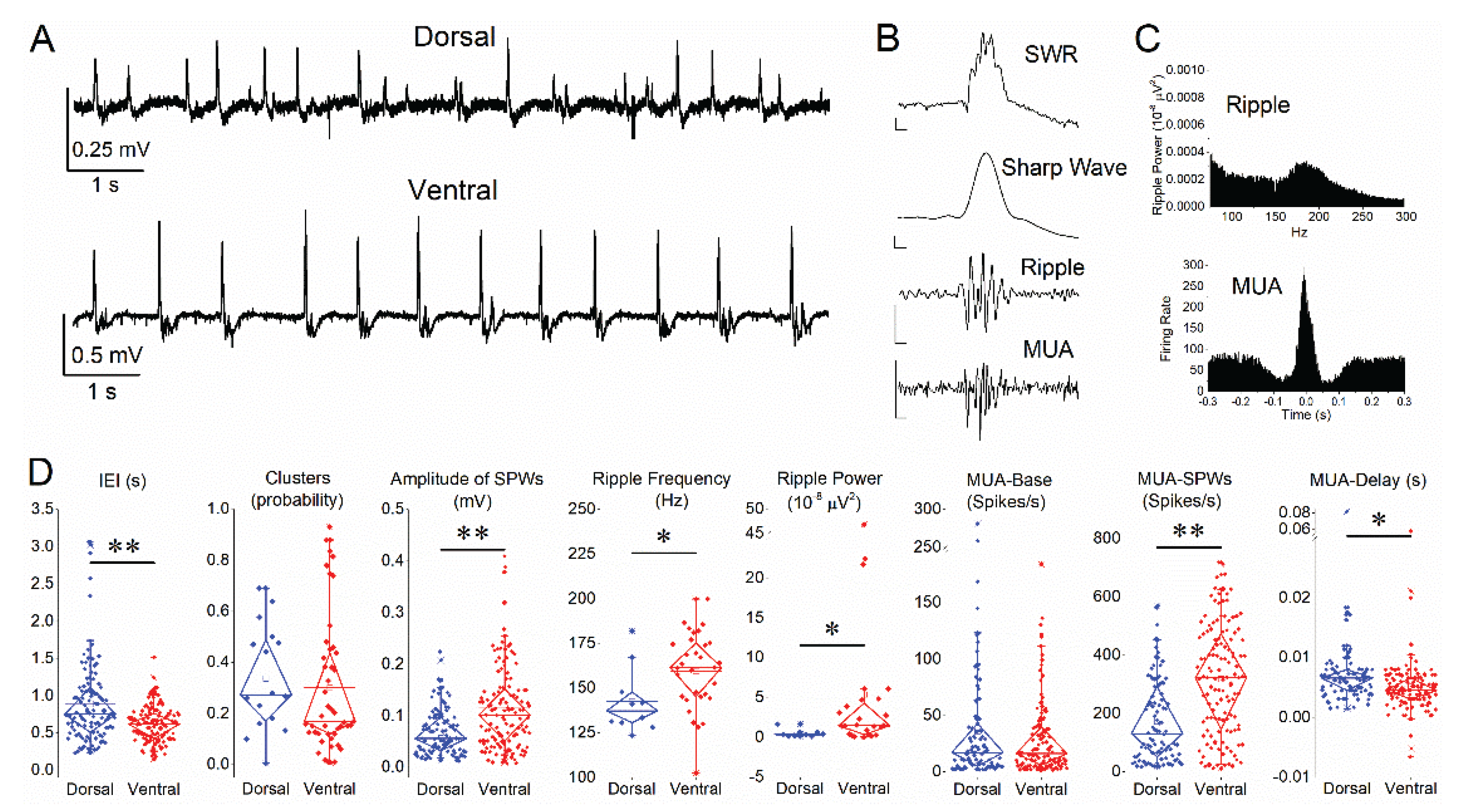
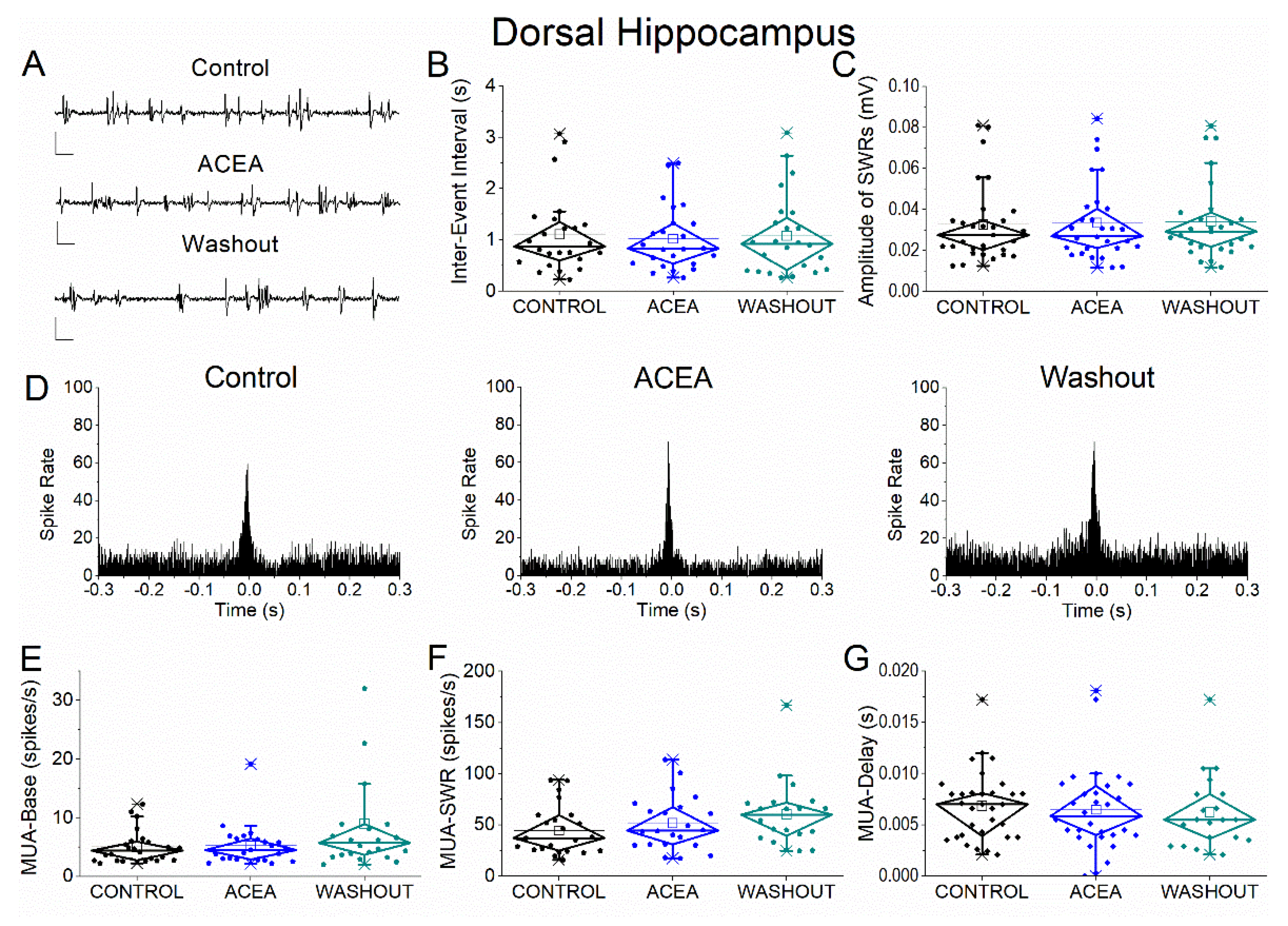
Figure 2.
Effects of the CB1 receptor agonist ACEA on SWRs and MUA in dorsal hippocampus. (A) Representative extracellular recordings from CA1 stratum pyramidale under control conditions (top), during ACEA application (middle), and after washout (bottom). Calibration bars: 50 μV, 0.5 s. (B–C) Quantification of SWR properties across conditions. (B) Inter-event interval (IEI). (C) Sharp wave amplitude. No significant effects of ACEA were observed. (D) Peri-event time histograms of MUA aligned to SWR peak under control (left), ACEA (middle), and washout (right) conditions. (E–G) Quantification of neuronal activity parameters. (E) Baseline firing rate (MUA-Base). (F) SWR-associated firing rate (MUA-SWR). (G) MUA delay relative to SWR peak (MUA-Delay). No significant effects of ACEA were detected for any parameter. Statistical analysis was performed using LMM (Control vs ACEA).
Figure 2.
Effects of the CB1 receptor agonist ACEA on SWRs and MUA in dorsal hippocampus. (A) Representative extracellular recordings from CA1 stratum pyramidale under control conditions (top), during ACEA application (middle), and after washout (bottom). Calibration bars: 50 μV, 0.5 s. (B–C) Quantification of SWR properties across conditions. (B) Inter-event interval (IEI). (C) Sharp wave amplitude. No significant effects of ACEA were observed. (D) Peri-event time histograms of MUA aligned to SWR peak under control (left), ACEA (middle), and washout (right) conditions. (E–G) Quantification of neuronal activity parameters. (E) Baseline firing rate (MUA-Base). (F) SWR-associated firing rate (MUA-SWR). (G) MUA delay relative to SWR peak (MUA-Delay). No significant effects of ACEA were detected for any parameter. Statistical analysis was performed using LMM (Control vs ACEA).
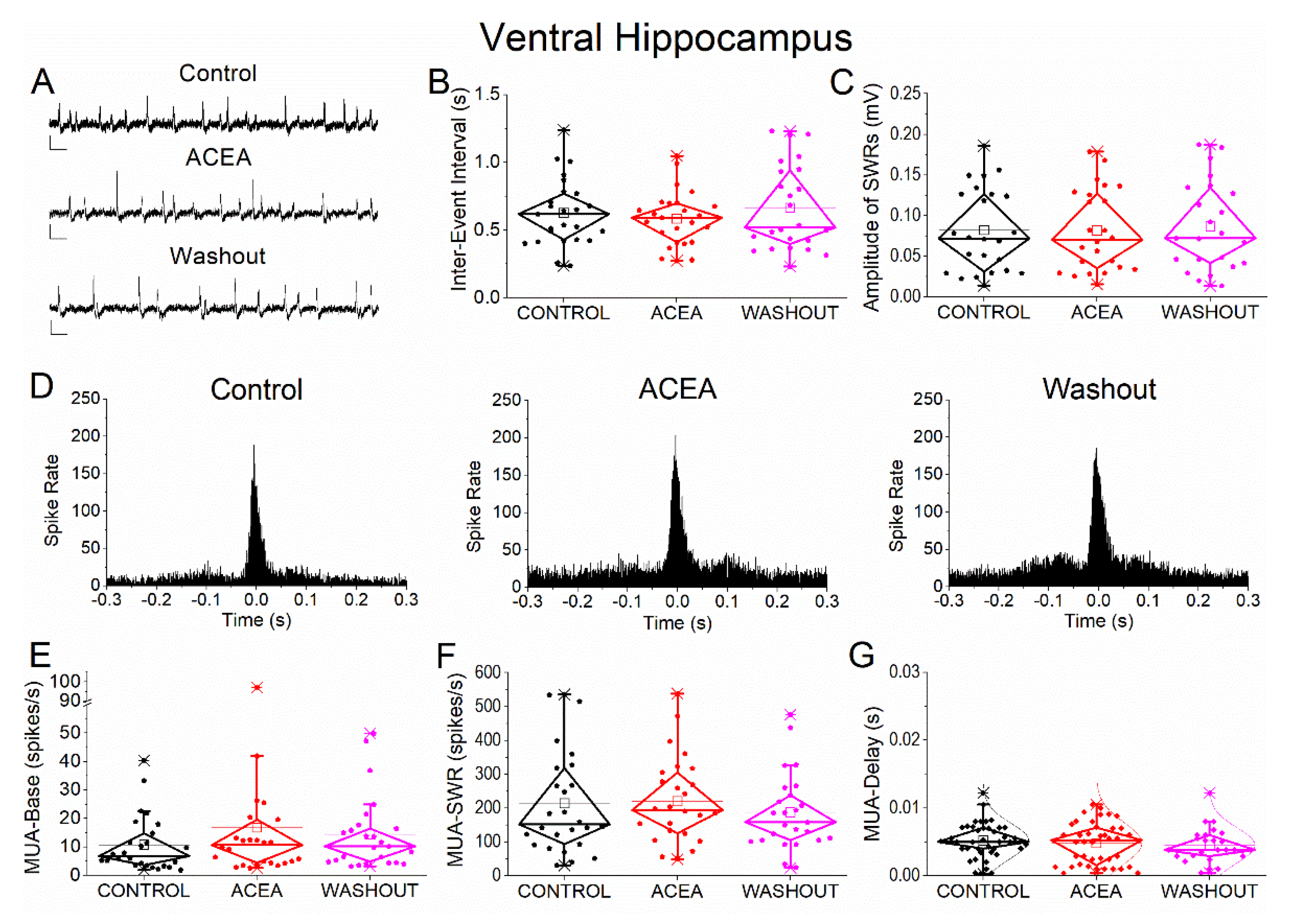
Figure 3.
Effects of the CB1 receptor agonist ACEA on SWRs and MUA in ventral hippocampus. (A) Representative extracellular recordings from CA1 stratum pyramidale under control conditions (top), during ACEA application (middle), and after washout (bottom). Calibration bars: 50 μV, 0.5 s. (B–C) Quantification of SWR properties across conditions. (B) Inter-event interval (IEI). (C) Sharp wave amplitude. No significant effects of ACEA were observed. (D) Peri-event time histograms of multiunit activity (MUA) aligned to SWR peak under control (left), ACEA (middle), and washout (right) conditions. (E–G) Quantification of MUA. (E) Baseline firing rate (MUA-Base). (F) SWR-associated firing rate (MUA-SWR). (G) MUA delay relative to SWR peak (MUA-Delay). No significant effects of ACEA were detected for any parameter. Statistical analysis was performed using LMM (Control vs ACEA). Washout data are shown for completeness but were not included in statistical comparisons.
Figure 3.
Effects of the CB1 receptor agonist ACEA on SWRs and MUA in ventral hippocampus. (A) Representative extracellular recordings from CA1 stratum pyramidale under control conditions (top), during ACEA application (middle), and after washout (bottom). Calibration bars: 50 μV, 0.5 s. (B–C) Quantification of SWR properties across conditions. (B) Inter-event interval (IEI). (C) Sharp wave amplitude. No significant effects of ACEA were observed. (D) Peri-event time histograms of multiunit activity (MUA) aligned to SWR peak under control (left), ACEA (middle), and washout (right) conditions. (E–G) Quantification of MUA. (E) Baseline firing rate (MUA-Base). (F) SWR-associated firing rate (MUA-SWR). (G) MUA delay relative to SWR peak (MUA-Delay). No significant effects of ACEA were detected for any parameter. Statistical analysis was performed using LMM (Control vs ACEA). Washout data are shown for completeness but were not included in statistical comparisons.
Figure 4.
Effects of the cannabinoid receptor agonist WIN55,212-2 on SWRs and MUA in dorsal hippocampus. (A) Representative extracellular recordings from CA1 stratum pyramidale under control conditions (top), during WIN55,212-2 application (middle), and after washout (bottom). Calibration bars: 50 μV, 0.5 s. (B–C) Quantification of SWR properties across conditions. (B) Inter-event interval (IEI). (C) Sharp wave amplitude. No significant effects of WIN55,212-2 were observed. (D) Peri-event time histograms of multiunit activity (MUA) aligned to SWR peak under control (left), WIN55,212-2 (middle), and washout (right) conditions. (E–G) Quantification of neuronal activity parameters. (E) Baseline firing rate (MUA-Base). (F) SWR-associated firing rate (MUA-SWR). (G) MUA delay relative to SWR peak (MUA-Delay). No significant effects of WIN55,212-2 were detected for any parameter. Statistical analysis was performed using LMM (Control vs WIN55,212-2). Washout data are shown for completeness but were not included in statistical comparisons.
Figure 4.
Effects of the cannabinoid receptor agonist WIN55,212-2 on SWRs and MUA in dorsal hippocampus. (A) Representative extracellular recordings from CA1 stratum pyramidale under control conditions (top), during WIN55,212-2 application (middle), and after washout (bottom). Calibration bars: 50 μV, 0.5 s. (B–C) Quantification of SWR properties across conditions. (B) Inter-event interval (IEI). (C) Sharp wave amplitude. No significant effects of WIN55,212-2 were observed. (D) Peri-event time histograms of multiunit activity (MUA) aligned to SWR peak under control (left), WIN55,212-2 (middle), and washout (right) conditions. (E–G) Quantification of neuronal activity parameters. (E) Baseline firing rate (MUA-Base). (F) SWR-associated firing rate (MUA-SWR). (G) MUA delay relative to SWR peak (MUA-Delay). No significant effects of WIN55,212-2 were detected for any parameter. Statistical analysis was performed using LMM (Control vs WIN55,212-2). Washout data are shown for completeness but were not included in statistical comparisons.
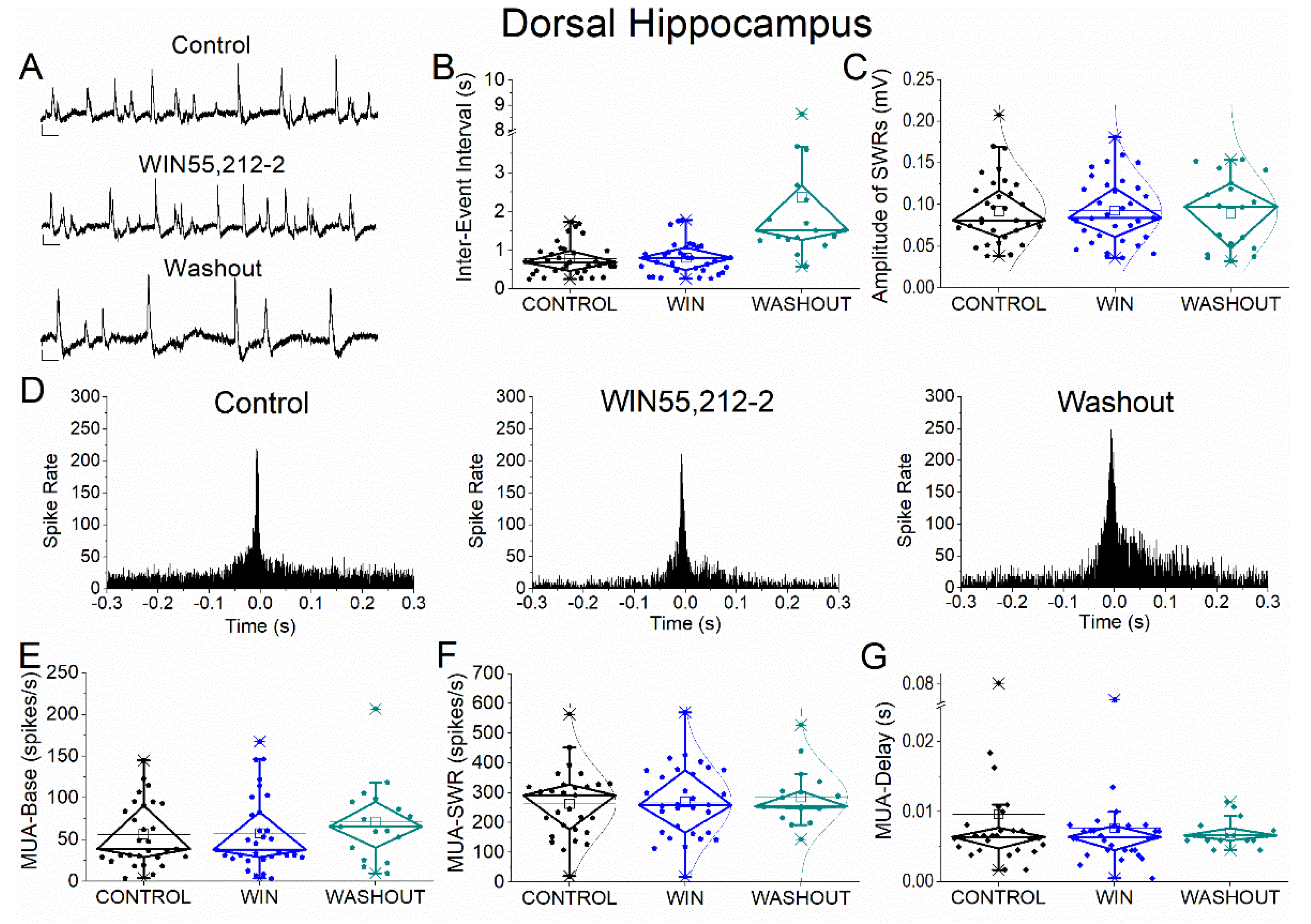
Figure 5.
Effects of the cannabinoid receptor agonist WIN55,212-2 on SWRs and MUA in ventral hippocampus. (A) Representative extracellular recordings from CA1 stratum pyramidale under control conditions (top), during WIN55,212-2 application (middle), and after washout (bottom). Calibration bars: 50 μV, 0.5 s. (B–C) Quantification of SWR properties across conditions. (B) Inter-event interval (IEI). (C) Sharp wave amplitude. No significant effects of WIN55,212-2 were observed. (D) Peri-event time histograms of multiunit activity (MUA) aligned to SWR peak under control (left), WIN55,212-2 (middle), and washout (right) conditions. (E–G) Quantification of MUA parameters. (E) Baseline firing rate (MUA-Base). (F) SWR-associated firing rate (MUA-SWR). (G) MUA delay relative to SWR peak (MUA-Delay). No significant effects of WIN55,212-2 were detected for any parameter. Statistical analysis was performed using LMM (Control vs WIN55,212-2). Washout data were not included in statistical comparisons.
Figure 5.
Effects of the cannabinoid receptor agonist WIN55,212-2 on SWRs and MUA in ventral hippocampus. (A) Representative extracellular recordings from CA1 stratum pyramidale under control conditions (top), during WIN55,212-2 application (middle), and after washout (bottom). Calibration bars: 50 μV, 0.5 s. (B–C) Quantification of SWR properties across conditions. (B) Inter-event interval (IEI). (C) Sharp wave amplitude. No significant effects of WIN55,212-2 were observed. (D) Peri-event time histograms of multiunit activity (MUA) aligned to SWR peak under control (left), WIN55,212-2 (middle), and washout (right) conditions. (E–G) Quantification of MUA parameters. (E) Baseline firing rate (MUA-Base). (F) SWR-associated firing rate (MUA-SWR). (G) MUA delay relative to SWR peak (MUA-Delay). No significant effects of WIN55,212-2 were detected for any parameter. Statistical analysis was performed using LMM (Control vs WIN55,212-2). Washout data were not included in statistical comparisons.
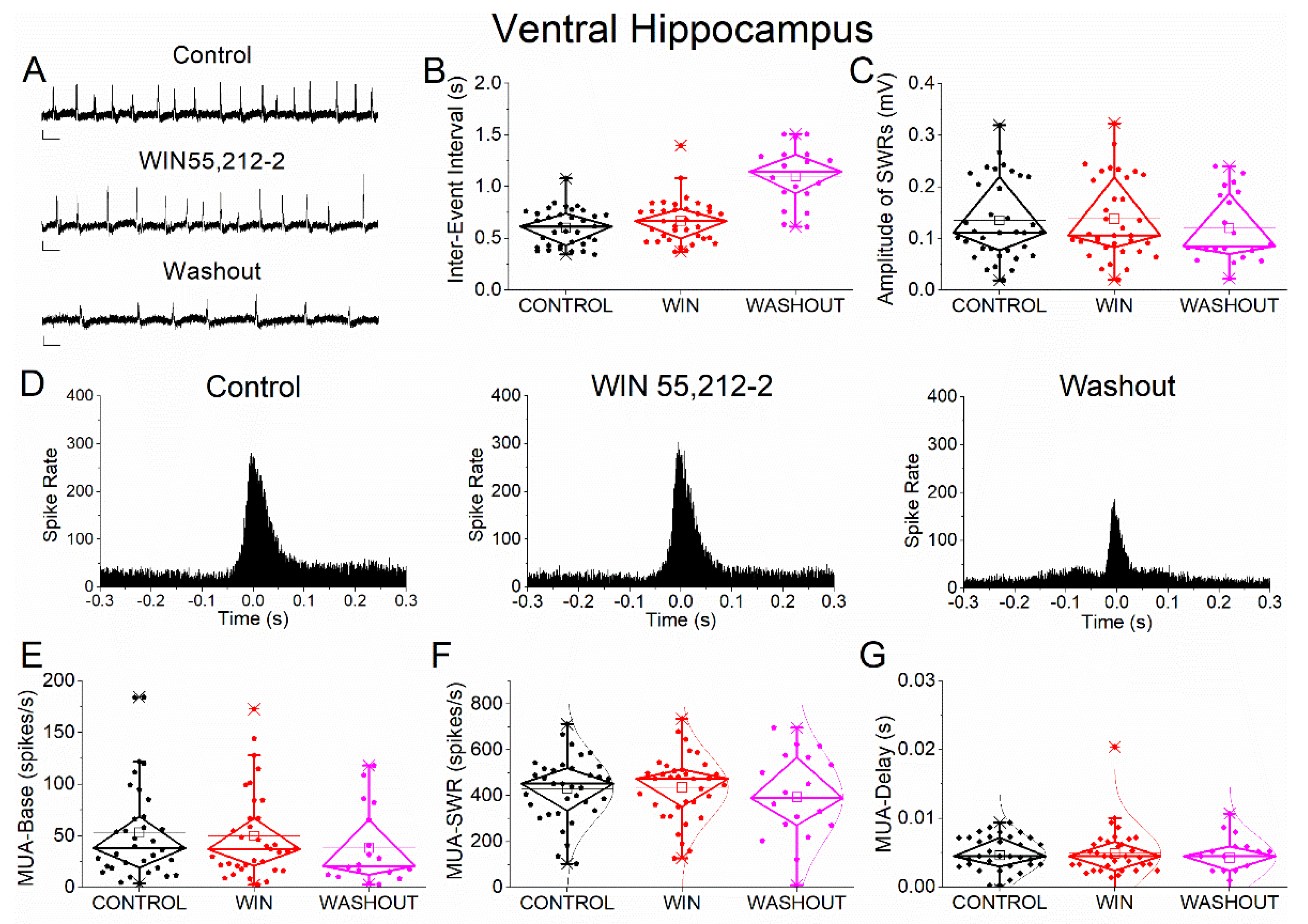
Figure 6.
Effects of vehicle (DMSO) and cannabidiol (CBD) on SWRs and MUA in dorsal hippocampus. (A) Representative extracellular recordings from CA1 stratum pyramidale under control conditions (top), during application of DMSO (second trace), CBD (third trace), and after washout (bottom). Calibration bars: 50 μV, 0.5 s. (B–C) Quantification of SWR properties across conditions. (B) Inter-event interval (IEI). (C) Sharp wave amplitude. Neither DMSO nor CBD produced significant effects. (D) Peri-event time histograms of multiunit activity (MUA) aligned to SWR peak under control, DMSO, CBD, and washout conditions. (E–G) Quantification of neuronal activity parameters. (E) Baseline firing rate (MUA-Base). (F) SWR-associated firing rate (MUA-SWR). (G) MUA delay relative to SWR peak (MUA-Delay). No significant effects of DMSO or CBD were detected for any parameter. Statistical analysis was performed using LMM (Control vs DMSO and Control vs CBD); washout data were not included in statistical comparisons.
Figure 6.
Effects of vehicle (DMSO) and cannabidiol (CBD) on SWRs and MUA in dorsal hippocampus. (A) Representative extracellular recordings from CA1 stratum pyramidale under control conditions (top), during application of DMSO (second trace), CBD (third trace), and after washout (bottom). Calibration bars: 50 μV, 0.5 s. (B–C) Quantification of SWR properties across conditions. (B) Inter-event interval (IEI). (C) Sharp wave amplitude. Neither DMSO nor CBD produced significant effects. (D) Peri-event time histograms of multiunit activity (MUA) aligned to SWR peak under control, DMSO, CBD, and washout conditions. (E–G) Quantification of neuronal activity parameters. (E) Baseline firing rate (MUA-Base). (F) SWR-associated firing rate (MUA-SWR). (G) MUA delay relative to SWR peak (MUA-Delay). No significant effects of DMSO or CBD were detected for any parameter. Statistical analysis was performed using LMM (Control vs DMSO and Control vs CBD); washout data were not included in statistical comparisons.
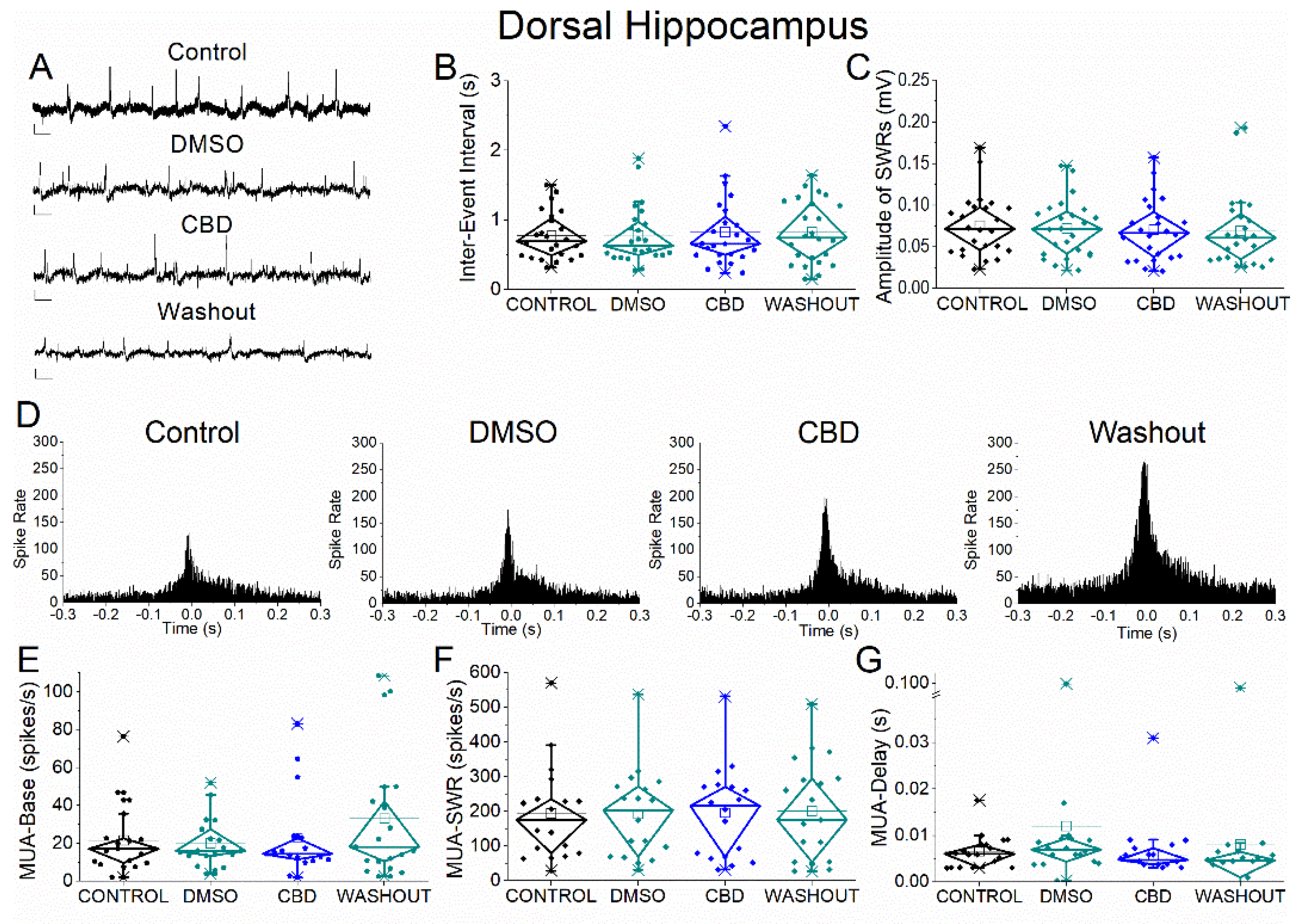
Figure 7.
Effects of vehicle (DMSO) and cannabidiol (CBD) on SWRs and associated neuronal activity in ventral hippocampus. (A) Representative extracellular recordings from CA1 stratum pyramidale under control conditions (top), during application of DMSO (second trace), CBD (third trace), and after washout (bottom). Calibration bars: 50 μV, 0.5 s. (B–C) Quantification of SWR properties across conditions. (B) Inter-event interval (IEI). (C) Sharp wave amplitude. Neither DMSO nor CBD produced significant effects. (D) Peri-event time histograms of multiunit activity (MUA) aligned to SWR peak under control, DMSO, CBD, and washout conditions. (E–G) Quantification of neuronal activity parameters. (E) Baseline firing rate (MUA-Base). (F) SWR-associated firing rate (MUA-SWR). (G) MUA delay relative to SWR peak (MUA-Delay). No significant effects of DMSO or CBD were detected for any parameter. Statistical analysis was performed using LMM (Control vs DMSO and Control vs CBD). Washout data were not included in statistical comparisons.
Figure 7.
Effects of vehicle (DMSO) and cannabidiol (CBD) on SWRs and associated neuronal activity in ventral hippocampus. (A) Representative extracellular recordings from CA1 stratum pyramidale under control conditions (top), during application of DMSO (second trace), CBD (third trace), and after washout (bottom). Calibration bars: 50 μV, 0.5 s. (B–C) Quantification of SWR properties across conditions. (B) Inter-event interval (IEI). (C) Sharp wave amplitude. Neither DMSO nor CBD produced significant effects. (D) Peri-event time histograms of multiunit activity (MUA) aligned to SWR peak under control, DMSO, CBD, and washout conditions. (E–G) Quantification of neuronal activity parameters. (E) Baseline firing rate (MUA-Base). (F) SWR-associated firing rate (MUA-SWR). (G) MUA delay relative to SWR peak (MUA-Delay). No significant effects of DMSO or CBD were detected for any parameter. Statistical analysis was performed using LMM (Control vs DMSO and Control vs CBD). Washout data were not included in statistical comparisons.
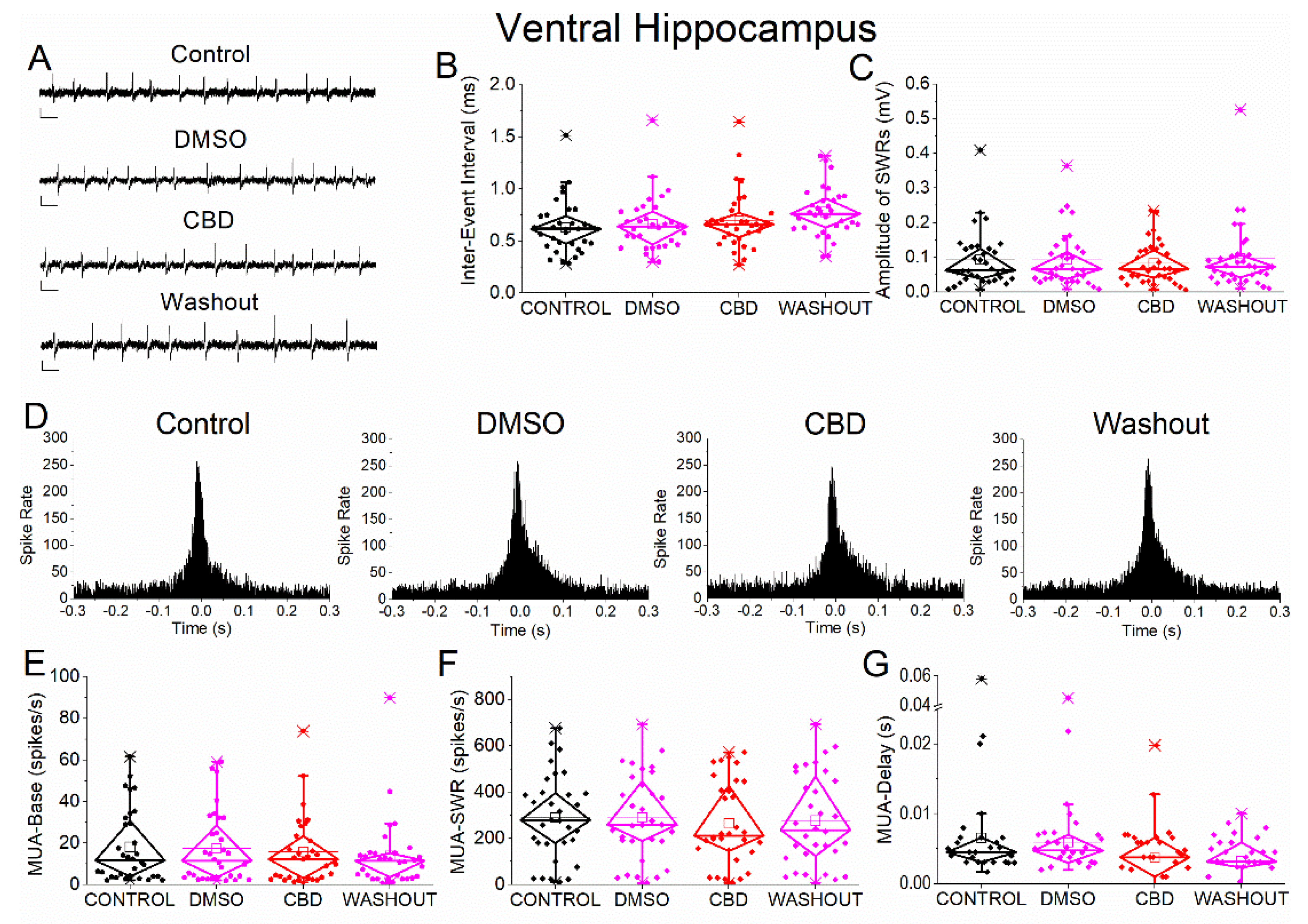
Figure 8.
Effects of GIRK channel blockade with TPN-Q and subsequent application of WIN55,212-2 on SWRs and associated neuronal activity in dorsal hippocampus. (A) Representative extracellular recordings from CA1 stratum pyramidale under control conditions (top), during TPN-Q application (middle), and after subsequent application of WIN55,212-2 (bottom). Calibration bars: 50 μV, 0.5 s. (B–C) Quantification of SWR properties across conditions. (B) Inter-event interval (IEI). (C) Sharp wave amplitude. TPN-Q produced no significant effects on SWR properties, and subsequent application of WIN55,212-2 did not alter these parameters. (D) Peri-event time histograms of MUA aligned to SWR peak under control (left), TPN-Q (middle), and WIN55,212-2 (right) conditions. (E–G) Quantification of neuronal activity parameters. (E) Baseline firing rate (MUA-Base). (F) SWR-associated firing rate (MUA-SWR). (G) MUA delay relative to SWR peak (MUA-Delay). TPN-Q produced only minor, non-significant changes, and no additional effects were observed following WIN55,212-2 application. Statistical analysis was performed using LMM (Control vs TPN-Q and TPN-Q vs WIN55,212-2).
Figure 8.
Effects of GIRK channel blockade with TPN-Q and subsequent application of WIN55,212-2 on SWRs and associated neuronal activity in dorsal hippocampus. (A) Representative extracellular recordings from CA1 stratum pyramidale under control conditions (top), during TPN-Q application (middle), and after subsequent application of WIN55,212-2 (bottom). Calibration bars: 50 μV, 0.5 s. (B–C) Quantification of SWR properties across conditions. (B) Inter-event interval (IEI). (C) Sharp wave amplitude. TPN-Q produced no significant effects on SWR properties, and subsequent application of WIN55,212-2 did not alter these parameters. (D) Peri-event time histograms of MUA aligned to SWR peak under control (left), TPN-Q (middle), and WIN55,212-2 (right) conditions. (E–G) Quantification of neuronal activity parameters. (E) Baseline firing rate (MUA-Base). (F) SWR-associated firing rate (MUA-SWR). (G) MUA delay relative to SWR peak (MUA-Delay). TPN-Q produced only minor, non-significant changes, and no additional effects were observed following WIN55,212-2 application. Statistical analysis was performed using LMM (Control vs TPN-Q and TPN-Q vs WIN55,212-2).
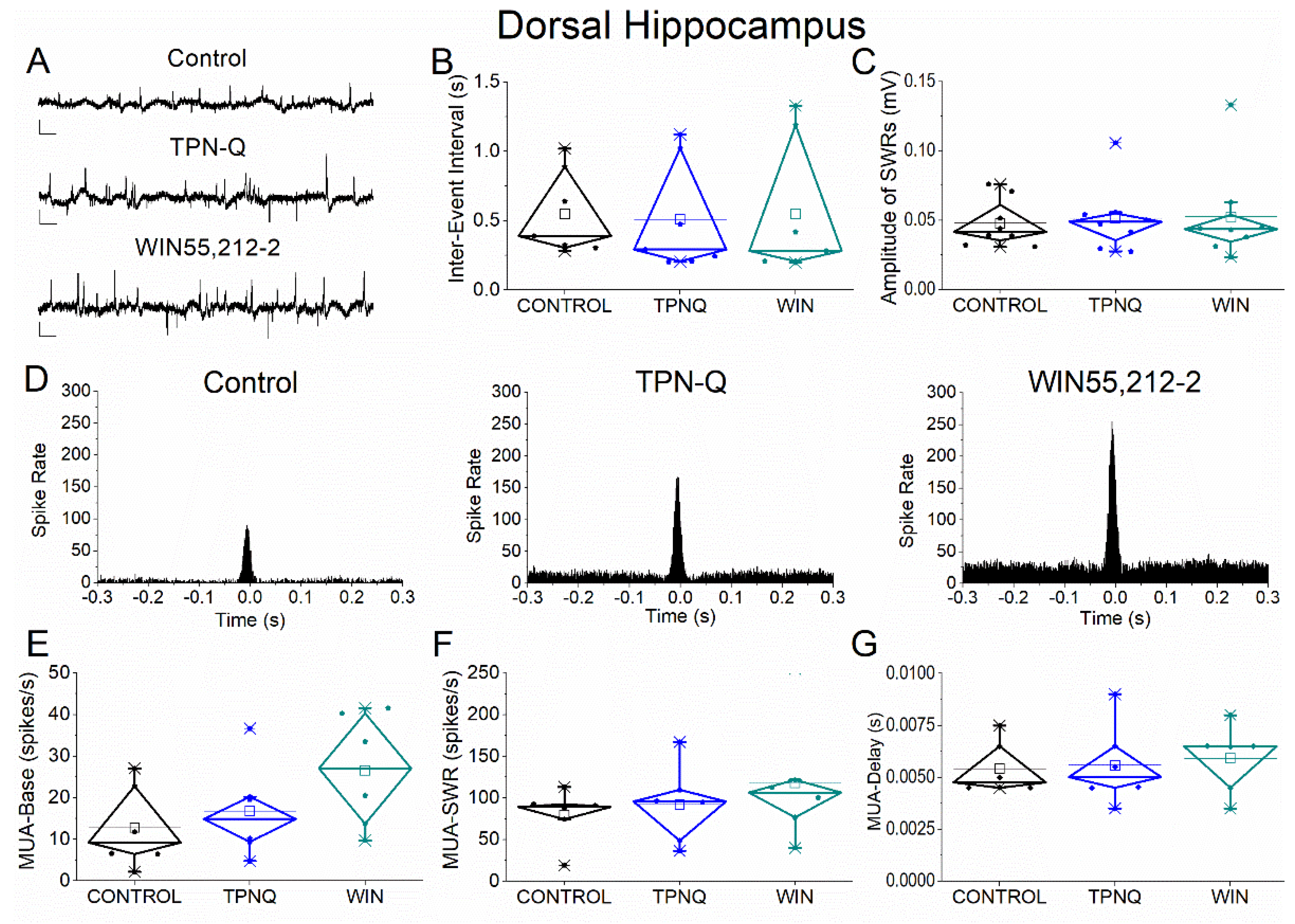
Figure 9.
Effects of GIRK channel blockade with TPN-Q and subsequent application of WIN55,212-2 on SWRs and MUA in ventral hippocampus. (A) Representative extracellular recordings from CA1 stratum pyramidale under control conditions (top), during TPN-Q application (middle), and after subsequent application of WIN55,212-2 (bottom). Calibration bars: 50 μV, 0.5 s. (B–C) Quantification of SWR properties across conditions. (B) Inter-event interval (IEI). (C) Sharp wave amplitude. TPN-Q produced no significant effects on SWR properties, and subsequent application of WIN55,212-2 did not alter these parameters. (D) Peri-event time histograms of MUA aligned to SWR peak under control (left), TPN-Q (middle), and WIN55,212-2 (right) conditions. (E–G) Quantification of MUA parameters. (E) Baseline firing rate (MUA-Base). (F) SWR-associated firing rate (MUA-SWR). (G) MUA delay relative to SWR peak (MUA-Delay). TPN-Q produced only limited, non-significant effects, and no additional effects were observed following WIN55,212-2 application. Statistical analysis was performed using LMM (Control vs TPN-Q and TPN-Q vs WIN55,212-2).
Figure 9.
Effects of GIRK channel blockade with TPN-Q and subsequent application of WIN55,212-2 on SWRs and MUA in ventral hippocampus. (A) Representative extracellular recordings from CA1 stratum pyramidale under control conditions (top), during TPN-Q application (middle), and after subsequent application of WIN55,212-2 (bottom). Calibration bars: 50 μV, 0.5 s. (B–C) Quantification of SWR properties across conditions. (B) Inter-event interval (IEI). (C) Sharp wave amplitude. TPN-Q produced no significant effects on SWR properties, and subsequent application of WIN55,212-2 did not alter these parameters. (D) Peri-event time histograms of MUA aligned to SWR peak under control (left), TPN-Q (middle), and WIN55,212-2 (right) conditions. (E–G) Quantification of MUA parameters. (E) Baseline firing rate (MUA-Base). (F) SWR-associated firing rate (MUA-SWR). (G) MUA delay relative to SWR peak (MUA-Delay). TPN-Q produced only limited, non-significant effects, and no additional effects were observed following WIN55,212-2 application. Statistical analysis was performed using LMM (Control vs TPN-Q and TPN-Q vs WIN55,212-2).
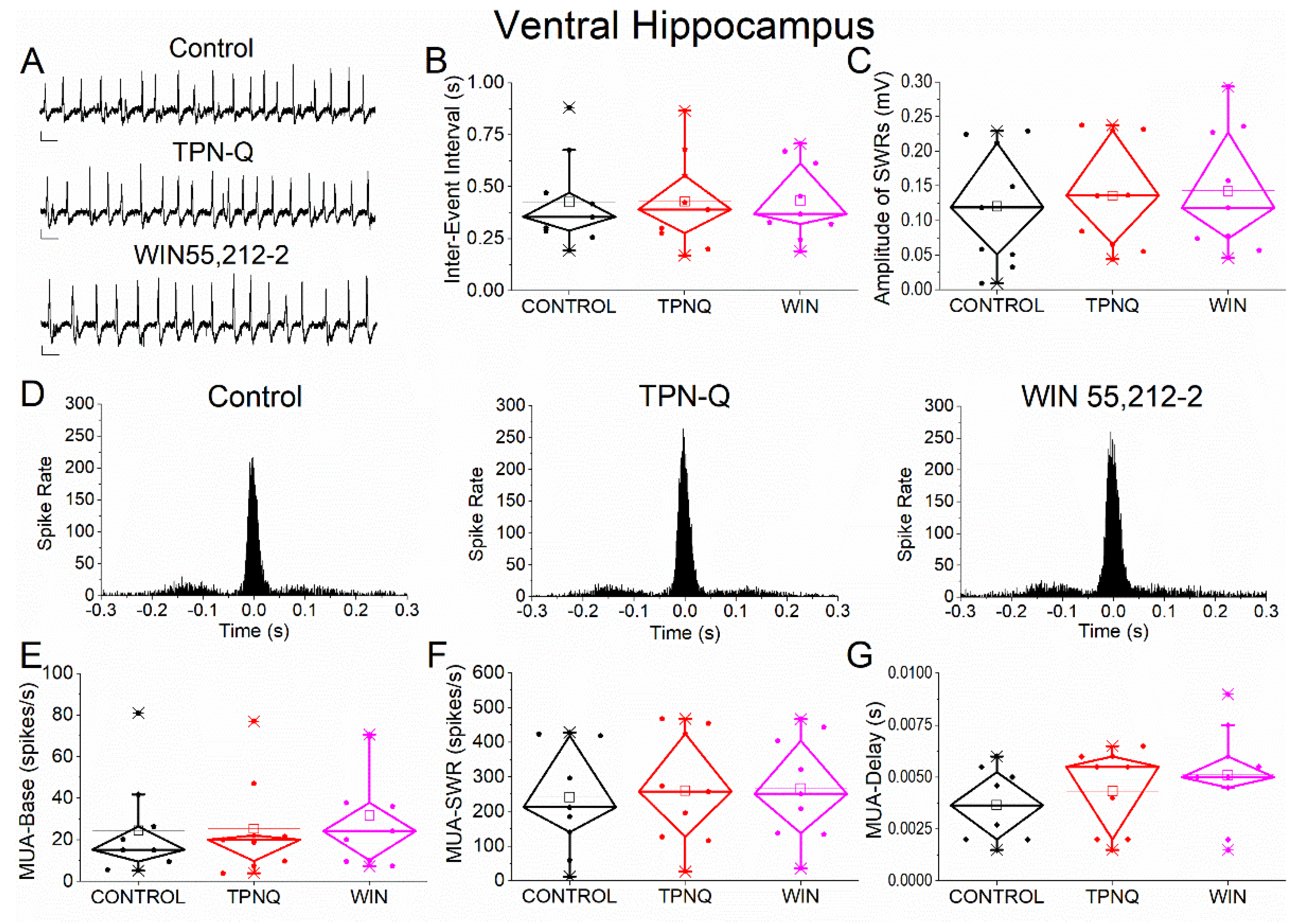
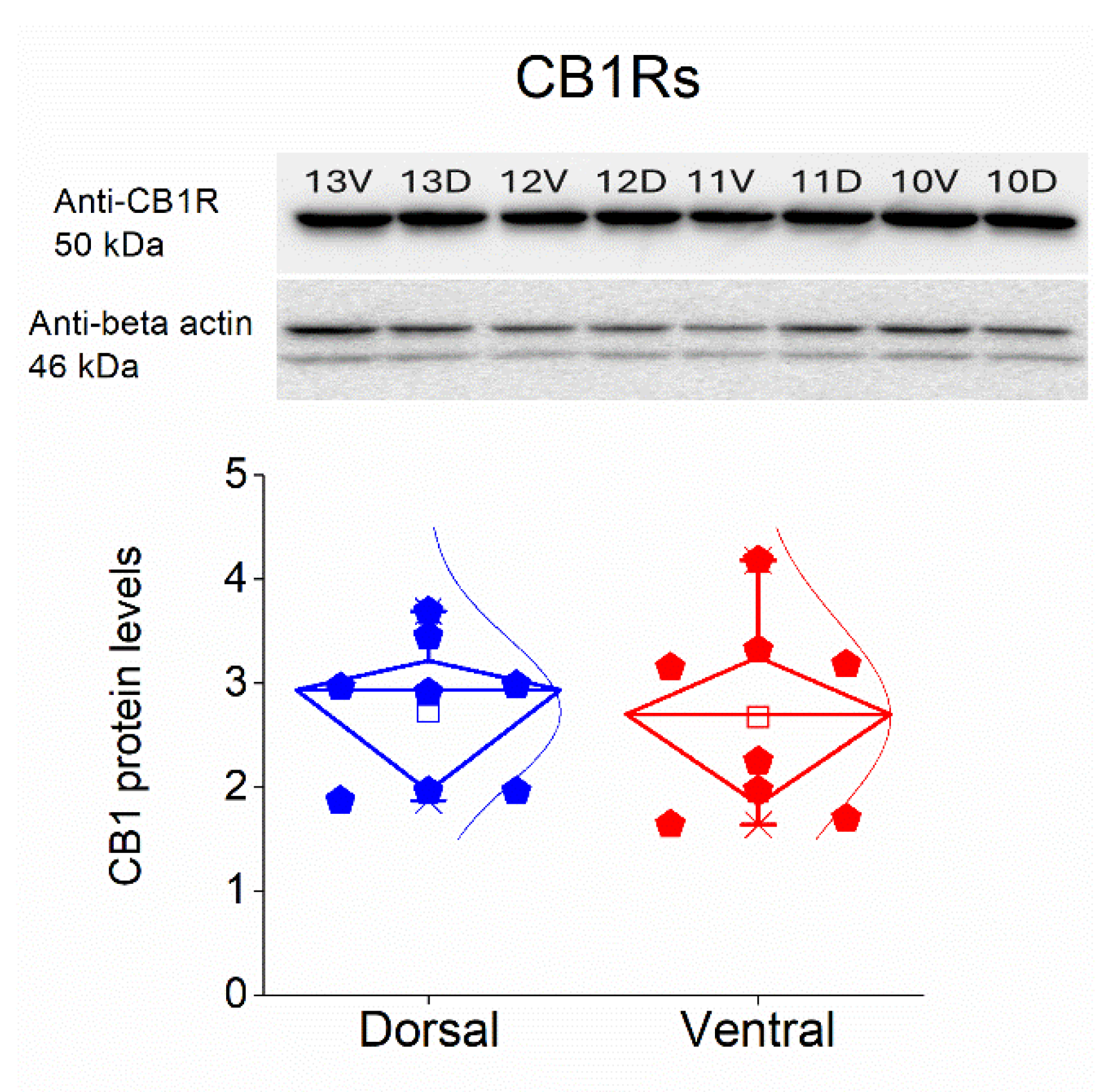
Figure 10.
Comparable CB1 receptor expression in CA3 dorsal and ventral hippocampal region. Representative Western blot showing CB1 receptor immunoreactivity (~50 kDa) in dorsal (D) and ventral (V) hippocampal samples, with β-actin (~46 kDa) used as loading control (top). Lane labels indicate individual samples from dorsal and ventral hippocampus. Quantification of CB1 protein levels (normalized to β-actin) revealed no significant difference between dorsal and ventral hippocampus. Data are presented as individual samples from corresponding rats, with distributions illustrated using box plots.
Figure 10.
Comparable CB1 receptor expression in CA3 dorsal and ventral hippocampal region. Representative Western blot showing CB1 receptor immunoreactivity (~50 kDa) in dorsal (D) and ventral (V) hippocampal samples, with β-actin (~46 kDa) used as loading control (top). Lane labels indicate individual samples from dorsal and ventral hippocampus. Quantification of CB1 protein levels (normalized to β-actin) revealed no significant difference between dorsal and ventral hippocampus. Data are presented as individual samples from corresponding rats, with distributions illustrated using box plots.