Submitted:
13 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
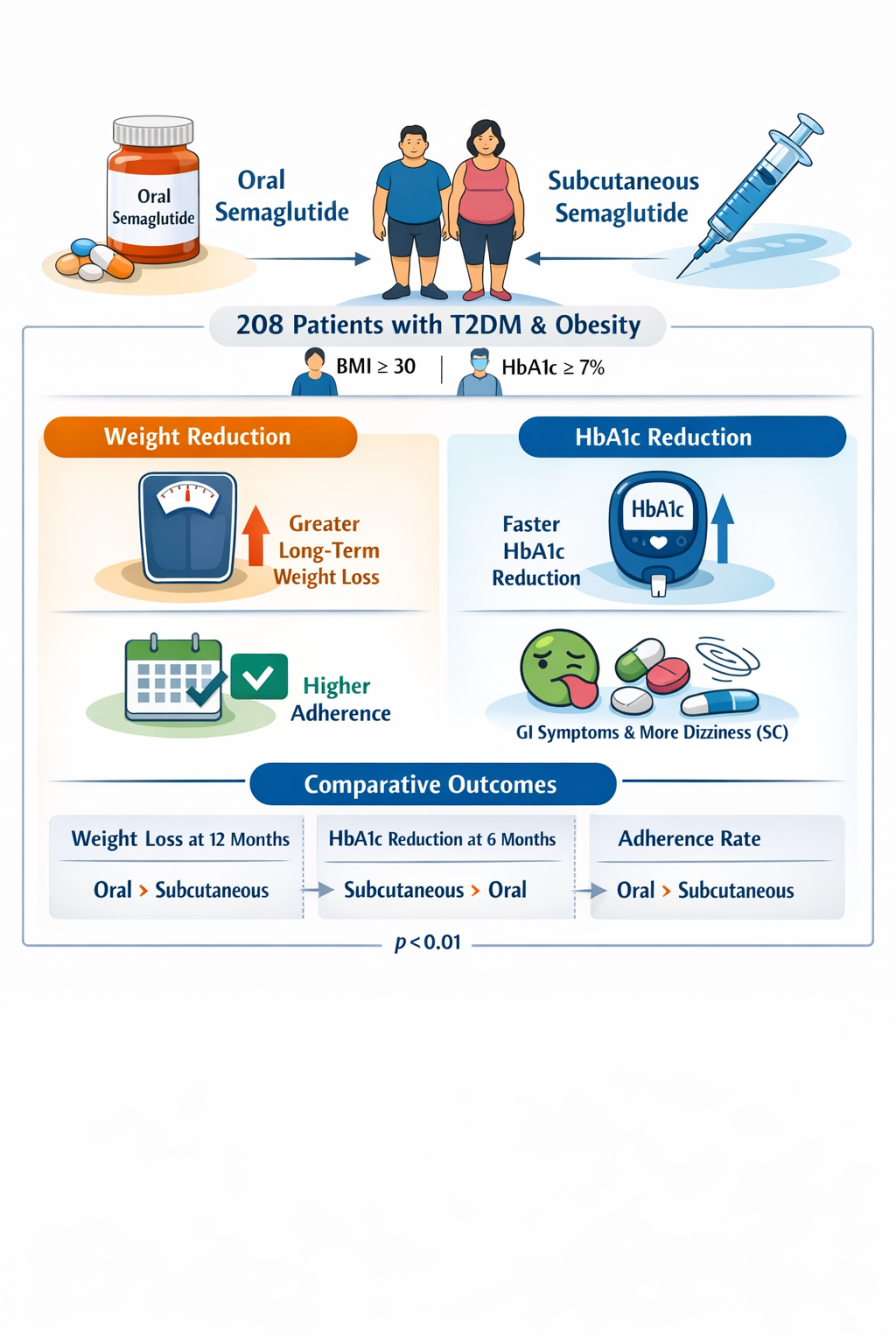
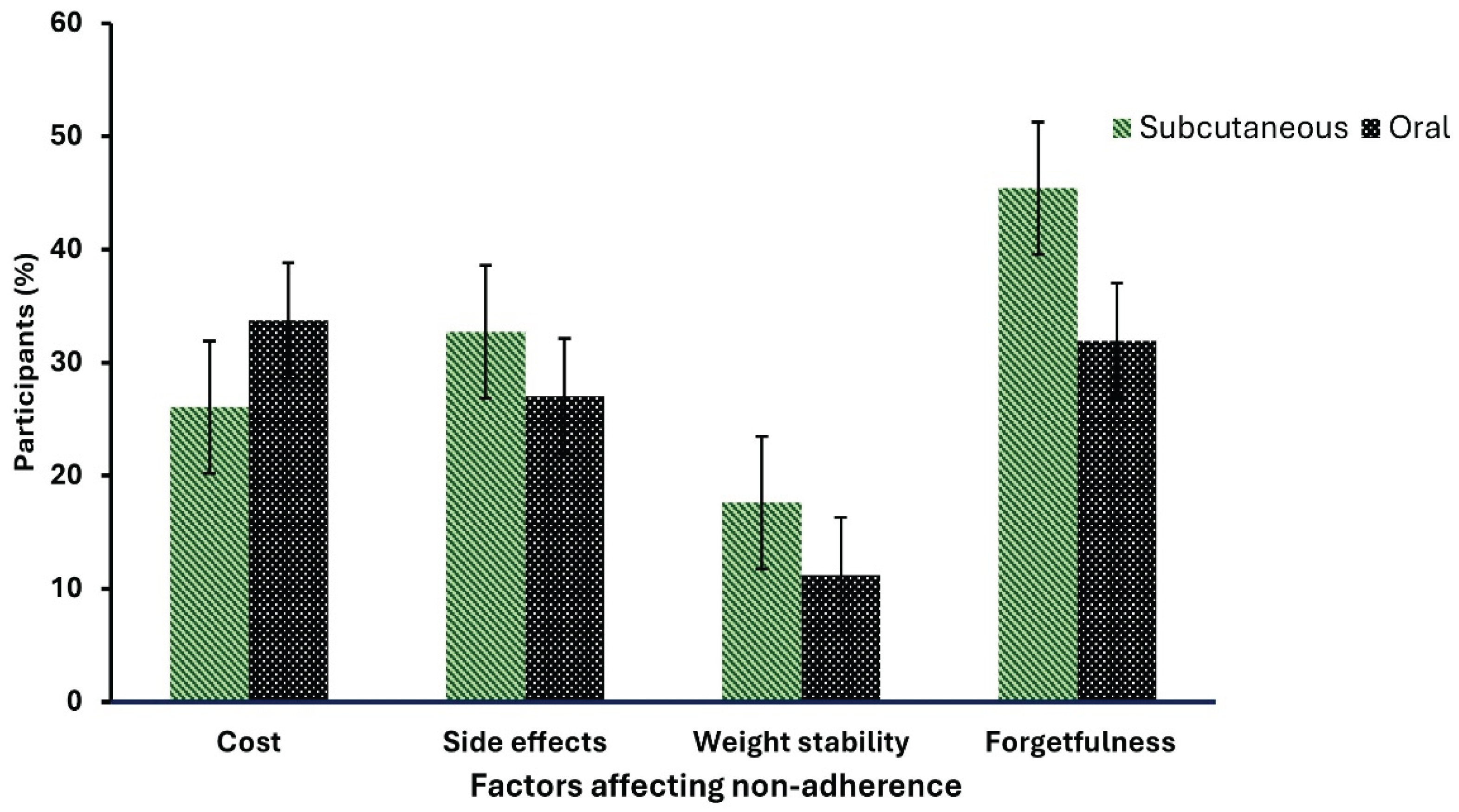
Objective: To compare the efficacy, safety, weight reduction and treatment adherence of oral versus subcutaneous semaglutide in adults with uncontrolled T2DM and obesity. Methods: A multicenter retrospective cohort study was conducted between January 2023 and January 2024. Adult patients (≥18 years) with T2DM (HbA1c ≥ 7%) and obesity (BMI ≥ 30) who received either oral or subcutaneous semaglutide were included. Demographic, clinical, and biochemical variables including body weight, BMI, HbA1c, side effects, and adherence were extracted from electronic medical records. Adverse effects were categorized by severity. Comparative analyses between groups used Chi-square and Mann Whitney U tests, with p<0.05 considered statistically significant. Results: A total of 208 patients were included: 89 on oral semaglutide and 119 on subcutaneous semaglutide. Baseline demographics, including gender, age, and physical activity, were comparable between groups (all p>0.05). The severity of adverse effects predominantly gastrointestinal symptoms such as nausea, vomiting, constipation, and diarrhea did not differ significantly between groups (p=0.994). However, dizziness was significantly more frequent in the subcutaneous group (p = 0.04). Adherence was markedly higher with oral semaglutide (p<0.05), with cost identified as the primary barrier among oral users, while subcutaneous users more frequently cited side effects, forgetfulness, and limited weight loss. Weight reduction was comparable at 3 months (p=0.23), but significantly greater with oral semaglutide at 6, 9, and 12 months (all p<0.01). Conversely, HbA1c reduction favored subcutaneous semaglutide at 3 and 6 months (p=0.03 and 0.02), although baseline glycemic control was similar. Conclusions: This study demonstrates that while subcutaneous semaglutide may provide a faster early HbA1c decline, oral semaglutide offers superior long-term weight reduction and significantly better adherence, likely attributable to easier administration. Both formulations exhibited comparable safety profiles.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Design and Setting
2.2. Population
2.3. Sampling and Tools
2.4. Pilot Study
2.5. Statistical Methods
3. Results
3.1. Sociodemographic Characteristics of Study Participants
3.2. Clinical Findings of Study Participants
3.3. Factors Affecting the Adherence to the Treatment
3.4. Therapeutic Effect of Semaglutide Formulations on Body Weight and HbA1c
3.5. Reported Side Effects in Both Test Groups
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| T2DM | Type 2 Diabetes Mellitus |
| HbA1c | Glycated Hemoglobin |
| BMI | Body Mass Index |
| GI | Gastrointestinal |
| GLP-1 RA | Glucagon-Like Peptide-1 Receptor Agonist |
| GLP-1 RAs | Glucagon-Like Peptide-1 Receptor Agonists |
| SNAC | Sodium N-(8-[2-hydroxybenzoyl] amino) caprylate |
| SPSS | Statistical Package for the Social Sciences |
| IBM | International Business Machines |
| NY | New York |
| χ² | Chi-square test |
| p | Probability value |
| Sc | Subcutaneous |
| CNS | Central Nervous System |
| IQ | Interquartile Range (Note: should be IQR in standard reporting) |
| yrs | Years |
| NB | Nota Bene (Note) |
References
- Xie, Y; Choi, T; Al-Aly, Z. GLP-1 receptor agonists in diabetes management: expanding therapeutic potential. Nat Med. 2025, 31(2), 240–8. [Google Scholar]
- Wilding, JPH; Batterham, RL. Mechanisms and clinical implications of GLP-1 receptor agonists in obesity and diabetes. Lancet Diabetes Endocrinol. 2024, 12(1), 45–58. [Google Scholar]
- Davies, MJ; et al. Semaglutide as a new option for the treatment of type 2 diabetes. Diabetes Care 2024, 47(3), 560–72. [Google Scholar]
- Zinman, B; Bain, SC; et al. Oral semaglutide in type 2 diabetes: the PIONEER clinical trial program. Lancet 2023, 402(10398), 2145–56. [Google Scholar]
- Nauck, MA; Meier, JJ. Oral GLP-1 receptor agonists: a new frontier in diabetes therapy. Nat Rev Endocrinol. 2025, 21(4), 320–33. [Google Scholar]
- Frontiers in Endocrinology. Oral semaglutide for the treatment of obesity: a real-world retrospective study. Front Endocrinol (Lausanne) 2024, 15, 1593334. [Google Scholar]
- Peyrot, M; et al. Patient adherence to diabetes therapy: barriers and strategies. Diabet Med. 2024, 41(2), 210–20. [Google Scholar]
- Pratley, RE; et al. Safety and tolerability of semaglutide across clinical trials. Clin Ther. 2023, 45(7), 1193–207. [Google Scholar]
- Marso, SP; et al. Cardiovascular and safety outcomes with semaglutide. N Engl J Med. 2024, 391(6), 490–503. [Google Scholar]
- Ahmed, A; et al. Cost-effectiveness of oral semaglutide versus injectable GLP-1 RAs in type 2 diabetes. Diabetes Obes Metab. 2025, 27(5), 1011–20. [Google Scholar]
- Husain, M.; Birkenfeld, A.L.; Donsmark, M.; Dungan, K.; Eliaschewitz, F.G.; Franco, D.R.; Jeppesen, O.K.; Lingvay, I.; Mosenzon, O.; Pedersen, S.D. Oral semaglutide and cardiovascular outcomes in patients with type 2 diabetes. New England Journal of Medicine 2019, 381, 841–851. [Google Scholar] [CrossRef] [PubMed]
- Marso, S.P.; Bain, S.C.; Consoli, A.; Eliaschewitz, F.G.; Jódar, E.; Leiter, L.A.; Lingvay, I.; Rosenstock, J.; Seufert, J.; Warren, M.L. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. New England Journal of Medicine 2016, 375, 1834–1844. [Google Scholar] [CrossRef] [PubMed]
- Pratley, R.; Amod, A.; Hoff, S.T.; Kadowaki, T.; Lingvay, I.; Nauck, M.; Pedersen, K.B.; Saugstrup, T.; Meier, J.J. Oral semaglutide versus subcutaneous liraglutide and placebo in type 2 diabetes (PIONEER 4): a randomised, double-blind, phase 3a trial. The Lancet 2019, 394, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Meier, J.J. Efficacy of Semaglutide in a Subcutaneous and an Oral Formulation. Frontiers in Endocrinology 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Kesavadev, J.; Shankar, A.; Saboo, B.D.; Ashik, A.; Naina, J.; Jothydev, K.; Basanth, A.; RAJ K, S.; Jothydev, S. 1708-P: Effect of Oral Semaglutide in Glycemic Control and Weight Reduction for People with Type 2 Diabetes Mellitus Having a BMI≥ 25 kg/m2—A Six-Month Real-World Study. Diabetes 2024, 73, 1708–P. [Google Scholar] [CrossRef]
- Sabaté, E. Adherence to long-term therapies: evidence for action; World health organization, 2003. [Google Scholar]
- Cramer, J.; Benedict, A.; Muszbek, N.; Keskinaslan, A.; Khan, Z. The significance of compliance and persistence in the treatment of diabetes, hypertension and dyslipidaemia: a review. International journal of clinical practice 2008, 62, 76–87. [Google Scholar] [CrossRef] [PubMed]
- Thethi, T.K.; Pratley, R.; Meier, J.J. Efficacy, safety and cardiovascular outcomes of once-daily oral semaglutide in patients with type 2 diabetes: the PIONEER programme. Diabetes, Obesity and Metabolism 2020, 22, 1263–1277. [Google Scholar] [CrossRef] [PubMed]
- Bain, S.C.; Belmar, N.; Hoff, S.T.; Husain, M.; Rasmussen, S.; Vilsbøll, T.; Petrie, M.C. Cardiovascular, Metabolic, and Safety Outcomes with Semaglutide by Baseline Age: Post Hoc Analysis of SUSTAIN 6 and PIONEER 6. Diabetes Therapy 2025, 16, 15–28. [Google Scholar] [CrossRef] [PubMed]
| Variable | Oral | Subcutaneous | P value |
|---|---|---|---|
| Gender | |||
| Male (%) | 44 (49.4) | 45 (37.8) | P = 0.07 |
| Female (%) | 45 (50.6) | 74 (62.2) | |
| Age | |||
| Range (yrs) | 14-64 | ||
| IQ (yrs) | 22.5-39 | P = 0.16 | |
| Median (yrs) | 32 | ||
| Exercise status | |||
| Yes (%) | 9 (10.1) | 18 (15.1) | P = 0.29 |
| No (%) | 80 (89.9) | 101 (84.9) |
| Variable | Oral Count (%) |
Subcutaneous Count (%) |
P value |
|---|---|---|---|
| Severity of side effects | |||
| Mild | 27 (30.3 %) | 37 (31.2 %) |
P = 0.994 |
| Moderate | 37 (41.6 %) | 49 (41.2 %) | |
| Severe | 25 (28.1%) | 34 (28.6 %) | |
| Patient adherence (Categories of dose missing) | |||
| Yes | 5 (5.6 %) | 109 (91.6 %) |
P << 0.05* |
| May be | 14 (15.7 %) | 2 (1.7 %) | |
| No | 70 (78.7 %) | 8 (6.7 %) | |
| Treatment period |
3 months | 6 months | 9 months | 12 months | |||||
|---|---|---|---|---|---|---|---|---|---|
| Weight reduction per treatment group |
Oral Count (%) |
Sc Count (%) |
Oral Count (%) |
Sc Count (%) |
Oral Count (%) |
Sc Count (%) |
Oral Count (%) |
Sc Count (%) |
|
| No | 0 | 3 (2.5) | 2 (2.2) | 0 | 2 (2.2) | 0 | 0 | 0 | |
| 0.1-5 | 14 (15.7) | 20 (16.8) | 0 | 6 (5) | 0 | 6 (5) | 2 (2.2) | 6 (5) | |
| 5.1-10 | 36 (40.5) | 38 (31.9) | 2 (2.2) | 0 | 2 (2.2) | 0 | 0 | 0 | |
| 10.1-15 | 38 (42.7) | 52 (43.7) | 9 (10.1) | 107 (90) | 9 (10.1) | 107 (90) | 11 (12.4%) | 95 (79.9) | |
| 15.1-20 | 1 (1.1%) | 6 (5.1) | 76 (85.5) | 6 (5) | 76 (85.5) | 6 (5) | 58 (65.2) | 13 (10.95) | |
| >20 | 0 | 0 | 0 | 0 | 0 | 0 | 18 (20.2) | 5 (4.2) | |
| p-value | 0.23 | << 0.01 | << 0.01 | << 0.01 | |||||
| Treatment Period |
0 months | 3 months | 6 months | ||||
|---|---|---|---|---|---|---|---|
| HbA1c Level by treatment group |
Oral Count (%) |
Sc Count (%) |
Oral Count (%) |
Sc Count (%) |
Oral Count (%) |
Sc Count (%) |
|
| Less than 5.7% | 18 (20.2) | 23 (19.3) | 52 (58.4) | 71 (59.7) | 55 (61.8) | 92 (77.3) | |
| 5.7-6.4% | 27 (30.3) | 34 (28.6) | 16 (18.0) | 34 (28.6) | 14 (15.7) | 7 (5.9) | |
| More than 6.4% | 44 (49.5) | 62 (52.1) | 21 (23.6) | 14 (11.7) | 20 (22.5) | 20 (16.8) | |
| p-value | >> 0.05 | 0.03 | 0.02 | ||||
| Side effect | Subcutaneous Counts (%) |
Oral Counts (%) |
p-value |
|---|---|---|---|
| Nausea | 32 (26.89%) | 35 (39.33%) | 0.06 |
| Constipation | 19 (15.97%) | 20 (22.47%) | 0.23 |
| Diarrhea | 28 (23.53%) | 19 (21.35%) | 0.71 |
| Vomiting | 39 (32.77%) | 32 (35.96%) | 0.63 |
| Abdominal Pain | 28 (23.53%) | 18 (20.22%) | 0.93 |
| fatigue | 26 (21.85%) | 11 (12.36%) | 0.08 |
| anxiety and depression | 22 (18.49%) | 8 (8.99%) | 0.05 |
| headache | 15 (12.61%) | 13 (14.61%) | 0.676 |
| dizziness | 17 (14.29%) | 5 (5.62%) | 0.04* |
| Joint pain | 8 (6.72%) | 5 (5.62%) | 0.74 |
| Hypoglycemia | 12 (10.08%) | 4 (4.49%) | 0.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




