Submitted:
13 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
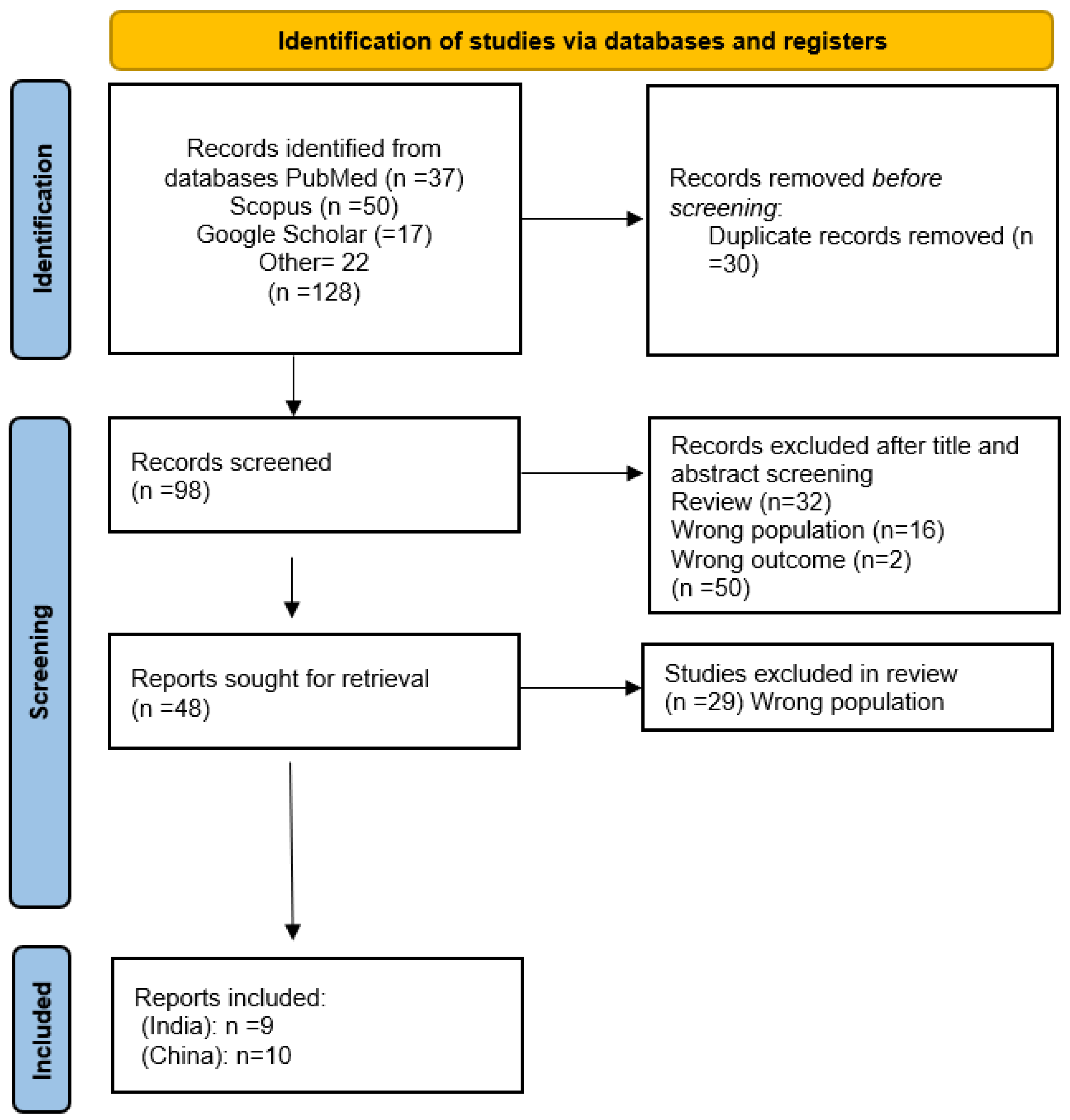
2. Materials and Methods
3. Results
3.1. China
3.2. India
4. Discussion
- (i)
- States Implementing NHS-UNHS Programs
- (ii)
- Proportion of Newborns Screened
- (iii)
- Screening Protocols and OAE Technologies
- (iv)
- Prevalence of Congenital and Bilateral HL
- (v)
- Causes Leading to HL and Intervention Strategies
Limitations of the Study
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AABR | Automated Auditory Brainstem Response |
| ABR | Auditory Brainstem Response |
| ASSR | Auditory Steady State Response |
| BD | bilateral deafness |
| CHL | Conductive Hearing Loss |
| CI | Cochlear Implant |
| DPOAE | Distortion Product OAE |
| EDHI | Early Hearing Detection and Intervention |
| ENT | Ear Nose and Throat |
| HL | Hearing Loss |
| LBW | Low Birth Weight |
| LTF | Loss To Follow-up |
| MHL | Mixed Hearing Loss |
| NHS | Neonatal Hearing Screening |
| NICU | neonatal intensive care unit |
| OAE | Otoacoustic Emissions |
| PTA | Pure Tone Average |
| SNHL | Sensorineural Hearing Loss |
| TEOAE | Transient Evoked OAE |
| UNHS | Universal Neonatal Hearing Screening |
Appendix A
| ID |
Country | Population | Studies selected | Region |
|---|---|---|---|---|
| 1 | China | 1,416,096,094 | East Asia | |
| 2 | India | 1,463,865,525 | - | South Asia |
| Selected | 2 | 2,879,961,619 | 16 | 1S; 1E |
References
- Yoshinaga- Itano, C. Benefits of early intervention for Children with Hearing Loss. Otolaryngol Clin North Am 1999, 32 (6), 1089-1102. [CrossRef]
- Downs, M.P.; Yoshinaga-Itano, C. The Efficacy of early identification and intervention in children with hearing impairment. Pediatr Clin North Am 1999 46(1), 79-87. [CrossRef]
- Kemp, D.T. Stimulated acoustic emissions from within the human auditory system. J Acoust Soc Am 1978; 64(5), 1386–1391. [CrossRef]
- Mason, J.A. and Hermann, KR. Universal infant hearing screening by automated auditory brainstem response measurement. Pediatrics 1998, 101:221-228. [CrossRef]
- Edmond, K.; Chadha, S.; Hunnicutt, C.; Strobel, N.; Manchaiah, V.; Yoshinga-Itano, C.; Universal Newborn Hearing Screening (UNHS) Review Group. Effectiveness of universal newborn hearing screening: A systematic review and meta-analysis. J. Glob. Health 2022, 12, 12006. [CrossRef]
- Hatzopoulos, S., Cardinali, L., Skarzynski, P. H., & Zimatore, G. (2025). The Otoacoustic Emissions in the Universal Neonatal Hearing Screening: An Update on the Asian Data (2004 to 2024). Children 2026, 13(1), 60. [CrossRef]
- Worlmeter website. Available online: https://www.worldometers.info/world-population/asia-population/ (assessed September 2025).
- Kapadia, M.; Vaid, N. & Vaze, V. UNHS: A Decade Long Feasibility and Sustenance Study from a Tertiary Care Hospital in India. Indian J Otolaryngol Head Neck Surg 2022, 74 (1), 624. [CrossRef]
- Hatzopoulos, S.; Cardinali, L.; Piotr Henryk Skarżyński; Zimatore, G. The Otoacoustic Emissions in the Universal Neonatal Hearing Screening: An Update on the European Data (2004 to 2024). Children 2024, 11 (11), 1276. [CrossRef]
- Hatzopoulos, S., Cardinali, L., Skarzynski, P. H., & Zimatore, G. The Otoacoustic Emissions in the Universal Neonatal Hearing Screening: A Scoping Review Update on the African Data (2004 to 2024). Children 2025, 12(2), 141. [CrossRef]
- Hatzopoulos, S., Cardinali, L., Skarzynski, P. H., Adewunmi, A. T., & Zimatore, G. (2025). Early Hearing Interventions for Children with Hearing Loss in Africa: A 21-Year Scoping Review (2004–2025). Children, 12(7), 864. Children 2024, 11. [CrossRef]
- Neumann, K.; Euler, H.A.; Chadha, S.; White, K.R. A Survey on the Global Status of Newborn and Infant Hearing Screening. JEHDI 2020, 2 (2): 63–84. [CrossRef]
- Wang, X.; et al. Concurrent Hearing and Genetic Screening of Newborns in China. Int. J. Pediatr. Otorhinolaryngol. 2011, 75, 1049–1053.
- Huang, L.-H.; Zhang, L.; Gai Tobe, R.-Y.; Qi, F.-H.; Sun, L.; Teng, Y.; Ke, Q.-L.; Mai, F.; Zhang, X.-F.; Zhang, M.; et al. Cost-Effectiveness Analysis of Neonatal Hearing Screening Program in China: Should Universal Screening Be Prioritized? BMC Health Serv. Res. 2012, 12, 97.
- Zhang, J.; et al. Combined Newborn Hearing and Genetic Screening in Tianjin, China. N. Engl. J. Med. 2013, 369, 1601–1603.
- Dai, P.; et al. Genetic Etiology of Nonsyndromic Hearing Loss in Chinese Patients. Clin. Genet. 2015, 87, 510–518.
- Peng, Q.; et al. Newborn Hearing Screening Combined with Genetic Screening in Southern China. BMC Pediatr. 2016, 16, 28.
- Zhou, Y.; et al. Association Between Gestational Diabetes Mellitus and Newborn Hearing Screening Outcomes. Int. J. Pediatr. Otorhinolaryngol. 2021, 147, 110782.
- Li, X.; et al. Audiological Outcomes of Universal Newborn Hearing Screening in Beijing: A Retrospective Study. Int. J. Audiol. 2023, 62, 795–804.
- Ren, Y.; et al. Metabolic Factors and Outcomes in Universal Newborn Hearing Screening: A Hospital-Based Study. Int. J. Pediatr. Otorhinolaryngol. 2025, 178, 111234.
- Wu, W.; Xiangrong, T.; Yun, L.; Jingrong, L.; Jianyong, C.; Xueling, W. & Hao, W. Neonatal hearing screening in remote areas of China: a comparison between rural and urban populations. J of Int Med Res 2018, 46(2), 637-651. [CrossRef]
- Chen, X.; Yuan, M.; Lu, J.; Zhang, Q.; Sun, M., & Chang, F.. Assessment of universal newborn hearing screening and intervention in Shanghai, China. Int. J. Technol. Assess. Health Care 2017, 33(2), 206-214. [CrossRef]
- John, M.; et al. Neonatal Hearing Screening in a Tertiary Care Hospital in India. Indian J. Pediatr. 2009, 76, 115–119.
- Bansal, S.; Gupta, A.; Nagarkar, A. Transient Evoked Otoacoustic Emissions in Hearing Screening Programs—Protocol for Developing Countries. Int. J. Pediatr. Otorhinolaryngol. 2008, 72, 1059–1063. [CrossRef]
- Sachdeva, K.; et al. Neonatal Hearing Screening in a Tertiary Care Hospital in India. Indian J. Otolaryngol. Head Neck Surg. 2017, 69, 420–425.
- Dar, L.; Namdeo, D.; Kumar, P.; Thakar, A.; Kant, S.; Rai, S.; Singh, P.K.; Kabra, M.; Fowler, K.B.; Boppana, S.B. Congenital Cytomegalovirus Infection and Permanent Hearing Loss in Rural North Indian Children. Pediatr. Infect. Dis. J. 2017, 36, 670–673. [CrossRef]
- Parab, S.; et al. Universal Newborn Hearing Screening in Rural India: Program Outcomes and Challenges. Int. J. Pediatr. Otorhinolaryngol. 2018, 107, 113–117.
- Upadhyay, S.; et al. Incidence of Hearing Loss in Newborns Undergoing Universal Screening in India. J. Family Med. Prim. Care 2022, 11, 1234–1239.
- Rawat, R.; et al. Evaluation of a Stepwise Newborn Hearing Screening Protocol in North India. Indian J. Otolaryngol. Head Neck Surg. 2023, 75, 215–221.
- Kapadia, M.; Vaid, N.; Vaze, V. Universal Newborn Hearing Screening: A Decade-Long Feasibility and Sustenance Study from a Tertiary Care Hospital in India. Indian Journal of Otolaryngology and Head & Neck Surgery 2022, 74(Suppl 1), 624–630. [CrossRef]
- Sahoo KC et al. Cost-Effectiveness of Portable-Automated ABR for Universal Neonatal Hearing Screening in India. Front Public Health. 2024, 12:1364226. [CrossRef]
| n | Country | Region/city or Town | Sample Size | Study Period | First Author |
Year |
|---|---|---|---|---|---|---|
| 1 | China | Multicenter | 14,913 | 2009–2010 | Wang | 2011 |
| 2 | China | Multicenter (8 provinces) | NR | 2007–2009 | Huang | 2012 |
| 3 | China | Tianjin | 58,397 | 2010–2012 | Zhang | 2013 |
| 4 | China | Multicenter | Patients with NSHL NR | NR | Dai | 2015 |
| 5 | China | Southern China | 9,317 | 2013–2014 | Peng | 2016 |
| 6 | China | Weifang | 666 | 2018–2019 | Zhou | 2021 |
| 7 | China | Beijing | 1,839 | 2011–2019 | Li | 2023 |
| 8 | China | Hospital-based | 8,631 | 2017–2024 | Ren | 2025 |
| 9 | China | Liuzhou (Guangxi, rural vs urban) | | 19,098 | 2012-2014 | Wu | 2017 |
| 10 | China | Shanghai | 1,574,380 | 2002-2012 | Chen | 2017 |
| 11 | India | Vellore | 500 | 2006–2007 | John | 2009 |
| 12 | India | Chandigarh | 2,659 | 2005–2007 | Bansal | 2008 |
| 13 | India | Jabalpur | 2,254 | 2015–2016 | Sachdeva | 2017 |
| 14 | India | Ballabgarh | 1,720 | 2011–2014 | Dar | 2017 |
| 15 | India | Maval, Pune District (rural Maharashtra) | 8,192 | 2014–2016 | Parab | 2018 |
| 16 | India | Lucknow | 2,676 | 2018–2020 | Upadhyay | 2022 |
| 17 | India | Panchkula, Haryana (North) | 506 | 2021–2022 | Rawat | 2023 |
| 18 | India | Pune (tertiary care hospital) | 5,542 |
2008-2018 | Kapadia | 2011 |
| 19 | India | Odisha (policy/program setting) | NR | NR | Sahoo | 2024 |
| n | State | Region/city or Town | Screening Protocol (OAE/ABR) |
Hearing Loss prevalence |
Causes / risk factors | Author (First) |
Year |
|---|---|---|---|---|---|---|---|
| 1 | China | Multicenter | OAE + genetics | 1.9/1000 | Hereditary | Wang | 2011 |
| 2 | China | Multicenter | OAE vs OAE+AABR | 3.0/1000 | Model | Huang | 2012 |
| 3 | China | Tianjin | OAE + genetics | 2.3/1000 | Pathogenic variants in GJB2, SLC26A4, mtDNA | Zhang | 2013 |
| 4 | China | Multicenter | OAE + genetics | NR | GJB2 mutations; non-syndromic HL | Dai | 2015 |
| 5 | China | Southern China | OAE + genetics | 2.7/1000 | GJB2 and mitochondrial mutations | Peng | 2016 |
| 6 | China | Shandong | Automated OAE | NR | gestational diabetes mellitus (GDM) | Zhou | 2021 |
| 7 | China | Beijing | UNHS referral | NR | Referral outcomes | Li | 2023 |
| 8 | China | Hospital-based | OAE+metabolic | NR | Metabolic disorders (e.g., hyperbilirubinemia) | Ren | 2025 |
| 9 | China | Liuzhou (Guangxi) | DPOAE + AABR (two-step UNHS) | 2.25/1000 (PCHL); 0.33% total HL | Metabolic disorders (e.g., hyperbilirubinemia) | Wu | 2017 |
| 10 | China | Shanghai | OAE + AABR | 1.66/1000 | Large-scale program; integrated screening–intervention–rehabilitation system; high coverage (93.6%) | Chen | 2017 |
| 11 | India | Vellore | DPOAE (2-stage) + AABR confirm DPOAE+ABR | 6.0/1000 | NICU | John | 2009 |
| 12 | India | Chandigarh | TEOAE+ABR | NR | Timing | Bansal | 2008 |
| 13 | India | Jabalpur | DPOAE+ABR | 8.9/1000 | High-risk | Sachdeva | 2017 |
| 14 | India | Ballabgarh | DPOAE (2-stage) + ABR confirmation | 5.0/1000 | CMV | Dar | 2017 |
| 15 | India | Pune | OAE+ABR | 3.54/1000 | Low birth weight, hyperbilirubinemia, craniofacial anomalies | Parab | 2018 |
| 16 | India | Lucknow | OAE+ABR | 7.0/1000 | Repeat OAE reduced referrals; program implementation feasibility | Upadhyay | 2022 |
| 17 | India | Panchkula | OAE→ ABR | 8.2/1000 | Prematurity | Rawat | 2023 |
| 18 | India | Pune | OAE + ABR (two-step UNHS) | 5.41/1000 (overall); 9.11/1000 high-risk; 1.49/1000 well-baby | NICU stay, low birth weight, IUGR, RDS, hyperbilirubinemia; high loss to follow-up | Kapadia | 2022 |
| 19 | India | Odisha (policy/program setting), | Decision-tree model: OAE vs portable AABR | NR (economic model; not an observed population prevalence) | Resource constraints; device portability; at-risk prevalence cited from literature | Sahoo | 2024 |
| Domain | China | India |
|---|---|---|
| Implementation | Large-scale UNHS | Mostly hospital-based |
| Coverage | High (>85–95%) | Variable |
| Prevalence range | 1.9–3.0/1000 | 3–9/1000 |
| Protocols | OAE + AABR + genetics | OAE + ABR |
| Main limitation | Regional inequality | Follow-up & infrastructure |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




