Submitted:
13 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Neuroprofile as the Basis for Intervention with Children
1.2. EEG-Based Markers of Disorders in Children from Alcohol-Affected and Abusive Families
1.3. Long-Term Consequences of the Lack of Intervention in Children from Alcohol-Affected Families
2. Materials and Methods
2.1. Participants and Recruitment
2.2. Procedure
2.3. EEG Measures (Functional Markers)
- 1)
- Theta–Beta Ratio (TBR)
- 2)
- Frontal Alpha Asymmetry (FAA)
- 3)
- Temporal Beta Stress
- 4)
- Parietal EMG Tension Index
- 5)
- SMR Composite Index
2.4. Data Processing
2.5. Statistical Analysis
3. Results
3.1. Sample Characteristics
3.2. Between-Group Differences in EEG Markers
3.3. EEG Profile of Children from Alcohol-Affected Families
4. Discussion
5. Conclusions
6. Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| MDPI | Multidisciplinary Digital Publishing Institute |
| DOAJ | Directory of open access journals |
| TLA | Three letter acronym |
| LD | Linear dichroism |
References
- Tokuhama-Espinosa, T. Neuroeducation: A Review of Theory and Practice; W.W. Norton: New York, NY, USA, 2019.
- Siegel, D.J. The Developing Mind: How Relationships and the Brain Interact to Shape Who We Are, 3rd ed.; Guilford Press: New York, NY, USA, 2020.
- Tokarz, A. Neuropsychologiczne podstawy uczenia się i wychowania; PWN: Warszawa, Poland, 2020.
- Diamond, A. Executive functions. Annu. Rev. Psychol. 2013, 64, 135–168.
- Zelazo, P.D.; Blair, C.B.; Willoughby, M.T. Executive function: Implications for education; NCER Report; National Center for Education Research: Washington, DC, USA, 2016.
- Perry, B.D.; Szalavitz, M. The Boy Who Was Raised as a Dog, 3rd ed.; Basic Books: New York, NY, USA, 2021.
- Immordino-Yang, M.H.; Damasio, A. We feel, therefore we learn. Mind Brain Educ. 2021, 15, 115–123.
- De Bellis, M.D.; Zisk, A. The biological effects of childhood trauma. Child Adolesc. Psychiatr. Clin. N. Am. 2014, 23, 185–222.
- Porges, S.W. The polyvagal theory. Clevel. Clin. J. Med. 2018, 85, S86–S90.
- Black, M.M.; Conway, A. Maternal and child nutrition. Lancet 2018, 391, 243–244.
- Grzybowski, P. Neuroeducation and cognitive development. Educ. Res. Rev. 2023, 38, 100501.
- Chojak, M. Neurodydaktyka w edukacji i wychowaniu; UMCS: Lublin, Poland, 2018.
- Im, H. Neurodevelopment and environmental adversity. Dev. Cogn. Neurosci. 2025, 62, 101250.
- Staton, M.; Leukefeld, C.; Webster, J.M. Substance use and family systems. J. Subst. Abuse Treat. 2024, 156, 108742.
- Helen, J.; Smith, A.; Brown, K. Social interventions and vulnerability reduction. Soc. Policy Adm. 2023, 57, 345–360.
- Rangaswamy, M.; Porjesz, B.; Chorlian, D.B.; Wang, K.; Jones, K.A.; Bauer, L.O.; Rohrbaugh, J.; O’Connor, S.J.; Kuperman, S.; Reich, T.; Begleiter, H. Beta power in the EEG of alcoholics. Biol. Psychiatry 2004, 55, 831–842.
- Kamarajan, C.; Porjesz, B.; Jones, K.A.; Chorlian, D.B.; Padmanabhapillai, A.; Rangaswamy, M.; Stimus, A.T.; Begleiter, H. Spatial-anatomical mapping of NoGo-P3 in offspring of alcoholics. Clin. Neurophysiol. 2005, 116, 1049–1061.
- Kamarajan, C.; Rangaswamy, M.; Manz, N.; Chorlian, D.B.; Pandey, A.K.; Roopesh, B.N.; Porjesz, B. Topography, current density, and neurocognitive correlates of P3 event-related potentials in alcohol use disorder. Brain Sci. 2015, 5, 360–388.
- Tarokh, L.; Carskadon, M.A.; Achermann, P. Developmental changes in brain connectivity assessed using sleep EEG. Sleep Med. Rev. 2009, 13, 203–210.
- Yu, X.; Zhang, L.; Li, Y. Neural correlates of emotional processing in maltreated children. Dev. Cogn. Neurosci. 2025, 63, 101280.
- Curtis, W.J.; Cicchetti, D. Emotion and resilience: Frontal EEG asymmetry in maltreated children. Dev. Psychopathol. 2007, 19, 811–840.
- Meiers, S.J.; et al. Child maltreatment and substance use outcomes. J. Adolesc. Health 2020, 67, 123–130.
- Chassin, L.; Pitts, S.C.; DeLucia, C.; Todd, M. A longitudinal study of children of alcoholics. J. Abnorm. Psychol. 1999, 108, 106–119.
- Felitti, V.J.; Anda, R.F.; Nordenberg, D.; Williamson, D.F.; Spitz, A.M.; Edwards, V.; Koss, M.P.; Marks, J.S. Relationship of childhood abuse and household dysfunction to leading causes of death in adults. Am. J. Prev. Med. 1998, 14, 245–258 (reprint 2019).
- Torvik, F.A.; Rognmo, K. Children of parents with alcohol problems. Soc. Psychiatry Psychiatr. Epidemiol. 2011, 46, 745–753.
- Raitasalo, K.; Holmila, M.; Autti-Rämö, I.; et al. Parental substance use and educational outcomes. Drug Alcohol Rev. 2020, 39, 559–567.
- Kearns-Bodkin, J.N.; Leonard, K.E. Relationship functioning among adult children of alcoholics. J. Stud. Alcohol Drugs 2008, 69, 149–157.
- Norman, R.E.; Byambaa, M.; De, R.; Butchart, A.; Scott, J.; Vos, T. The long-term health consequences of child abuse. PLoS Med. 2012, 9, e1001349.
- Gilbert, R.; Widom, C.S.; Browne, K.; Fergusson, D.; Webb, E.; Janson, S. Burden and consequences of child maltreatment. Lancet 2009, 373, 68–81.
- Hensch, T.K. Critical period regulation. Annu. Rev. Neurosci. 2004, 27, 549–579.
- Hensch, T.K. Critical period plasticity. Nat. Rev. Neurosci. 2005, 6, 877–888.
- Marshall, P.J.; Fox, N.A.; Bucharest Early Intervention Project Core Group. A comparison of the electroencephalogram between institutionalized and community children in Romania. J. Cogn. Neurosci. 2004, 16, 1327–1338.
- Wilkinson, P.O.; Goodyer, I.M.; et al. Frontal alpha asymmetry and stress-related neural regulation in children. Biol. Psychol. 2024, 190, 108600.
- Marshall, P.J.; Reeb, B.C.; Fox, N.A.; Nelson, C.A.; Zeanah, C.H. Effects of early intervention on EEG power in previously institutionalized children. Dev. Psychopathol. 2008, 20, 861–880.
- Chassin, L.; Hussong, A.; Barrera, M.; Molina, B.; Trim, R.; Ritter, J. Adolescent substance use. Dev. Psychopathol. 2016, 28, 795–819.
- Peled, M.; Sacks, M. Neurodevelopment in children from high-risk families. Child Dev. 2018, 89, 156–170.
- Perry, B.D. Neurosequential model of therapeutics. J. Child Adolesc. Trauma 2021, 14, 1–15.
- FDA-NIH Biomarker Working Group. BEST Resource: Biomarkers, EndpointS, and other Tools; FDA-NIH: Silver Spring, MD, USA, 2016.
- Arns, M.; Conners, C.K.; Kraemer, H.C. A decade of EEG Theta/Beta Ratio research. J. Atten. Disord. 2013, 17, 374–383.
- Bussalb, A.; Congedo, M.; Barthélemy, Q.; Ojeda, D. Clinical utility of TBR. Biol. Psychol. 2019, 146, 107734.
- Nezhad, M.; et al. EEG biomarkers of attention. Neurosci. Biobehav. Rev. 2025, 158, 105562.
- Davidson, R.J. Frontal EEG asymmetry and emotion. Psychophysiology 1990, 27, 1–17.
- Coan, J.A.; Allen, J.J.B. Frontal EEG asymmetry. Biol. Psychol. 2004, 67, 7–49.
- Smith, E.E.; et al. Affective neuroscience and EEG markers. Neurosci. Biobehav. Rev. 2016, 68, 555–570.
- Goncharova, I.I.; McFarland, D.J.; Vaughan, T.M.; Wolpaw, J.R. EMG contamination. Clin. Neurophysiol. 2003, 114, 1580–1593.
- Whitham, E.M.; et al. Muscle artifacts in EEG. Clin. Neurophysiol. 2007, 118, 1877–1888.
- Trujillo-Barreto, N.J.; et al. EEG and EMG interaction. Neuroimage 2020, 219, 117048.
- Sterman, M.B.; et al. Sensorimotor rhythm. Electroencephalogr. Clin. Neurophysiol. 1974, 36, 1–7.
- Hammond, D.C. Neurofeedback treatment. J. Neurother. 2011, 15, 305–336.
- Gruzelier, J.H. EEG-neurofeedback for cognition. Neurosci. Biobehav. Rev. 2014, 44, 124–141.
- Snyder, S.M.; Hall, J.R. A meta-analysis of quantitative EEG power. J. Clin. Neurophysiol. 2006, 23, 440–455.
- Loo, S.K.; Makeig, S. Clinical utility of EEG in attention disorders. J. Atten. Disord. 2012, 16, 513–520.
- Thibodeau, R.; Jorgensen, R.S.; Kim, S. Depression, anxiety, and frontal EEG asymmetry. J. Abnorm. Psychol. 2006, 115, 715–729.
- McLaughlin, K.A.; Sheridan, M.A. Beyond cumulative risk. Curr. Dir. Psychol. Sci. 2016, 25, 239–245.
| Metric | Definition | ROI/canal |
| Global TBR | θ/(β1+β2) | mean z 19 canals |
| Frontal TBR | θ/(β1+β2) | Fp1, Fp2, F3, F4, F7, F8, Fz |
| Prefrontal TBR | θ/(β1+β2) | Fp1, Fp2, F3, F4 |
| TBR midline | θ/(β1+β2) | Fz, Cz |
| Frontal Alpha Asymmetry FAA | ln(α right) − ln(α left) | Fp2–Fp1 (± F4–F3) |
| Temporal beta stress | mean(β1+β2) | T3, T4 (≈T7/T8) |
| Parietal EMG tension | β2 | P3, P4, Pz |
| SMR composite | Z (Temporal β stress, global θ, symptoms of stress) | — |
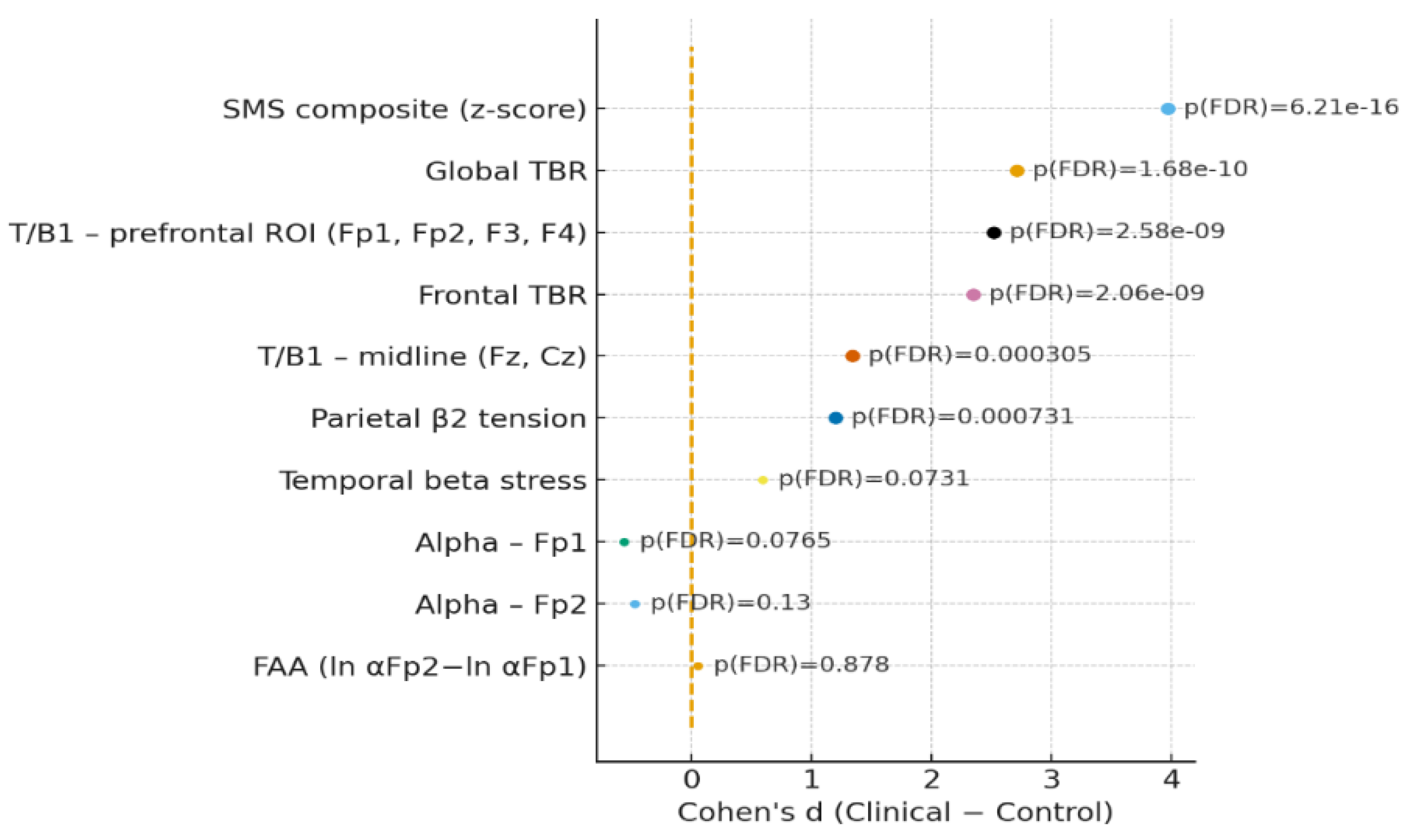
| Marker | M_K | SD_K | M_Z | SD_Z | t | p | d | p_adj |
| SMS composite | 0.814 | 0.438 | -0.651 | 0.374 | 11.891 | 0.0 | 3.6 | 0.000* |
| Frontal TBR | 1.43 | 0.101 | 1.153 | 0.099 | 9.225 | 0.0 | 2.77 | 0.000* |
| Global TBR | 1.367 | 0.068 | 1.189 | 0.065 | 8.905 | 0.0 | 2.68 | 0.000* |
| Prefrontal Theta Beta1 | 2.745 | 0.355 | 1.957 | 0.263 | 8.271 | 0.0 | 2.52 | 0.000* |
| Motivation TBR midline | 2.699 | 0.52 | 2.107 | 0.342 | 4.389 | 0.000 | 1.35 | 0.000* |
| Temporal beta stress | 19.774 | 2.224 | 17.435 | 2.204 | 3.52 | 0.001 | 1.06 | 0.002* |
| Alpha Fp1 | 39.11 | 9.738 | 47.724 | 8.8 | -3.077 | 0.004 | -0.93 | 0.005* |
| Parietal beta2 tension | 15.824 | 2.74 | 13.721 | 2.043 | 2.856 | 0.007 | 0.87 | 0.009* |
| Alpha Fp2 | 42.271 | 7.549 | 46.568 | 7.693 | -1.882 | 0.067 | -0.56 | 0.074 |
| FAA ln | 0.088 | 0.303 | -0.021 | 0.24 | 1.309 | 0.199 | 0.4 | 0.199 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




