Submitted:
10 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Controlled Drug Release and Targeted Drug Delivery
2.1. Kinetics of Controlled Drug Release
2.2. Targeted Drug Delivery
3. Polymethacrylates and Oral Drug Delivery
4. Nanoparticles Containing Eudragit for Oral Drug Delivery
4.1. Inorganic Nanoparticles
4.2. Nanocrystals
4.3. Lipid Nanoparticles
4.3.1. Liposomes
4.3.2. Solid Lipid Nanoparticles
4.3.3. Nanostructured Lipid Carriers
4.4. Polymeric Nanoparticles
5. Discussion
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| 5-ASA | 5-Aminosalicylic acid |
| 5-FUO | 5-Fluorouracil |
| 5-FUO-E-NLCs | 5-Fluorouracil loaded, Eudragit coated nanostructured lipid carriers |
| AUC | Area under the curve |
| AZM | Acetazolamide |
| AZM-SH | Acetazolamide derivative |
| BCS | Biopharmaceutical Classification System |
| SG/LP | Bile salt containing liposomes |
| BIBF | Nintedanib |
| BIBF-NCs | Nintedanib nanocrystals |
| BIBF-NCs@L100 | Eudragit-coated BIBF-NCs |
| BUD | Budenoside |
| BUD-A-MSNPs | Budesonide loaded, amino-functionalised MSNP |
| BUD-A-E-MSNPs | Budesonide loaded, Eudragit-coated, amino-functionalised MSNP |
| BUD-E-LPs | Budesonide-loaded, Eudragit-coated liposomes |
| BUD-E-BSLPs | Budesonide-loaded, bile salt-containing liposomes |
| CHT | Catechin |
| CTAB | Cetyl trimethyl ammonium bromide |
| DAI | Disease Activity Index |
| DMBA | 7,12-Dimethylbenz[a]anthracene |
| DOX | Doxorubicin hydrochloride |
| DDS | Drug delivery system |
| DSS | Dextran sulphate sodium |
| DTN | Dasatinib |
| DTN-E-PNPs | Dasatinib loaded, Eudragit coated polymeric nanoparticles |
| ES@PND-PEG-TPP/DOX | Eudragit coated PND-PEG-TPP/DOX |
| EE | Entrapment efficiency |
| EL100 | Eudragit L100 |
| EL100-55 | Eudragit L100-55 |
| EL30D | Eudragit L30D-55 |
| EGFR | Endothelial growth factor receptor |
| E-PNPs | Iridoid glycoside loaded pH-sensitive polymeric nanoparticles |
| EPR | Enhanced permeability and retention |
| ES100 | Eudragit S100 |
| ES-SG/LP | Eudragit coated, bile salt containing liposomes |
| E@MSND-BUD | Enteric-Coated MSNDs-BUD |
| Eud-MER-MCM-NH2 | Eudragit coated, MER-MCM-NH2 |
| FaSSIF | Fasted-state simulated intestinal fluid |
| FDA | Food and Drug Administration |
| FeSSIF | Fed-state simulated intestinal fluid |
| FS30D | Eudragit FS30D |
| FS100 | Eudragit FS100 |
| hCA IX | Human Carbonic Anhydrase IX |
| HPMC-E5 | Hydroxypropyl methylcellulose E5 |
| IBD | Inflammatory bowel disease |
| IG | Iridoid glycoside |
| INP | Inorganic nanoparticle |
| IVM | Ivermectin |
| LNP | Lipid nanoparticle |
| LP | Liposome |
| MCM | Mesoporous silica nanoparticles MCM 41 |
| MCM-NH2 | Amino-functionalised MCM |
| MER | Meropenem |
| MER-MCM | MER loaded-MCM |
| MER-MCM-NH2 | MER loaded-amino-functionalised MCM |
| MER-MCM-PO3 | MER loaded-phosphonate functionalised MCM |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| MSNP | Mesoporous silica nanoparticle |
| MSND | Dendritic MSNP |
| MSND-BUD | BUD-Loaded MSND |
| NC | Nanocrystal |
| ND | Nanodiamond |
| NDNP | Nanodiamond-based nanoparticle |
| NH2-MNSPs | unloaded amino-functionalized MSNPs |
| NH2-MNSPs@EUS-100 | NH2-MNSPs coated with ES100 |
| NH2-MSNPs/CHT | Catechin-loaded NH2-MNSPs |
| NH2-MSNPs/CHT@EUS-100 | NH2-MNSPs/CHT coated with ES100 |
| NIR | Near Infrared |
| NLC | Nanostructured lipid carrier |
| NP | Nanoparticle |
| MSNPs/CHT | Catechin-loaded MNSPs |
| OFLO | Ofloxacin |
| OXA | Oxaliplatin |
| P-gp | P-glycoprotein |
| PCR | Polymerase chain reaction |
| PDI | Polydispersity index |
| PEG | Polyethylene glycol |
| PLGA | Poly (lactic-co-glycolic acid) |
| PLGA-E-PNPs | Iridoid glycoside loaded, Eudragit coated, PLGA-based polymeric NPs |
| PMMA | Poly (methyl methacrylate) |
| PM | Physical mixture |
| PND | ND coated with polydopamine |
| PND-PEG | PEG-Functionalized PND |
| PND-PEG-TPP | TPP-functionalized PND-PEG |
| PND-PEG-TPP/DOX | Doxorrubicin loaded- PND-PEG-TPP |
| PNP | Polymeric NP |
| Pred | Prednisolone |
| Pred-A-MSNPs | Prednisolone loaded, amino-functionalised mesoporous silica NPs |
| Pred-A-E-MSNPs | Prednisolone loaded, Eudragit coated, amino-functionalised mesoporous silica NPs |
| PS | Particle size |
| RS100 | Eudragit RS100 |
| SAX | Saxagliptin |
| SLN | Solid lipid NPs |
| SLS | Sodium lauryl sulphate |
| SGF | Simulated gastric fluid |
| TAC | Tacrolimus |
| TAC-NLCs | Tacrolimus-loaded nanostructured lipid carriers |
| TAC-NLCs/E FS100 | Tacrolimus-loaded, Eudragit-coated nanostructured lipid carriers |
| TDNPs | Iridoid glycoside loaded time-dependent polymeric NPs |
| TEOS | Tetraethoxysilane |
| TPP | Triphenylphosphonium |
| ZP | Zeta potential |
References
- Milián-Guimerá, C.; McCabe, R.; Thamdrup, L.H. E.; Ghavami, M.; Boisen, A. Smart pills and drug delivery devices enabling next generation oral dosage forms. Journal of Controlled Release 2023, 364, 227–245. [Google Scholar] [CrossRef]
- Wang, D.; Jiang, Q.; Dong, Z.; Meng, T.; Hu, F.; Wang, J.; Yuan, H. Nanocarriers transport across the gastrointestinal barriers: The contribution to oral bioavailability via blood circulation and lymphatic pathway. Advanced Drug Delivery Reviews 2023, 203, 115130. [Google Scholar] [CrossRef] [PubMed]
- Sant, S.; Tao, S.L.; Fisher, O.Z.; Xu, Q.; Peppas, N.A.; Khademhosseini, A. Microfabrication technologies for oral drug delivery. Advanced Drug Delivery Reviews 2012, 64, 496–507. [Google Scholar] [CrossRef]
- Ahadian, S.; Finbloom, J.A.; Mofidfar, M.; Diltemiz, S.E.; Nasrollahi, F.; Davoodi, E.; Hosseini, V.; Mylonaki, I.; Sangabathuni, S.; Montazerian, H.; et al. Micro and nanoscale technologies in oral drug delivery. Advanced Drug Delivery Reviews 2020, 157, 37–62. [Google Scholar] [CrossRef] [PubMed]
- Lou, J.; Duan, H.; Qin, Q.; Teng, Z.; Gan, F.; Zhou, X.; Zhou, X. Advances in Oral Drug Delivery Systems: Challenges and Opportunities. Pharmaceutics 2023, 15, 484. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; Xie, Z.; Li, D.; Kassymova, M.; Zang, S.Q.; Jiang, H.L. Nano-sized metal-organic frameworks: Synthesis and applications. Coordination Chemistry Reviews 2020, 417, 213366. [Google Scholar] [CrossRef]
- Johnsen, H.M.; Filtvedt, W.; Klaveness, J.; Hiorth, M. Nano-strategies for advancing oral drug delivery: Porous silicon particles and cyclodextrin encapsulation for enhanced dissolution of poorly soluble drugs. International journal of pharmaceutics 2024, 666, 124809. [Google Scholar] [CrossRef]
- Singh, K.; Singhal, S.; Pahwa, S.; Sethi, V.A.; Sharma, S.; Singh, P.; Kale, R.D.; Ali, S.W.; Sagadevan, S. Nanomedicine and drug delivery: A comprehensive review of applications and challenges. Nano-Structures and Nano-Objects 2024, 40, 101403. [Google Scholar] [CrossRef]
- Aggas, J.R.; Guiseppi-Elie, A. Responsive Polymers in the Fabrication of Enzyme-Based Biosensors. In Biomaterials Science: An Introduction to Materials in Medicine; Elsevier, 2020; pp. 1267–1286. [Google Scholar]
- Nikam, A.; Sahoo, P.R.; Musale, S.; Pagar, R.R.; Paiva-Santos, A.C.; Giram, P.S. A Systematic Overview of Eudragit® Based Copolymer for Smart Healthcare. Pharmaceutics 2023, 15, 587. [Google Scholar] [CrossRef]
- NCI, Immediate Release Dosage Form ( Code - C42669 ). 2026. Available online: https://evsexplore.semantics.cancer.gov/evsexplore/concept/ncit/C42669.
- Thesaurus, N.C.I. Pharmaceutical Quality/CMC Terminology Files in the National Cancer Institute Thesaurus (NCIt).
- 2026-02-23, N. T.-V. d. R. D., Controlled Release Dosage Form ( Code - C42731 ). Available online: https://evsexplore.semantics.cancer.gov/evsexplore/concept/ncit/C42731.
- 2026-02-23, N. T.-V. d. R. D., Modified Release Dosage Form ( Code - C42712 ). Available online: https://evsexplore.semantics.cancer.gov/evsexplore/concept/ncit/C42712.
- Hong Wen, K.P. Introduction And Overview Of Oral Controlled Release Formulation Design. In Oral Controlled Release Formulation Design and Drug Delivery: Theory to Practice; Hong Wen, K.P., Ed.; Wiley, 2010. [Google Scholar]
- Laracuente, M.L.; Yu, M.H.; McHugh, K.J. Zero-order drug delivery: State of the art and future prospects. Journal of Controlled Release 2020, 327, 834–856. [Google Scholar] [CrossRef]
- Bader, R.A.; Putnam, D.A. Engineering polymer systems for improved drug delivery; John Wiley & Sons, 2014; pp. 492–492. [Google Scholar]
- Attia, M.F.; Anton, N.; Wallyn, J.; Omran, Z.; Vandamme, T.F. An overview of active and passive targeting strategies to improve the nanocarriers efficiency to tumour sites. Journal of Pharmacy and Pharmacology 2019, 71, 1185–1198. [Google Scholar] [CrossRef]
- Amiji, A.S.M.M. Stimuli-responsive Drug Delivery Systems 2018.
- Mitchell, M.J.; Billingsley, M.M.; Haley, R.M.; Wechsler, M.E.; Peppas, N.A.; Langer, R. Engineering precision nanoparticles for drug delivery. Nature Reviews Drug Discovery 2021, 20, 101–124. [Google Scholar] [CrossRef]
- Frey, M.; Bobbala, S.; Karabin, N.; Scott, E. Influences of nanocarrier morphology on therapeutic immunomodulation. Nanomedicine 2018, 13, 1795–1795. [Google Scholar] [CrossRef]
- Garapaty, A.; Champion, J.A. Tunable particles alter macrophage uptake based on combinatorial effects of physical properties. Bioengineering & Translational Medicine 2017, 2, 92–92. [Google Scholar] [CrossRef] [PubMed]
- Huynh, E.; Zheng, G. Cancer nanomedicine: Addressing the dark side of the enhanced permeability and retention effect. Nanomedicine 2015, 10, 1993–1995. [Google Scholar] [CrossRef] [PubMed]
- Poole-Warren, L.; Martens, P.J.; Green, R.A. Biosynthetic polymers for medical applications; Woodhead Publishing, 2016. [Google Scholar]
- Fenton, O.S.; Olafson, K.N.; Pillai, P.S.; Mitchell, M.J.; Langer, R. Advances in Biomaterials for Drug Delivery. Advanced Materials 2018, 30. [Google Scholar] [CrossRef]
- Ekladious, I.; Colson, Y.L.; Grinstaff, M.W. Polymer–drug conjugate therapeutics: advances, insights and prospects. Nature Reviews Drug Discovery 2019, 18, 273–294. [Google Scholar] [CrossRef]
- Hardenia, A.; Maheshwari, N.; Hardenia, S.S.; Dwivedi, S.K.; Maheshwari, R.; Tekade, R.K. Scientific rationale for designing controlled drug delivery systems. In Basic Fundamentals of Drug Delivery; Elsevier, 2018; pp. pp 1–28. [Google Scholar]
- Depan, D.; Shah, J.; Misra, R.D.K. Controlled release of drug from folate-decorated and graphene mediated drug delivery system: Synthesis, loading efficiency, and drug release response. Materials Science and Engineering: C 2011, 31, 1305–1312. [Google Scholar] [CrossRef]
- Zhang, X.; Xing, H.; Zhao, Y.; Ma, Z. Pharmaceutical dispersion techniques for dissolution and bioavailability enhancement of poorly water-soluble drugs. Pharmaceutics 2018, 10, 74. [Google Scholar] [CrossRef]
- Pandi, J.S.; Pavadai, P.; Sundar, L.M.; Sankaranarayanan, M.; Panneerselvam, T.; Pandian, S.R.K.; Kunjiappan, S. Pharmacokinetics and Brain Tumor Delivery Studies of Thymoquinone-Encapsulated Eudragit L100-Coated Solid-Lipid Nanoparticles. Journal of Cluster Science 2025, 36. [Google Scholar] [CrossRef]
- Al-Shaeli, M.; Benkhaya, S.; Al-Juboori, R.A.; Koyuncu, I.; Vatanpour, V. pH-responsive membranes: Mechanisms, fabrications, and applications. Science of the Total Environment 2024, 946. [Google Scholar] [CrossRef]
- Huang, T.; Su, Z.; Hou, K.; Zeng, J.; Zhou, H.; Zhang, L.; Nunes, S.P. Advanced stimuli-responsive membranes for smart separation. Chemical Society Reviews 2023, 52, 4173–4207. [Google Scholar] [CrossRef]
- Zhang, J.; Guo, M.; Luo, M.; Cai, T. Advances in the development of amorphous solid dispersions: The role of polymeric carriers. Asian Journal of Pharmaceutical Sciences 2023, 18, 100834. [Google Scholar] [CrossRef]
- Chen, Z.; Wang, X.; Zhao, N.; Chen, H.; Guo, G. Advancements in pH-responsive nanocarriers: enhancing drug delivery for tumor therapy. Expert Opinion on Drug Delivery 2023, 20, 1623–1642. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Yue, R.; Guan, G.; Zhang, C.; Zhou, Y.; Song, G. Recent development of pH-responsive theranostic nanoplatforms for magnetic resonance imaging-guided cancer therapy. Exploration 2023, 3. [Google Scholar] [CrossRef]
- Patra, C.N.; Priya, R.; Swain, S.; Jena, G.K.; Panigrahi, K.C.; Ghose, D. Pharmaceutical significance of Eudragit: A review. Future Journal of Pharmaceutical Sciences 2017, 3, 33–45. [Google Scholar] [CrossRef]
- Convention, U.S.P. United States Pharmacopeia 32 - National Formulary 27 (USP32-NF27); United States Pharmacopeial Convention, 2009. [Google Scholar]
- Rowe, C.; Sheskey, P.J.; Quinn, M.E. Handbook of Pharmaceutical Excipients, 6th ed.; RPS Publishing, 2009. [Google Scholar]
- Thakral, S.; Thakral, N.K.; Majumdar, D.K. Eudragit®: A technology evaluation. Expert Opinion on Drug Delivery 2013, 10, 131–149. [Google Scholar] [CrossRef]
- Brewer, K.; Blencowe, A. Composition-Property Relationships of pH-Responsive Poly[(2-vinylpyridine)-co-(butyl methacrylate)] Copolymers for Reverse Enteric Coatings. 2023. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Shen, L.; Yang, Y.; Shen, J. Development of nanoparticle-based orodispersible palatable pediatric formulations. International journal of pharmaceutics 2021, 596, 120206. [Google Scholar] [CrossRef]
- EUDRAGIT® L 100 & S 100: Specs & Tests. 14 Aug 2025. Available online: https://studylib.net/doc/25298564/evonik-eudragit-l-100-and-eudragit-s-100-specificationsheet.
- The Global Market Leader for Enteric Release Coatings | EUDRAGIT® - Evonik Industries. 14 Aug 2025. Available online: https://healthcare.evonik.com/en/drugdelivery/oral-drug-delivery/oral-excipients/eudragitportfolio/delayed-release.
- EUDRAGIT® L 100. 14 Aug 2025. Available online: https://www.evonik.com/en/products/hc/pr_52000884.html.
- Evonik, Functional excipients to take control of your release profile Versatility and reliability for oral solid dosage forms. 13 Aug 2025. Available online: www.evonik.com/healthcare.
- EUDRAGIT® Functional Polymers for Sustained Release - Evonik Industries. In [Internet]. 14 Aug 2025. Available online: https://healthcare.evonik.com/en/drugdelivery/oral-drug-delivery/oral-excipients/eudragit-portfolio/sustained-release.
- Zhang, F. Melt-Extruded Eudragit® FS-Based Granules for Colonic Drug Delivery. AAPS PharmSciTech 2015, 17, 56. [Google Scholar] [CrossRef]
- Cardoso, A.M. L.; Oliveira, E.E.; Machado, B.A.S.; Marcelino, H.R. Eudragit®-based nanoparticles for controlled release through topical use. Journal of Nanoparticle Research 2023, 25. [Google Scholar] [CrossRef]
- Nassar, T.; Rom, A.; Nyska, A.; Benita, S. Novel double coated nanocapsules for intestinal delivery and enhanced oral bioavailability of tacrolimus, a P-gp substrate drug. Journal of Controlled Release 2009, 133, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Huang, I.P.; Sun, S.P.; Cheng, S.H.; Lee, C.H.; Wu, C.Y.; Yang, C.S.; Lo, L.W.; Lai, Y.K. Enhanced chemotherapy of cancer using pH-sensitive mesoporous silica nanoparticles to antagonize P-glycoprotein-mediated drug resistance. Molecular Cancer Therapeutics 2011, 10, 761–769. [Google Scholar] [CrossRef] [PubMed]
- Mohammadzadeh, R.; Baradaran, B.; Valizadeh, H.; Yousefi, B.; Zakeri-Milani, P. Reduced ABCB1 Expression and Activity in the Presence of Acrylic Copolymers. Advanced Pharmaceutical Bulletin 2014, 4, 219–219. [Google Scholar] [CrossRef]
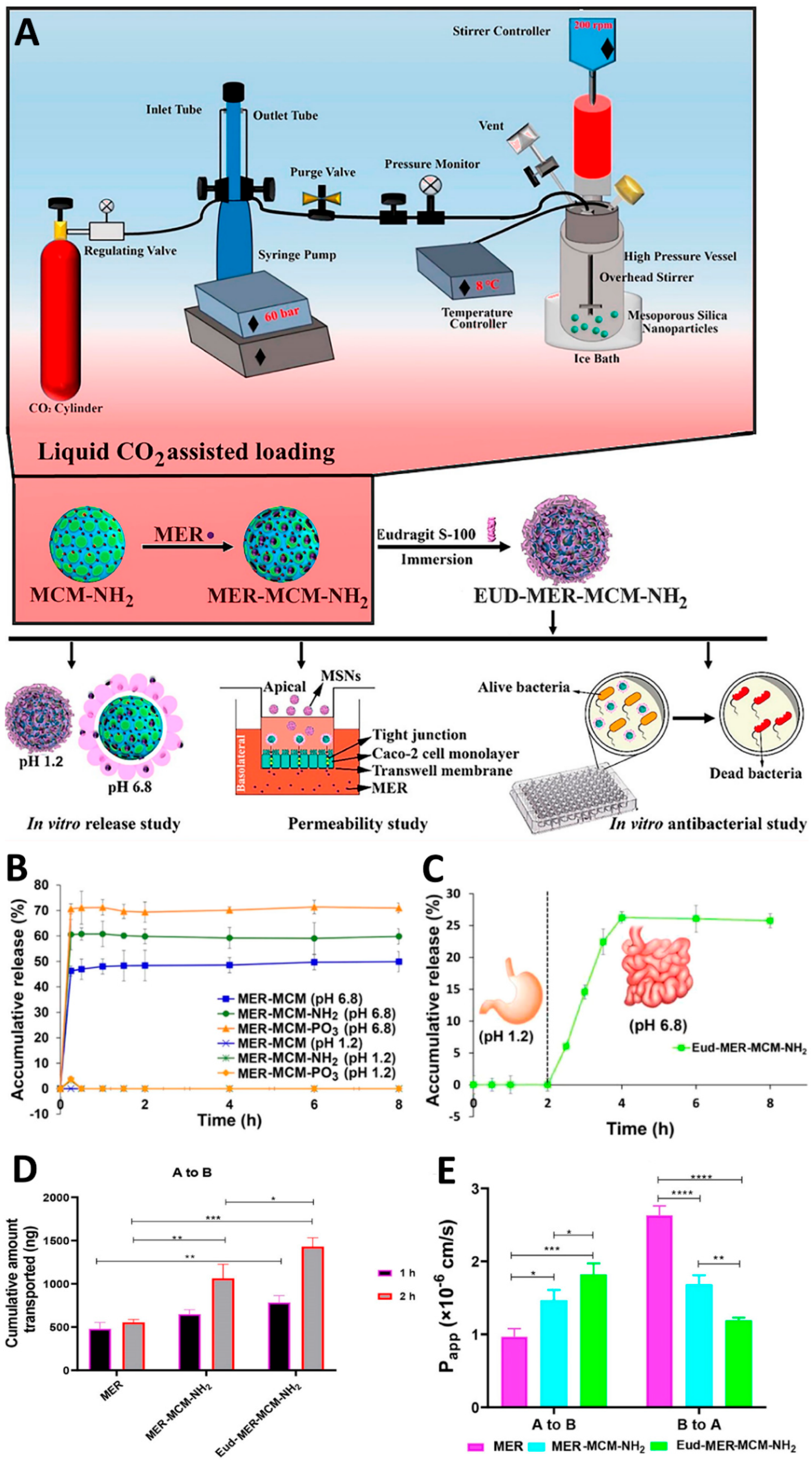
- Raza, A.; Sime, F.B.; Cabot, P.J.; Roberts, J.A.; Falconer, J.R.; Kumeria, T.; Popat, A. Liquid CO2 formulated mesoporous silica nanoparticles for pH-responsive oral delivery of meropenem. ACS Biomaterials Science and Engineering 2021, 7, 1836–1853. [Google Scholar] [CrossRef]
- Gupta, A.; Sood, A.; Dhiman, A.; Shrimali, N.; Singhmar, R.; Guchhait, P.; Agrawal, G. Redox responsive poly(allylamine)/eudragit S-100 nanoparticles for dual drug delivery in colorectal cancer. Biomaterials Advances 2022, 143, 213184. [Google Scholar] [CrossRef]
- Gupta, A.; Dhiman, A.; Sood, A.; Bharadwaj, R.; Silverman, N.; Agrawal, G. Dextran/eudragit S-100 based redox sensitive nanoparticles for colorectal cancer therapy. Nanoscale 2023, 15, 3273–3283. [Google Scholar] [CrossRef]
- Khalid, H.M. B.; Rasul, A.; Shah, S.; Abbas, G.; Mahmood, A. Disulfide Bridged Nanoparticles of Thiolated Sodium Alginate and Eudragit RS100 for Oral Delivery of Paclitaxel: In Vitro and In Vivo Evaluation. ACS Omega 2023, 8, 9662–9672. [Google Scholar] [CrossRef]
- Leichner, C.; Jelkmann, M.; Bernkop-Schnürch, A. Thiolated polymers: Bioinspired polymers utilizing one of the most important bridging structures in nature. Advanced Drug Delivery Reviews 2019, 151-152, 191–221. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, O.; Shah, S.; Abbas, G.; Rasul, A.; Hanif, M.; Ashfaq, M.; Afzal, Z. Moxifloxacin loaded nanoparticles of disulfide bridged thiolated chitosan-eudragit RS100 for controlled drug delivery. International journal of biological macromolecules 2021, 182, 2087–2096. [Google Scholar] [CrossRef] [PubMed]
- Bhat, S.S.; Mukherjee, D.; Sukharamwala, P.; Dehuri, R.; Murali, A.; Teja, B.V. Thiolated polymer nanocarrier reinforced with glycyrrhetinic acid for targeted delivery of 5-fluorouracil in hepatocellular carcinoma. Drug delivery and translational research 2021, 11, 2252–2269. [Google Scholar] [CrossRef]
- Abbas, N.; Rasul, A.; Abbas, G.; Shah, S.; Hanif, M. Targeted delivery of aspirin and metformin to colorectal cancer using disulfide bridged nanoparticles of thiolated pectin and thiolated Eudragit RL100. Materials Today Communications 2023, 35, 105586. [Google Scholar] [CrossRef]
- EUDRAGIT® FS 100 - Pharma Excipients. 16 Aug 2025. Available online: https://www.pharmaexcipients.com/product/eudragit-fs-100/.
- EUDRAGIT®, E.P.O. 29 Aug 2025. Available online: https://www.evonik.com/en/products/hc/pr_52011259.html.
- EUDRAGIT® E 100. 29 Aug 2025. Available online: https://www.evonik.com/en/products/hc/pr_52000110.html.
- Balogh, A.; Farkas, B.; Domokos, A.; Farkas, A.; Démuth, B.; Borbás, E.; Nagy, B.; Marosi, G.; Nagy, Z.K. Controlled-release solid dispersions of Eudragit® FS 100 and poorly soluble spironolactone prepared by electrospinning and melt extrusion. European Polymer Journal 2017, 95, 406–417. [Google Scholar] [CrossRef]
- Oral Drug Delivery Market Size & Industry Growth Forecast By 2029. 19 Feb 2025. Available online: https://www.databridgemarketresearch.com/reports/global-oral-drug-delivery-market.
- Sharma, A.; Jangra, N.; Dheer, D.; Jha, S.K.; Gupta, G.; Puri, V.; Kesharwani, P. Understanding the journey of biopolymeric nanoformulations for oral drug delivery: Conventional to advanced treatment approaches. European Polymer Journal 2024, 218, 113338–113338. [Google Scholar] [CrossRef]
- Mok, Z.H. The effect of particle size on drug bioavailability in various parts of the body. Pharmaceutical Science Advances 2024, 2, 100031–100031. [Google Scholar] [CrossRef]
- Tarn, D.; Ashley, C.E.; Xue, M.; Carnes, E.C.; Zink, J.I.; Brinker, C.J. Mesoporous silica nanoparticle nanocarriers: Biofunctionality and biocompatibility. Accounts of Chemical Research 2013, 46, 792–801. [Google Scholar] [CrossRef]
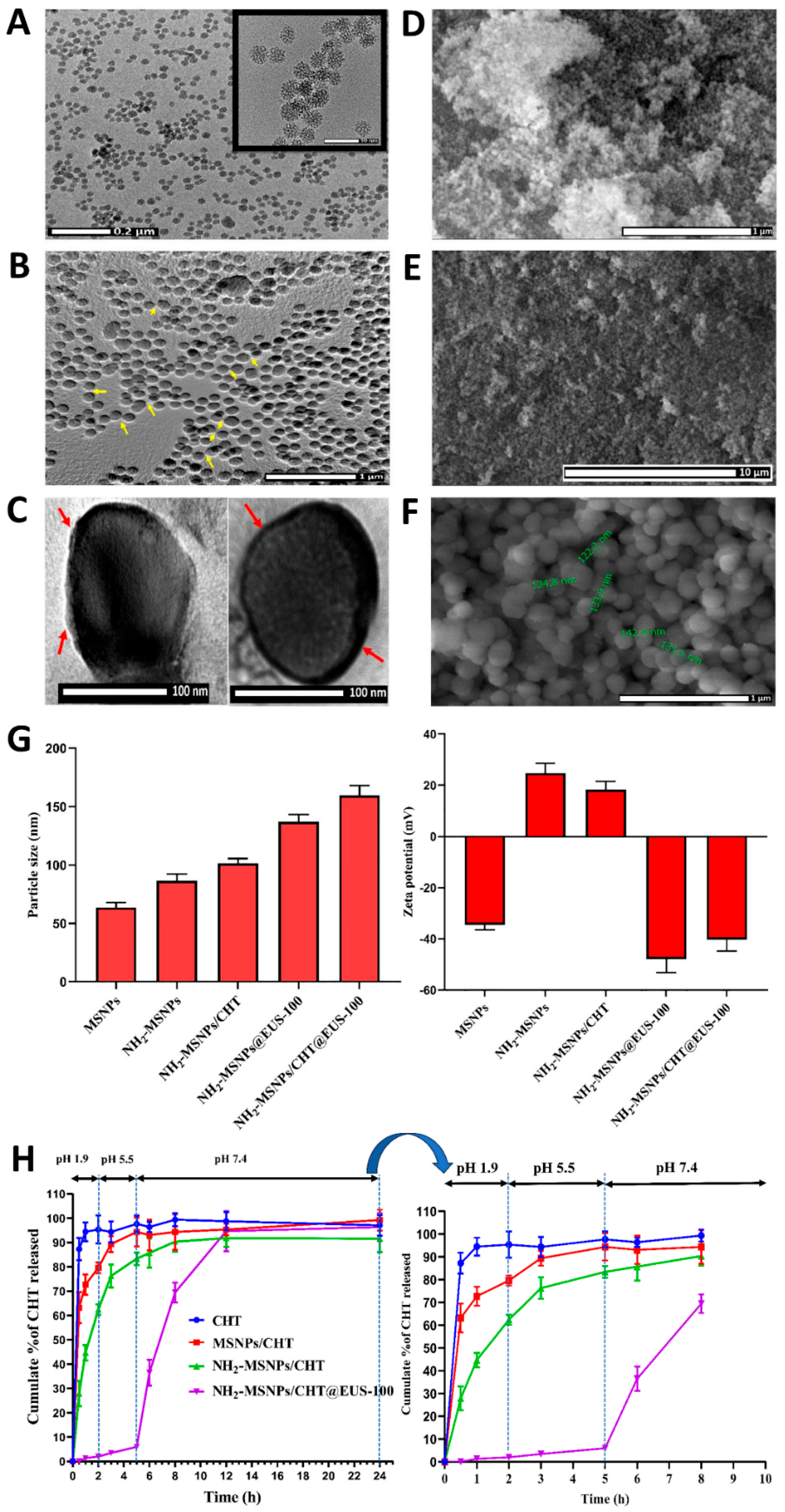
- Kassem, A.M.; Almukainzi, M.; Faris, T.M.; Ibrahim, A.H.; Anwar, W.; Elbahwy, I.A.; El-Gamal, F.R.; Zidan, M.F.; Akl, M.A.; Abd-ElGawad, A.M.; et al. A pH-sensitive silica nanoparticles for colon-specific delivery and controlled release of catechin: Optimization of loading efficiency and in vitro release kinetics. European Journal of Pharmaceutical Sciences 2024, 192, 106652. [Google Scholar] [CrossRef]
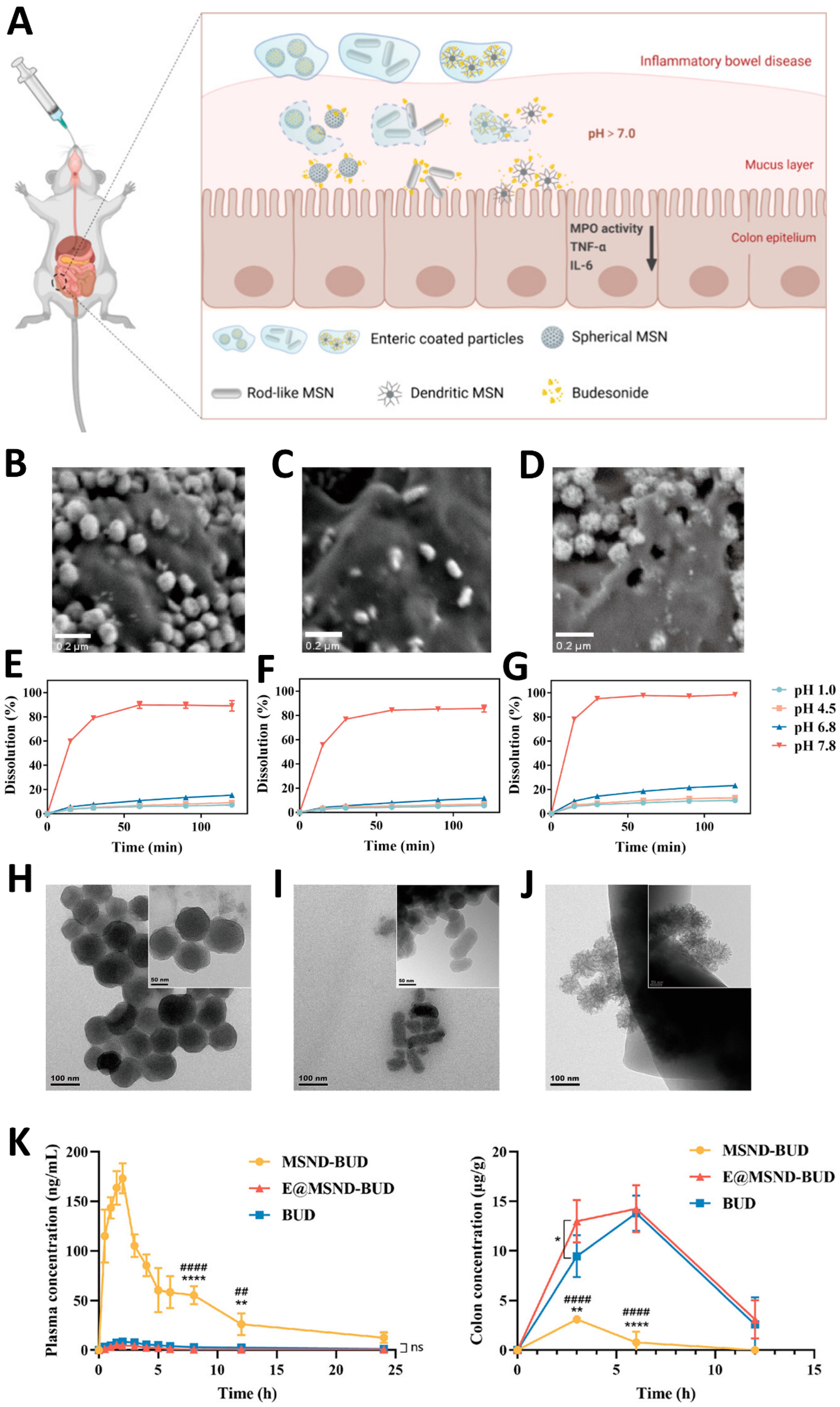
- Li, Q.; Liu, W.; Liu, K.; Dong, Z.; Kong, W.; Lu, X.; Wei, Y.; Wu, W.; Yang, J.; Qi, J. The Role of Nanoparticle Morphology on Enhancing Delivery of Budesonide for Treatment of Inflammatory Bowel Disease. ACS Applied Materials and Interfaces 2024, 16, 33081–33092. [Google Scholar] [CrossRef]
- Qu, Z.; Wong, K.Y.; Moniruzzaman, M.; Begun, J.; Santos, H.A.; Hasnain, S.Z.; Kumeria, T.; McGuckin, M.A.; Popat, A. One-Pot Synthesis of pH-Responsive Eudragit-Mesoporous Silica Nanocomposites Enable Colonic Delivery of Glucocorticoids for the Treatment of Inflammatory Bowel Disease. Advanced Therapeutics 2021, 4. [Google Scholar] [CrossRef]
- Marcelo, G.A.; Montpeyó, D.; Galhano, J.; Martínez-Máñez, R.; Capelo-Martínez, J.L.; Lorenzo, J.; Lodeiro, C.; Oliveira, E. Development of New Targeted Nanotherapy Combined with Magneto-Fluorescent Nanoparticles against Colorectal Cancer. International journal of molecular sciences 2023, 24, 6612. [Google Scholar] [CrossRef]
- Su, Y.; Pan, H.; Wang, J.; Liu, D.; Pan, W. Eudragit S100 coated nanodiamond-based nanoparticles as an oral chemo-photothermal delivery system for local treatment of colon cancer. Colloids and Surfaces B: Biointerfaces 2024, 237. [Google Scholar] [CrossRef]
- Lopez-Vidal, L.; Parodi, P.; Actis, M.R.; Camacho, N.; Real, D.A.; Paredes, A.J.; Irazoqui, F.J.; Real, J.P.; Palma, S.D. Formulation and optimization of pH-sensitive nanocrystals for improved oral delivery. Drug delivery and translational research 2024, 14, 1301–1318. [Google Scholar] [CrossRef]
- Che, J.; Fu, Y.; Li, Y.; Zhang, Y.; Yin, T.; Gou, J.; Tang, X.; Wang, Y.; He, H. Eudragit L100-coated nintedanib nanocrystals improve oral bioavailability by reducing drug particle size and maintaining drug supersaturation. International journal of pharmaceutics 2024, 658, 124196. [Google Scholar] [CrossRef]
- Alghurabi, H.; Muhammad, H.J.; Tagami, T.; Ogawa, K.; Ozeki, T. Optimization, cellular uptake, and in vivo evaluation of Eudragit S100-coated bile salt-containing liposomes for oral colonic delivery of 5-aminosalicylic acid. International journal of pharmaceutics 2023, 648, 123597. [Google Scholar] [CrossRef] [PubMed]
- Alghurabi, H.; Tagami, T.; Ogawa, K.; Ozeki, T. Preparation, Characterization, and In Vitro Evaluation of Eudragit S100-Coated Bile Salt-Containing Liposomes for Oral Colonic Delivery of Budesonide. Polymers 2022, 14, 2693. [Google Scholar] [CrossRef] [PubMed]
- Alhamhoom, Y.; Ravi, G.; Osmani, R.A. M.; Hani, U.; Prakash, G.M. Formulation, Characterization, and Evaluation of Eudragit-Coated Saxagliptin Nanoparticles Using 3 Factorial Design Modules. Molecules 2022, 27, 7510. [Google Scholar] [CrossRef] [PubMed]
- Golla, V.S. K.; Boddu, P.; Nageswara, S. Statistical Experimental Approach in Designing pH-Sensitive Oxaliplatin Lipid Nanoparticles for Application in Colorectal Cancer Therapy. BioNanoScience 2023, 13, 1100–1109. [Google Scholar] [CrossRef]
- Altaf, S.; Zeeshan, M.; Ali, H.; Zeb, A.; Afzal, I.; Imran, A.; Mazhar, D.; Khan, S.; Khan, F.A. pH-Sensitive Tacrolimus loaded nanostructured lipid carriers for the treatment of inflammatory bowel disease. European Journal of Pharmaceutics and Biopharmaceutics 2024, 204, 114461. [Google Scholar] [CrossRef]
- Borderwala, K.; Rathod, S.; Yadav, S.; Vyas, B.; Shah, P. Eudragit S-100 Surface Engineered Nanostructured Lipid Carriers for Colon Targeting of 5-Fluorouracil: Optimization and In Vitro and In Vivo Characterization. AAPS PharmSciTech 2021, 22. [Google Scholar] [CrossRef]
- Gao, C.; Yu, S.; Zhang, X.; Dang, Y.; Han, D.D.; Liu, X.; Han, J.; Hui, M. Dual functional eudragit® s100/l30d-55 and plga colon-targeted nanoparticles of iridoid glycoside for improved treatment of induced ulcerative colitis. International journal of nanomedicine 2021, 16, 1405–1422. [Google Scholar] [CrossRef]
- G, H.; Patil, A.; Mg, H.; Redhwan, M.A. M.; Guha, S. Development, optimization, and characterization of Eudragit-based nanoparticles for Dasatinib delivery. Journal of Biomaterials Science, Polymer Edition 2024. [Google Scholar] [CrossRef]
- Yaqoob, A.A.; Ahmad, H.; Parveen, T.; Ahmad, A.; Oves, M.; Ismail, I.M.I.; Qari, H.A.; Umar, K.; Ibrahim, M.N.M. Recent Advances in Metal Decorated Nanomaterials and Their Various Biological Applications: A Review. Frontiers in Chemistry 2020, 8, 528583–528583. [Google Scholar] [CrossRef]
- Noreen, S.; Maqbool, A.; Maqbool, I.; Shafique, A.; Khan, M.M.; Junejo, Y.; Ahmed, B.; Anwar, M.; Majeed, A.; Abbas, M.; et al. Multifunctional mesoporous silica-based nanocomposites: Synthesis and biomedical applications. Materials Chemistry and Physics 2022, 285, 126132. [Google Scholar] [CrossRef]
- Elmowafy, M.; Alruwaili, N.K.; Ahmad, N.; Kassem, A.M.; Ibrahim, M.F. Quercetin-Loaded Mesoporous Silica Nanoparticle–Based Lyophilized Tablets for Enhanced Physicochemical Features and Dissolution Rate: Formulation, Optimization, and In Vitro Evaluation. AAPS PharmSciTech 2023, 24. [Google Scholar] [CrossRef]
- Juère, E.; Favero, G.D.; Masse, F.; Marko, D.; Popat, A.; Florek, J.; Caillard, R.; Kleitz, F. Gastro-protective protein-silica nanoparticles formulation for oral drug delivery: In vitro release, cytotoxicity and mitochondrial activity. European Journal of Pharmaceutics and Biopharmaceutics 2020, 151, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, Z.; Subramaniam, S.; Khan, G.M.; Abeer, M.M.; Qu, Z.; Janjua, T.; Kumeria, T.; Batra, J.; Popat, A. Encapsulation and Controlled Release of Resveratrol Within Functionalized Mesoporous Silica Nanoparticles for Prostate Cancer Therapy. Frontiers in Bioengineering and Biotechnology 2019, 7, 482266–482266. [Google Scholar] [CrossRef]
- Estevão, B.M.; Miletto, I.; Hioka, N.; Marchese, L.; Gianotti, E. Mesoporous Silica Nanoparticles Functionalized with Amino Groups for Biomedical Applications. ChemistryOpen 2021, 10, 1251–1259. [Google Scholar] [CrossRef]
- Catechin | C15H14O6 | CID 9064 - PubChem.
- Reina, G.; Zhao, L.; Bianco, A.; Komatsu, N. Chemical Functionalization of Nanodiamonds: Opportunities and Challenges Ahead. Angewandte Chemie - International Edition 2019, 58, 17918–17929. [Google Scholar] [CrossRef] [PubMed]
- Kushwaha, A.K.; John, M.; Misra, M.; Menezes, P.L. Nanocrystalline Materials: Synthesis, Characterization, Properties, and Applications. Crystals 2021, 11, 1317–1317. [Google Scholar] [CrossRef]
- How Lipid Nanoparticles Overcome Solubility Challenges For Oral And Injectable Formulations. 27 Feb 2025. Available online: https://www.pharmaceuticalonline.com/doc/how-lipid-nanoparticles-overcome-solubility-challenges-for-oral-and-injectable-formulations-0001.
- Exploring Lipid Nanoparticles For Drug Delivery. 25 Feb 2025. Available online: https://www.pharmaceuticalonline.com/doc/exploring-lipid-nanoparticles-for-drug-delivery-0001.
- Lipid Nanoparticles Are Having A Breakout Moment. 4 Mar 2025. Available online: https://www.pharmaceuticalonline.com/doc/lipid-nanoparticles-are-having-a-breakout-moment-0001.
- Liposomes Challenges And Opportunities. 27 Feb 2025. Available online: https://www.pharmaceuticalonline.com/doc/liposomes-challenges-and-opportunities-0001.
- Alavi, M.; Karimi, N.; Safaei, M. Application of various types of liposomes in drug delivery systems. Advanced Pharmaceutical Bulletin 2017, 7, 3–9. [Google Scholar] [CrossRef]
- Berends, S.E.; Strik, A.S.; Löwenberg, M.; D’Haens, G.R.; Mathôt, R.A.A. Clinical Pharmacokinetic and Pharmacodynamic Considerations in the Treatment of Ulcerative Colitis. Clinical Pharmacokinetics 2018, 58, 15–37. [Google Scholar] [CrossRef]
- Nakashima, J.; Patel, P.; Preuss, C.V. Mesalamine (USAN). StatPearls 2024. [Google Scholar]
- Duan, Y.; Dhar, A.; Patel, C.; Khimani, M.; Neogi, S.; Sharma, P.; Siva Kumar, N. A brief review on solid lipid nanoparticles: part and parcel of contemporary drug delivery systems. RSC advances 2020, 10, 26777–26791. [Google Scholar] [CrossRef] [PubMed]
- Elmowafy, M.; Al-Sanea, M.M. Nanostructured lipid carriers (NLCs) as drug delivery platform: Advances in formulation and delivery strategies. Saudi Pharmaceutical Journal SPJ 2021, 29, 999–999. [Google Scholar] [CrossRef]
- Beloqui, A.; Solinís, M.Á; Rodríguez-Gascón, A.; Almeida, A.J.; Préat, V. Nanostructured lipid carriers: Promising drug delivery systems for future clinics. Nanomedicine: Nanotechnology, Biology, and Medicine 2016, 12, 143–161. [Google Scholar] [CrossRef]
- Gurjar, R.; Chan, C.Y. S.; Curley, P.; Sharp, J.; Chiong, J.; Rannard, S.; Siccardi, M.; Owen, A. Inhibitory Effects of Commonly Used Excipients on P-Glycoprotein in Vitro. Molecular pharmaceutics 2018, 15, 4835–4842. [Google Scholar] [CrossRef]
- Mady, O.Y.; Khaled, S.; Hedaya, A.A.; Abdine, N.; Haggag, Y. Formulation and characterization of novel oral pH-sensitive electrospun nanofibers for boosting dissolution and penetration of model class IV drug. Pharmaceutical Development and Technology 2025, 30, 712–727. [Google Scholar] [CrossRef] [PubMed]
- Cano, A.; Sánchez-López, E.; Ettcheto, M.; López-Machado, A.; Espina, M.; Souto, E.B.; Galindo, R.; Camins, A.; García, M.L.; Turowski, P. Current advances in the development of novel polymeric nanoparticles for the treatment of neurodegenerative diseases. Nanomedicine 2020, 15, 1239–1261. [Google Scholar] [CrossRef] [PubMed]
- Zielinska, A.; Carreiró, F.; Oliveira, A.M.; Neves, A.; Pires, B.; Venkatesh, D.N.; Durazzo, A.; Lucarini, M.; Eder, P.; Silva, A.M.; et al. Polymeric Nanoparticles: Production, Characterization, Toxicology and Ecotoxicology. Molecules 2020, 25, 3731–3731. [Google Scholar] [CrossRef] [PubMed]
- Naeem, M.; Bae, J.; Oshi, M.A.; Kim, M.S.; Moon, H.R.; Lee, B.L.; Im, E.; Jung, Y.; Yoo, J.W. Colon-targeted delivery of cyclosporine A using dual-functional eudragit® FS30D/PLGA nanoparticles ameliorates murine experimental colitis. International journal of nanomedicine 2018, 13, 1225–1240. [Google Scholar] [CrossRef]
- Hassouneh, W.B.; Al-Ghazawi, M.A.; Saleh, M.I.; Najib, N. Population Pharmacokinetics of Dasatinib in Healthy Subjects. Pharmaceuticals 2024, 17, 671–671. [Google Scholar] [CrossRef]
- 70 years of EUDRAGIT - Coatings for targeted drug delivery. 8 Sep 2025. Available online: https://www.pharmaexcipients.com/news/70-years-eudragit/?utm_source=chatgpt.com.
- Center For Drug Evaluation And Research Application Number: 203389Orig1s000 PHARMACOLOGY REVIEW(S).
- Eisele, J.; Haynes, G.; Rosamilia, T. Characterisation and toxicological behaviour of Basic Methacrylate Copolymer for GRAS evaluation. Regulatory Toxicology and Pharmacology 2011, 61, 32–43. [Google Scholar] [CrossRef]
- Esporrín-Ubieto, D.; Sonzogni, A.S.; Fernández, M.; Acera, A.; Matxinandiarena, E.; Cadavid-Vargas, J.F.; Calafel, I.; Schmarsow, R.N.; Müller, A.J.; Larrañaga, A.; et al. The role of Eudragit® as a component of hydrogel formulations for medical devices. Journal of Materials Chemistry B 2023, 11, 9276–9289. [Google Scholar] [CrossRef]
- Mathes, D.; Macedo, L.B.; Pieta, T.B.; Maia, B.C.; Rolim, C.M.B.; Nogueira-Librelotto, D.R. The role of polymer type and surfactant composition on the toxicological profile of nanoparticles: an in vitro comparative study. Journal of Biomaterials Science, Polymer Edition 2025. [Google Scholar] [CrossRef]
- Ronzani, C.; Belle, C.V.; Didier, P.; Spiegelhalter, C.; Pierrat, P.; Lebeau, L.; Pons, F. Lysosome mediates toxicological effects of polyethyleneimine-based cationic carbon dots. Journal of Nanoparticle Research 2019, 21, 1–17. [Google Scholar] [CrossRef]
- Machado, F.R.; Bortolotto, V.C.; Araujo, S.M.; Dahleh, M.M.M.; Fernandes, E.J.; Musachio, E.A.S.; Funguetto-Ribeiro, A.C.; Haas, S.E.; Guerra, G.P.; Prigol, M.; et al. Toxicological analysis of chronic exposure to polymeric nanocapsules with different coatings in Drosophila melanogaster. Comparative Biochemistry and Physiology Part - C: Toxicology and Pharmacology 2024, 283. [Google Scholar] [CrossRef]
- Tehrani, S.F.; Bharadwaj, P.; Chain, J.L.; Roullin, V.G. Purification processes of polymeric nanoparticles: How to improve their clinical translation? Journal of Controlled Release 2023, 360, 591–612. [Google Scholar] [CrossRef] [PubMed]
- Vaughn, N.K.V.M. Oral rapamycin nanoparticle preparations. 2014. [Google Scholar]
- Vaughn, N.K.V.M. Oral rapamycin nanoparticle preparations and use. 2014.
- Sirkar, Kamalesh; Singh, R.P. Dhananjay; Chen, Dengyue. System and method for continuous polymer coating of particles. 2014. [Google Scholar]
- Yu Qian, L. J. Z., Zhi Min WU, Li Ying ZHOU, Wei Jiang, Li Ling, Qian Luo, Xin Dong GUO Enteric-coated capsule containing cationic nanoparticles for oral insulin delivery. Enteric-coated capsule containing cationic nanoparticles for oral insulin delivery 2012.
- A kind of enteric solubility nano-particle of load insulin and its preparation method and application. 2018.
- Chopra, Sunandini; Wang, R.K. Amy; Farokhzad, Omid C.; Zhang, Xue-Qing. Nanoparticles with pH Triggered Drug Release. 2016. [Google Scholar]
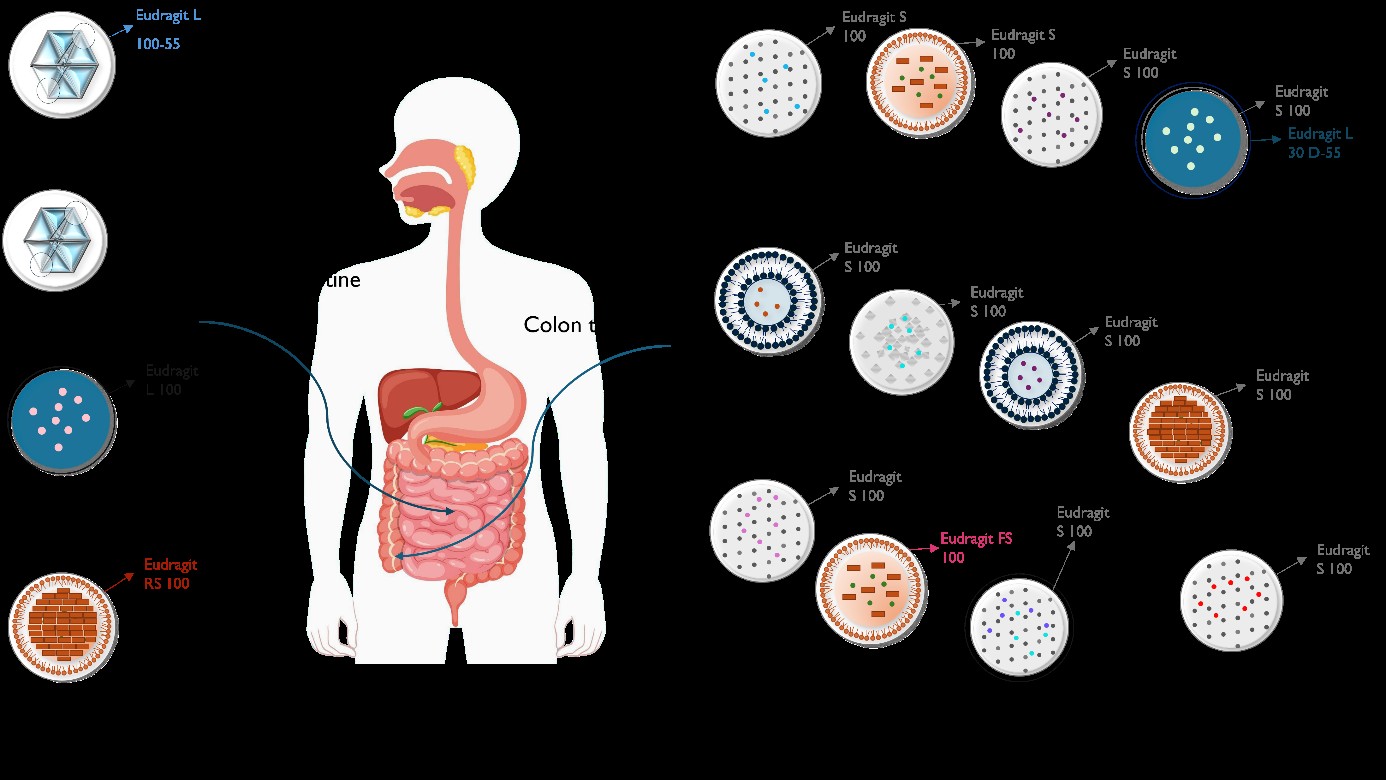
| Grade | Chemical name | pH-dependent solubility | Glass Transition Temperature (ºC) | Physical appearance | Applications | Reference |
| Eudragit E100 | Poly (butyl methacrylate, (2-dimethylaminoethyl) methacrylate, methyl methacrylate) 1:2:1 | Soluble in gastric fluid below pH 5 | 48 |
Granules | Increased bioavailability and dissolution profile; high oral bioavailability. | [10,38,39,60,61,62,63]. |
| Eudragit E PO | Powder | |||||
|
Eudragit L 100 |
Poly (methacrylic acid, methyl methacrylate) 1:1 | Soluble in intestinal fluid from pH 6 | 150 | Powder | Increased oral absorption, increased taste masking, controlled release, enteric targeted drug delivery, delayed release profile, high oral bioavailability. | |
| Eudragit L30 D-55 | Poly (methacrylic acid, ethyl acrylate) 1:1 | Soluble in intestinal fluid from pH 5.5 (resistant to gastric juice but readily dissolves at pH above 5.5) | 110 | Aqueous dispersion | Enteric coating | |
| Eudragit S100 | Poly (methacrylic acid, methyl methacrylate) 1:2 | Soluble in intestinal fluid from pH 7 | 150 | Powder | Increased oral absorption, increased taste masking, controlled release, colonic-specific drug delivery, delayed release profile, high oral bioavailability. | |
| Eudragit FS30 D | Poly (methyl acrylate, methyl methacrylate, methacrylic acid) 7:3:1 | Soluble in intestinal fluid from pH 7 | 48 | Aqueous dispersion | Increased oral absorption, increased taste masking, controlled release, colonic-specific drug delivery, delayed release profile, high oral bioavailability. | |
| Eudragit FS100 | Soluble in intestinal fluid from pH 7 | 55.6 | Granules | |||
| Eudragit RL100 | Poly (ethyl acrylate, methyl methacrylate, trimethylammonioethyl methacrylate chloride) 1:2:0.2 | High permeability, insoluble (pH-independent) | 70 | Granules | Sustained release, improved permeation and increased bioavailability and shelf life. | |
| Eudragit RS100 | Poly (ethyl acrylate, methyl methacrylate, trimethylammonioethyl methacrylate chloride) 1:2:0.1 | Low permeability, insoluble (pH-independent) | 64 |
Granules | Sustained release, improved permeation and increased bioavailability and shelf life. | |
| Eudragit RS PO | Powder |
| Nanoparticle Type | Drug | Eudragit Grade | Production Method | Physicochemical Properties | Target Site | Reference |
| Mesoporous Silica Nanoparticles | Catechin |
Eudragit S100 | Rotary evaporation method followed by polymeric coating | Size:150 nm ZP: - 40.25 mV PDI: 0.23±0.09 AE: 82% |
Colon | [68] |
| Budesonide | Eudragit S100 | CTAB-templated method followed by polymeric coating |
Size: 110 nm (spherical); 115 x 45 nm (rod-like); 100 nm (dendritic) (these values correspond to uncoated particles) ZP: -28±0.38 mV (spherical); -21.61±1.35 mV (rod-like); -22.23±1.21 mV (dendritic) EE: 44% (spherical, rod-like, dendritic) |
Colon | [69] | |
| Prednisolone and Budesonide | Eudragit S100 | Rotary evaporation method followed by polymeric coating | Size: ≈238 nm (Pred); ≈242 nm (BUD) ZP: -19.2±2.8 mV (Pred); -19.5±3.5 mV (BUD) PDI: 0.42 ± 0.11 (Pred); 0.45 ± 0.16 (BUD) LE: 95.2% (Pred); 82.0% (BUD) |
Colon | [70] | |
| Meropenem | Eudragit S100 | CTAB-templated method followed by polymeric coating | Size: 645 nm ZP: -9 mV LC: 24.1% |
Small intestine | [52] | |
| Ofloxacin and Doxorubicin | Eudragit S100 Eudragit L100 | Modified co-precipitation, silica coating and rhodamine labelling, followed by polymeric coating | HD: ≈450 nm ZP: -24.0±1.7 mV LC: 287.5 ugDox/mgNP; 274.0 ugDox/mgNP + 140 ugOflo/mgNP; 182.8 ugOflo/mgNP |
Colon | [71] | |
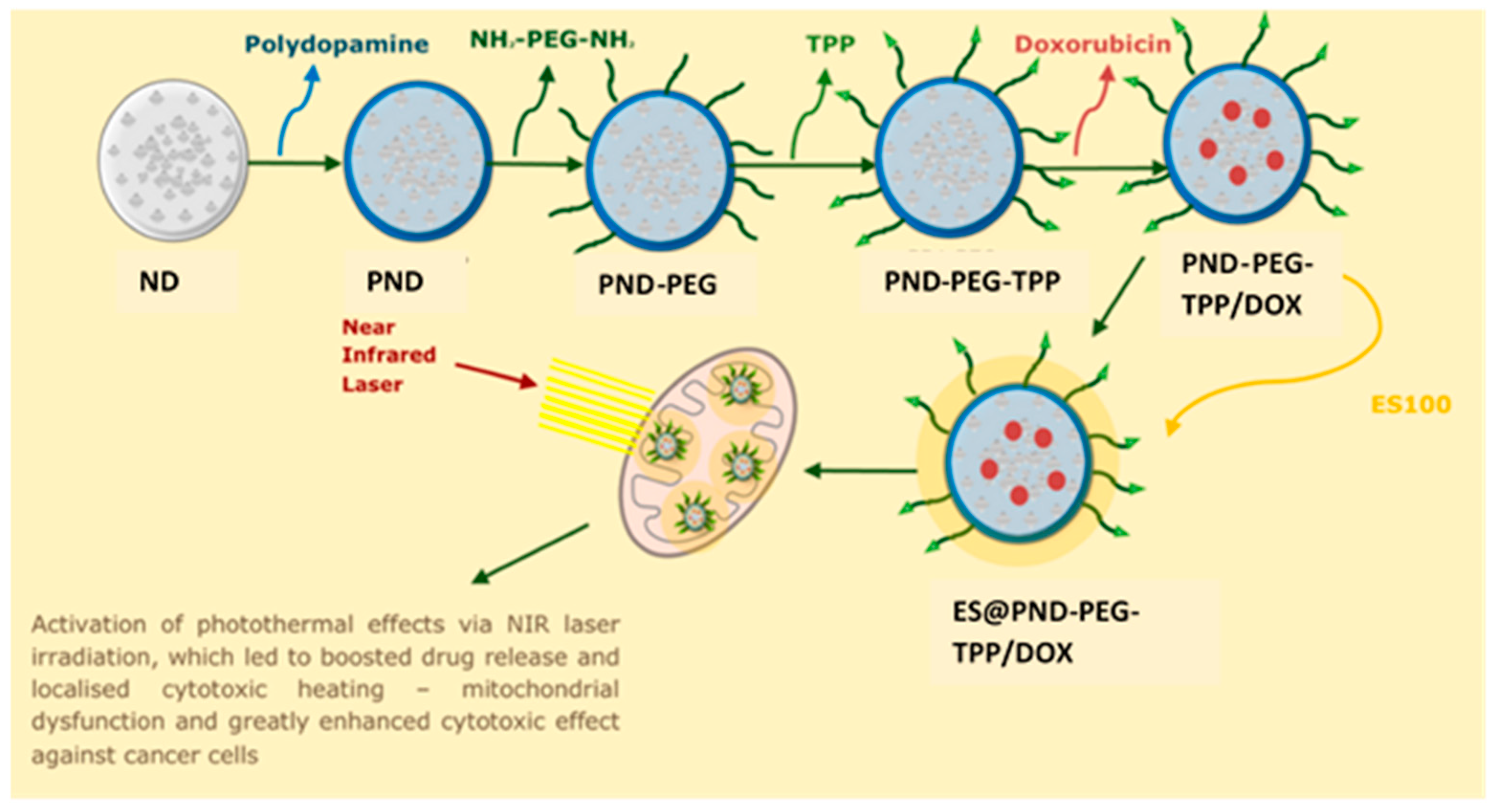
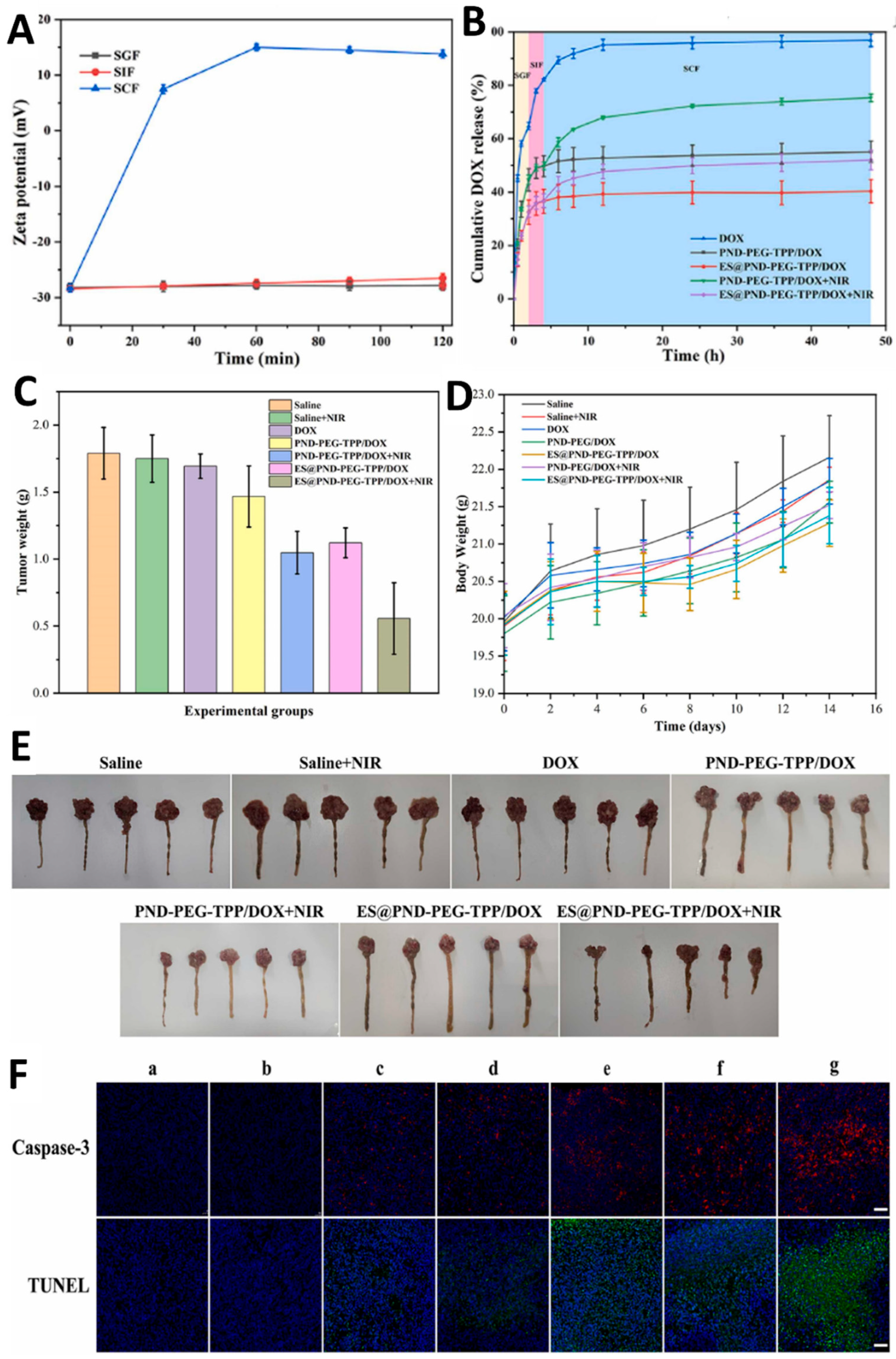
| Nanodiamond-Based Nanoparticles | Doxorubicin | Eudragit S100 | Polydopamine coating followed by solvent evaporation | Size: 221.4 ± 5.86 nm ZP: -28.1 ± 0.7 PDI: 0.207 LC: 12.86 ± 0.61% |
Colon | [72] |
| Nanocrystals | Ivermectin | Eudragit L100-55 | Wet milling, followed by spray drying | Size: 294 ± 4 nm PDI: 0.278 EE: 92.3 ± 1.23% (polymer concentration of 25%) |
Small intestine | [73] |
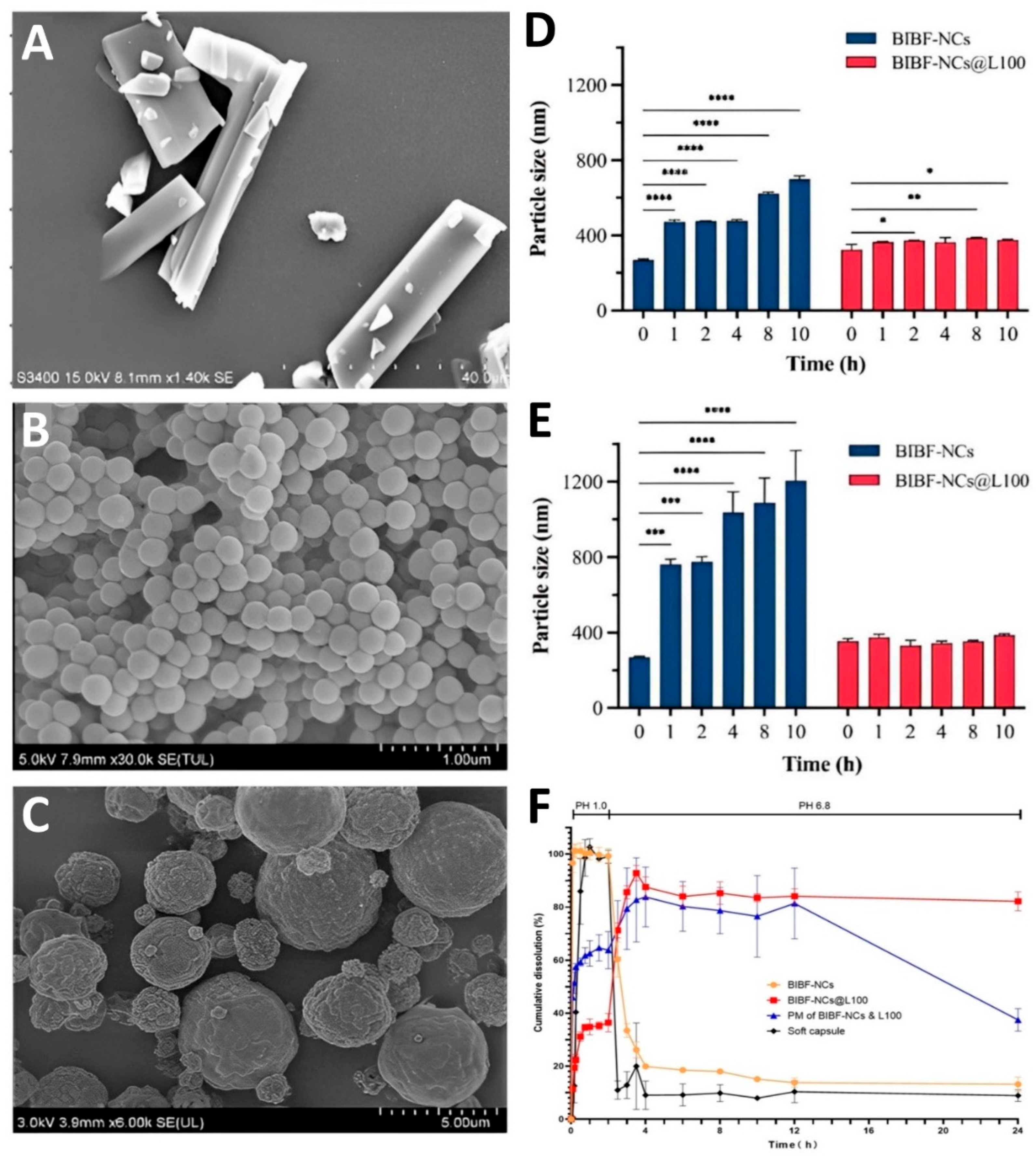
| Nintedanib | Eudragit L100 | Wet milling followed by spray drying | Size: 295.72 ± 3.04 nm ZP: -34.33 ± 0.68 mV PDI: 0.186 ± 0.054 |
Small intestine | [74] | |
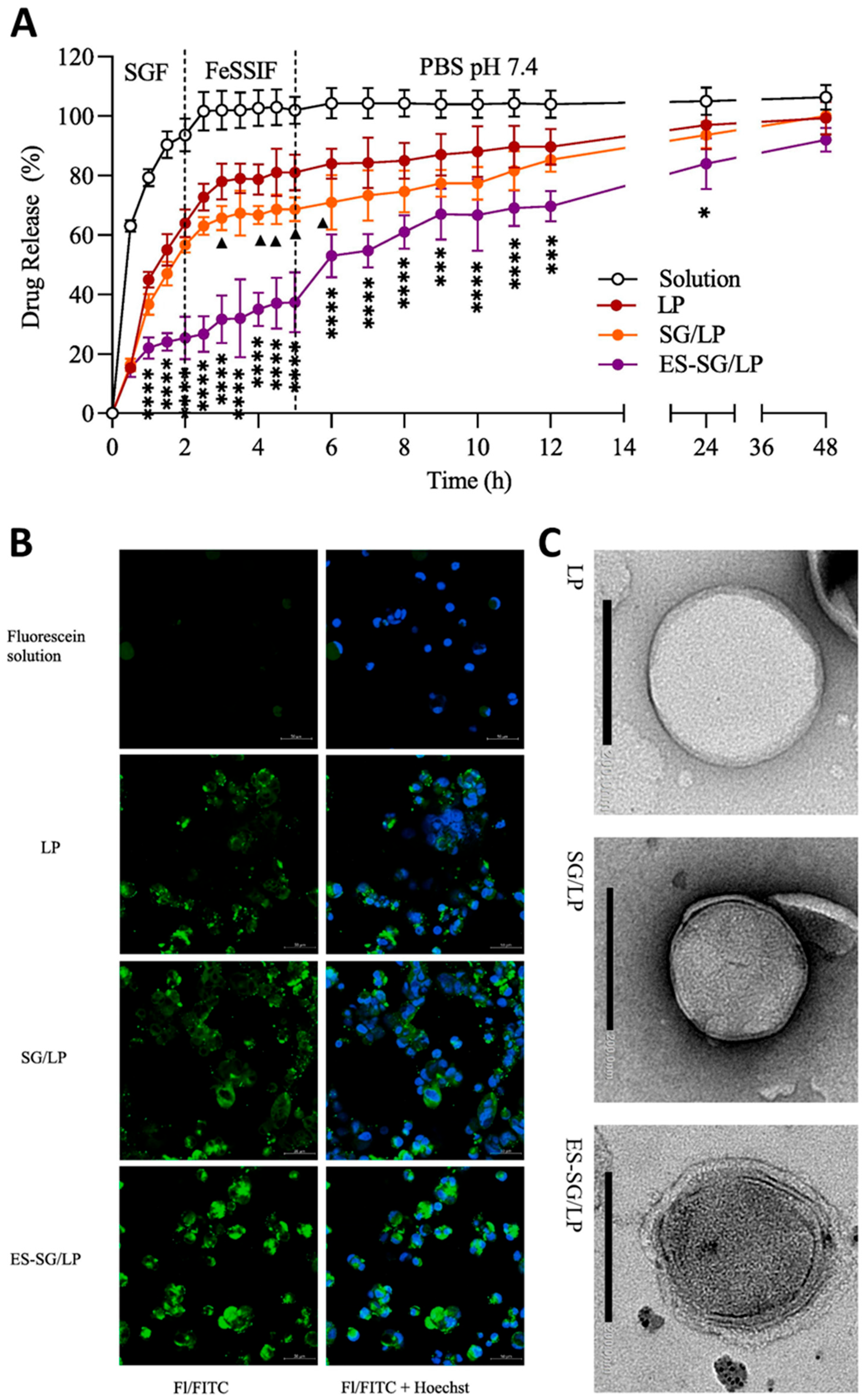
| Liposomes | 5-aminosalicylic acid | Eudragit S100 | Thin-film hydration followed by probe sonication and layer-by-layer electrostatic deposition | Size: ≈ 240 nm ZP: ≈ - 26 mV PDI: ≈ 0.25 EE: ≈ 50-55% (values of ES100 coated LPs with 1mg/mL of sodium glycocholate) |
Colon | [75] |
| Budesonide | Eudragit S100 | Thin-film hydration followed by probe sonication and layer-by-layer electrostatic deposition | Size: 275 nm ZP: -38 mV PDI: 0.128 |
Colon | [76] | |
| Solid Lipid Nanoparticles | Saxagliptin | Eudragit RS100 | Modified solvent injection | Size: 212 to 442 nm ZP: - 41.09 ± 0.11 to 30.86 ± 0.63 mV PDI: 0.26 ± 0.051 to 0.45 ± 0.017 |
Small intestine | [77] |
| Oxaliplatin | Eudragit S100 | Solvent emulsification followed by pelletisation | Size: 116.81 ± 1.37 nm EE: 81.12 ± 0.26 % |
Colon | [78] | |
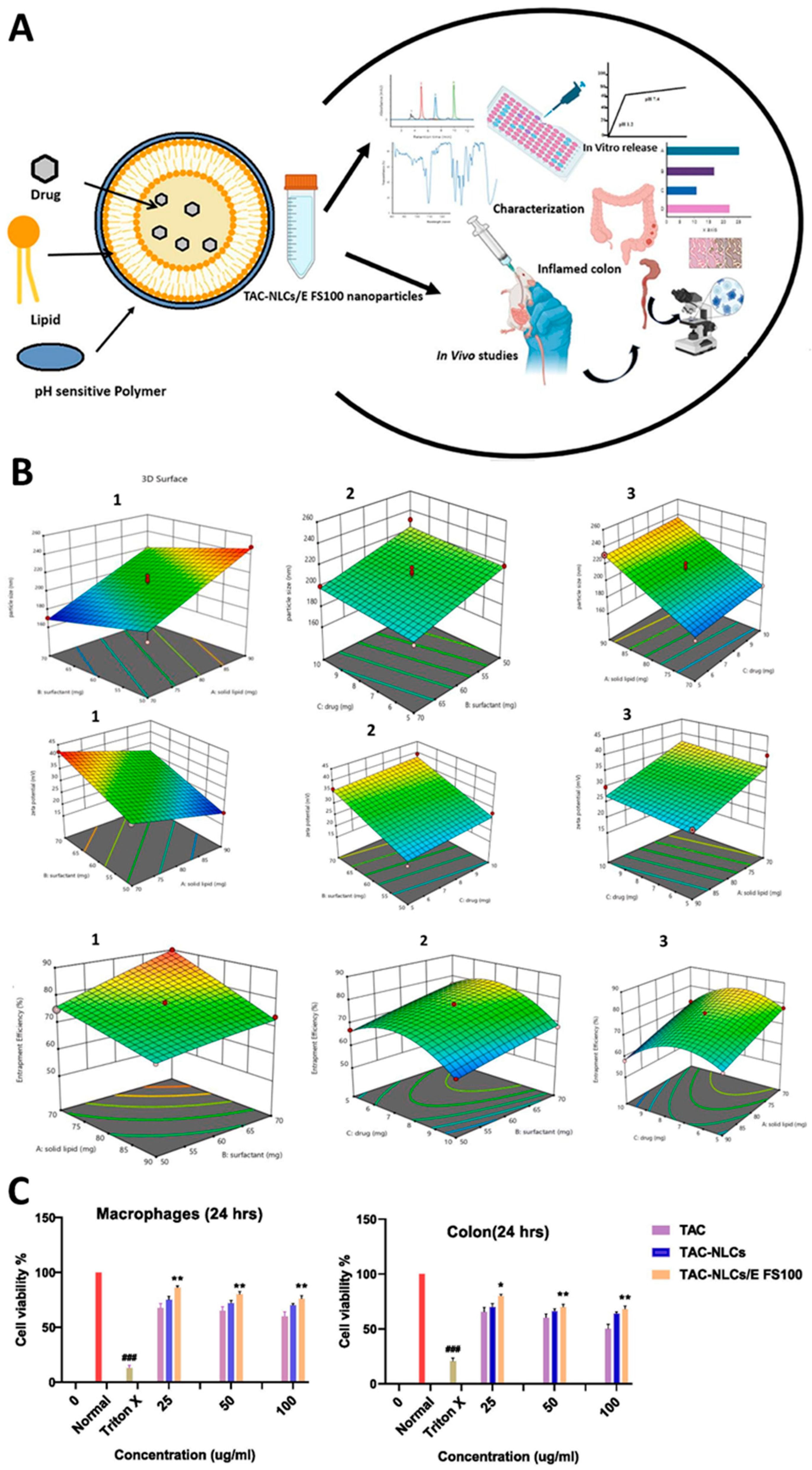
| Nanostructured Lipid Carriers | Tacrolimus | Eudragit FS100 | Modified microemulsion method followed by polymeric coating | Size: 198.7 nm ZP: -47.6 mV PDI: 0.176 EE: 78% |
Colon | [79] |
| 5-Fluorouracil | Eudragit S100 | High pressure homogenisation followed by polymeric coating | Size: 154 ± 3.17 nm ZP: -21.7 ± 2.02 mV PDI: 0.29 ± 0.07 EE: 89.81 ± 2.6% |
Colon | [80] | |
| Polymeric Nanoparticles | Iridoid glycoside | Eudragit S100 Eudragit L30-D 55 |
Single emulsion solvent evaporation followed by polymeric coating | Size: 247 ± 26 nm ZP: -22.4 ± 1.76 mV PDI: 0.21± 0.05 EE: 39.47 ± 2.69% |
Colon | [81] |
| Dasatinib | Eudragit L100 | Single emulsion solvent evaporation followed by polymeric coating | Size: 202.1 ± 5.7 nm ZP: -18 ± 1.01 mV EE: 93.11 ± 0.2% |
Small intestine | [82] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




