Submitted:
12 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. The Characteristics of ALE
- (1)
- Periodicity. The entire process operates in cycles. The process in each cycle is the same, and the total amount of material etching can be determined by the number of cycles.
- (2)
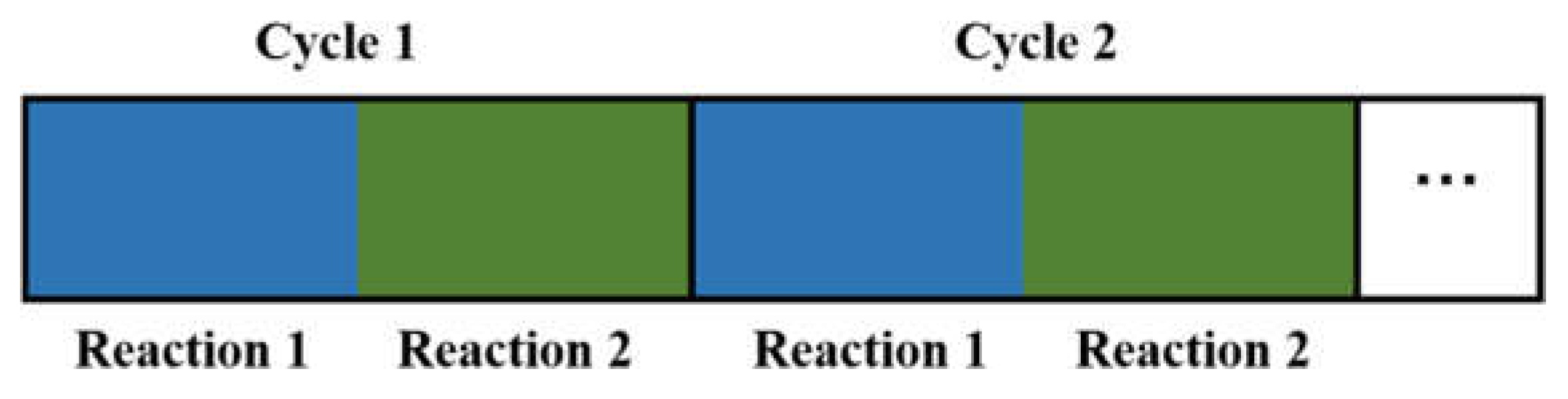
- Independence. Each cycle contains independent and continuous sub-steps, as shown in Figure 1.
- (3)
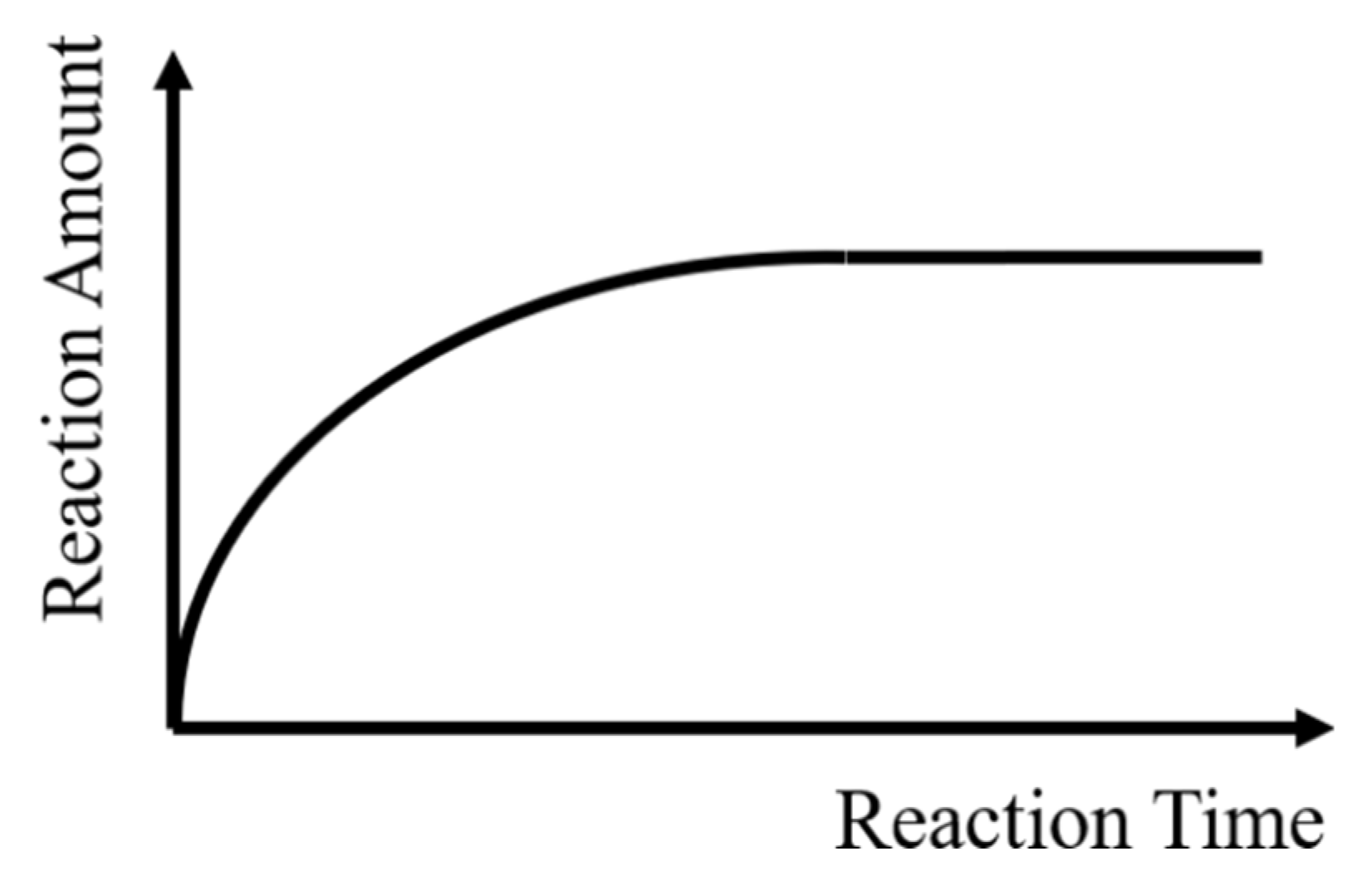
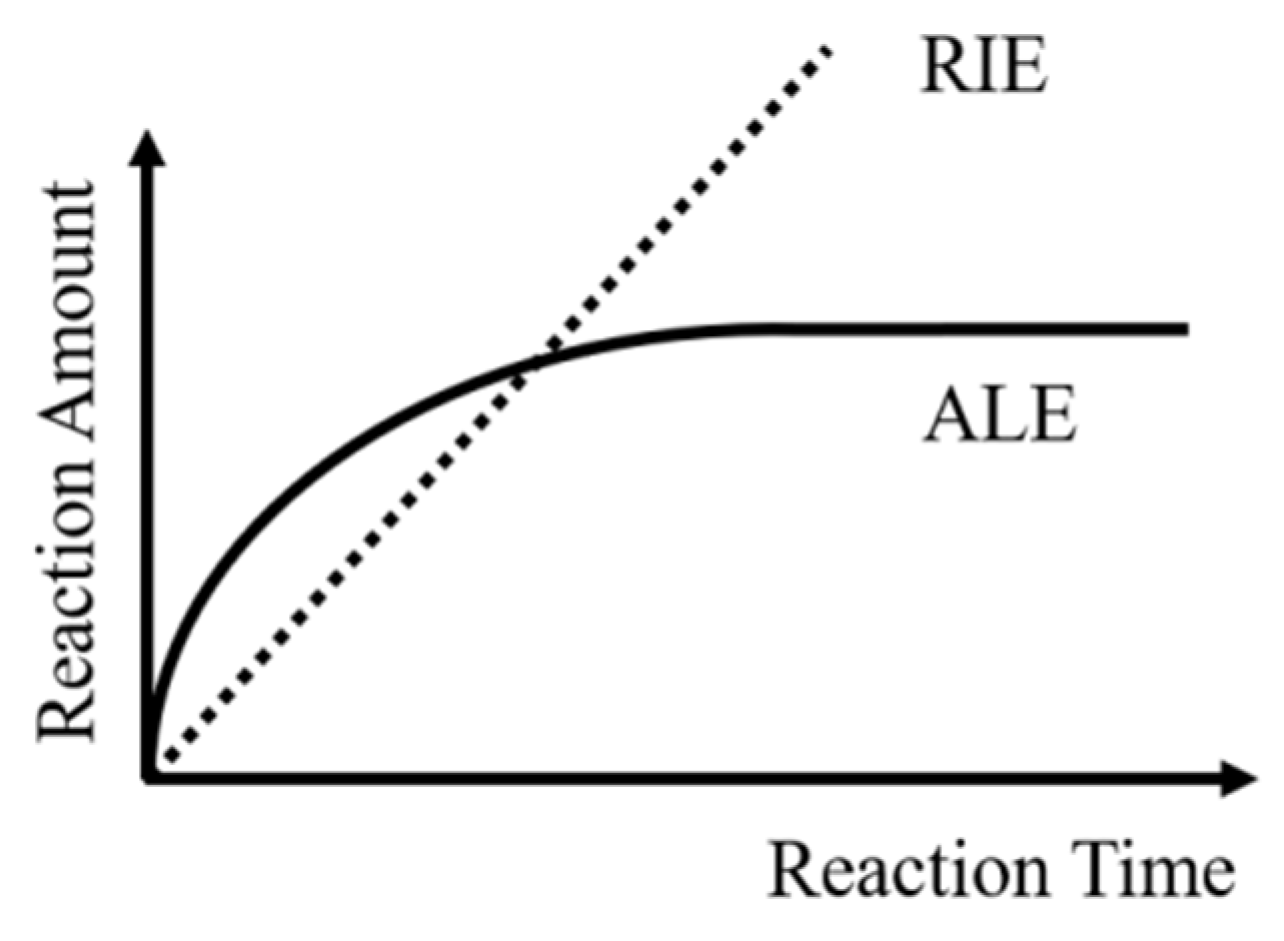
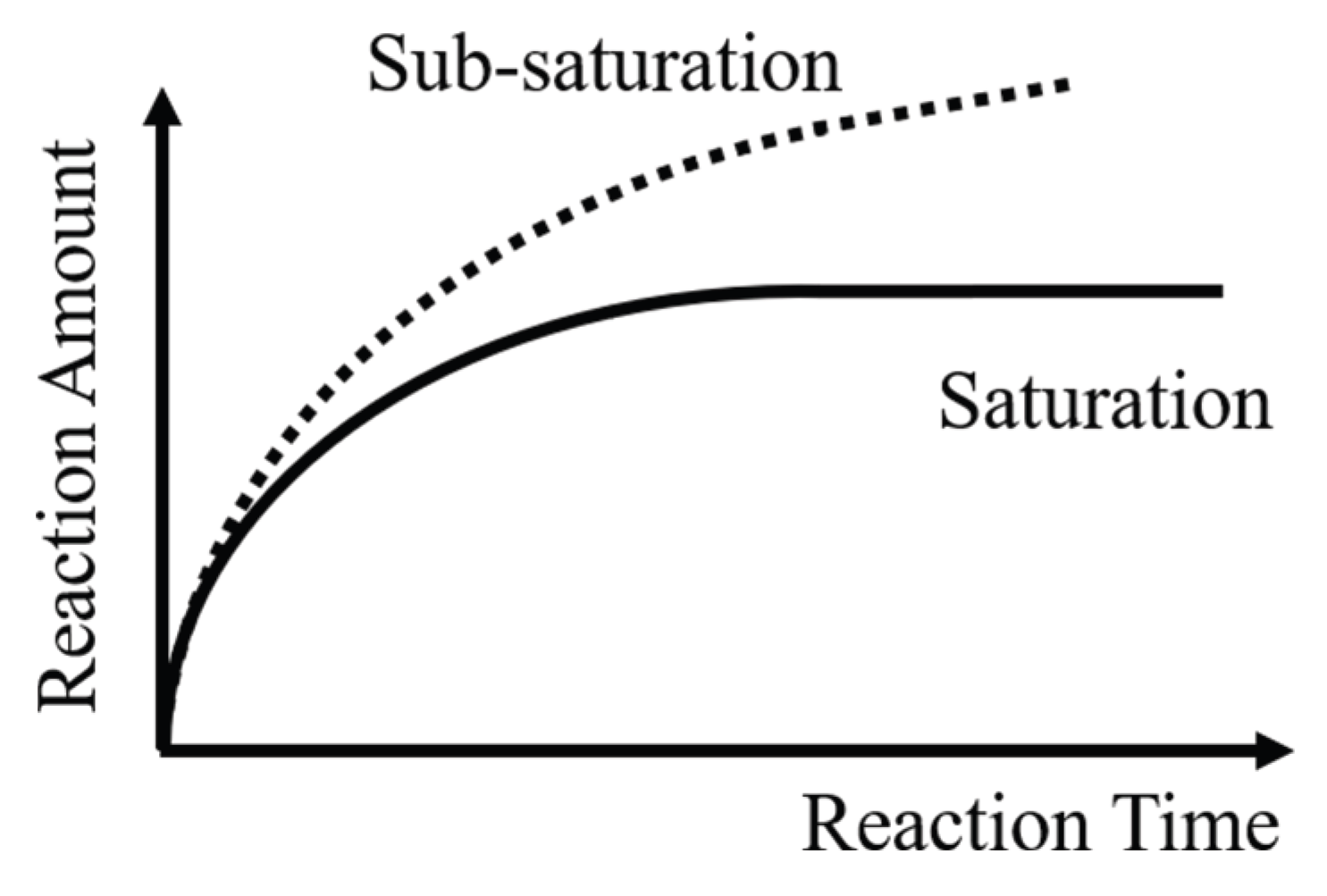
- Self-limitation. At least one sub-step in each cycle meets self-limitation. The total amount of etching in this sub-step can reach saturation over time, which means the reaction can stop by itself, as shown in Figure 2.
- (1)
- Isotropic etching (IALE), etching uniformly in all directions. Convert a certain thickness of a single layer of raw material into a volatile substance through a chemical reaction, and then heat it within a certain temperature range to remove the volatile substance.
- (2)
- Anisotropic etching (AALE), uniform etching in the specified direction. Plasma is used to assist chemical reactions so that the raw materials in a specific area are transformed into other solid substances, and then the solid substances are bombarded with ion beams to separate them from the surface of the raw materials. Generally, because AALE uses plasma to accelerate the adsorption reaction, the anisotropic etching rate is much higher than that of isotropic etching.
- (1)
- Preprocessing. To reduce the potential barrier of the substrate surface, so that the reactants can contact the surface of the substrate through physical or chemical adsorption.
- (2)
- Modification. The adsorbed atoms and the atomic layer with a specified thickness on the substrate surface can be transformed into another material layer through a single or multiple chemical reactions that can reach saturation by themselves. The modified layer is much easier to be removed than unmodified material in the next step.
- (3)
- Removal. The final material generated in the previous step desorbs and volatilizes under ion bombardment or heating, without damaging the original material underneath.
- (4)
- Cleaning. After each step of the reaction, the chamber must be purged with N2 to remove all gaseous reactants and prepare for the next reaction cycle.
- 1)
- High precision. It can achieve etching on an atomic-scale precision, with good etching uniformity and small standard deviation;
- 2)
- Strong controllability. Compared with traditional continuous etching, ALE divides the reaction into multiple independent and self-limiting sub-steps, which enhances the ability to control the etching process.
- 3)
- Good uniformity. The same etching effect can be achieved on a batch of wafers, ensuring high fidelity of circuit pattern transfer.
- 4)
- High selectivity. For example, aluminum oxide and hafnium oxide have a selectivity of more than 1000:1 relative to silicon, silicon dioxide, and nitride;
- 5)
- Wide applicability. It can etch metals and metal composites, which is difficult to achieve with traditional RIE.
- 6)
- The reaction conditions for IALE are relatively simple. Compared with AALE, the temperature and pressure is easier to control than plasma;
- 7)
- AALE has good directionality. AALE can be used to etch high-aspect-ratio graphics.
- 1)
- Compared with other etching techniques, ALE requires a longer time and higher cost;
- 2)
- The reaction rate will be different at different depths of the material. Because of the diffusion of reactants and other mechanisms, the concentration of reactants at different depths is different, so the rate of reaction is also different, and finally, the actual etching pattern does not match the design;
- 3)
- The accuracy of AALE is affected by plasma parameters, including plasma exposure time, power, and bias.
- 4)
- IALE is sensitive to temperature, which limits the application of IALE. The upper limit of the adjustable temperature is about 650 °C, and over high temperature may damage the device;
3. Application of ALE on Different Materials
4. The Self-Limiting Mechanism of ALE
5. Five Improvement Methods of ALE
5.1. Reduce the Preprocessing Time by Lowering the Surface Barrier of the Substrate
5.2. Reduce the Roughness of the Material Surface After Etching
5.3. To Achieve Better IALE on the Substrate Which Is Covered by Masks
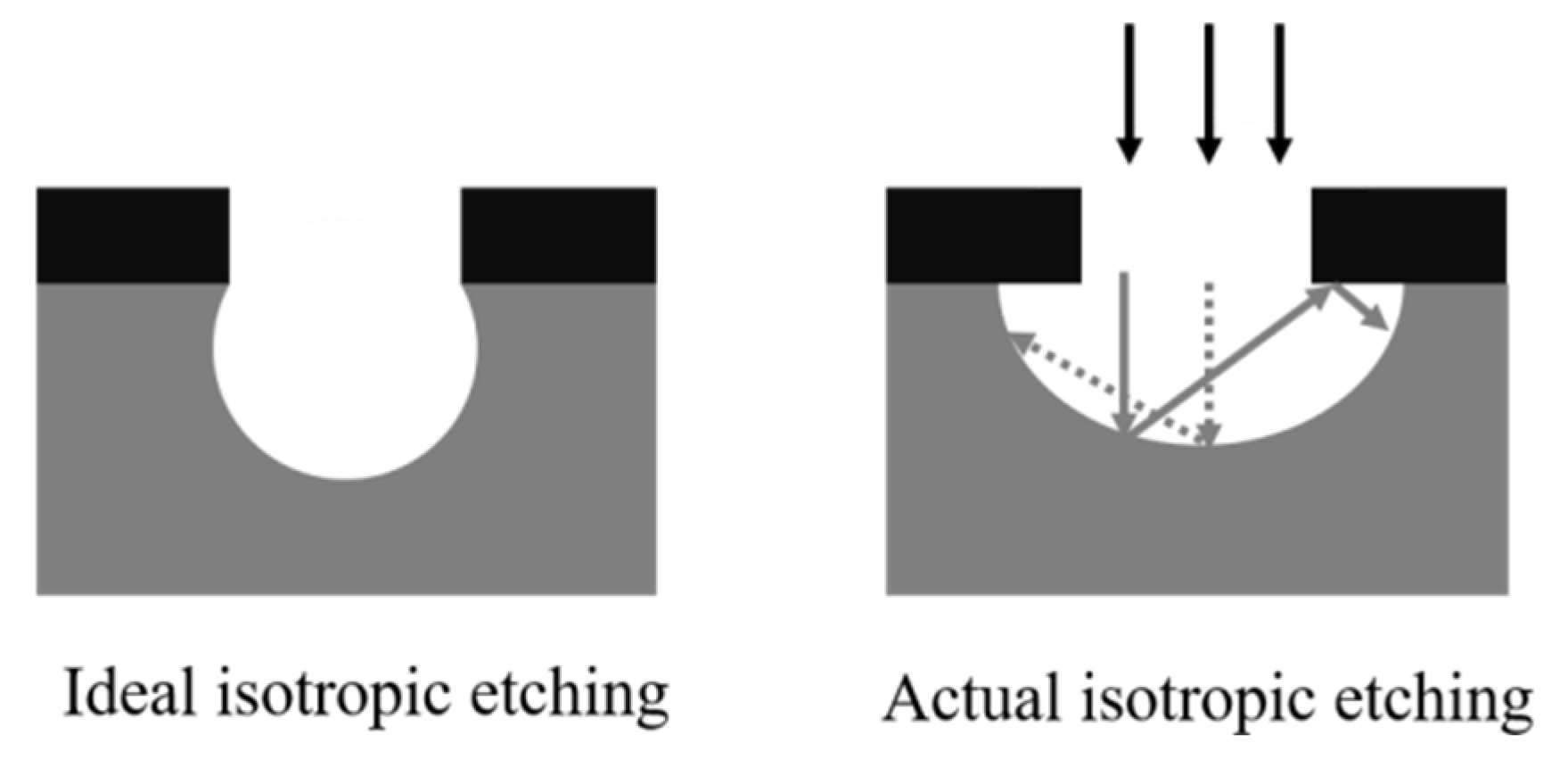
- (1)
- To control the reaction area of the substrate, the mask is used to cover part of the substrate material. As shown in Figure 4, When reactant ions are incident, the exposed part of the substrate reacts uniformly, but the substrates on both sides cannot directly receive the reactant. The reactants on both sides indirectly come from the reflective particles in the middle part of the substrate surface. Obviously, the reactant ions received on both sides are far less than the middle part, so the reaction rate on both sides is much slower than the middle part of the substrate surface. Therefore, the ideal IALE cannot be achieved.
- (2)
- The reactant molecules diffuse unevenly at each depth of the substrate. When the surface material is transformed into other substances, its lattice structure changes, which imposes compressive or tensile stress on the lattice structure of the underlying original substrate material, thereby changing the diffusion rate of reactant molecules at various depths.
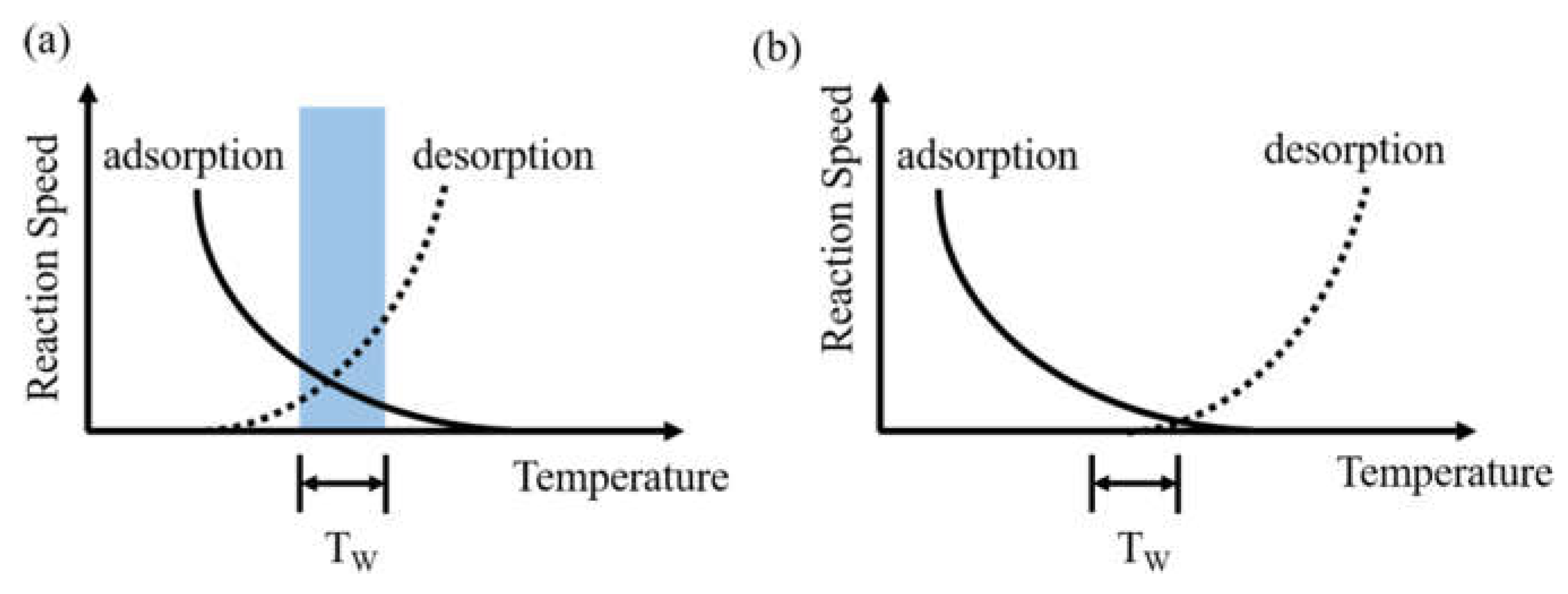
5.4. Use Multiple Temperature Windows to Increase the Etch Rate of ALE and Expand Its Application Range
5.5. Shorten the Etching Time of the Entire Etching Process by Introducing a Temperature-Changing Mechanism While Cleaning the Chamber
6. Conclusion
References
- Kanarik, K.J.; et al. Overview of atomic layer etching in the semiconductor industry. Journal of Vacuum Science & Technology A: Vacuum, Surfaces, and Films 2015, 33. [Google Scholar] [CrossRef]
- Fang, C.; Cao, Y.; Wu, D.; Li, A. Thermal atomic layer etching: Mechanism, materials and prospects. Progress in Natural Science: Materials International 2018, 28, 667–675. [Google Scholar] [CrossRef]
- Atomic Layer Etching: Directional. In Encyclopedia of Plasma Technology; 2016; pp. 133–142.
- Sang, X.; Chang, J.P. Physical and chemical effects in directional atomic layer etching. Journal of Physics D: Applied Physics 2020, 53. [Google Scholar] [CrossRef]
- Fischer, A.; Routzahn, A.; George, S.M.; Lill, T. Thermal atomic layer etching: A review. Journal of Vacuum Science & Technology A 2021, 39. [Google Scholar] [CrossRef]
- C. You, C. Mannequin, J. G., T. Chevolleau, and D. C. Development of Atomic Layer Etching (ALEt) for GaN-based materials. In International Workshop on Nitride Semiconductors; 2018.
- Ohba, T.; Yang, W.; Tan, S.; Kanarik, K.J.; Nojiri, K. Atomic layer etching of GaN and AlGaN using directional plasma-enhanced approach. Japanese Journal of Applied Physics 2017, 56. [Google Scholar] [CrossRef]
- Lee, Y.; DuMont, J.W.; George, S.M. Trimethylaluminum as the Metal Precursor for the Atomic Layer Etching of Al2O3 Using Sequential, Self-Limiting Thermal Reactions. Chemistry of Materials 2016, 28, 2994–3003. [Google Scholar] [CrossRef]
- Oh, C.K.; Park, S.D.; Lee, H.C.; Bae, J.W.; Yeom, G.Y. Surface Analysis of Atomic-Layer-Etched Silicon by Chlorine. Electrochemical and Solid-State Letters 2007, 10. [Google Scholar] [CrossRef]
- Abdulagatov, A.; George, S. Thermal Atomic Layer Etching of Silicon Using O2, HF and Al(CH3)3 as the Reactants. Chemistry of Materials 2018, 30. [Google Scholar] [CrossRef]
- Kanarik, K.J.; et al. Predicting synergy in atomic layer etching. Journal of Vacuum Science & Technology A: Vacuum, Surfaces, and Films 2017, 35. [Google Scholar] [CrossRef]
- Lee, Y.; DuMont, J.W.; George, S.M. Atomic Layer Etching of HfO2Using Sequential, Self-Limiting Thermal Reactions with Sn(acac)2and HF. ECS Journal of Solid State Science and Technology 2015, 4, N5013–N5022. [Google Scholar] [CrossRef]
- Shinoda, K.; et al. Self-limiting reactions of ammonium salt in CHF3/O2 downstream plasma for thermal-cyclic atomic layer etching of silicon nitride. Journal of Vacuum Science & Technology A 2019, 37. [Google Scholar] [CrossRef]
- Johnson, N.R.; Hite, J.K.; Mastro, M.A.; Eddy, C.R.; George, S.M. Thermal atomic layer etching of crystalline GaN using sequential exposures of XeF2 and BCl3. Applied Physics Letters 2019, 114. [Google Scholar] [CrossRef]
- Hwang, I.-H.; Cha, H.-Y.; Seo, K.-S. Low-Damage and Self-Limiting (Al)GaN Etching Process through Atomic Layer Etching Using O2 and BCl3 Plasma. Coatings 2021, 11. [Google Scholar] [CrossRef]
- Johnson, N.R.; George, S.M. WO3 and W Thermal Atomic Layer Etching Using “Conversion-Fluorination” and “Oxidation-Conversion-Fluorination” Mechanisms. ACS Appl Mater Interfaces 2017, 9, 34435–34447. [Google Scholar] [CrossRef] [PubMed]
- Park, J.W.; Kim, D.S.; Lee, W.O.; Kim, J.E.; Yeom, G.Y. Atomic layer etching of chrome using ion beams. Nanotechnology 2019, 30, 085303. [Google Scholar] [CrossRef] [PubMed]
- Sakaue, H.; Iseda, S.; Asami, K.; Yamamoto, J.; Hirose, M.; Horiike, Y. Atomic Layer Controlled Digital Etching of Silicon. Japanese Journal of Applied Physics 1990, 29, 2648–2652. [Google Scholar] [CrossRef]
- Donnelly, V.M.; Kornblit, A. Plasma etching: Yesterday, today, and tomorrow. Journal of Vacuum Science & Technology A: Vacuum, Surfaces, and Films 2013, 31. [Google Scholar] [CrossRef]
- Tan, S.; et al. Highly Selective Directional Atomic Layer Etching of Silicon. ECS Journal of Solid State Science and Technology 2015, 4, N5010–N5012. [Google Scholar] [CrossRef]
| Question | RIE | ALE |
|---|---|---|
| Speed | Fast | Slow |
| Isotropic/anisotropic? | Anisotropic | Both |
| Are chemistry and ion bombardment separated? | No | Yes |
| Is it easy to control desirable species fluxes? | Partially | Yes |
| Is it easy to control surface morphology (Reduce roughness and thick mixed layer)? |
No | Yes |
| Does it depend on the delivery of species (neutrals or ions)? |
Yes | No |
| Can it maintain stoichiometry during directional etching? |
No | Yes |
| Etching | Isotropic | Process Sequence | Best | ref |
|---|---|---|---|---|
| Material | Or | Etching | ||
| Anisotropic | Rate/(Å/c | |||
| ycle) | ||||
| Si | Anisotropic | Chlorination (Cl2) → Ar+ bombardment | 1.36/7 | [1,9] |
| Isotropic | Oxidation(O2) → Fluorination (HF) → Ligand-exchange (TMA) | 0.4 | [10] | |
| Ge | Anisotropic | Chlorination (Cl2) → Ar+ bombardment | 8 | [11] |
| Al2O3 | Isotropic | Fluorination (HF) → Ligand-exchange (TMA) |
0.75 | [8] |
| HfO2 | Isotropic | Ligand-exchange (Sn(acac)2) → Fluorination (HF) | 0.124 | [12] |
| Si3N4 | Isotropic | Oxidation (O2/O3) → Fluorination (HF) → Ligand-exchange (TMA) | 0.47 | [13] |
| GaN | Isotropic | Fluorination (XeF2) → Ligand-exchange (BCl3) | 0.72 | [14] |
| Anisotropic | Chlorination (Cl2) → Ar+ bombardment | 4 | [7,15] | |
| WO3 | Isotropic | Chlorination (BCl3) → Fluorination (HF) | 4.19 | [16] |
| W | Isotropic | Oxidation (O3) → Chlorination (BCl3) → Fluorination (HF) | 2.5 | [16] |
| AlGaN | Anisotropic | Chlorination (BCl3) → Ar+ bombardment | 7 | [7] |
| Cr | Anisotropic | Cl/O radical adsorption → Ar+ bombardment |
1.6 | [17] |
| Bond | Bong Energy/(kJ/mol) |
|---|---|
| Si-O | 452 |
| Si-Cl | 381 |
| Si-F | 565 |
| Reactant (R) | Reaction Intermediate (I) |
Product (P) | |
|---|---|---|---|
| Adsorption rate (A) | RA | IA | PA |
| Desorption rate (D) | RD | ID | PD |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




