Submitted:
11 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Settings
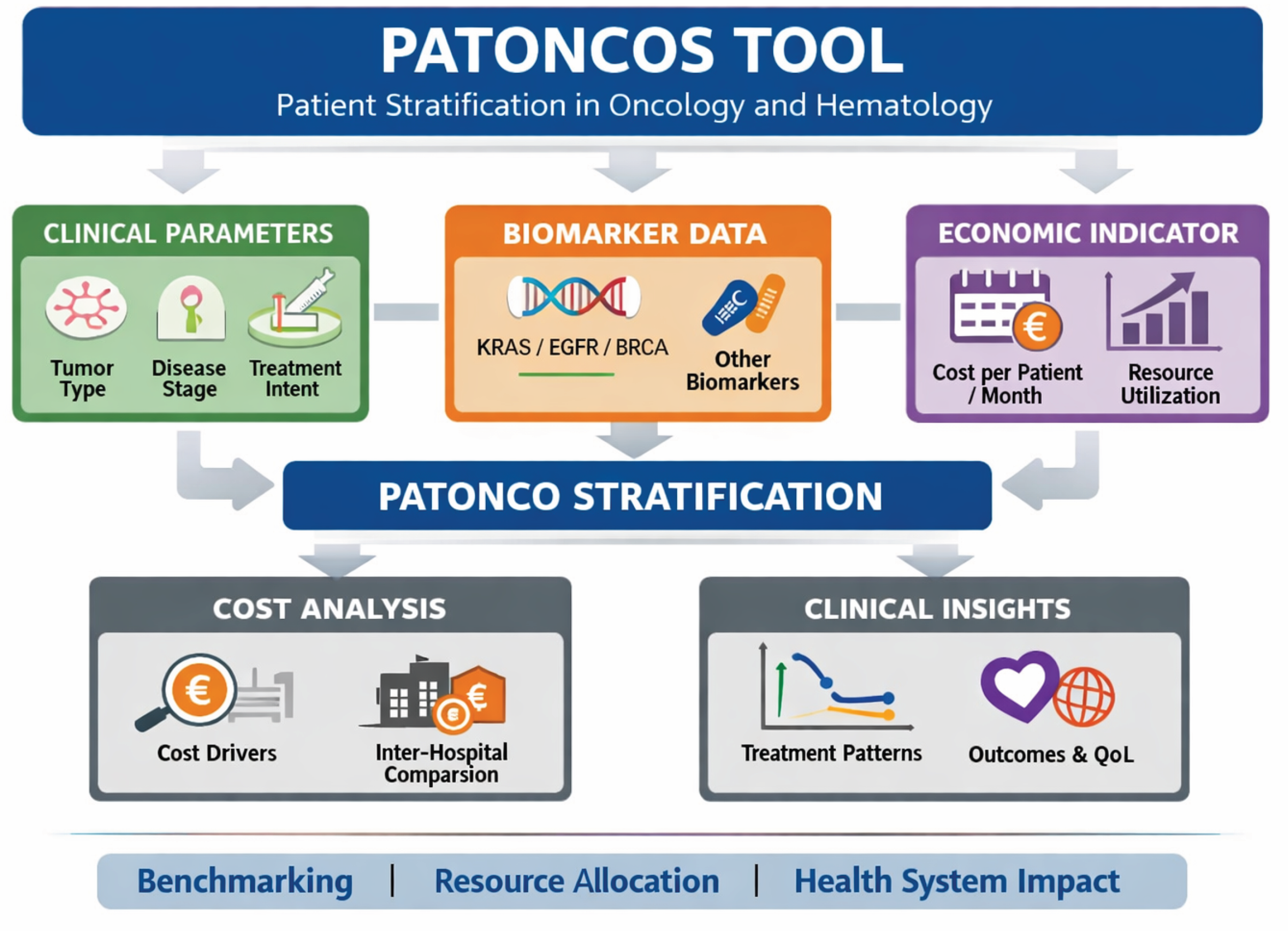
2.2. Development of the PATONCO Classification System
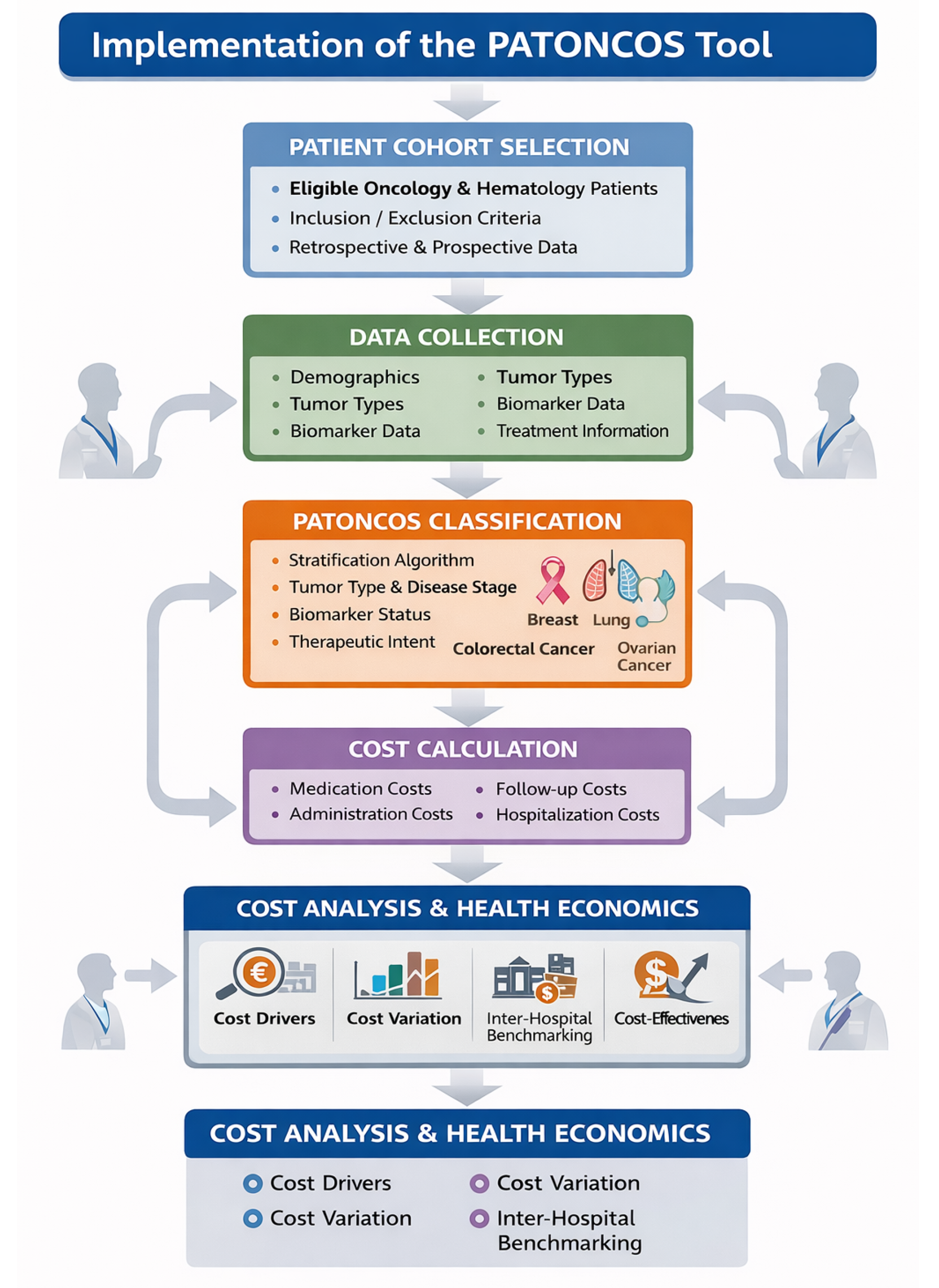
2.3. Implementation and Data Collection
2.4. Cost Definition and Economic Indicator
2.5. Statistical Analysis
2.6. Study Period and Benchmarking
3. Results
3.1. PATONCOS Tool Development
3.2. PATONCO Classification System
3.3. Study Population
- Metastatic breast cancer HER2(−) HR(+)
- Metastatic non-squamous non-small cell lung cancer ALK(−) EGFR(−)
- Adjuvant colon cancer
- Metastatic colorectal cancer KRAS/NRAS mutated
- Metastatic colorectal cancer KRAS/NRAS wild-type
- Castration-resistant metastatic prostate cancer
- Multiple myeloma transplant candidate
3.4. Cost Indicator and Inter-Hospital Comparison
3.5. Drivers of Cost Variability
3.6. Budget Impact Distribution
4. Discussion
5. Conclusions
6. Future directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACC | Adjuvant Colon Cancer |
| AJCC | American Joint Committee on Cancer |
| ASCO | American Society of Clinical Oncology |
| CRMPC | Castrate Resistant Metastasic Prostate Cancer |
| ESMO | European Society for Medical Oncology |
| ESMO-MCBS | European Society for Medical Oncology- Magnitude of Clinical Benefit Scale |
| GEJ | Gastric and gastresophageal Junction |
| ICD-O | International Classification of Disease for Oncology |
| MBC-RH | Metastasic Breast Cancer Postive Hormonal receptor |
| MCC-KNM | Metastasic Colorectal Cancer Kras Nras Mutate |
| MCC-KNN | Metastasic Colorectal Cancer Kras Nras Native |
| MMTC | Multiple Myeloma Transplant Candidate |
| MNSCLC | Metastasic Non Small Cell Lung Cancer No Squamous |
| NSCLC | Non Small Cell Lung Cancer |
| RH | Hormonal Receptor |
| TDABC | Time Driven Activity-Based Costing |
References
- Del Paggio, J.C.; Azariah, B.; Sullivan, R.; Hopman, W.M.; James, F.V.; Roshni, S.; et al. Do Contemporary Randomized Controlled Trials Meet ESMO Thresholds for Meaningful Clinical Benefit? Annals of Oncology 2017, 28, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Grumberg, V.; Cotté, F.E.; Giroux-Leprieur, E.; Gaudin, A.F.; Lebbé, C.; Borget, I. Clinical benefit of anti-PD-(L)1 immunotherapies in advanced cancer in France: a population-based estimate from 2014 to 2021. ESMO Open 2024, 9, 102240. [Google Scholar] [CrossRef] [PubMed]
- Capri, S.; Russo, A. Cost of breast cancer based on real-world a cancer registry study in Italy. BMC Health Services Research 2017, 17, 84. [Google Scholar] [CrossRef] [PubMed]
- Vargas Alves, R.J.; Beck da Silva Etges, A.P.; Tiscoski, K.A.; de Lara, L.R.; de Medeiros Zelmanowicz, A.; Polanczyk, C.A. The cost of metastatic prostate cancer using time-driven activity-based costing. International Journal of Technology Assessment in Health Care 2021, 37, e60. [Google Scholar] [CrossRef]
- Tew, M.; Clarke, P.; Thursky, K.; Dalziel, K. Incorporating Future Medical Costs: Impact on Cost-Effectiveness Analysis in Cancer Patients. PharmacoEconomics 2019, 37, 931–941. [Google Scholar] [CrossRef]
- Hari, P.; Latremouille-Viau, D.; Lin, P.; Guerin, A.; Sasane, M. Clinical and economic burden of Medicare beneficiaries with multiple myeloma and renal impairment: An observational study. Medicine 2024, 103, e38609. [Google Scholar] [CrossRef]
- Freitag, A.; Sarri, G.; Ta, A.; Gurskyte, L.; Cherepanov, D.; Hernandez, L.G. A Systematic Review of Modeling Approaches to Evaluate Treatments for Relapsed Refractory Multiple Myeloma: Critical Review and Considerations for Future Health Economic Models. PharmacoEconomics 2024, 42, 955–1002. [Google Scholar] [CrossRef]
- Jönsson, B.; Walter, E. Cost of Cancer: Healthcare Expenditures and Economic Impact. In Regulatory and Economic Aspects in Oncology; Walter, E., Ed.; Springer International Publishing: Cham, 2019; pp. 7–23. [Google Scholar] [CrossRef]
- Carreras, M.J.; Tomás-Guillén, E.; Farriols, A.; Renedo-Miró, B.; Valdivia, C.; Vidal, J.; et al. Use of Drugs in Clinical Practice and the Associated Cost of Cancer Treatment in Adult Patients with Solid Tumors: A 10-Year Retrospective Cohort Study. Current Oncology 2023, 30, 7984–8004. [Google Scholar] [CrossRef]
- Harfouche, A.; Silva, S.; Faria, J.; Araújo, R.; Gouveia, A.; Lacerda, M.; et al. Breast Cancer: Value-Based Healthcare, Costs and Financing. Acta Médica Portuguesa 2017, 30, 762–768. [Google Scholar] [CrossRef]
- Desai, A.P.; Scheckel, C.J.; Soderberg, L.C.; Jensen, C.J.; Orme, J.J.; Tella, S.H.; et al. Economic Cost and Sustainability of Oral Therapies in Precision Oncology. JCO Oncology Practice 2022, 18, 1247–1254. [Google Scholar] [CrossRef]
- Datta, S.S.; Sharma, V.; Mukherjee, A.; Agrawal, S.; Sirohi, B.; Gyawali, B. What constitutes meaningful benefit of cancer drugs in the context of LMICs? A mixed-methods study of oncologists’ perceptions on endpoints, benefit, price, and value of cancer drugs. ESMO Open 2024, 9, 103976. [Google Scholar] [CrossRef] [PubMed]
- Sociedad Española de Oncología Médica. Cifras del cáncer en España 2021. Internet. 2021. Available online: https://seom.org/images/Cifras_del_cancer_en_Espnaha_2021.pdf.
- Mariotto, A.B.; Yabroff, K.R.; Shao, Y.C.; Feuer, E.J.; Brown, M.L. Projections of the cost of cancer care in the United States: 2010–2020. Journal of the National Cancer Institute 2011, 103, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Bhimani, J.; Philipps, L.; Simpson, L.; Lythgoe, M.; Soultati, A.; Webb, A.; Savage, P. The impact of new cancer drug therapies on site specialised cancer treatment activity in a UK cancer network 2014–2018. Journal of Oncology Pharmacy Practice 2020, 26, 93–98. [Google Scholar] [CrossRef]
- Schnipper, L.E.; Davidson, N.E.; Wollins, D.S.; Blayney, D.W.; Dicker, A.P.; Ganz, P.A.; et al. Updating the American Society of Clinical Oncology Value Framework: Revisions and Reflections in Response to Comments Received. Journal of Clinical Oncology 2016, 34, 2925–2934. [Google Scholar] [CrossRef]
- Cherny, N.I.; Sullivan, R.; Dafni, U.; Kerst, J.M.; Sobrero, A.; Zielinski, C.; et al. A standardised, generic, validated approach to stratify the magnitude of clinical benefit that can be anticipated from anti-cancer therapies: the European Society for Medical Oncology Magnitude of Clinical Benefit Scale (ESMO-MCBS). Annals of Oncology 2015, 26, 1547–1573. [Google Scholar] [CrossRef]
- Warren, J.L.; Brown, M.L.; Fay, M.P.; Schussler, N.; Potosky, A.L.; Riley, G.F. Costs of Treatment for Elderly Women With Early-Stage Breast Cancer in Fee-for-Service Settings. Journal of Clinical Oncology 2002, 20, 307–316. [Google Scholar] [CrossRef]
- Tan, P.H.; Ellis, I.; Allison, K.; Brogi, E.; Fox, S.B.; Lakhani, S.; et al. The 2019 World Health Organization classification of tumours of the breast. Histopathology 2020, 77, 181–185. [Google Scholar] [CrossRef]
- Cardoso, F.; Senkus, E.; Costa, A.; Papadopoulos, E.; Aapro, M.; André, F.; et al. ESO-ESMO International Consensus Guidelines for Advanced Breast Cancer (ABC 4). Annals of Oncology 2018, 29, 1634–1657. [Google Scholar] [CrossRef] [PubMed]
- Garrido, C.; Grávalos, C. Memoria del Registro de Tumores de Madrid (RTMAD). Año 2020. Informe técnico Acceso en línea. 2021. [Google Scholar]
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer Statistics, 2021. CA: A Cancer Journal for Clinicians 2021, 71, 7–33. [Google Scholar] [CrossRef]
- Buja, A.; Pasello, G.; De Luca, G.; Bortolami, A.; Zorzi, M.; Rea, F.; et al. Non–Small-Cell Lung Cancer: Real-World Cost Consequence Analysis. JCO Oncology Practice 2021, 17, e1085–e1093. [Google Scholar] [CrossRef] [PubMed]
- Korytowsky, B.; Radtchenko, J.; Nwokeji, E.D.; Tuell, K.W.; Kish, J.K.; Feinberg, B.A. Understanding total cost of care in advanced non-small cell lung cancer pre- and postapproval of immuno-oncology therapies. The American Journal of Managed Care 2018, 24, S439–S447. [Google Scholar] [PubMed]
- Siegel, R.L.; Giaquinto, A.N.; Jemal, A. Cancer statistics, 2024. CA: A Cancer Journal for Clinicians 2024, 74, 12–49. [Google Scholar] [CrossRef]
- Braal, C.L.; Jongbloed, E.M.; Wilting, S.M.; Mathijssen, R.H.J.; Koolen, S.L.W.; Jager, A. Inhibiting CDK4/6 in Breast Cancer with Palbociclib, Ribociclib, and Abemaciclib: Similarities and Differences. Drugs 2021, 81, 317–331. [Google Scholar] [CrossRef]
- ESMO. ESMO-Magnitude of Clinical Benefit Scale. Internet. Citado. 2021.
- Khan, S.A.; Hernandez-Villafuerte, K.; Hernandez, D.; Schlander, M. Estimation of the stage-wise costs of breast cancer in Germany using a modeling approach. Frontiers in Public Health 2022, 10, 946544. [Google Scholar] [CrossRef]
- Hassett, M.J.; Li, H.; Burstein, H.J.; Punglia, R.S. Neoadjuvant treatment strategies for HER2-positive breast cancer: cost-effectiveness and quality of life outcomes. Breast Cancer Research and Treatment 2020, 181, 43–51. [Google Scholar] [CrossRef]
- Seefat, M.R.; Cucchi, D.G.J.; Groen, K.; Donker, M.L.; van der Hem, K.G.; Westerman, M.; et al. Treatment sequences and drug costs from diagnosis to death in multiple myeloma. European Journal of Haematology 2024, 112, 360–366. [Google Scholar] [CrossRef] [PubMed]
| Tumour Type | Representative PATONCO Categories |
|---|---|
| Breast cancer | Adjuvant HER2(+) HR(-); Adjuvant HER2(-) HR(+); Metastatic HER2(-) HR(+); Metastatic triple-negative |
| Colorectal cancer | Adjuvant colon cancer; Metastatic KRAS/NRAS mutated; Metastatic KRAS/NRAS wild-type |
| Lung cancer (NSCLC) | Adjuvant NSCLC; Metastatic non-squamous ALK(-)/EGFR(-); Metastatic ALK(+)/EGFR(+) |
| Ovarian cancer | Adjuvant ovarian cancer; Metastatic BRCA(+) first line; Platinum-resistant second line |
| Prostate cancer | Hormone-sensitive metastatic; Castration-resistant metastatic |
| Melanoma | Adjuvant melanoma; Metastatic BRAF(+); Metastatic BRAF(-) |
| Gastroesophageal cancer (GEJ) | Adjuvant adenocarcinoma; Metastatic HER2(+); Metastatic HER2(-) |
| Haematological malignancies | Hodgkin lymphoma; Large B-cell lymphoma; Multiple myeloma (transplant candidate) |
| PATONCO Category | H1 | H2 | H3 | H4 | Dispensed | Patients | H | p |
|---|---|---|---|---|---|---|---|---|
| MBC HER2(-) HR(+) | 1546.76 (1198.78) | 1192.50 (865.80) | 1497.64 (1233.16) | 1426.45 (861.11) | 2079 | 285 | 21.63** | .000 |
| MNSCLC ALK(-) EGFR(-) | 1851.02 (1773.10) | 2327.83 (1626.00) | 1832.98 (1584.21) | 1797.35 (1592.73) | 1130 | 208 | 25.29** | .000 |
| Adjuvant colon cancer | 60.43 (61.49) | 117.08 (568.66) | 151.57 (546.73) | 62.73 (32.37) | 655 | 191 | 126.60** | .000 |
| MCRC KRAS/NRAS mutated | 972.40 (997.52) | 791.77 (717.42) | 502.62 (698.64) | 542.36 (713.52) | 999 | 175 | 94.04** | .000 |
| MCRC KRAS/NRAS wild-type | 1632.86 (1067.64) | 1650.30 (1311.47) | 1058.25 (1313.52) | 1520.21 (1201.89) | 1209 | 170 | 92.61** | .000 |
| Castration-resistant metastatic | ||||||||
| prostate cancer | 2387.42 (2030.45) | 1915.45 (1944.07) | 2962.25 (2228.91) | 1780.68 (1311.32) | 940 | 166 | 52.37** | .000 |
| Multiple myeloma | ||||||||
| (transplant candidate) | 2009.50 (2958.81) | 1106.19 (2286.92) | 2613.81 (3965.06) | 3272.27 (4932.95) | 1012 | 153 | 11.56** | .009 |
| Adjuvant breast cancer | ||||||||
| HER2(-) HR(+) | 238.04 (566.73) | 41.90 (45.75) | 465.41 (883.28) | 182.58 (492.69) | 457 | 117 | 71.04** | .000 |
| Hormone-sensitive metastatic | ||||||||
| prostate cancer | 2265.70 (2062.00) | 2621.13 (1940.00) | 2763.11 (2145.70) | 1354.96 (971.46) | 498 | 101 | 40.22** | .000 |
| Metastatic small cell lung cancer | 1163.57 (1296.99) | 1283.81 (1243.63) | 715.06 (875.21) | 1253.02 (1272.92) | 353 | 101 | 18.45** | .000 |
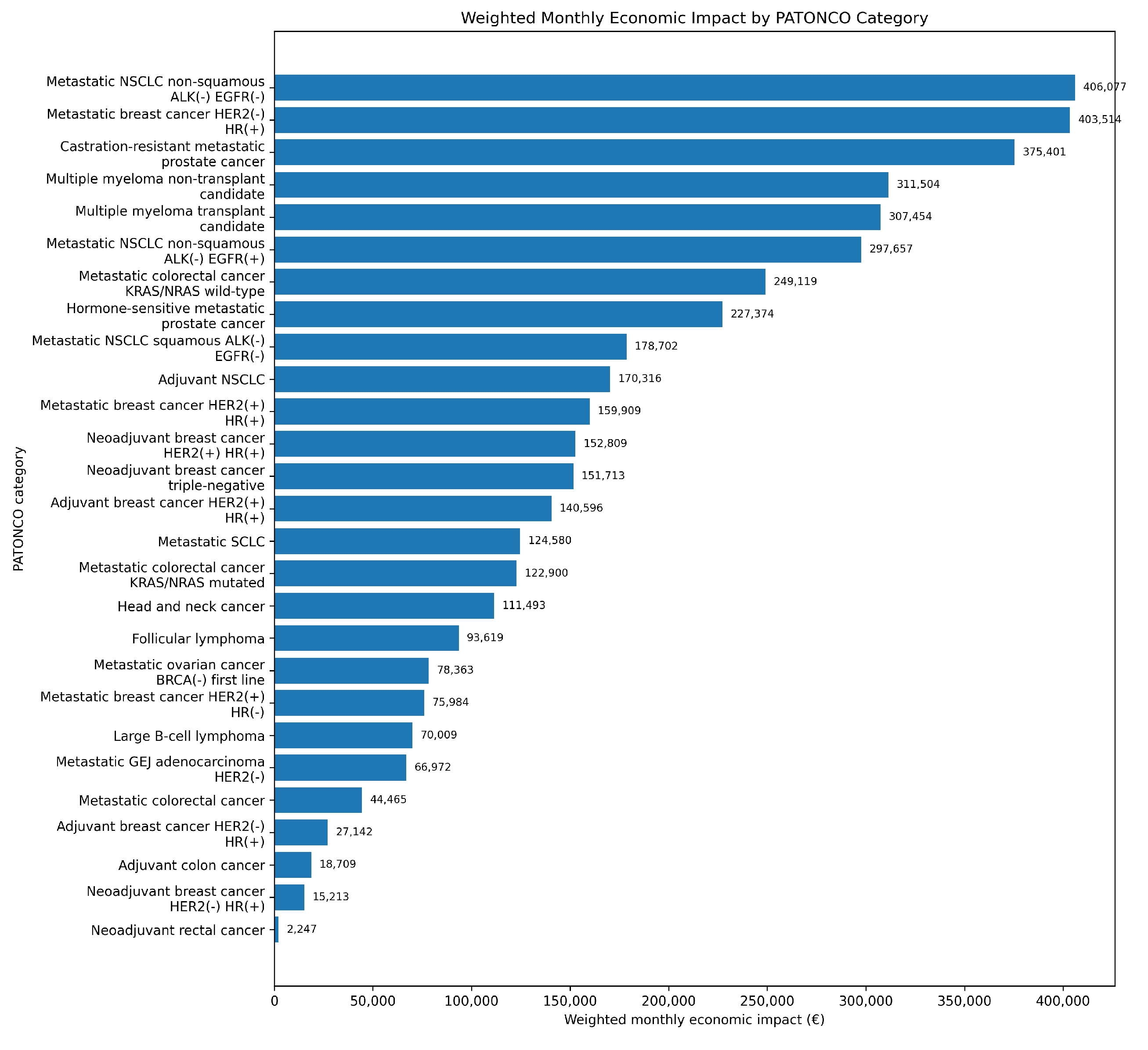
| PATONCO Category | H1 | H2 | H3 | H4 | Avg (€) | Dispensed | Patients | H | Impact (€) |
|---|---|---|---|---|---|---|---|---|---|
| MNSCLC ALK(-) EGFR(-) | 1851.02 | 2327.83 | 1832.98 | 1797.35 | 1952.30 | 1130 | 208 | 25.29** | 406077.36 |
| MBC HER2(-) HR(+) | 1546.76 | 1192.50 | 1497.64 | 1426.45 | 1415.84 | 2079 | 285 | 21.63** | 403513.69 |
| CR metastatic prostate cancer | 2387.42 | 1915.45 | 2962.25 | 1780.68 | 2261.45 | 940 | 166 | 52.37** | 375400.70 |
| MM non-transplant | 4002.05 | 2238.41 | 2219.96 | 2664.74 | 2781.29 | 815 | 112 | 61.33** | 311504.48 |
| MM transplant candidate | 2009.50 | 1106.19 | 2613.81 | 3272.27 | 2009.50 | 1012 | 153 | 11.56** | 307453.50 |
| MNSCLC ALK(-) EGFR(+) | 4507.12 | 3501.82 | 3749.08 | 4117.00 | 3968.76 | 424 | 75 | 8.35* | 297656.63 |
| MCRC KRAS/NRAS wt | 1632.86 | 1650.30 | 1058.25 | 1520.21 | 1465.41 | 1209 | 170 | 92.61** | 249118.85 |
| HS metastatic prostate cancer | 2265.70 | 2621.13 | 2763.11 | 1354.96 | 2251.23 | 498 | 101 | 40.22** | 227373.73 |
| NSCLC squamous | 2055.38 | 2218.76 | 1532.76 | 1562.25 | 1842.29 | 392 | 97 | 20.71** | 178701.89 |
| Adjuvant NSCLC | 1313.05 | 286.05 | 1045.29 | 2163.33 | 1507.22 | 377 | 113 | 8.06* | 170316.24 |
| MBC HER2(+) HR(+) | 2685.88 | 2425.44 | 2495.37 | 2233.89 | 2460.15 | 625 | 65 | 7.19NS | 159909.43 |
| Neoadjuvant breast HER2(+) | 2649.06 | 2208.15 | 1896.48 | 3266.54 | 2505.06 | 320 | 61 | 8.25* | 152808.51 |
| Neoadjuvant breast TN | 2446.99 | 58.35 | 101.94 | 423.67 | 2446.99 | 295 | 62 | 141.47** | 151713.38 |
| Adjuvant breast HER2(+) | 2143.24 | 1670.44 | 1766.15 | 1449.95 | 1757.45 | 510 | 80 | 28.42** | 140595.60 |
| Metastatic SCLC | 1163.57 | 1283.81 | 715.06 | 1253.02 | 1233.47 | 353 | 101 | 18.45** | 124580.13 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




