Submitted:
12 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
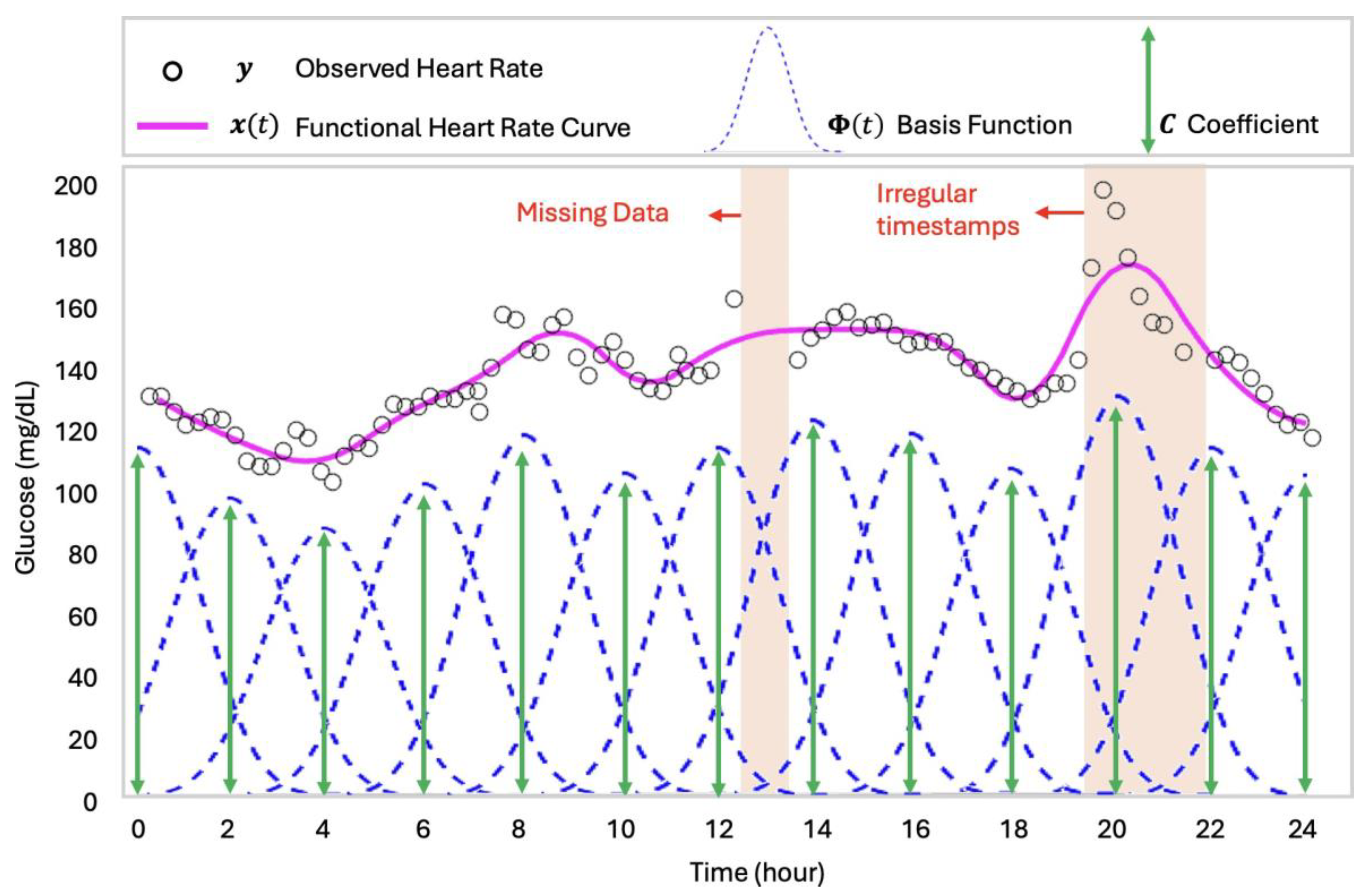
2.2. Continuous Glucose Monitoring via Functional Data Analysis
3. Results
3.1. Continuous Glucose Monitoring via Functional Data Analysis
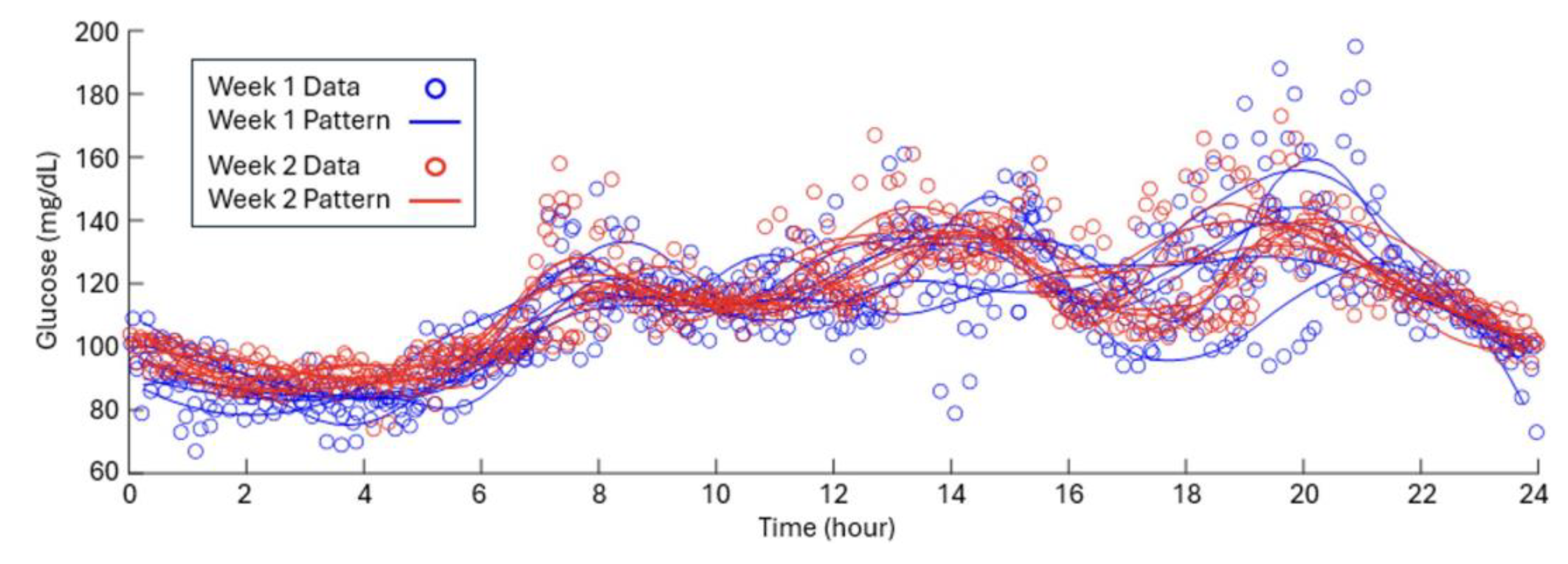
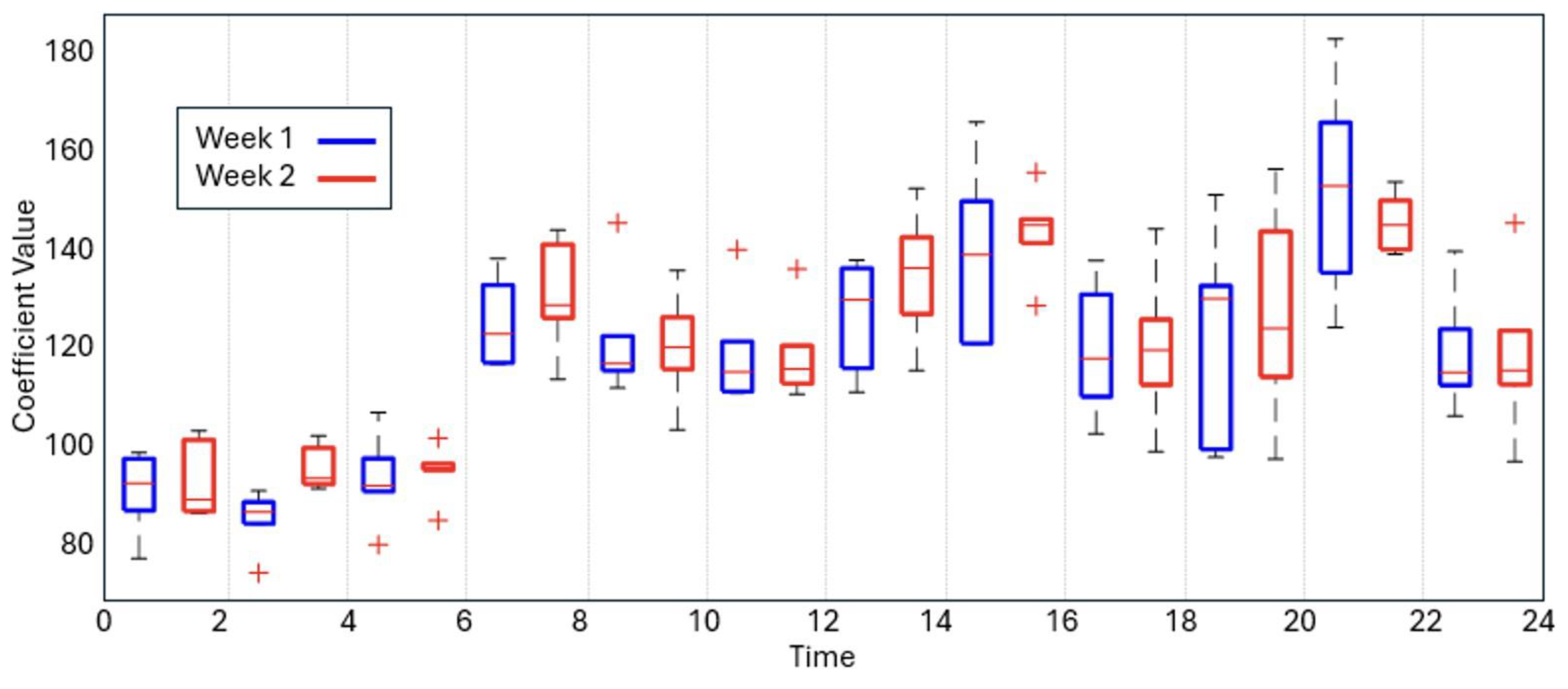
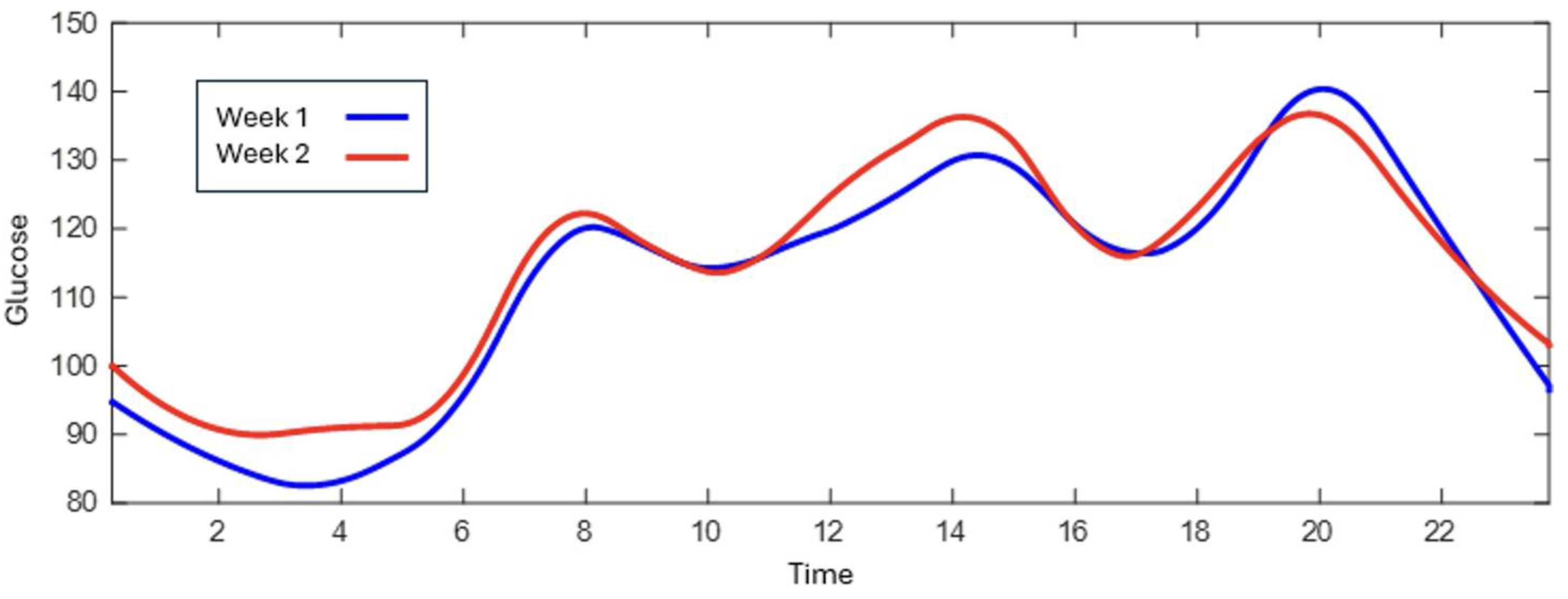
3.2. Pre/Post-Intervention Comparison
4. Conclusions
References
- Diabetes — who . int. Available online: https://www.who.int/news-room/fact-sheets/detail/diabetes? (accessed on 19-10-2025).
- Stafford, L. K.; Gage, A.; Xu, Y. Y. Global, regional, and national cascades of diabetes care. The Lancet Diabetes & Endocrinology 2025. [Google Scholar]
- Galicia-Garcia, U.; Benito-Vicente, A.; Jebari, S. Pathophysiology of type 2 diabetes mellitus. International journal of molecular sciences 2020, vol. 21(no. 17), 6275. [Google Scholar] [CrossRef] [PubMed]
- Echouffo-Tcheugui, J. B.; Perreault, L.; Ji, L.; Dagogo-Jack, S. Diagnosis and management of prediabetes: A review. Jama 2023, vol. 329(no. 14), 1206–1216. [Google Scholar] [CrossRef] [PubMed]
- for Disease Control, C.; Prevention. National diabetes fact sheet: National estimates and general information on diabetes and prediabetes in the united states, 2011. Atlanta, GA: US department of health and human services, centers for disease control and prevention 2011, vol. 201(no. 1), 2568–2569. [Google Scholar]
- CDC Newsroom — archive.cdc.gov . Available online: https:// archive . cdc . gov / www _ cdc _ gov / media / releases/2017/p0718-diabetes-report.html (accessed on 19-10-2025).
- Lagisetty, P. A.; Priyadarshini, S.; Terrell, S. Culturally targeted strategies for diabetes prevention in minority population: A systematic review and framework. The Diabetes Educator 2017, vol. 43(no. 1), 54–77. [Google Scholar] [CrossRef]
- Jung, H. S. Clinical implications of glucose variability: Chronic complications of diabetes. En- docrinology and Metabolism 2015, vol. 30(no. 2), 167–174. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Pan, Y.; Zhou, K.; Liu, H.; Zhong, S. Correlation between glycemic variability and diabetic complications: A narrative review. International Journal of General Medicine 2023, 3083–3094. [Google Scholar] [CrossRef] [PubMed]
- Hall, H.; Perelman, D.; Breschi, A. Glucotypes reveal new patterns of glucose dysregulation. PLoS biology 2018, vol. 16(no. 7), e2005143. [Google Scholar] [CrossRef]
- Joseph, J. I. Review of the long-term implantable senseonics continuous glucose monitoring system and other continuous glucose monitoring systems. Journal of Diabetes Science and Technology 2021, vol. 15(no. 1), 167–173. [Google Scholar] [CrossRef] [PubMed]
- Vettoretti, M.; Cappon, G.; Acciaroli, G.; Facchinetti, A.; Sparacino, G. Continuous glucose monitoring: Current use in diabetes management and possible future applications. Journal of diabetes science and technology 2018, vol. 12(no. 5), 1064–1071. [Google Scholar] [CrossRef]
- Gecili, E.; Huang, R.; Khoury, J. C. Functional data analysis and prediction tools for continuous glucose-monitoring studies. Journal of clinical and translational science 2021, vol. 5(no. 1), e51. [Google Scholar] [CrossRef] [PubMed]
- Xia, S.; Nishat, T. A. H.; Jo, H.; Liu, J. Regularized tensor completion for structural health monitoring data imputation. in 2025 11th International Conference on Computing and Artificial Intelligence (ICCAI) 2025, 648–653. [Google Scholar]
- Wang, J.-L.; Chiou, J.-M.; Müller, H.-G. Functional data analysis. Annual Review of Statistics and its application 2016, vol. 3(no. 1), 257–295. [Google Scholar] [CrossRef]
- Matabuena, M.; Pazos-Couselo, M.; Alonso-Sampedro, M.; Fernández-Merino, C.; González- Quintela, A.; Gude, F. Reproducibility of continuous glucose monitoring results under real-life conditions in an adult population: A functional data analysis. Scientific Reports 2023, vol. 13(no. 1), 13 987. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Jiang, M.; Li, C.; Luo, S.; Crowley, M. J.; Shaw, R. J. Predicting health outcomes with intensive longitudinal data collected by mobile health devices: A functional principal component regression approach. BMC Medical Research Methodology 2024, vol. 24(no. 1), 69. [Google Scholar] [CrossRef] [PubMed]
- Xia, S.; Wung, S.-F.; Chen, C.-C.; Coompson, J. L. K.; Roveda, J.; Liu, J. Data-fusion-based quality enhancement for hr measurements collected by wearable sensors. Sensors 2024, vol. 24(no. 10), 2970. [Google Scholar] [CrossRef] [PubMed]
- Xia, S.; Zhang, Y.; Lansey, K.; Liu, J. Penalized spatial-temporal sensor fusion for detecting and localizing bursts in water distribution systems. Information Fusion vol. 117, 102 912, 2025. [CrossRef]
 |
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




