Submitted:
13 April 2026
Posted:
14 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
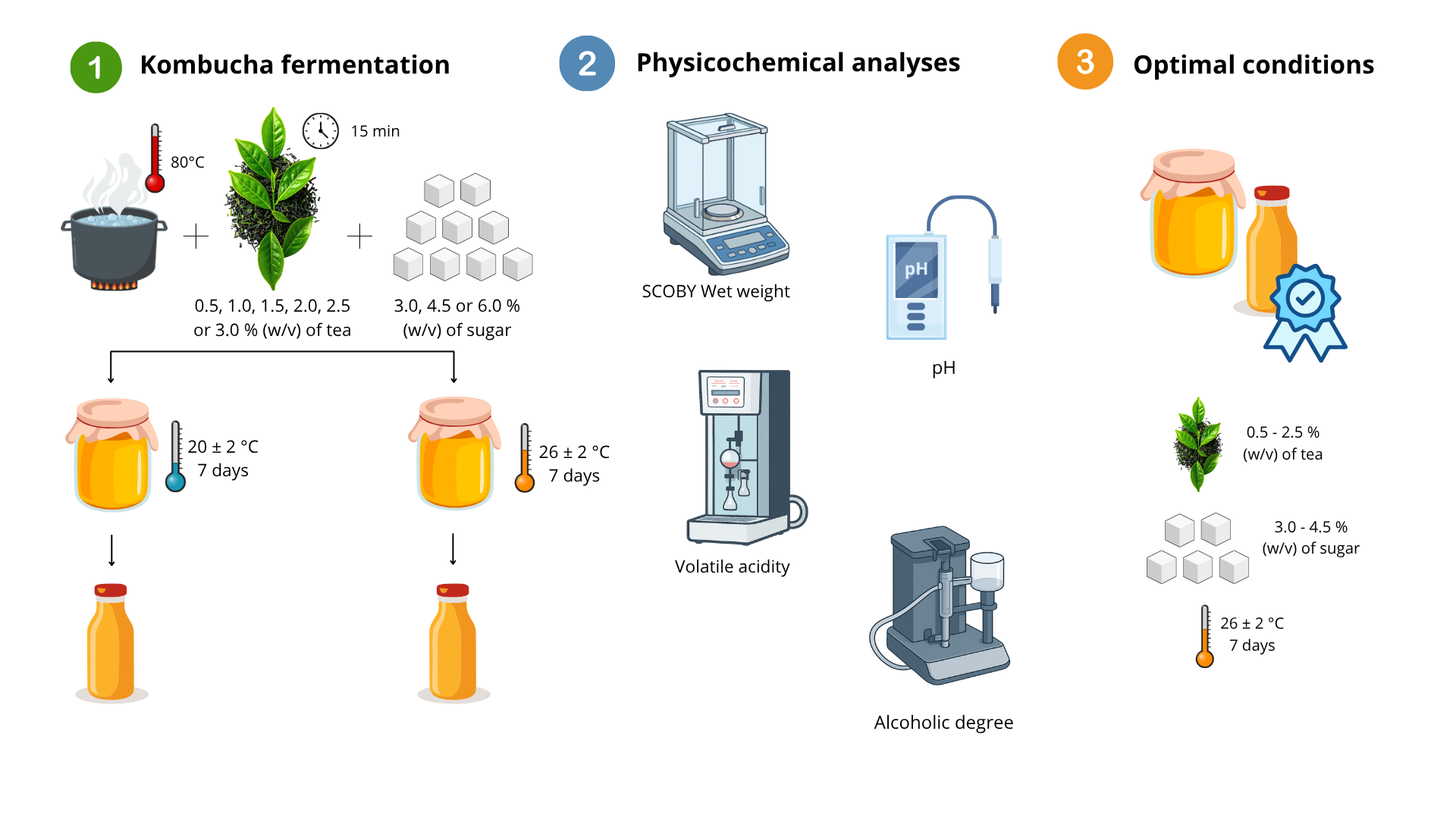
2. Materials and Methods
2.1. Kombucha Preparation
2.2. SCOBY Growth
2.3. pH, Alcohol Content, and Volatile Acidity
2.4. Data Analysis
3. Results and Discussion
3.1. Kombucha Production at 20 °C ± 2 °C
3.2. Kombucha Production at 26 ± 2 °C
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| SCOBY | Symbiotic Culture of Bacteria and Yeast |
References
- Onsun, B.; Toprak, K.; Sanlier, N. Kombucha Tea: A Functional Beverage and All Its Aspects. Curr. Nutr. Rep. 2025, 14, 69. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, C.D.; Baqueta, M.R.; Santos, W.C.R.; Gomes, D.; Alvarenga, V.O.; Teixeira, P.; Albano, H.; Rosa, C.A.; Valderrama, P.; Lacerda, I.C. Data fusion of UPLC data, NIR spectra and physicochemical parameters with chemometrics as an alternative to evaluating kombucha fermentation. LWT 2020, 133, 109875. [Google Scholar] [CrossRef]
- Treviso, R.L.; Sant’Anna, V.; Fabricio, M.F.; Ayub, M.A.Z.; Brandelli, A.; Hickert, L.R. Time and temperature influence on physicochemical, microbiological, and sensory profiles of yerba mate kombucha. J. Food Sci. Technol. 2024, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Saito, M.S.; Ferreira, R.M.S.; de Jesus, A.C.A.; Mamede, M.E.D.O. Analysis of innovations in kombucha production and the protection of intellectual property. Biotech. Resear. Innov. 2024, 8(2), e2024016. [Google Scholar] [CrossRef]
- Cheepchirasuk, N.; Kaewkod, T.; Suriyaprom, S.; Intachaisri, V.; Ngamsaard, P.; Tragoolpua, Y. Functional Metabolites and Inhibitory Efficacy of Kombucha Beverage on Pathogenic Bacteria, Free Radicals and Inflammation. Sci. Rep. 2025, 15, 19187. [Google Scholar] [CrossRef] [PubMed]
- Brasil. Ministério da Agricultura e Pecuária. Instrução Normativa n° 41, de 17 de Setembro de 2019. Estabelece o Padrão de Identidade e Qualidade da Kombucha em todo o território nacional. Diário Oficial da União, Brasília, 18 de Setembro de 2019. Seção 1. Available online: https://pesquisa.in.gov.br/imprensa/jsp/visualiza/index.jsp?data=18/09/2019&jornal=515&pagina=13&totalArquivos=76 (accessed on 1 February 2026).
- Kombucha Brewers International. Kombucha Code of Practice. Available online: https://kombuchabrewers.org/kombucha-code-of-practice/ (accessed on 1 February 2026).
- Laavanya, D.; Shirkole, S.; Balasubramanian, P. Current challenges, applications and future perspectives of SCOBY cellulose of Kombucha fermentation. J. Clean. Prod. 2021, 295, 126454. [Google Scholar] [CrossRef]
- Goh, W. N.; Rosma, A.; Kaur, B.; Fazilah, A.; Karim, A. A.; Bhat, R. Fermentation of black tea broth (Kombucha): I. Effects of sucrose concentration and fermentation time on the yield of microbial cellulose. Int. Food Res. J. 2012, 19(1). [Google Scholar]
- Cohen, G.; Sela, D.A.; Nolden, A.A. Sucrose Concentration and Fermentation Temperature Impact the Sensory Characteristics and Liking of Kombucha. Foods 2023, 12, 3116. [Google Scholar] [CrossRef] [PubMed]
- Saito, M.S.; dos Santos, W.A.; Mamede, M.E.d.O. Coffee-Flavoured Kombucha: Development, Physicochemical Characterisation, and Sensory Analysis. Fermentation 2024, 10, 334. [Google Scholar] [CrossRef]
- Villarreal-Soto, S.A.; Beaufort, S.; Bouajila, J.; Souchard, J.P.; Taillandier, P. Understanding Kombucha Tea Fermentation: A Review. J. Food Sci. 2018, 83, 580–588. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, C.D.; Costa, I.M.; Santos, W.C.R.; Alvarenga, V.O.; Lacerda, I.C.A.; Simiqueli, G.F. Acetic Acid Bacteria Analysis Using Non-Conventional Culture Media in Kombucha Fermentation. Ensai. e Ciên.: Ciên. Biolo., Agrar. e da Sau. 2023, 27(3), 370–375. [Google Scholar] [CrossRef]
- Priyadharshini, T.; Nageshwari, K.; Vimaladhasan, S.; Prakash, S.P.; Balasubramanian, P. Machine Learning Prediction of SCOBY Cellulose Yield from Kombucha Tea Fermentation. Bioresour. Technol. Rep. 2022, 18, 101027. [Google Scholar] [CrossRef]
- Dartora, B.; Hickert, L. R.; Fabricio, M. F.; Ayub, M. A. Z.; Furlan, J. M.; Wagner, R; Perez, K. J.; Sant’Anna, V. Understanding the effect of fermentation time on physicochemical characteristics, sensory attributes, and volatile compounds in green tea kombucha. Food Res. Int. 2023, 174, 113569. [Google Scholar] [CrossRef] [PubMed]
- Noronha, M.C.D.; Cardoso, R.R.; D’Almeida, C.T.S.; do Carmo, M.A.V.; Azevedo, L.; Maltarollo, V.G.; Ribeiro Júnior, J.I.; Eller, M.R.; Cameron, L.C.; Ferreira, M.S.L.; et al. Black tea kombucha: Physicochemical, microbiological and comprehensive phenolic profile changes during fermentation, and antimalarial activity. Food Chem. 2022, 384, 132515. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, J.T.; da Costa, F.M.; da Silva, T.G.; Simoes, G.D.; Pereira, E.S.; da Costa, P.Q.; Andreazza, R.; Schenkel, P.C.; Pieniz, S. Green tea and kombucha characterization: Phenolic composition, antioxidant capacity and enzymatic inhibition potential. Food Chem. 2023, 408, 135206. [Google Scholar] [CrossRef] [PubMed]
- Azfaralariff, A.; Vohra, B.; Fazry, S; Law, D.; Sairi, F.; Othman, B.A. Effects of starter culture and sweetener on biochemical compounds and microbial diversity of kombucha tea. Sains Malaysiana 2022, 51(11), 3715–3729. [Google Scholar]
| Parameters | Sugar (% w/v) | |||
|---|---|---|---|---|
| Tea (% w/v) | 3.0 | 4.5 | 6.0 | |
| SCOBY growth (g%) | 0.5 | 9 | 8 | 0 |
| 1.0 | 27 | 7 | 0 | |
| 1.5 | 32 | 17 | 0 | |
| 2.0 | 35 | 20 | 0 | |
| 2.5 | 14 | 18 | 0 | |
| 3.0 | 20 | 8 | 0 | |
| pH | 0.5 | 2.89 ± 0.00eC | 3.32 ± 0.01bA | 3.11 ± 0.01eB |
| 1.0 | 2.97 ± 0.01dC | 3.28 ± 0.02cA | 3.20 ± 0.00bB | |
| 1.5 | 3.02 ± 0.01cC | 3.21 ± 0.01eA | 3.13 ± 0.01dB | |
| 2.0 | 3.03 ± 0.02cC | 3.25 ± 0.01dA | 3.19 ± 0.01bB | |
| 2.5 | 3.09 ± 0.00bC | 3.27 ± 0.00cA | 3.17 ± 0.01cB | |
| 3.0 | 3.17 ± 0.00aC | 3.41 ± 0.01aA | 3.24 ± 0.01aB | |
| Volatile acidity (mEq L-1) | 0.5 | 93.52 ± 0.38dA | 38.88 ± 4.06dC | 78.18 ± 0.39eB |
| 1.0 | 102.55 ± 0.14cA | 63.12 ± 0.00cC | 82.14 ± 6.27eB | |
| 1.5 | 111.51 ± 1.29bA | 91.82 ± 0.89aB | 114.46 ± 0.73dA | |
| 2.0 | 123.83 ± 2.77aA | 89.12 ± 0.67aB | 124.39 ± 1.27cA | |
| 2.5 | 127.02 ± 0.63aA | 90.55 ± 0.81aB | 130.11 ± 0.64bA | |
| 3.0 | 107.46 ± 0.15bB | 75.74 ± 0.76bC | 135.24 ± 0.29aA | |
| Alcohol content (% v/v) | 0.5 | ND | ND | ND |
| 1.0 | ND | ND | 0.23 ± 0.05a | |
| 1.5 | ND | ND | 0.16 ± 0.03b | |
| 2.0 | ND | 0.14 ± 0.00c | ND | |
| 2.5 | ND | 0.22 ± 0.04ª,b | ND | |
| 3.0 | ND | 0.19 ± 0.02b | ND | |
|
Parameters |
Sugar (% w/v) | |||
|---|---|---|---|---|
| Tea (% w/v) | 3.0 | 4.5 | 6.0 | |
| SCOBY growth (g%) | 0.5 | 10 | 23 | 45 |
| 1.0 | 4 | 21 | 66 | |
| 1.5 | 32 | 31 | 24 | |
| 2.0 | 43 | 93 | 58 | |
| 2.5 | 65 | 146 | 203 | |
| 3.0 | 128 | 19 | 99 | |
| pH | 0.5 | 3.32 ± 0.01eC | 3.36 ± 0.01eB | 3.38 ± 0.01eA |
| 1.0 | 3.50 ± 0.01cA | 3.36 ± 0.00eB | 3.26± 0.01fC | |
| 1.5 | 3.28 ± 0.01fC | 3.40 ± 0.00dB | 3.47 ± 0.00cA | |
| 2.0 | 3.60 ± 0.00aA | 3.50 ± 0.01bC | 3.58 ± 0.01aB | |
| 2.5 | 3.57 ± 0.01bA | 3.44 ± 0.00cB | 3.44 ± 0.01dB | |
| 3.0 | 3.49 ± 0.01dC | 3.56 ± 0.00aA | 3.53 ± 0.01bB | |
| Volatile acidity (mEq L-1) | 0.5 | 38.31 ± 0.28eA | 41.91 ± 0.91eA | 34.42 ± 0.64eB |
| 1.0 | 31.23 ± 0.79fC | 63.54 ± 0.15c,dB | 69.09 ± 6.69bA | |
| 1.5 | 79.83 ± 0.14bA | 66.15 ± 2.52cB | 46.12 ± 0.39dC | |
| 2.0 | 50.96 ± 0.14dB | 59.75 ± 1.62dA | 51.42 ± 0.53cB | |
| 2.5 | 71.68 ± 1.12cB | 83.23 ± 0.77aA | 83.74 ± 0.53aA | |
| 3.0 | 94.26 ± 0.28aA | 71.03 ± 2.26bB | 73.30 ± 0.29bB | |
| Alcohol content (% v/v) | 0.5 | ND | 0.13 ± 0.00e | ND |
| 1.0 | 0.19 ± 0.01c | 0.16 ± 0.01d,e | 0.18 ± 0.01c | |
| 1.5 | 0.16 ± 0.02c | 0.21 ± 0.03d | ND | |
| 2.0 | 0.35 ± 0.05b | 0.26 ± 0.04c,d | 0.23 ± 0.04c | |
| 2.5 | 0.19 ± 0.03c | 0.38 ± 0.04b | 0.43 ± 0.05b | |
| 3.0 | 0.44 ± 0.02a | 0.50 ± 0.06a | 0.51 ± 0.07c | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




