Submitted:
10 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Patients and Methods
- puls determination on the remaining distal segment of the splenic artery 15-30 minutes after clamping;
- assessment of changes in the color and consistency of the spleen. Significant darkening and cyanosis, as well as pronounced swelling of the spleen were contraindications for its preservation.
- intraoperative ultrasound for the assessment of intraparenchymal arterial blood flow in the spleen. Detection of arterial blood flow in the splenic parenchyma was considered sufficient to recognize it as viable [30];
- assessment of the retrograde blood flow after removing the clamp from the remaining distal segment of the splenic artery (Video S1. Collateral blood flow test 1);
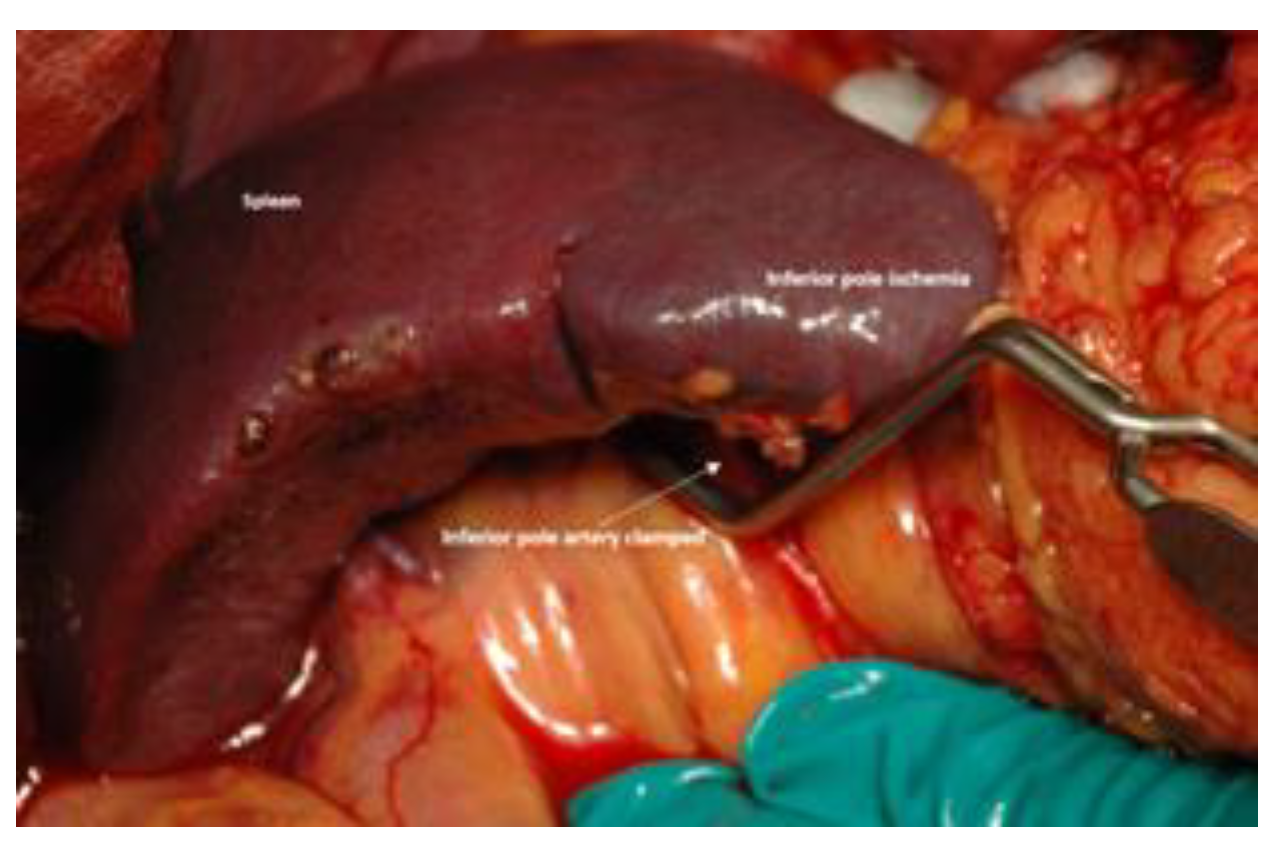
- If, after clamping both splenic vessels, the spleen acquired a darker color and assessment of its arterial blood flow became difficult, the following technique used. A soft vascular clamp (“bulldog”) occluded one of the terminal (usually the lower pole) arteries of the spleen. If within 15-20 minutes the sector of the spleen supplied though the clamped branch changed color to a darker one (see Figure 1) compared to the rest of the spleen, then the arterial blood flow of the spleen (subject to the above conditions) was considered adequate:
- In case of preservation of the distal pancreas during right-sided pancreatectomy, besides the above steps, an additional transection of the pancreatic stump was performed. When arterial bleeding appeared from the cut surface, and the first four tests were positive, the blood supply to the pancreatic stump was considered sufficient (Video S2. Collateral blood flow test 2).
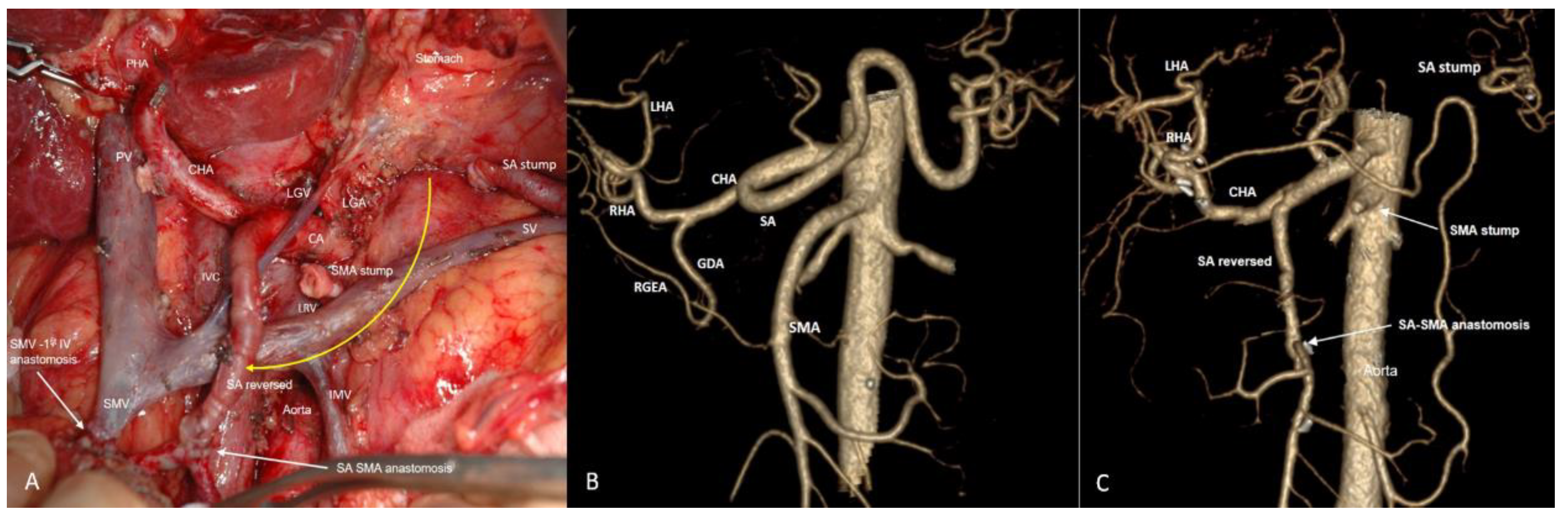
- A mandatory requirement for performing PD with resection of the splenic artery with its rotation, was (a) a hard pancreas; (b) wide (at least 5 mm) pancreatic tail duct, and removal of the pancreatic body
Statistics
3. Results
3.1. Spleen-Preserving Distal Pancreatectomies (SPDP)
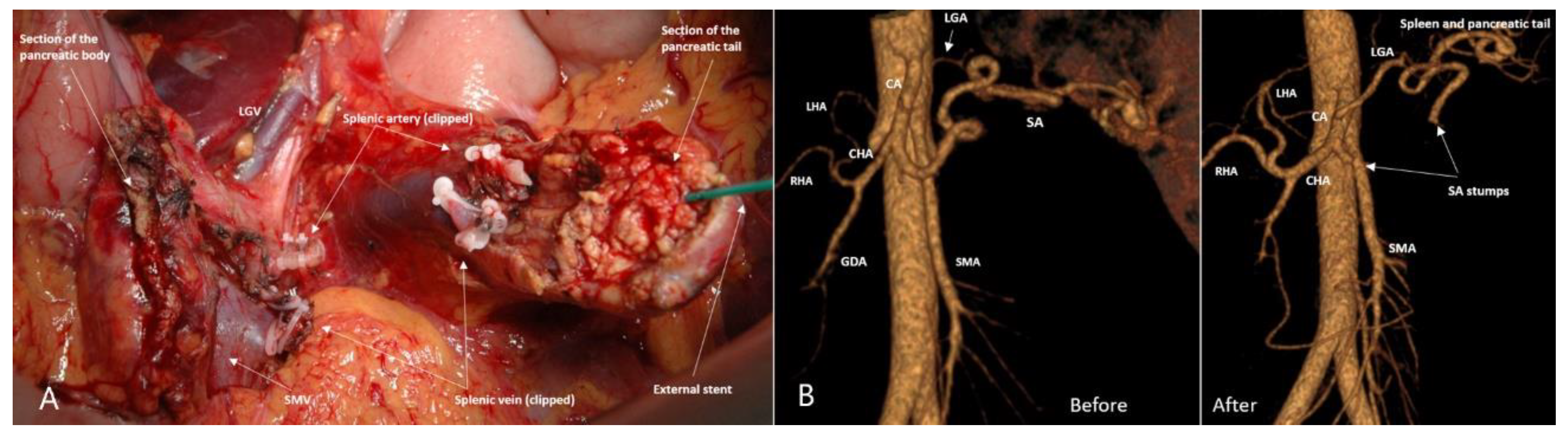
3.2. Spleen-Preserving Pancreatectomies with Preservation of the Splenic Vessels, Other than Distal Resections, Were Performed in Seven Cases
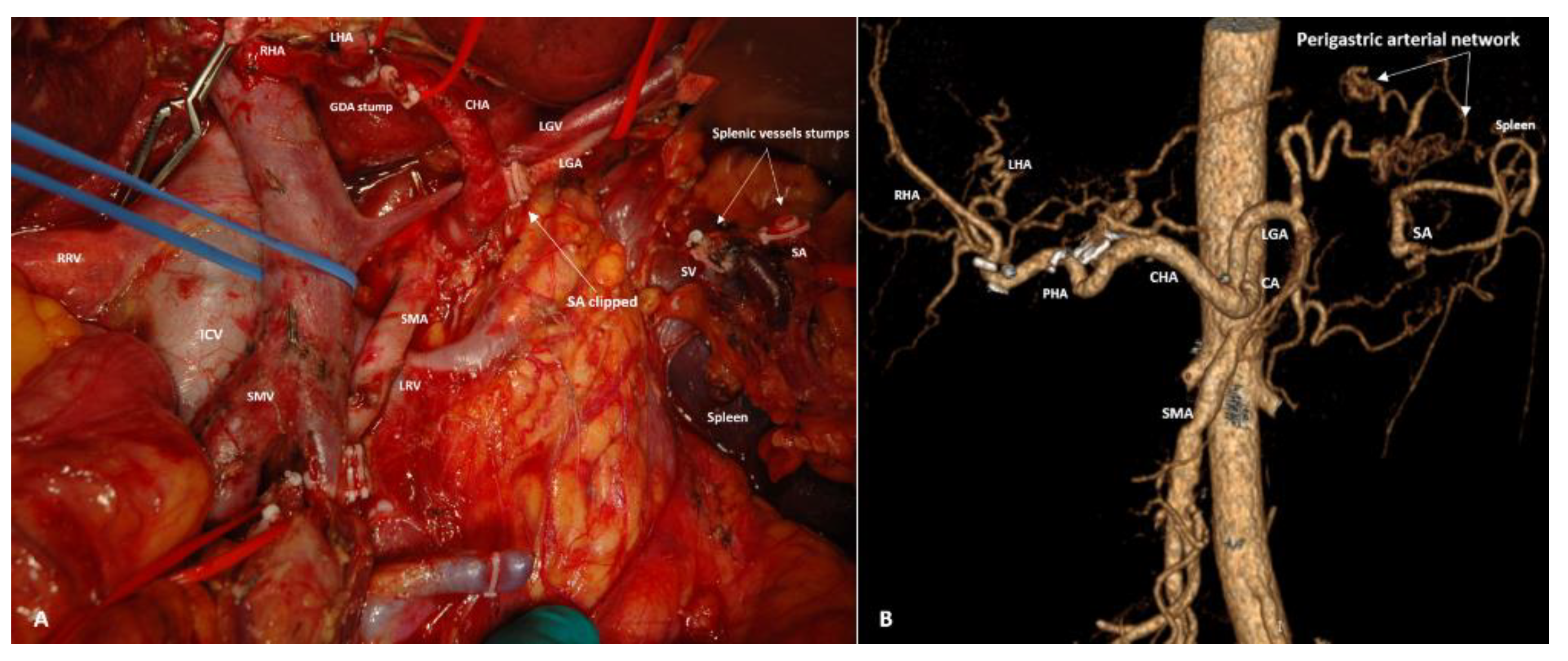
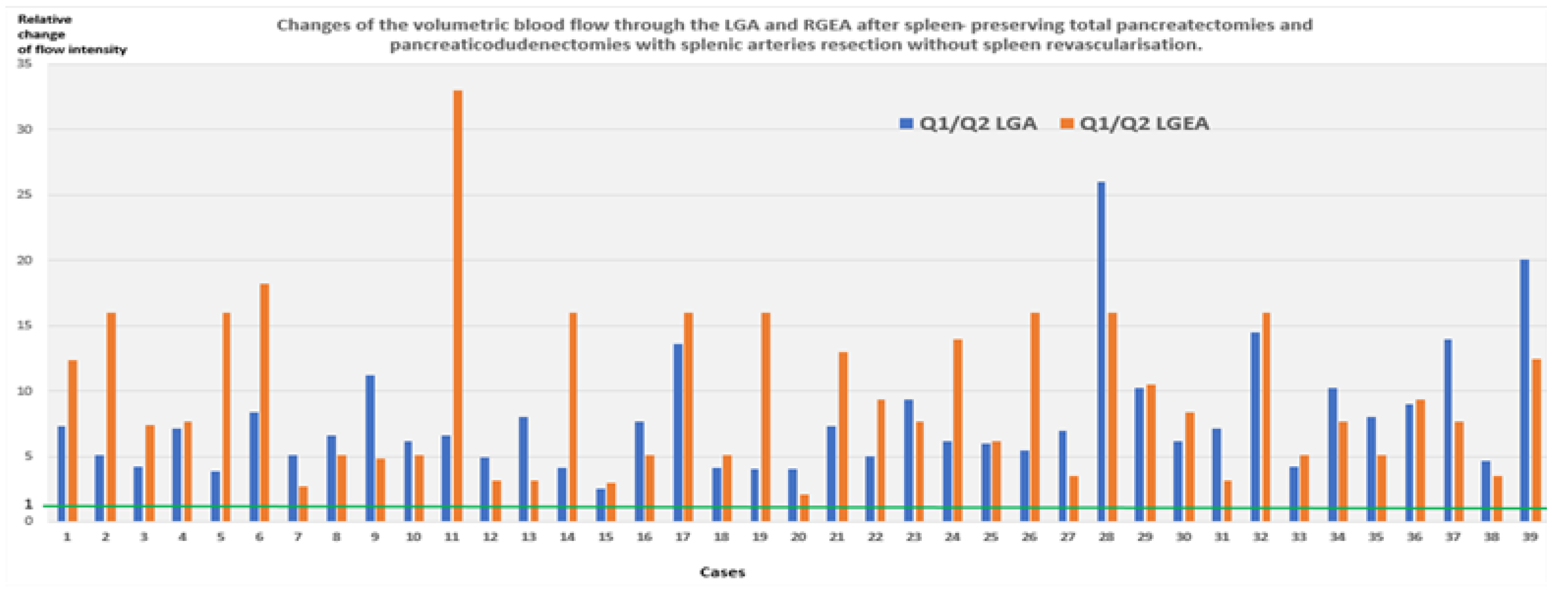
3.3. Spleen-Preserving Pancreatectomies with Resection of the Splenic Vessels, Other than Distal Resections
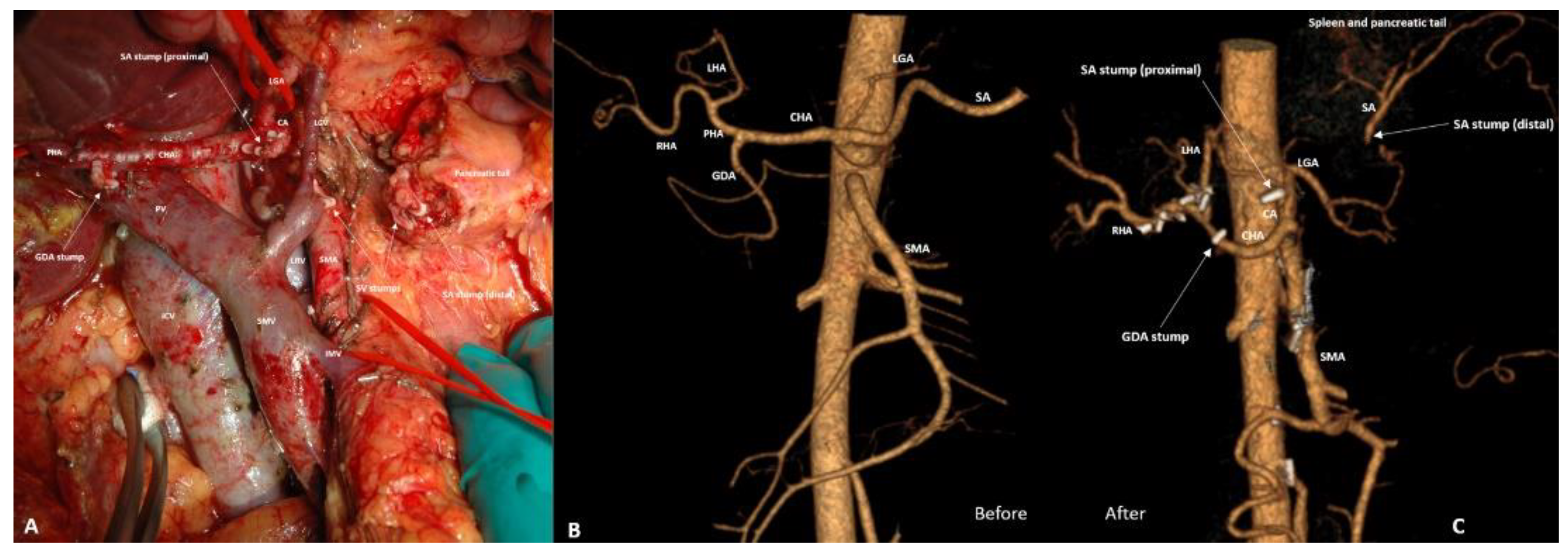
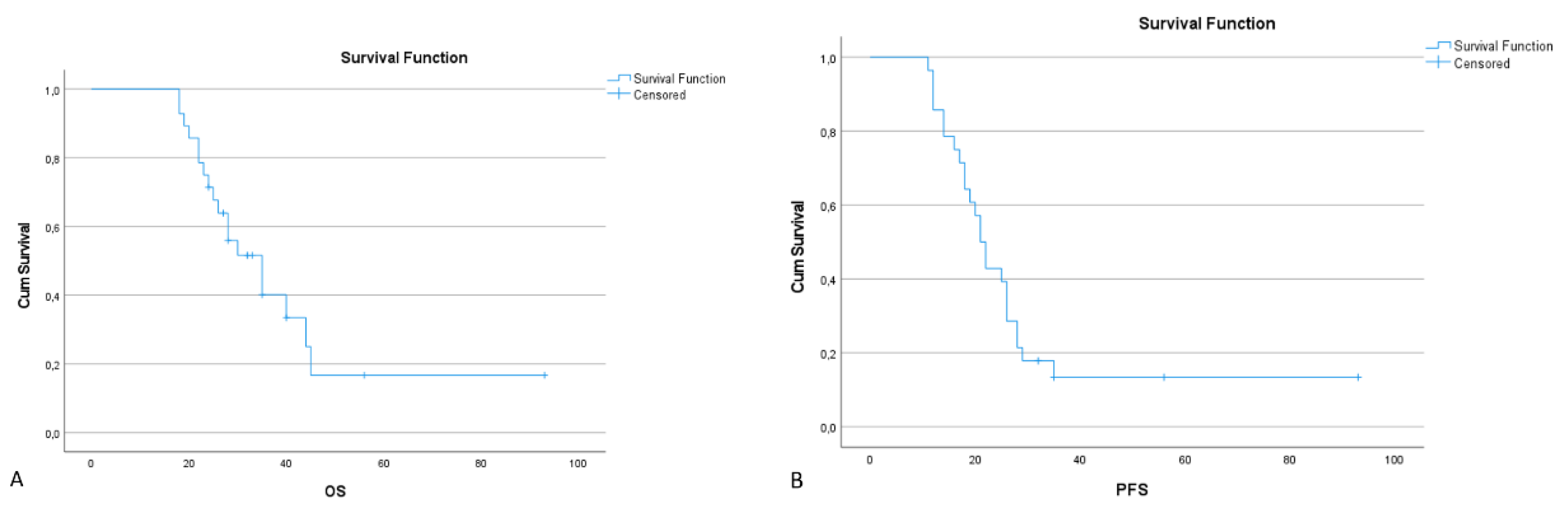
3.4. Spleen-Preserving Pancreatectomies with Resection of the Splenic Artery (Vessels) for Border Line Resectable and Locally Advanced Pancreatic Head Cancers
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Sutherland, DERS; Goetz, FC; Najarian, JS. Living-related donor segmental pancreatectomy for transplantation. Transplant Proc 1980, 12 (Suppl. 2), 19–25. [Google Scholar] [PubMed]
- Warshaw, AL. Conservation of the spleen with distal pancreatectomy. Arch Surg 1988, 123, 550–3. [Google Scholar] [CrossRef]
- Korrel, M; Lof, S; Al Sarireh, B; et al. Short-term outcomes after spleen-preserving minimally invasive distal pancreatectomy with or without preservation of splenic vessels: a pan-European retrospective study in high-volume centers. European consortium on minimally invasive pancreatic surgery (E-MIPS). Ann Surg 2023, 277(1), e119–e125. [Google Scholar] [PubMed]
- He, Z; Qian, D; Hua, J; et al. Clinical comparison of distal pancreatectomy with or without splenectomy: a meta-analysis. PlosOne 2014, 9(3), e91593. [Google Scholar] [CrossRef] [PubMed]
- Timmerhuis, HC; Ngongoni, RF; Jensen, CW; et al. Comparison of spleen-preservation versus splenectomy in minimally invasive distal pancreatectomy. Gastrointest Surg 2023, 27(10), 2166–2176. [Google Scholar] [CrossRef]
- Șandra-Petrescu, F; Tzatzarakis, E; Mansour Basha, M; et al. Impact of spleen preservation on the incidence of postoperative pancreatic fistula after distal pancreatectomy: Is less More? Pancreatology 2022, 22(7), 1013–1019. [Google Scholar] [CrossRef]
- Sun, LM; Chen, HJ; Jeng, LB; et al. Splenectomy and increased subsequent cancer risk: a nationwide population-based cohort study. Am J Surg 210, 243–251. [CrossRef]
- Xu, JL; Tang, CW; Feng, WM; et al. Hematological benefits after spleen-preserving distal pancreatectomy: perioperative and long-term outcome analysis. Hepatogastroenterol 2013, 60, 2103–21069. [Google Scholar]
- Cui, M; Liu, JK; Zheng, B; et al. Dynamic hematological changes in patients undergoing distal pancreatectomy with or without splenectomy: a population-based cohort study. BMC Surg. 2020, 20(1), 265. [Google Scholar] [CrossRef]
- Clément, E; Addeo, P; Sauvanet, A; et al. Benefit of splenectomy in distal pancreatectomy for neuroendocrine tumours: multicentre retrospective study. Collaborators. BJS Open 2025, 9(3), zraf038. [Google Scholar] [CrossRef]
- Codjia, T; Hobeika, C; Platevoet, P; et al. Distal pancreatectomy for body pancreatic ductal adenocarcinoma: Is splenectomy necessary? A propensity score matched study. Ann Surg Oncol 2024, 31(7), 4611–4620. [Google Scholar] [CrossRef] [PubMed]
- Zhou, E; Shi, G; Shi, H; et al. Outcome after spleen- preserving distal pancreatectomy by Warshaw technique for pancreatic body cancer. Ann Hepatobiliary Pancreat Surg 2025, 29(2), 177–186. [Google Scholar] [CrossRef]
- Mallet-Guy, P; Vachon, A. Pancreatites chroniques gauches; Masson & Cie: Paris, 1943. [Google Scholar]
- Kimura, W; Inoue, T; Futakawa, N; et al. Spleen-preserving distal pancreatectomy with conservation of the splenic artery and vein. Surgery 1996, 120, 885–90. [Google Scholar] [CrossRef] [PubMed]
- Ferrone, CR; Konstantinidis, IT; Sahani, DV; et al. Twenty-three years of the Warshaw operation for distal pancreatectomy with preservation of the spleen. Ann Surg 2011, 253, 1136–9. [Google Scholar] [CrossRef] [PubMed]
- Yu, X; Li, H; Jin, C; et al. Splenic vessel preservation versus Warshaw’s technique during spleen-preserving distal pancreatectomy: a meta-analysis and systematic review. Langenbecks Arch Surg 2015, 400(2), 183–91. [Google Scholar] [CrossRef]
- Li, BQ; Qiao, YX; Li, J; et al. Preservation or ligation of splenic vessels during spleen-preserving distal pancreatectomy: a meta-analysis. J Invest Surg 2019, 32(7), 654–669. [Google Scholar] [CrossRef]
- Hang, K; Zhou, L; Liu, H; et al. Splenic vessels preserving versus Warshaw technique in spleen preserving distal pancreatectomy: a systematic review and meta-analysis. Int J Surg 2022, 103, 106686. [Google Scholar] [CrossRef]
- Granieri, S; Bonomi, A; Frassini, S; et al. Kimura’s vs Warshaw’s technique for spleen preserving distal pancreatectomy: a systematic review and meta-analysis of high-quality studies. HPB (Oxford) 2023, 25(6), 614–624. [Google Scholar] [CrossRef]
- World Medical Association. World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA 2013, 310, 2191–4. [Google Scholar] [CrossRef]
- Mathew, G; Sohrabi, C; Franchi, T; Nicola, M; Kerwan, A; Agha, R; PROCESS Group. Preferred Reporting Of Case Series in Surgery (PROCESS) 2023 guidelines. Int J Surg. 2023, 109(12), 3760–3769. [Google Scholar] [CrossRef]
- Mathew, G; Agha, R. for the STROCSS Group. STROCSS 2021: strengthening the reporting of cohort, cross-sectional and case-control studies in surgery. Int J Surg 2021, 96, 106165. [Google Scholar] [CrossRef] [PubMed]
- Strasberg, SM; Sanchez, LA; Hawkins, WG; et al. Resection of tumors of the neck of the pancreas with venous invasion: the “Whipple at the Splenic Artery (WATSA)” procedure. J Gastrointest Surg 2012, 16(5), 1048–54. [Google Scholar] [CrossRef] [PubMed]
- Dindo, D; Demartines, N; Clavien, PA; et al. Classification of surgical complications: A new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 2004, 240, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Bassi, C; Marchegiani, G; Dervenis, C; et al. The 2016 update of the International Study Group (ISGPS) definition and grading of postoperative pancreatic fistula: 11 years after. Surgery 2017, 161, 584–591. [Google Scholar] [CrossRef]
- Wente, MN; Veit, JA; Bassi, C; et al. Postpancreatectomy hemorrhage (PPH) - An International Study Group of pancreatic surgery (ISGPS) definition. Surgery 2007, 142, 20–25. [Google Scholar] [CrossRef]
- Truty, M; Kendrick, M; Nagorney, D; et al. Factors predicting response, perioperative outcomes, and survival following total neoadjuvant therapy for borderline/locally advanced pancreatic cancer. Ann Surg 2021, 273, 341–349. [Google Scholar] [CrossRef]
- Katz, MH; Shi, Q; Ahmad, SA; et al. Preoperative modified FOLFIRINOX treatment followed by capecitabine-based chemoradiation for borderline resectable pancreatic cancer: Alliance for clinical trials in oncology trial A021101. JAMA Surg 2016, 151, e161137. [Google Scholar] [CrossRef]
- National Comprehensive Cancer Network (NCCN) guidelines for pancreatic.
- adenocarcinoma. Version 2. 2023 – June 19, 2023. Available online: https://www.nccn.org/professionals/physician_gls/pdf/pancreatic.pdf (accessed on 16 July 2023).
- Egorov, VI; Yashina, NI; Zhurenkova, TV; et al. Spleen-preserving distal pancreatectomy with resection of the splenic vessels. Should one rely on the short gastric arteries? JOP 2011, 12(5), 445–57. [Google Scholar]
- Yang, HK; Park, MS; Choi, M; et al. Systematic review and meta-analysis of diagnostic performance of CT imaging for assessing resectability of pancreatic ductal adenocarcinoma after neoadjuvant therapy: importance of CT criteria. Abdom Radiol (NY) 2021, 46(11), 5201–5217. [Google Scholar] [CrossRef]
- Michels, NA. Blood supply and anatomy of the upper abdominal organs with a descriptive atlas; Lippincott: Philadelphia, PA, USA, 1955. [Google Scholar]
- Murray, CD. The physiological principle of minimum work. I. The vascular system and the cost of blood volume. Proc Natl Acad Sci, USA 1926, 12, 207–14. [Google Scholar] [CrossRef]
- Zamir, M. Shear forces and blood vessel radii in the cardiovascular system. J Gen Physiol 1977, 69, 449–461. [Google Scholar] [CrossRef]
- Shang, Q. Separation and segmentation of the hepatic vasculature in CT Images. Ph.D. Thesis, Faculty of the Graduate School of Vanderbilt University, Nashville, TN, USA, 2010; p. 113. [Google Scholar]
- Helliwell, T; Woolgar, JA. Standards and minimum datasets for reporting cancers. Minimum dataset for the histopathological reporting of pancreatic, ampulla of Vater and bile duct carcinoma; The Royal College of Pathologists: London, UK, 2002. [Google Scholar]
- Diagnosis and classification of diabetes: standards of care in diabetes. Diabetes Care 2026, 49 (Supplement_1), S27–S49. [CrossRef]
- European evidence-based guidelines on pancreatic cystic neoplasms. European Study Group on Cystic Tumours of the Pancreas Gut 2018, 67(5), 789–804.
- Tomioka, M; Murakami, A; Saito, H; et al. Anatomical and surgical evaluation of gastroepiploic artery. Okajimas Folia Anat Jpn 2016, 92(3–4), 49–52. [Google Scholar] [CrossRef] [PubMed]
- Pasinato, G; Laferté, Q; Pannet, M; et al. Predominant role of the left gastroomental artery in distal pancreatectomy with preservation of the spleen and splenic vessels resection (Warshaw technique): an anatomoradioclinical study. Surg Radiol Anat 2025, 47(1), 209. [Google Scholar] [CrossRef] [PubMed]
- Napoli, N; Ripolli, A; Kauffmann, EF; et al. Survival benefit of surgery versus oncology-only therapy in artery-involving borderline resectable and locally advanced pancreatic cancer. Ann Surg Oncol 2025, 32(13), 9995–10006. [Google Scholar] [CrossRef]
- Bachellier, P; Addeo, P; Paul, C; et al. Prognostic Impact of type of arterial resection on outcomes of pancreatectomy with arterial resection for pancreatic adenocarcinoma. Ann Surg 2025. [Google Scholar] [CrossRef]
- Truty, MJ; Colglazier, JJ; Mendes, BC; et al. En bloc celiac axis resection for pancreatic cancer: Classification of anatomical variants based on tumor extent. J Am Coll Surg 2020, 231, 8–29. [Google Scholar] [CrossRef]
- Truty, M; Kendrick, M; Nagorney, D; et al. Factors predicting response, perioperative outcomes, and survival following total neoadjuvant therapy for borderline/locally advanced pancreatic cancer. Ann Surg 2021, 273, 341–349. [Google Scholar] [CrossRef]
- Napoli, N; Kauffmann, EF; Lombardo, C; et al. Postoperative results, learning curve, and outcomes of pancreatectomy with arterial resection: a single-center retrospective cohort study on 236 procedures. Int J Surg 2024, 110(10), 6111–6125. [Google Scholar] [CrossRef]
- Loos, M; Kester, T; Klaiber, U; et al. Arterial Resection in Pancreatic Cancer Surgery: Effective After a Learning Curve. Ann Surg 2022, 275(4), 759–768. [Google Scholar] [CrossRef] [PubMed]
- Kwon, J; Shin, SH; Yoo, D; et al. Arterial resection during pancreatectomy for pancreatic ductal adenocarcinoma with arterial invasion: A single-center experience with 109 patients. Medicine (Baltimore) 2020, 99(37), e22115. [Google Scholar] [CrossRef] [PubMed]
- Kelvin, L; Khang, D; Ricky, L; et al. Systematic review and meta-analysis of current and novel approaches in the management of borderline resectable and locally advanced Pancreatic Cancer. Cancers 2026, 18(7), 1139. [Google Scholar] [CrossRef]
- Sutherland, DER; Gruessner, RWG; Dunn, DL; et al. Lessons learned from more than 1,000 pancreas transplants at a single institution. Ann Surg 2001, 233, 463–501. [Google Scholar] [CrossRef]
- Yang, F; Jin, C; Warshaw, AL; Mizuno, S; Isaji, S; Ohsawa, I; et al.; Total pancreatectomy for pancreatic malignancy with preservation of the spleen Pancreaticoduodenectomy with resection of the splenic artery and splenectomy for pancreatic double cancers after total gastrectomy. Preservation of the pancreatic function via the blood supply from the posterior epiploic artery: report of a case. J Surg Oncol;Surg Today 2019, 119(6) 42(5), 784-793 482-8. [Google Scholar]
- Desaki, R; Mizuno, S; Tanemura, A; et al. A new surgical technique of pancreaticoduodenectomy with splenic artery resection for ductal adenocarcinoma of the pancreatic head and/or body invading splenic artery: impact of the balance between surgical radicality and QOL to avoid total pancreatectomy. Biomed Res Int 2014, 2014, 219038. [Google Scholar] [CrossRef]
- Kuo, TC; Wu, CH; Chen, BB; et al. Most total pancreatectomies for ductal adenocarcinoma potentially can be replaced by Whipple over the splenic artery: a before and after study. Int J Surg 2024, 110(10), 6461–6469. [Google Scholar] [CrossRef]
- Kimura, Y; Imamura, M; Itoh, T; et al. Conversion pancreaticoduodenectomy with dual arterial reconstructions for locally advanced pancreatic cancer: Case report and literature review. Int J Surg Case Rep 2021, 80, 105692. [Google Scholar] [CrossRef]
- Kondo, S; Ambo, Y; Katoh, H; et al. Middle colic artery-gastroepiploic artery bypass for compromised collateral flow in distal pancreatectomy with celiac artery resection. Hepatogastroenterology 2003, 50(50), 305–7. [Google Scholar]
- Seelig, MH; Belyaev, O; Uhl, WJ. Reconstruction of the common hepatic artery at the time of total pancreatectomy using a splenohepatic bypass. Gastrointest Surg 2010, 14(5), 913–5. [Google Scholar] [CrossRef]
- Hideharu, T; Hisashi, I; Toshiya, H; et al. Splenic artery transposition for hepatic arterial reconstruction in conversion surgery of an initially unresectable, locally advanced pancreatic cancer after gemcitabine/nab-paclitaxel: A case report. International Journal of Surgery Case Reports 2021, 78, 192–196. [Google Scholar]
- Kießler, M; Jäger, C; Mota Reyes, C; et al. A retrospective, multicentric, nationwide analysis of the impact of splenectomy on survival of pancreatic cancer patients. Langenbecks Arch Surg 2024, 410(1), 14. [Google Scholar] [CrossRef] [PubMed]
- Seykora, TF; Ecker, BL; McMillan, MT; et al. The beneficial effects of minimizing blood loss in pancreatoduodenectomy. Ann Surg 2019, 270(1), 147–157. [Google Scholar] [CrossRef] [PubMed]
- Loos, M; Al-Saeedi, M; Hinz, U; et al. Categorization of differing types of total pancreatectomy. JAMA Surg 2022, 157(2), 120–128. [Google Scholar] [CrossRef] [PubMed]
- Rebelo, A; Andersson, B; Bandyopadhyay, SK; et al. Oncologic multivisceral resections involving the pancreas. Int J Surg 2025. [Google Scholar] [CrossRef]
- Kulemann, B; Hoeppner, J; Wittel, U; et al. Perioperative and long-term outcome after standard pancreaticoduodenectomy, additional portal vein and multivisceral resection for pancreatic head cancer. J Gastrointest Surg 2015, 19(3), 438–44. [Google Scholar] [CrossRef]
- Weber, R; Kuehni, FLM; Aeschbacher, P; et al. Initiation and completion of adjuvant chemotherapy after total versus partial pancreaticoduodenectomy for pancreatic cancer. Ann Surg Oncol 2025, 32(13), 10017–10024. [Google Scholar] [CrossRef]
- Stoop, TF; Javed, AA; Oba, A; et al. Pancreatic Cancer. Lancet 2025, 405(10485), 1182–1202. [Google Scholar] [CrossRef]
- Teoule, P; Tombers, K; Rahbari, M; et al. Definition and treatment of superior mesenteric artery revascularization and dissection- associated diarrhea (SMARD syndrome) in Germany]. Chirurg 2022, 93(2), 173–181. [Google Scholar] [CrossRef]
- Loos, M; Mehrabi, A; Ramouz, A; et al. Gastric venous congestion after total pancreatectomy is frequent and dangerous. Ann Surg 2022, 276(6), e896–e904. [Google Scholar] [CrossRef]
- Napoli, N; Kauffmann, E; Cacace, C. Factors predicting survival in patients with locally advanced pancreatic cancer undergoing pancreatectomy with arterial resection. Updates Surg. 2021, 73(1), 233–249. [Google Scholar] [CrossRef]
- Beger, HG; Poch, B; Mayer, B; et al. New Onset of Diabetes and Pancreatic Exocrine Insufficiency After Pancreaticoduodenectomy for Benign and Malignant Tumors: A Systematic Review and Meta-Analysis of Long-Term Results. Ann Surg 2018, 267(2), 259–270. [Google Scholar] [CrossRef] [PubMed]
- Scholten, L; Mungroop, TH; Haijtink, SAL; et al. New-onset diabetes after Pancreatoduodenectomy: A systematic review and meta-analysis. S0039-6060(18)30081-3; Surgery. 2018. [Google Scholar]
- Wu, L; Nahm, CB; Jamieson, NB; et al. Risk factors for development of diabetes mellitus (Type 3c) after partial pancreatectomy: a systematic review. Clin Endocrinol (Oxf) 2020, 92(5), 396–406. [Google Scholar] [CrossRef] [PubMed]
- Gaujoux, S; de Ponthaud, C; Amouyal, C. How new diabetes technology will improve outcomes after total pancreatectomy? Gland Surg 2026, 15(1), 1. [Google Scholar] [CrossRef] [PubMed]
- van Veldhuisen, CL; Latenstein, AEJ; Blauw, H; et al. Bihormonal artificial pancreas with closed-loop glucose control vs current diabetes care after total pancreatectomy: a randomized clinical trial. JAMA Surg 2022, 157(10), 950–957. [Google Scholar] [CrossRef]
- Scholten, L; Stoop, TFR; Chiaro, MD; et al. Systematic review of functional outcome and quality of life after total pancreatectomy. BJS 2019, 106, 1735–46. [Google Scholar] [CrossRef]
- Suto, H; Kamei, K; Kato, H; et al. Diabetic control and nutritional status up to 1 year after total pancreatectomy: a nationwide multicentre prospective study. BJS 2021, 108, e237–8. [Google Scholar] [CrossRef]
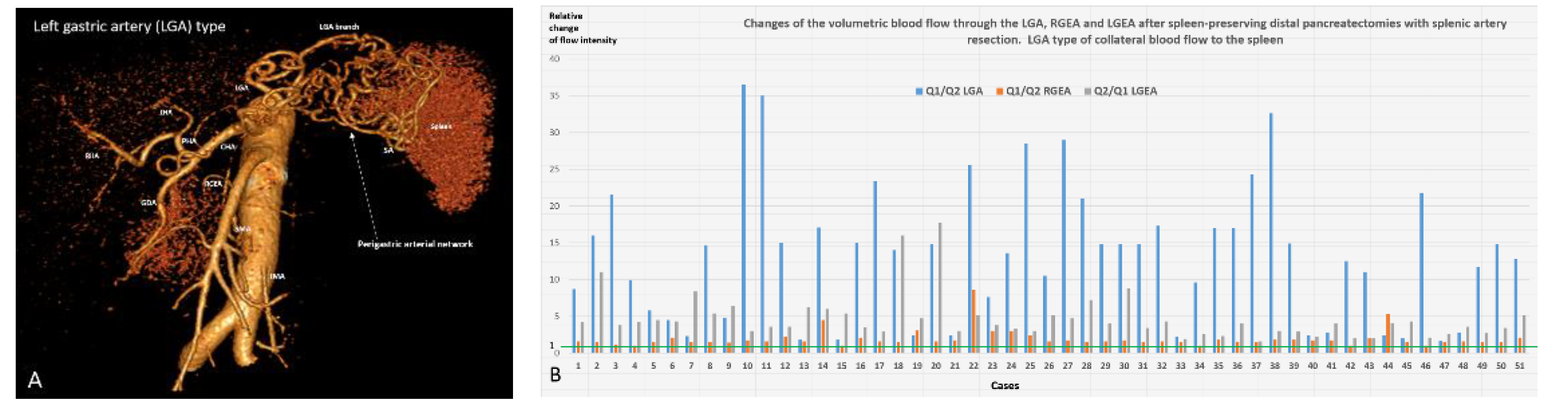
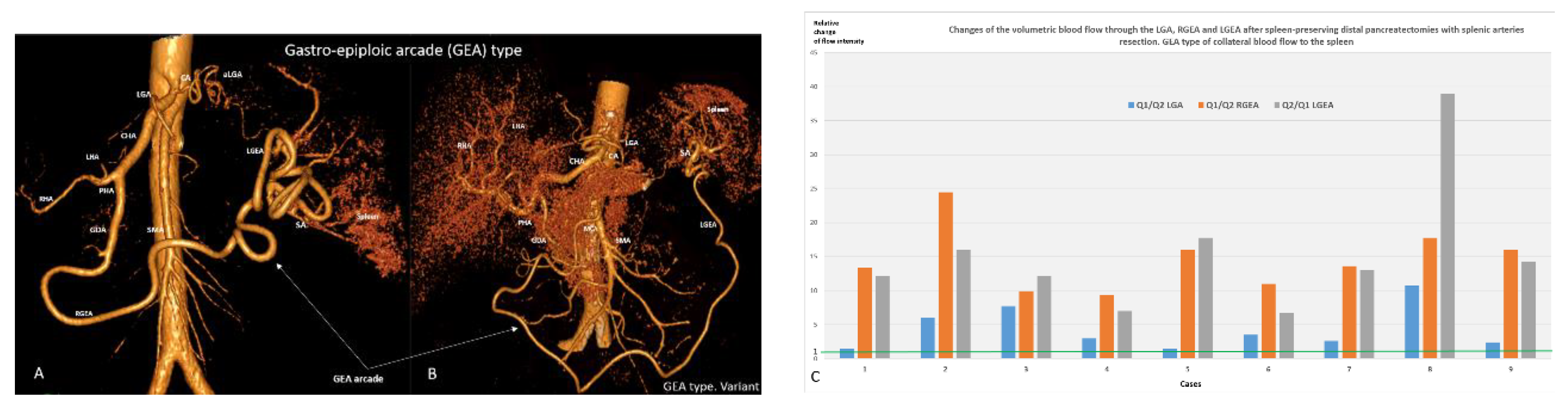
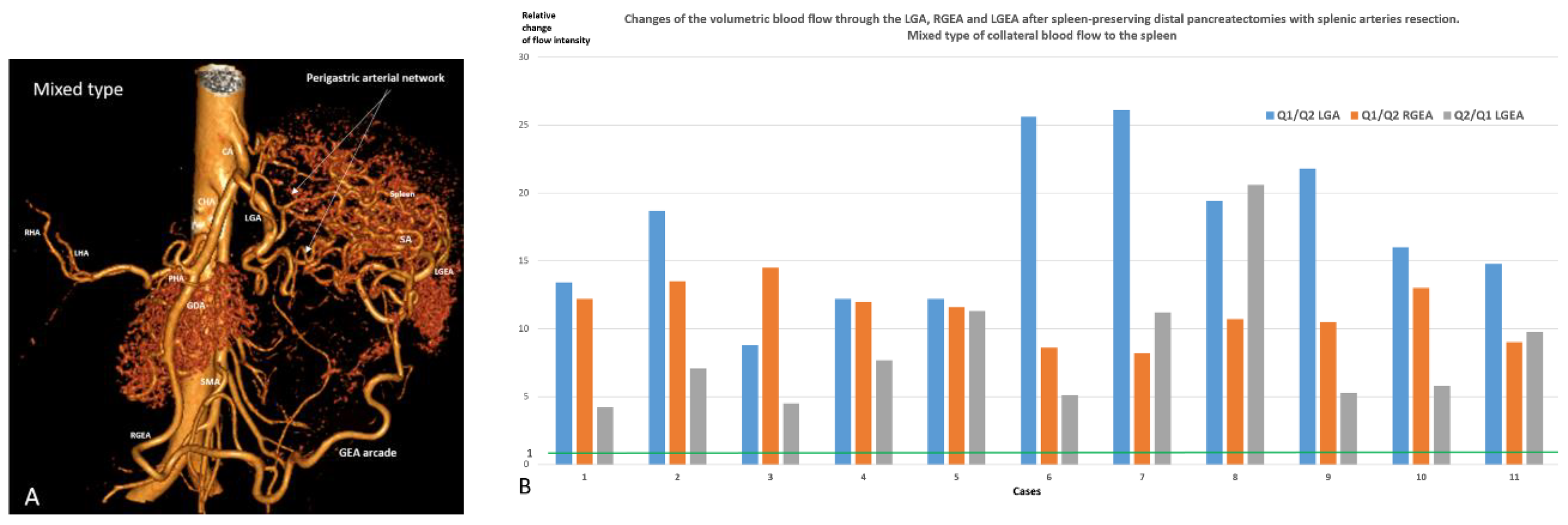
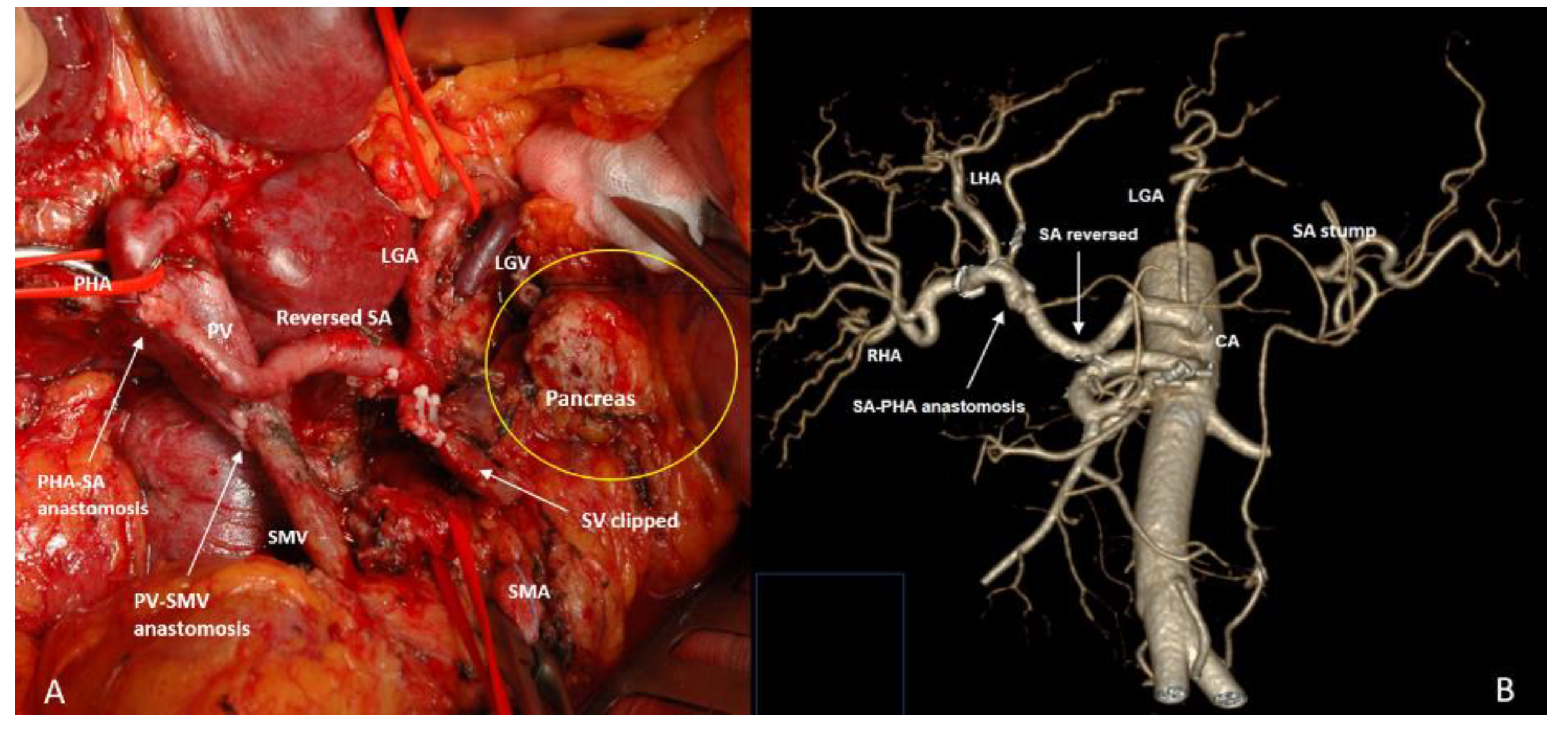
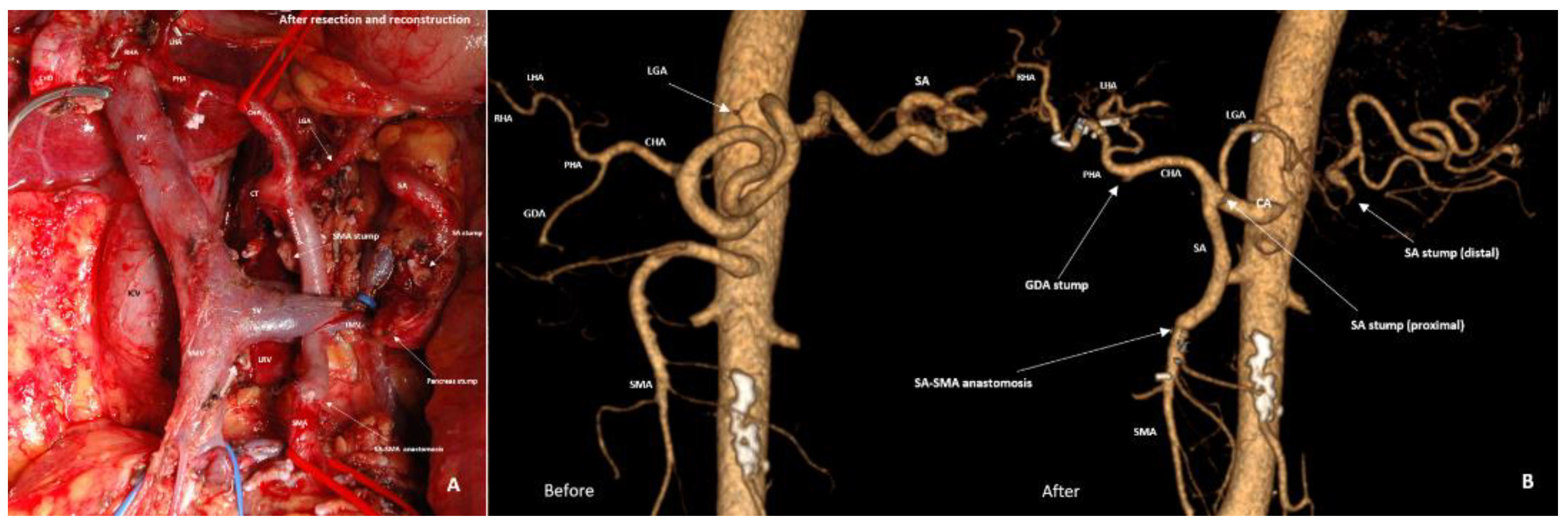
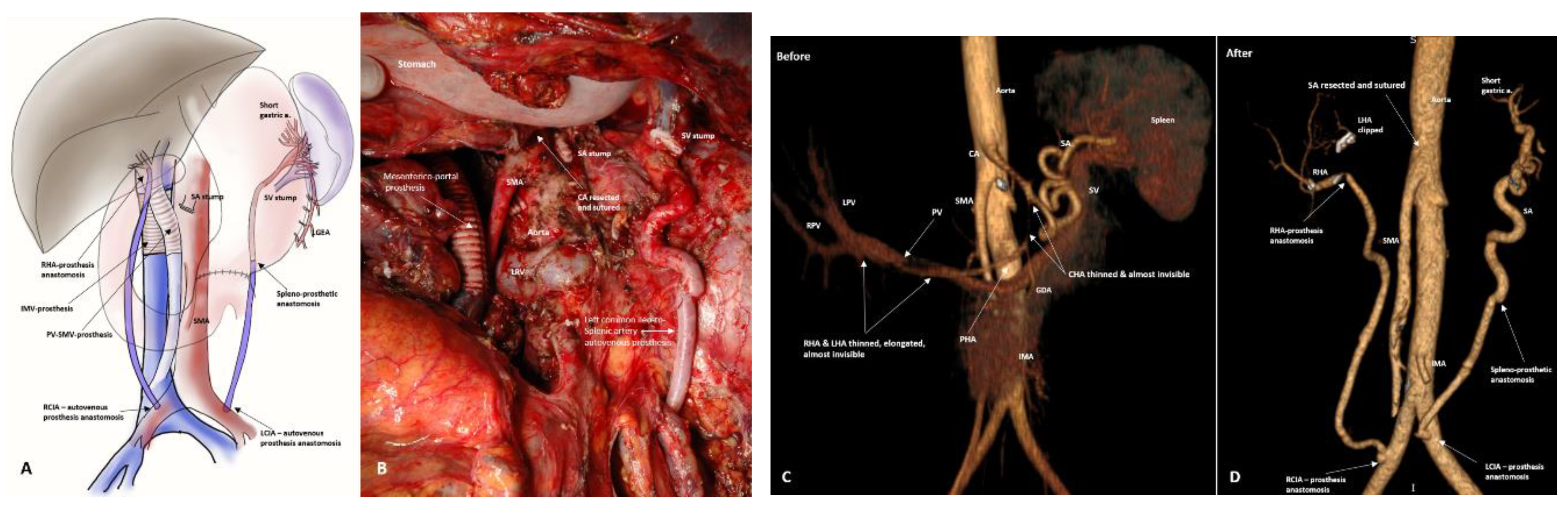
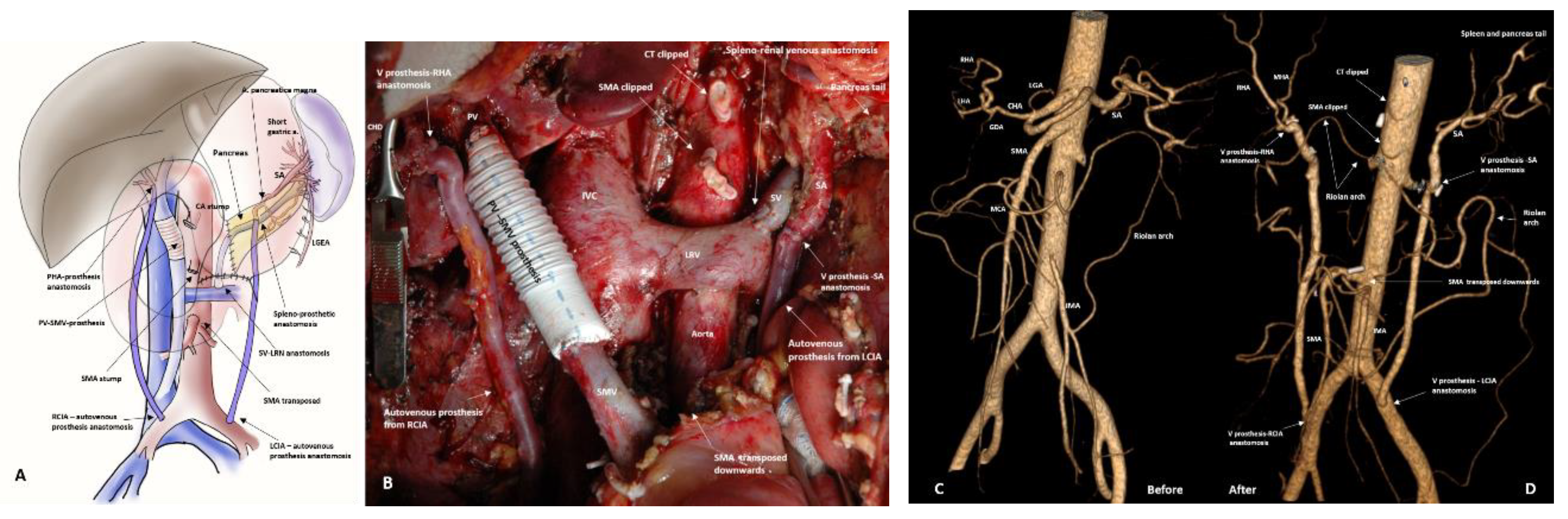
| Indications for SP pancreatectomies/Diagnosis | Number | |
|---|---|---|
| 1 | Mucinous cystic neoplasm (MCN) | 41 |
| 2 | Branch duct intraepithelial mucinous neoplasia (bdIPMN) | 9 |
| 3 | Main duct intraepithelial mucinous neoplasia (mdIPMN)* | 10 |
| 4 | Cerous cystic adenoma (CSA) | 3 |
| 5 | Well-differentiated neuroendocrine neoplasms (NEN) | 23 |
| 6 | Solid-pseudopapillary neoplasm (SPPN) | 8 |
| 7 | Chronic calculous pancreatitis (CP) | 1 |
| 8 | Autoimmune pancreatitis (local form) (AIP) | 1 |
| 9 | Malignant peripheral nerve sheath tumor (MPNST) | 1 |
| 10 | BR and LA pancreatic head ductal adenocarcinoma (PHDAC) | 40 |
| Total | 137 | |
| Types of surgery | Number | |
| Spleen-preserving pancreatectomies with splenic vessels preservation (SPPE SVP) | 19 | |
| 1. Distal pancreatectomy (SPDP SVP) | 12 | |
| 2. Total pancreatectomy (TP SVP) | 6 | |
| 3. Center-preserving pancreatectomy with splenic vessels preservation | 1 | |
| Spleen-preserving pancreatectomies with splenic vessels resection (SPPE SPR) | 115 | |
| 1. Distal pancreatectomy (SPDP SVR) | 71 | |
| 2. Central pancreatectomy with splenic vessels resection (CP SVR) | 3 | |
| 3. Total pancreatectomy with splenic vessels resection (TP SVR) | 2 | |
| 4. Total pancreatectomy with SMA resection and splenic artery rotation (TP SAR) | 6 | |
| 5. Total pancreatectomy with HA/CA/LGA resection and splenic artery revascularization. | 1 | |
| 6. Pancreaticoduodenectomy with splenic vessels resection (PD SVR) | 3 | |
| 7. Pancreaticoduodenectomy with SMA or CHA resection and splenic artery rotation (PD SAR) | 28 | |
| 8. Pancreaticoduodenectomy with SMA and CA/HA/LGA resection and splenic artery revascularization. | 1 | |
| Total | 134 | |
| Age (mean ± SD) | 44.23± 13,9 |
|---|---|
| Female gender, n (%) | 61 (76.25%) |
| BMI (mean ± SD) | 22.58±3,2 |
| Tumor size (mean ± SD) (mm) | 61.88±40,0 |
| OP time (min) (mean ± SD) | 208.54±143,6 |
| E blood Loss (ml) (mean ± SD) | 108.05±146 |
| Mean hospital stay (days) | 5.2 ± 0,75 |
| Median F-up time (months) | 88 [58,116] |
| Type of surgery | # | POPF Grade B | D-C ≥ 3 | Spleen infarction (n,%, cr) | Any gastric/ epigastric varices | Late morbidity* | Postop DM |
|---|---|---|---|---|---|---|---|
| SPDPSVR | 71 | 14 (20%) of 33 |
0 | 18 (25%), 0 cr | 11(17.5%) of 63 | 1 | 9 (13%) |
| SPDPSVP | 12 | 2 (22%) of 7 |
1 | 0 | 1 (8%) | 2 | 1 (8%) |
| Total | 83 | 16 (19%) | 1 | 18 (22%), 0 cr | 12 (14.5%) | 3 | 10 (12%) |
| Age (years) | 57,8 ± 10.1 (33-73) |
| Gender (m/f) | 18/23 (44%/56%) |
| PDAC/MPNST | 40/1 |
| Neoadjuvant chemotherapy (NACHT) (yes/no) | 41/0 (100%/0) |
| Mean number of NACHT courses | 11.6 ± 2.95 |
| Adjuvant chemotherapy (ACHT) (yes/no) | 38/3 (93%/7%) |
| Completed ACHT, n38 | 31 (82%) |
| Incompleted ACHT. In process/incompleted, n7 | 5/2 (13,5% / 2,5%) |
| Mean number of ACHT courses | 4.43 ± 2,56 |
| CCI – 2/3/4/5/ (score) | 5/19/14/3 (12%/46%/34%/8%) |
| OP time (min.) | 560 ± 146 (195- 1260) |
| Estimated blood loss (ml) | 358 ± 211 (100- 1200) |
| Operative packed RBC transfusion, n (cases) | 9 (22%) |
| No. of packed RBC units transfused, ≥ 2 />2, n | 6/3 |
| PV/SMV resection (yes/no) | 38/3(93%/7%) |
| CA 19-9 before NACHT, U/ml, n40 | 308,7±352 |
| CA 19.9 before surgery, n40 | 49,5±69,6 |
| CA 19-9 decreasing ratio after/before NACHT, n40 | 6,3 |
| Tumor size at pathology (mm), n40 | 34,6±13,3 (0-81) |
| Tumor grade, G1,G2,G3, n40 | 3, 33, 4 (7,5%,82,5, 10%) |
| Contact on CT > 180° with SA, HA/CA, SMA, CA&SMA | 5, 15, 19, 2 (12%, 36,5%, 46,5%, 5%) |
| Arteries resected in addition to SA: HA/CA, SMA, CA&SMA | 15 / 19 / 2 |
| LA tumors (T4)/ BR tumors(T3) | 36/5 |
| Arterial invasion at pathology (yes/no), n40 | 27/14 (66%/34%) |
| PV-SMV invasion at pathology yes/no), n40 | 34/4 (90,5%/9,5%) |
| Perineural invasion (yes/no) (n40) | 34/6 (85%/15%) |
| R0/R1-resection | 38/3 (92,7/7,3%) |
| Number of lymph nodes removed (n40) | 37,6±13 (21-75) |
| Lymph nodes involvement, pN0/pN1/pN2 (n40) | 7/20/13 (17,5%/50%/32,5%) |
| Tumor regression score, 0/1/2/3, n40 | 2,2,14,22 (5%, 5%, 34%, 56%) |
| Pancreatectomy type, n (%): PD/total pancreatectomy | 32/9 (78%/22%) |
| Mean time for anastomosis with the SA (min) | 21,9±1,5 min |
| Recurrence type, n (% total, % all recurrence), n40 | |
| Local | 3 (7.5, 11,5) |
| Peritoneal | 5 (12,5, 19) |
| Distant (liver, lungs, lymph nodes, bone) | 18 (45, 69) |
| Multisite | 8 (20, 30.8) |
| Vital status at last follow-up, n (%) | |
| Alive, with disease | 8 (20) |
| Alive, no evidence of disease | 14 (35) |
| Died | 19 (47.5) * |
| Complications (С-D), <3 / III,IV | 32 / 7,2 (88%/17%, 5%) |
| POPF, Grade B, n32 | 1 (3%) |
| Diarrhea (n) | 17 (42%) |
| Length of stay (days) | 16,4±4,7 (9-29) |
| Lymphorrhea (n) | 12 (29%) |
| cr DGE, Grade 2/3 | 8 / 2 (20%,5%) |
| Postpancreatectomy hemorrhage (n) | 1 (2,4%) |
| Any cr ischemic abdominal complications | 0 |
| Reoperation (n) | 2 (7,5%) |
| Readmission (n) | 7 (17%) |
| Postoperative diabetes mellitus, n31 | 7 (22,6%) |
| Mortality, 90-days | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




