Submitted:
09 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
- Relationship Between Macrophyte Richness and the Evaluated Mesohabitats. The richness of macrophytes was evaluated across the different mesohabitats within the study area. Six mesohabitats were classified based on their vegetation composition and the ecosystems present: flooded lowlands, swamp, wetland, ponds, naturalized ponds, and the riparian zone of streams.
- Relationship Between Macrophyte Richness and the Vulnerability Status of Habitats to Climate Change. The climate change vulnerability model [16] was used to assess the impact of anthropogenic activities and climatic variations on aquatic ecosystems. Vulnerability was associated with vegetation composition, type of productive activity, and environmental conditions of each mesohabitat.
- Principal Component Analysis (PCA). A Principal Component Analysis (PCA) was performed to evaluate the relationship between macrophyte composition and the productive activities present in the region. This multivariate analysis allowed the identification of patterns in the presence and abundance of macrophyte species and their association with different land uses and productive activities, such as livestock farming, agriculture, and aquaculture. Additionally, the PCA facilitated the identification of ecological gradients related to anthropogenic pressures affecting aquatic ecosystems in the study area.
- Statistical Analysis of Richness Patterns. To quantitatively assess differences in macrophyte richness among mesohabitats and its relationship with vulnerability levels, inferential statistical analyses were conducted. A Generalized Linear Model (GLM) with a Poisson distribution was applied to evaluate the effect of mesohabitat type and vulnerability on species richness. Additionally, a non-parametric Kruskal–Wallis test was performed to assess differences in richness among mesohabitats due to the non-normal distribution of the data. These analyses provided a robust statistical framework to support the ecological patterns observed in the study.
3. Results
3.1. Characterization of Monitoring Sites
3.2. Species Composition
| Family | Genus | Common name | Scientific name |
|---|---|---|---|
| Poaceae | Brachiaria | Canutillo | Brachiaria plantaginea (Link) Hitchc. |
| Salviniaceae | Salvinia | Lenteja De Agua | Salvinia minima Baker |
| Cyperaceae | Cyperus | Coquito | Cyperus odoratus L. |
| Pontederiaceae | Eichhornia | Tarulla | Eichhornia crassipes Solms |
| Onagraceae | Ludwigia | Palito De Agua | Ludwigia decurrens Walter |
| Marantaceae | Thalia | Bocachica | Thalia geniculata L. |
| Cyperaceae | Cyperus | Coquito | Cyperus diffusus Vahl |
| Marantaceae | Calathea | Bijao | Calathea lutea |
| Malvaceae | Sida | Escobillo | Sida acuta Burm.f. |
| Blechnaceae | Telmatoblechnum | Helecho | Telmatoblechnum serrulatum (L.C.Rich.) |
| Solanaceae | Solanum | Lulo De Perro | Solanum pseudolulo Heiser |
| Heliconiaceae | Heliconia | Heliconia | Heliconia psittacorum L.f. |
| Alismataceae | Limnocharis | Buchon | Limnocharis flava Buchenau |
| Araceae | Colocasia | Oreja De Elefante | Colocasia esculenta (L.) Schott |
| Cyperaceae | Rhynchospora | Ciperacea | Rhynchospora corymbosa (L.) Britton |
| Pontederiaceae | Heteranthera | Oreja De Ratón | Heteranthera reniformis Ruiz & Pav. |
| Nymphaeaceae | Nymphaea | Ninfa De Agua | Nymphaea alba L. |
| Commelinaceae | Commelina | Hierba De Pollo | Commelina diffusa Burm.f. |
| Cyperaceae | Cyperus | Zacate De Estrella | Cyperus luzulae (L.) Retz. |
| Cyperaceae | Eleocharis | Junco | Eleocharis palustris (L.) Roem. & Schult |
| Cyperaceae | Fimbristylis | Ciperacea | Fimbristylis quinquangularis (M.Vahl) Kunth |
| Osmundaceae | Osmundastrum | Helecho Marrano | Osmundastrum cinnamomeum (L.) C.Presl |
| Lentibuliriaceae | Utricularia | Perrito De Agua | Utricularia gibba L. |
| Plantaginaceae | Bacopa | Abánico De Agua | Bacopa egensis (Poepp.) Pennell |
| Alismataceae | Sagittaria | Lirio De Agua | Sagittaria guayanensis Kunth |
| Poaceae | Pennisetum Purpureum | Pasto Elefante | Pennisetum purpureum Schumach. |
| Rubiaceae | Isertia | Tabaquillo | Isertia haenkeana DC. |
| Lycopodiaceae | Lycopodium | Colchon De Pobre | Lycopodium clavatum L. |
| Marantaceae | Ischnosiphon | Ischnosiphon arouma Körn. | |
| Heliconiaceae | Heliconia | Heliconia | Heliconia brachyantha L.Andersson |
| Araceae | Anthurium | Anturio | Anthurium Schott |
| Fabaceae | Pueraria | Pueraria phaseoloides Benth. | |
| Heliconiaceae | Heliconia | Heliconia | Heliconia latispatha Benth. |
| Araceae | Dieffenbachia | Galatea | Dieffenbachia Schott |
| Poaceae | Rottboellia | Rottboellia exaltata (L.) L.f. | |
| Onagraceae | Ludwigia | Palito De Agua | Ludwigia erecta (L.) H.Hara |
| Urticaceae | Cecropia | Guarumo | Cecropia peltata L. |
| Euphorbiaceae | Croton | Croton hirtus L’Hér. | |
| Dennstaedtiaceae | Pteridium | Helecho Marrano | Pteridium aquilinum (L.) Kuhn |
| Cucurbitaceae | Momordica | Balsamina | Momordica charantia L. |
| Cyperaceae | Scleria | Scleria melaleuca Rchb. ex Schltdl. & Cham. | |
| Menyanthaceae | Nymphoides | Lirio De Agua | Nymphoides indica (L.) Griseb. |
| Marsileaceae | Marsilea | Helecho Trebol | Marsilea polycarpa Hook. & Grev. |
| Fabaceae | Chamaecrista | Guajito | Chamaecrista nictitans Moench |
| Convolvulaceae | Ipomoea | Campanilla | Ipomoea cairica (L.) Sweet |
| Vitaceae | Cissus | Cissus erosa Rich. | |
| Asteraceae | Emilia | Pincel | Emilia sonchifolia (L.) DC. |
| Fabaceae | Vigna | Vigna longifolia (Benth.) Verdc. | |
| Onagraceae | Ludwigia | Rosita De Agua | Ludwigia helminthorrhiza (Mart.) H.Hara |
| Cyperaceae | Eleocharis | Junco | Eleocharis elegans Roem. & Schult. |
| Cyperaceae | Cyperus | Junco | Cyperus blepharoleptos Steud. |
| Pteridaceae | Ceratopteris | Helecho De Agua | Ceratopteris pteridoides (Hook.) Hieron. |
| Lentibuliriaceae | Utricularia | Perrito De Agua | Utricularia foliosa L. |
| Poaceae | Panicum | Pasto Espiga | Panicum maximum Jacq. |
| Cyperaceae | Fuirena | Junco | Fuirena umbellata Rottb. |
| Cyperaceae | Eleocharis | Junco | Eleocharis geniculata (L.) Roem. & Schult. |
| Malvaceae | Hibiscus | Hibisco | Hibiscus furcellatus Desr. |
| Malvaceae | Hibiscus | Hibisco | Hibiscus sororius L. |
| Poaceae | Andropogon | Cola De Zorro | Andropogon bicornis L. |
| Nymphaeaceae | Nymphaea | Ninfa De Agua | Nymphaea ampla DC. |
| Fabaceae | Senna | Senna obtusifolia (L.) H.S.Irwin & Barneby | |
| Cyperaceae | Schoenoplectus | Totora | Schoenoplectus californicus (C.A.Mey.)Soják |
| Poaceae | Luziola | Pasto Acuático | Luziola subintegra Swallen |
3.3. Macrophyte Species Richness Across Different Mesohabitats
3.3.1. Floodable Lowlands
3.3.2. Swamps
3.3.3. Naturalized Ponds
3.3.4. Stream Riparian Zones
3.4. Frequency of Occurrence of Macrophytes
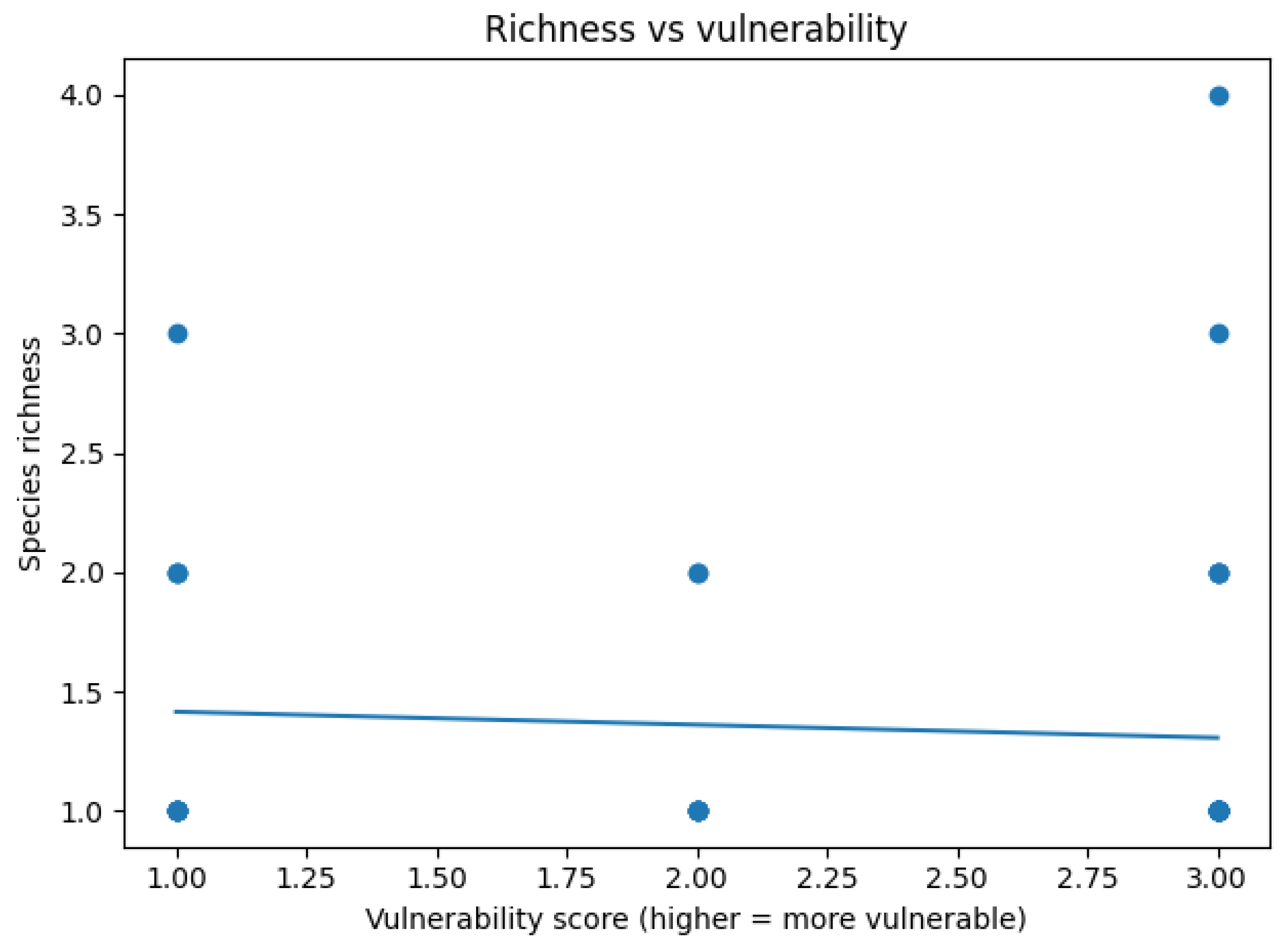
3.5. Relationship Between Species Richness and the Climate Change Vulnerability Index
3.5.1. Species Indicative of Anthropogenic Disturbance
3.5.2. Species Tolerant to Eutrophication and Organic Enrichment
3.5.3. Species Indicative of Ecosystems with Greater Ecological Stability

3.6. Principal Component Analysis (PCA)
3.7. Influence of Productive Activities on Macrophyte Richness
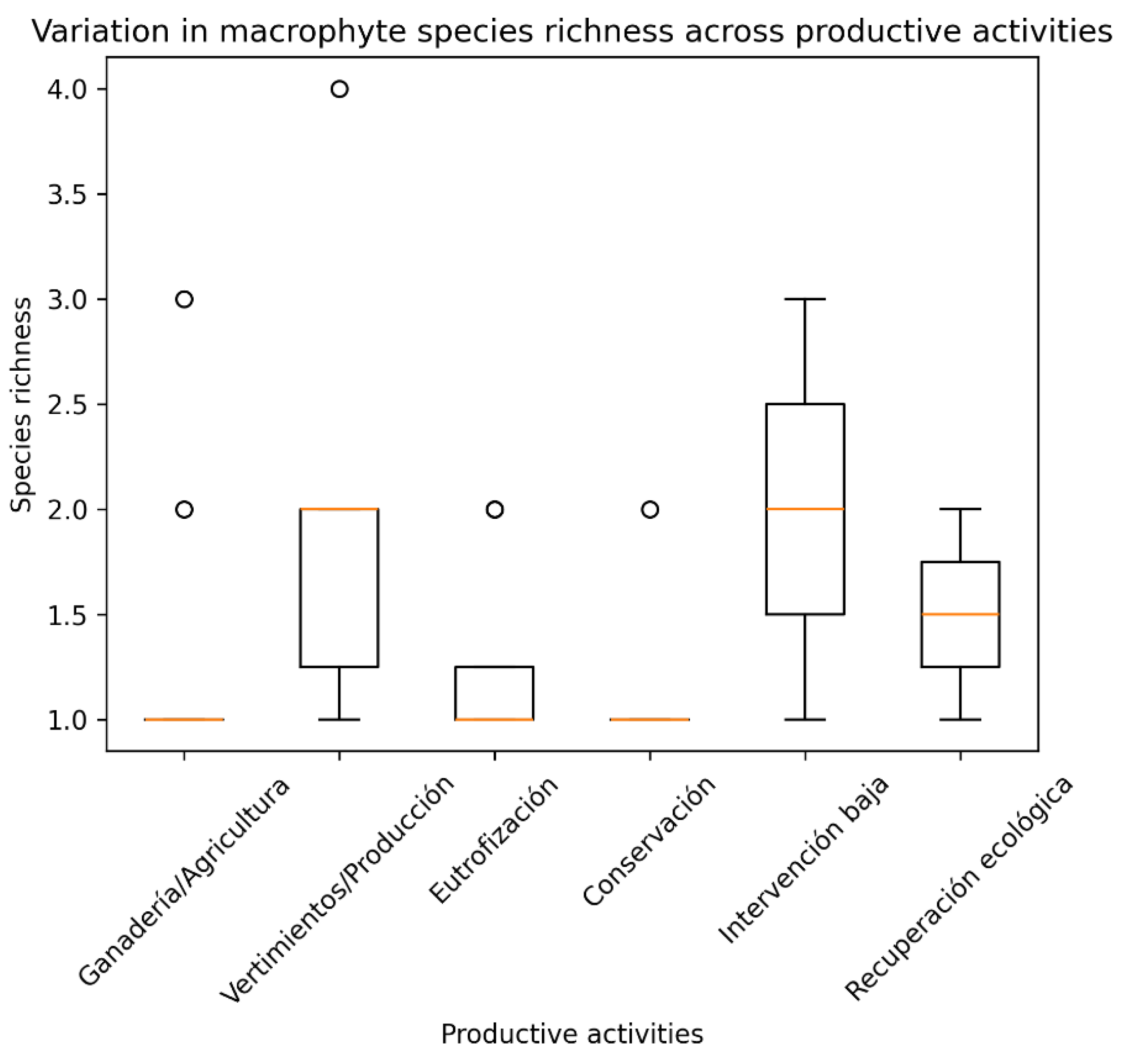
3.8. Macrophyte Richness Across Mesohabitats and Vulnerability Levels
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Abbreviations
| MDPI | Multidisciplinary Digital Publishing Institute |
| DOAJ | Directory of open access journals |
| TLA | Three letter acronym |
| LD | Linear dichroism |
| MINCIENCIAS | Ministry of Science, Technology and Innovation of Colombia |
| UNIPAZ | University Institute of Peace |
| BIOCON | Research Group on Conservation and Use of Biodiversity |
References
- Zedler, J. B.; Kercher, S. Wetland resources: Status, trends, ecosystem services, and restorability. Annual Review of Environmental Resources 2005, 30, 39–74. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2014: Impacts, Adaptation, and Vulnerability; Cambridge University Press, 2014. [Google Scholar]
- Acreman, M.; Arthington, A. H.; Colloff, M. J.; Couch, C.; Crossman, N. D.; Dyer, F.; Overton, I.; Pollino, C. A.; Stewardson, M. J.; Young, W. Environmental flows for natural, hybrid, and novel riverine ecosystems in a changing world. Frontiers in Ecology and the Environment 2014, 12, 466–473. [Google Scholar] [CrossRef]
- Duarte, C. M.; Holmer, M.; Olsen, Y.; Soto, D. Will the oceans help to fight global warming? Frontiers in Ecology and the Environment 2008, 6, 504–508. [Google Scholar]
- Alahuhta, J. M. L. H. P. Wetland plants as indicators of ecological status: Implications of climate change. Hydrobiologia 2017, 804, 57–68. [Google Scholar]
- Weigel, B.; L. P. A. M. & J. P. The role of aquatic plants in the vulnerability of wetland ecosystems to climate change. Ecological Indicators 2020, 110, 105933. [Google Scholar]
- Mitsch, W. J.; Gosselink, J. G. Wetlands, 5th ed.; Wiley, 2015. [Google Scholar]
- Brix, H. Functions of macrophytes in constructed wetlands. Water Science and Technology 1994, 29, 1–16. [Google Scholar] [CrossRef]
- Richardson, D. M.; Pyšek, P. Plant invasions: Merging the concepts of species invasiveness and community invasibility. Progress in Physical Geography 2006, 30, 409–431. [Google Scholar] [CrossRef]
- Gamboa, M.; Reyes, R.; Arrivillaga, J. Macroinvertebrados bentónicos como bioindicadores de salud ambiental. Boletín de malariología y salud ambiental 2008, 48, 109–120. [Google Scholar]
- Cairns, J.; McCormick, P. V.; Niederlehner, B. R. A proposed framework for developing indicators of ecosystem health. Hydrobiologia 1993, 263, 1–44. [Google Scholar] [CrossRef]
- Bonada, N.; Prat, N.; Resh, V. H.; Statzner, B. Developments in aquatic insect biomonitoring: A comparative analysis of recent approaches. Annual Review of Entomology 2006, 51, 495–523. [Google Scholar] [CrossRef]
- Ministerio De Ambiente Y Desarrollo Sostenible. Guía metodológica para la formulación de los planes de manejo ambiental de microcuencas—PMAM. 2018. Available online: http://www.andi.com.co/Uploads/GUÍA%20PMA%20de%20Microcuenca.pdf.
- Lasso, C. Descripción Metodológica Para La Evaluación Biológica En Los Complejos De Humedales. Instituto de Investigación de Recursos Biológicos Alexander von Humboldt. 2015. Available online: http://repository.humboldt.org.co/bitstream/handle/20.500.11761/9591/2211%20Descripci%C3%B3n%20metodologica%20Bd_Humedales_PazAriporo.pdf?sequence=1&isAllowed=y.
- Instituto de Hidrología; Meteorología y Estudios Ambientales (IDEAM). Protocolo para el monitoreo de la vegetación acuática y ribereña en humedales continentales de Colombia; IDEAM, 2021. [Google Scholar]
- NatureServe. Guidelines for using the Climate Change Vulnerability Index (CCVI). NatureServe. 2011. Available online: https://www.natureserve.org/sites/default/files/2024-10/CCVI_Guidelines_4.0_USGS%20approved.pdf.
- Bernal, R.; Gradstein, S.R.; Celis, M. (Eds.) Catálogo de Plantas y Líquenes de Colombia. v1.1. Universidad Nacional de Colombia. Dataset/Checklist. 2020. [Google Scholar] [CrossRef]
- Mora Marín, M. A.; Ríos Pescador, L.; Ríos Ramos, L.; Almario Charry, J. L. Impacto de la actividad ganadera sobre el suelo en Colombia. Ingeniería Y Región 2017, 17, 1–12. [Google Scholar] [CrossRef]
- Carrillo, G. A. C. Evaluación de Salvinia minima Baker como potencial… [Tesis de maestría, Centro de Investigación Científica de Yucatán, A. C.]. Repositorio Institucional CICY. 2017. Available online: https://cicy.repositorioinstitucional.mx/jspui/bitstream/1003/433/1/PCB_RN_M_Tesis_2017_Carrillo_%20Gerardo.pdf.
- Beyra, Ángela; Reyes Artiles, G. Revisión taxonómica de los géneros Phaseolus y Vigna (Leguminosae-Papilionoideae) en Cuba. Anales Del Jardín Botánico De Madrid 2004, 61, 135–154. [Google Scholar] [CrossRef]
- Sanclemente, Maria Angélica; Peña, Enrique Javier. Crecimiento y eficiencia fotosintética de ludwigia decurrens walter (onagraceae) bajo diferentes concentraciones de nitrógeno. Acta Biológica Colombiana 2008, 13, 175–186. Available online: http://www.scielo.org.co/scielo.php?script=sci_arttext&pid=S0120-548X2008000100012&lng=en&tlng=es.
- Barton, D. R.; de Senerpont Domis, L. N.; Hillebrand, H.; Bendor, R. Aquatic plant diversity and its role in ecosystem resilience. Hydrobiologia 2019, 831, 45–58. [Google Scholar]
- Strayer, D. L.; Eviner, V. T.; Jeschke, J. M.; Pace, M. L. Understanding the role of biodiversity in ecosystem function: The relationship between aquatic plant diversity and ecosystem resilience. Ecology Letters 2003, 6, 612–620. [Google Scholar] [CrossRef]
- Jansen, M. A.; Pol, D. D.; Wijgergangs, M. Effects of grazing and agriculture on aquatic macrophyte diversity in temperate wetlands. Environmental Management 2012, 50, 447–455. [Google Scholar]
- Gómez, C.; González, L.; Sánchez, E. Impacto de la eutrofización en ecosistemas acuáticos tropicales. Revista de Biología Tropical 2014, 62, 1–14. [Google Scholar]
- Vilà, M.; Garcı́a-Berthou, E.; Chave, J. Impacts of invasive species on ecosystem functioning. Ecology 2011, 92, 1708–1717. [Google Scholar]
- Mishra, V. K.; Tripathi, B. D. Concurrent removal and accumulation of heavy metals by three aquatic macrophytes. Bioresource Technology 2008, 99, 7091–7097. [Google Scholar] [CrossRef]
- Carvalho, P. N.; Basto, M. C. P.; Almeida, C. M. R.; Brix, H. A review of plant–pharmaceutical interactions: from uptake and effects in plants to phytoremediation in constructed wetlands. Environmental Science and Pollution Research 2013, 21, 11729–11763. [Google Scholar] [CrossRef]
- Gray, J. S. [Rev. of B.A. Markert, A.M. Breure, H.G. Zechmeister, Review of Bioindicators and Biomonitors: Principles, Concepts and Applications, 2003]. Marine Pollution Bulletin 2005, 50, 360–361. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




