Submitted:
09 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Material and Methods
2.1. Histological and Immunohistochemical Examination
2.2. Integrated Morphological Indices
2.3. TMS Scoring System
- 1 point – tubular structures;
- 2 points – cribriform structures;
- 3 points – large solid structures;
- 4 points – small solid structures;
- 5 points – discohesive tumor cells.
- 3 points – pronounced lymphoid infiltration (TILs > 40%);
- 2 points – moderate lymphoid infiltration (TILs 10% to 40%);
- 1 point – absent or weak lymphoid infiltration (TILs < 10%).
- 0 points – absence of lymphovascular invasion;
- 1 point – up to 3 emboli in the lymphatic vessels of the stroma in one field of view;
- 2 points – 3 to 10 emboli in one field of view;
- 3 points – more than 10 emboli in one field of view.
2.4. Statistical Analysis
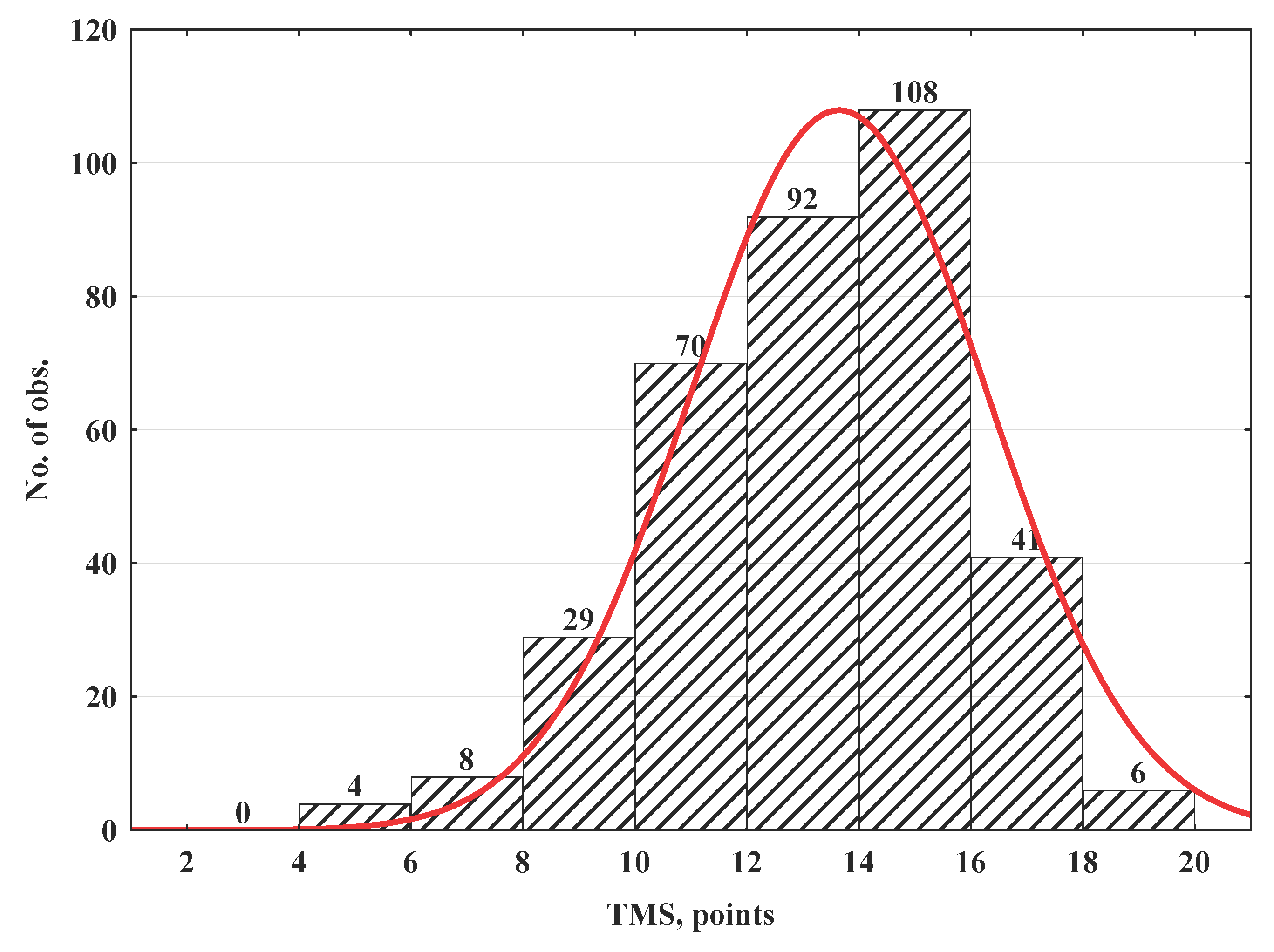
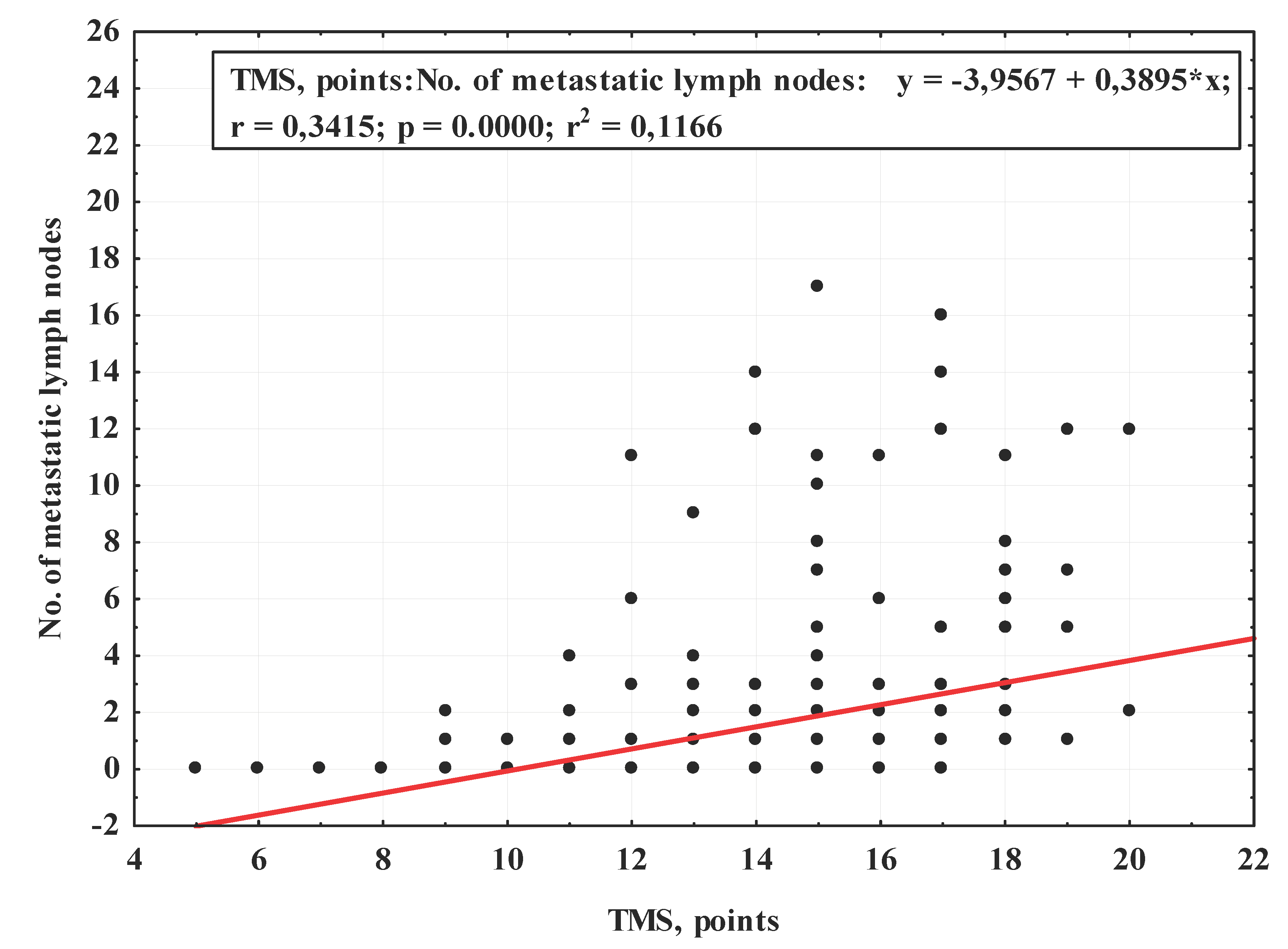
3. Results
4. Discussion
Funding
Authors’ contributions
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74(3):229–263. [CrossRef]
- Malignant tumors in Russia in 2023 (incidence and mortality). Ed. by A.D. Kaprin, V.V. Starinskiy, A.O. Shakhzadova. Moscow: P.A. Gerzen Moscow Oncology Research Center of Radiology, Ministry of Health of Russia, 2024. 276 p. (In Russian).
- Galea MH, Blamey RW, Elston CE, Ellis IO. The Nottingham Prognostic Index in primary breast cancer. Breast Cancer Res Treat. 1992;22(3):207–219.
- Rakha EA, Soria D, Green AR, Lemetre C, Powe DG, Nolan CC, et al. Nottingham Prognostic Index Plus (NPI+): a modern clinical decision making tool in breast cancer. Br J Cancer. 2014;110(7):1688–1697. [CrossRef]
- Gray E, Donten A, Payne K, Hall PS. Survival estimates stratified by the Nottingham Prognostic Index for early breast cancer: a systematic review and meta-analysis of observational studies. Syst Rev. 2018;7(1):142. [CrossRef]
- Cardoso F, Paluch-Shimon S, Senkus E, Curigliano G, Aapro MS, André F, et al. 5th ESO-ESMO international consensus guidelines for advanced breast cancer (ABC 5). Ann Oncol. 2020;31(12):1623–1649. [CrossRef]
- Kometova VV, Zankin VV, Khairullin RM, Rodionov VV. Method for predicting five-year survival of patients with infiltrating breast cancer by determining the total malignancy score. Russian Federation patent No 2013156541/15. 2015 Apr 10. (In Russian).
- Green AR, Soria D, Stephen J, Powe DG, Nolan CC, Kunkler I, et al. Nottingham Prognostic Index Plus: validation of a clinical decision making tool in breast cancer in an independent series. J Pathol Clin Res. 2016;2(1):32–40. [CrossRef]
- Kometova V, Rodionov V, Rodionova M, Dergunova Y, Panchenko S. New prognostic integrated pathological index in breast cancer patients. Virchows Arch. 2017;471(Suppl 1):PS-01-032.
- Podyacheva OA, Rodionov VV, Tupitsyn NN, Petrov SV, Kometova VV, Sharafutdinov MG. Problems of carcinomyelomatosis in breast cancer patients. Ulyanovsk: Ulyanovsk State University; 2016. 106 p. (In Russian).
- Rodionov VV, Torosyan MK, Kometova VV, Idrisova SR, Bogdasarov AU. Personification of drug therapy for patients with breast cancer based on integrated morphological index and immunohistochemical tumor characteristics. Mod Probl Sci Educ. 2014;(6). Available from: https://science-education.ru/ru/article/view?id=17023. (In Russian).
| NPI value | Prognosis | 5-year survival, % |
| 2.0–2.4 | Excellent | 93 |
| 2.4–3.4 | Good | 85 |
| 3.4–5.4 | Moderate | 70 |
| > 5.4 | Poor | 50 |
| TMS, points | Prognosis | Comments |
| 4–9 | Excellent | Tumors that rarely metastasize |
| 10–13 | Good | Predominantly lymphogenous metastasis |
| 14–17 | Moderate | Tendency to lymphogenous as well as hematogenous metastasis |
| 18–20 | Poor | High capacity for lymphogenous and hematogenous metastasis |
| Characteristic | All patients | Pathological LN0 | Pathological LN+ | P value | |
| Number of patients | 358 | 226 (63.1%) | 132 (36.9%) | ||
| Age, years (median [Q1;Q3]) | 58.0 (48.0‒65.0) | 60.0 (49.0‒66.8) | 56.0 (45.0‒62.2) | 0.005 | |
| Pathological tumor size, cm (median [Q1;Q3]) | 1.8 (1.4‒2.5) | 1.6 (1.2‒2.1) | 2.1 (1.5‒2.9) | <0.001 | |
| Histological type (n,%) | Invasive Carcinoma of No Special Type (NST) | 243 (67.9%) | 146 (64.6%) | 97 (73.4%) | 0.137 |
| Invasive Lobular Carcinoma (ILC) | 54 (15.1%) | 34 (15.0%) | 20 (15.2%) | ||
| Special Types | 61 (17.0%) | 46 (20.4%) | 15 (11.4%) | ||
| Grade (n,%) | 1 | 49 (13.7%) | 34 (15.0%) | 15 (11.4%) | 0,407 |
| 2 | 218 (60.9%) | 139 (61.5%) | 79 (59.8%) | ||
| 3 | 91 (25.4%) | 53 (23.5%) | 38 (28.8%) | ||
| Nottingham Prognostic Index (NPI) (median [Q1;Q3]) | 3.8 (3.3‒4.5) | 3.4 (3.2‒4.2) | 4.6 (4.3‒5.5) | <0.001 | |
| Total malignancy score (TMS), points (median [Q1;Q3]) | 14.0 (12.0‒15.0) | 13.0 (11.0‒15.0) | 15.0 (14.0‒16.0) | <0.001 | |
| ER status (n,%) | 0 | 51 (14.2) | 37 (16.4) | 14 (10.6) | 0.177 |
| >0 | 307 (85.8) | 189 (83.6) | 118 (89.4) | ||
| PgR status (n,%) | 0 | 93 (26.0) | 61 (27.0) | 32 (24.2) | 0.655 |
| >0 | 265 (74.0) | 165 (73) | 100 (75.8) | ||
| HER2 status (n,%) | Not overexpressed | 318 (88.8) | 201 (88.9) | 117 (88.6) | 0.503 |
| Overexpressed | 40 (11.2) | 25 (11.1) | 15 (11.4) | ||
| Ki-67 index, % (median [Q1;Q3]) | 18.0 (12.0‒37.0) | 17.0 (12.0‒37.8) | 22.0 (14.5‒35.0) | 0.559 | |
| Surrogate subtype (n,%) | Luminal А | 202 (56.4) | 130 (57.5) | 72 (54.5) | 0.159 |
| Luminal В HER2 negative | 89 (24.9) | 49 (21.7) | 40 (30.3) | ||
| Luminal В HER2 positive | 18 (5.0) | 11 (4.9) | 7 (5.3) | ||
| Non luminal В HER2 positive | 15 (4.2) | 9 (4.0) | 6 (4.5) | ||
| Triple-negative | 34 (9.5) | 27 (11.9) | 7 (5.3) | ||
| Groups | LN+ (No=132) | LN0 (No=226) | P value | ||
| NPI | TMS | NPI | TMS | <0.001 | |
| 1 | 1 (0.8%) | 2 (1.5%) | 36 (15.9%) | 27 (11.9%) | |
| 2 | 13 (9.8%) | 17 (12.9%) | 110 (48.7%) | 65 (28.8%) | |
| 3 | 83 (62.9%) | 96 (72.7%) | 79 (35.0%) | 134 (59.3%) | |
| 4 | 35 (26.5%) | 17 (12.9%) | 1 (0.4%) | 0 (0%) | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




