Submitted:
09 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Setting
2.3. Study Population
2.4. Inclusion Criteria
- Adults aged ≤ 18 years old
- Individuals without HIV diagnosis or not on HAART.
2.5. Excluding Criteria
- Adults aged ≥ 18 years diagnosed with HIV and receiving HAART at the selected facilities.
2.6. Sampling Size and Sampling Strategy
2.6.1. Sampling Strategy
2.6.2. Sample Size Calculation
2.7. Statistical Analysis
2.7. Patients Records Review for Clinical Data Collection
2.8. Specimen Collection, Storage, Transport, and Handling
2.9. Laboratory Procedure (Serum Lipid Profile)
2.8. Ethical Considerations and Data Protection
3. Results
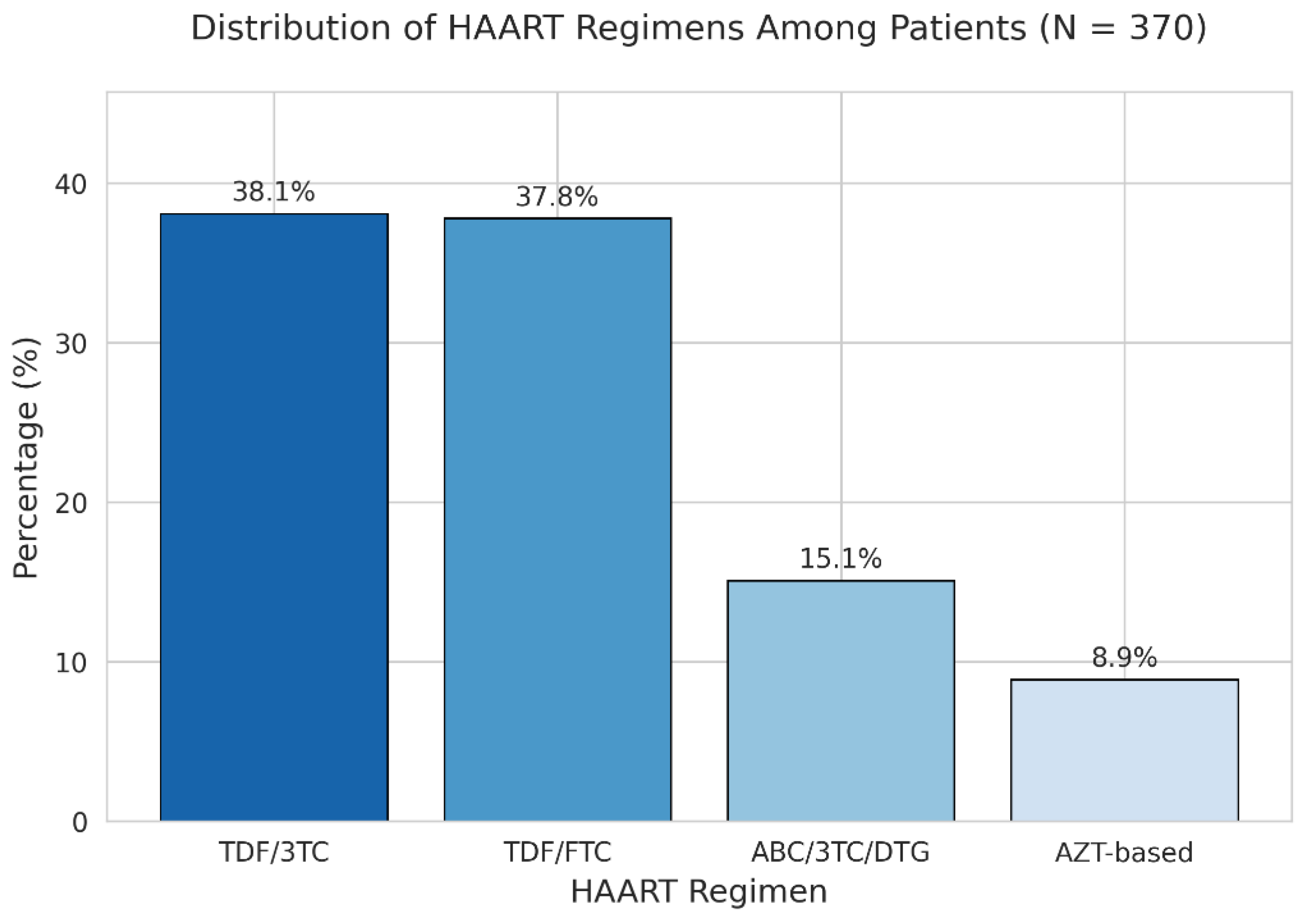
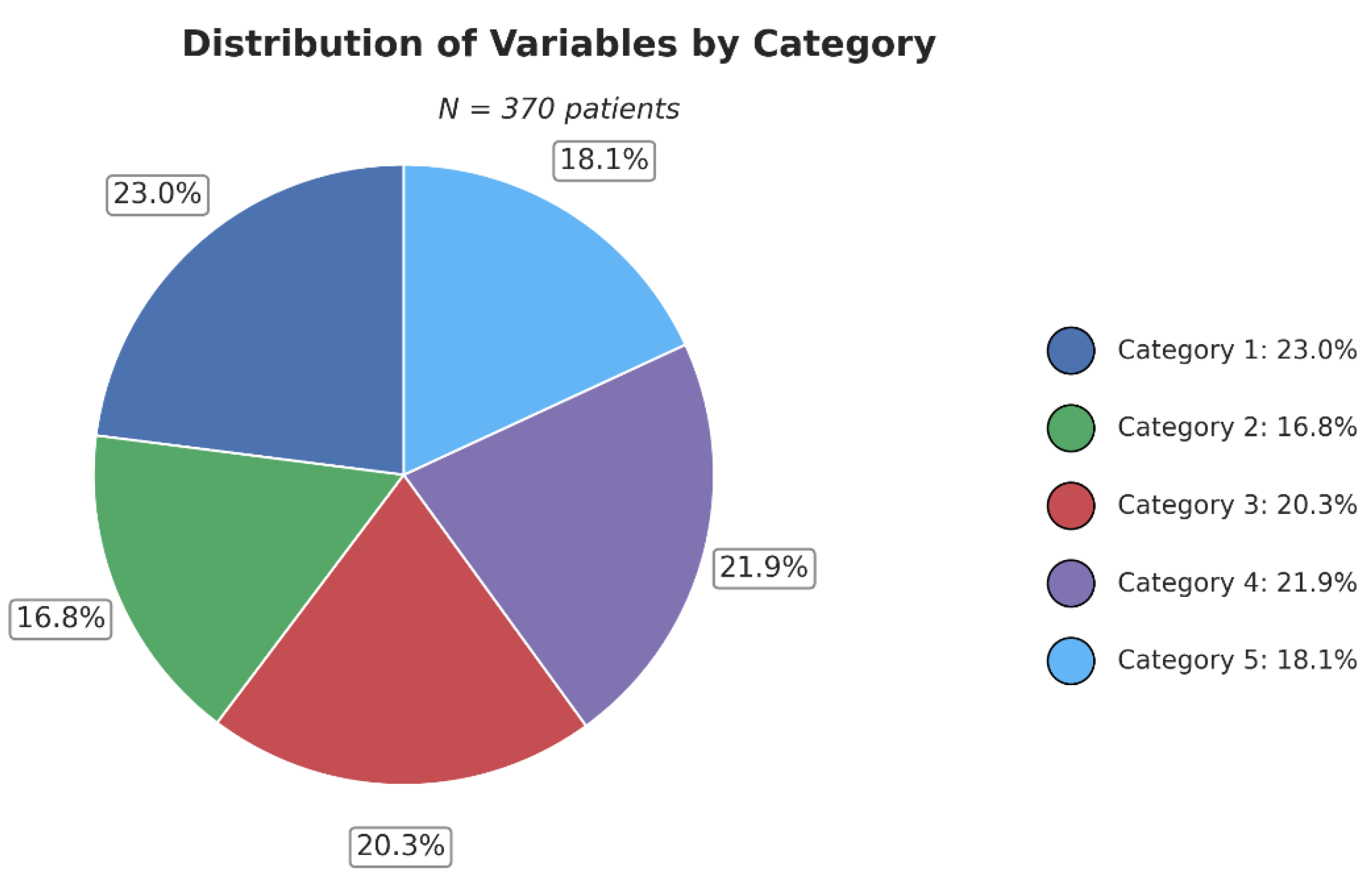
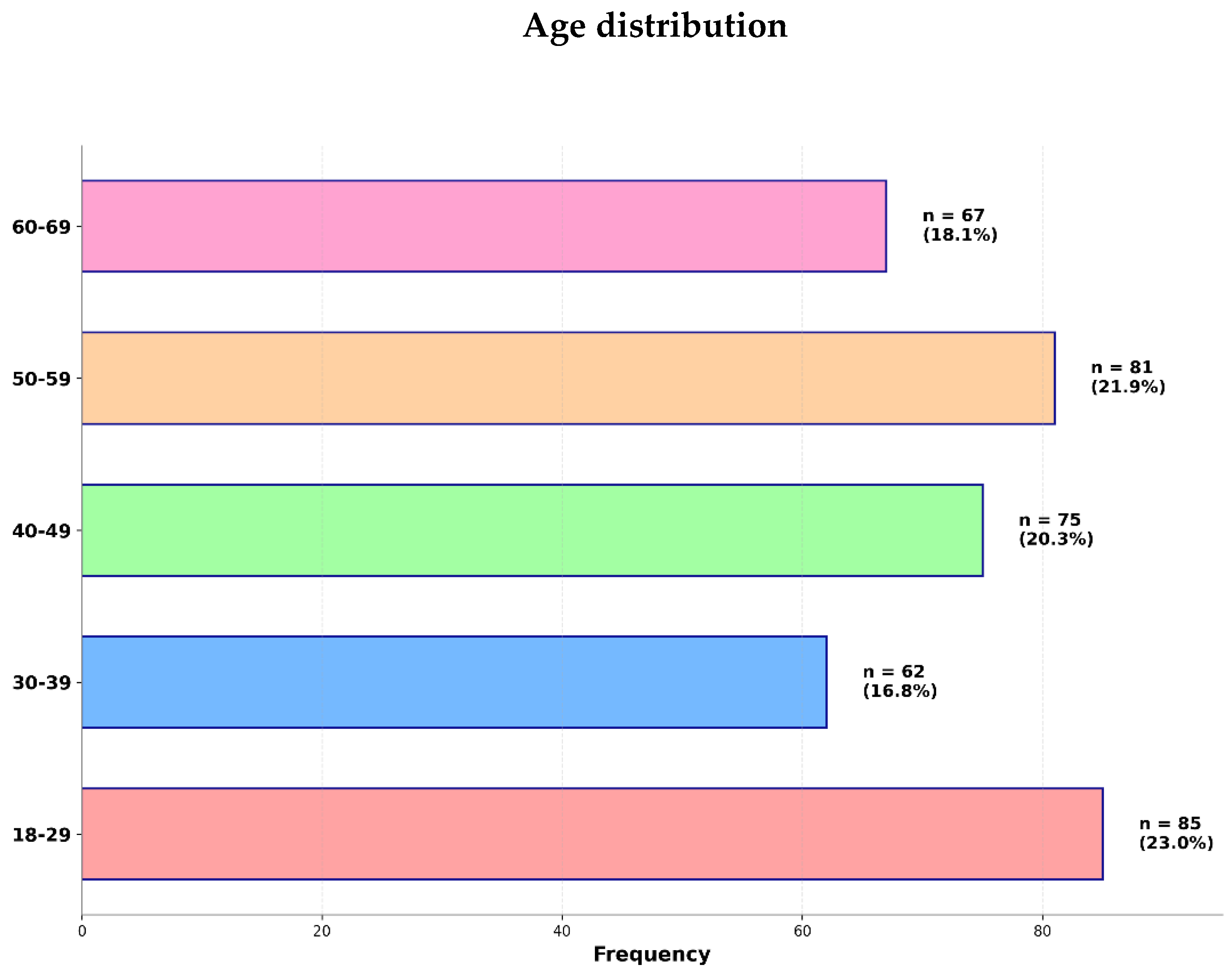
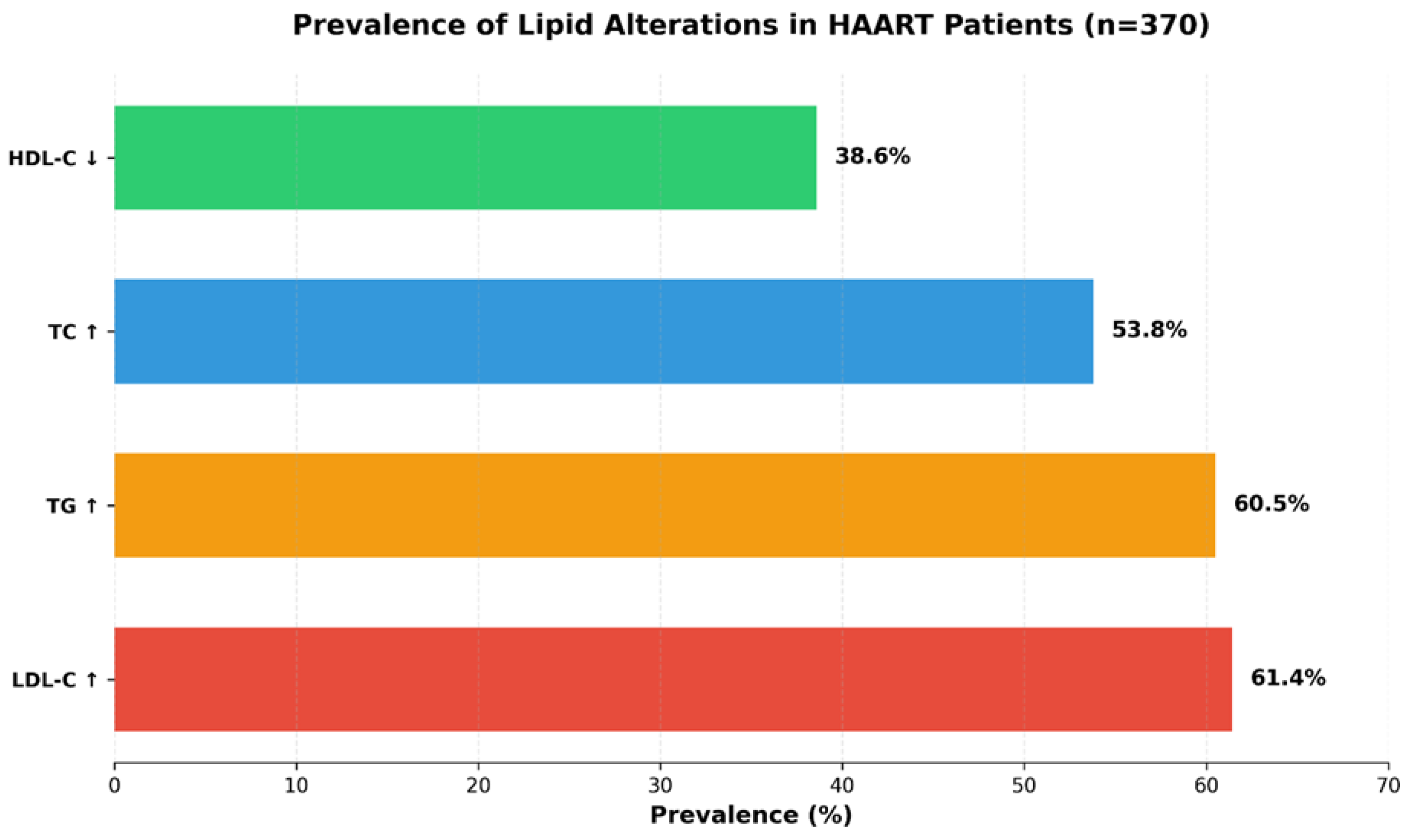
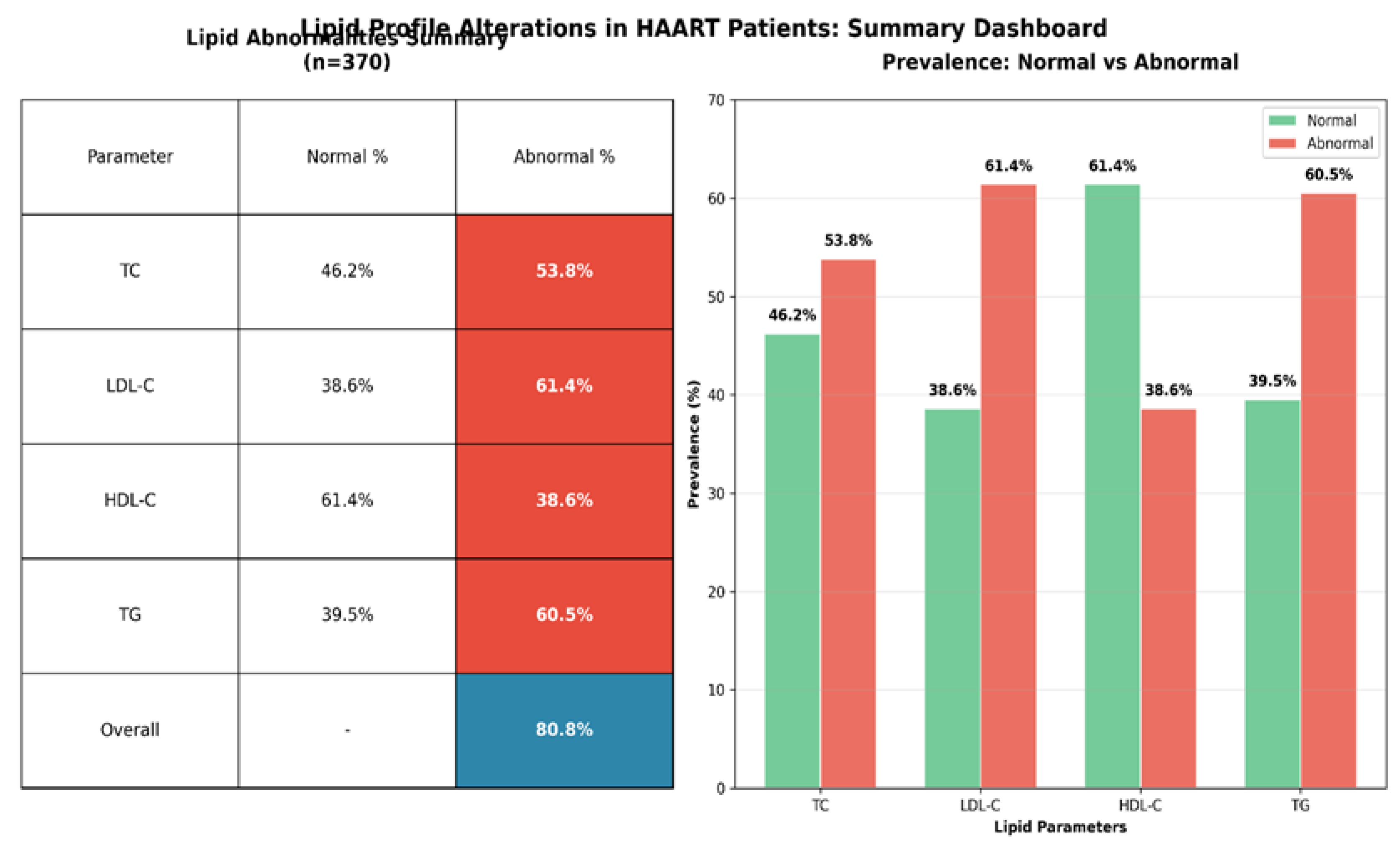
4. Discussion
4.1. Comparison with Existing Literature
4.2. Clinical and Public Health Implications
4.3. Strengths
4.4. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. HIV statistics, globally and by WHO region, 2023 (No. WHO/UCN/HHS/SIA/2023.01); World Health Organization, 2023. [Google Scholar]
- Phaswana-Mafuya, N.; Seager, J.; Peltzer, K.; Jooste, S.; Mkhonto, S. Social determinants of HIV in the Eastern Cape; Eastern Cape Socio-Economic Consultative Council, 2010. [Google Scholar]
- Huibers, M. HIV-infection in sub-Saharan Africa. Details not provided.
- Bradshaw, D.; Msemburi, W.; Dorrington, R.; Pillay-van Wyk, V.; Laubscher, R.; Groenewald, P. and South African National Burden of Disease Study team, 2016. HIV/AIDS in South Africa: how many people died from the disease between 1997 and 2010? Aids 30(5), 771–778. [CrossRef] [PubMed]
- Das, S.; Grant, L.; Fernandes, G. Task shifting healthcare services in the post-COVID world: a scoping review. PLOS Glob Public Health 2023, 3(12), e0001712. [Google Scholar] [CrossRef] [PubMed]
- Syengo, S.M. Lipid profiles, cardiovascular disease risk and dyslipidemia in HIV-positive patients on HAART at Machakos Level Five Hospital, Machakos County, Kenya. dissertation, Kenyatta University, 2023. [Google Scholar]
- Bari, R. Dyslipidemia in human immunodeficiency virus. In Manual of Lipidology; 2025; p. 297. [Google Scholar]
- Zeleke, T.M.; Liben, F.E.; Tesfaye, T.S.; Munea, A.L.; Kumsa, F.A. Prevalence and associated factors of dyslipidemia among adults with coexisting chronic disease in Ethiopia: a systematic review and meta-analysis. PLoS One 2025, 20(4), e0320119. [Google Scholar] [CrossRef] [PubMed]
- Nyabera, R.A.O. Risk factors associated with cardiovascular disease among people living with human immunodeficiency virus in Nairobi City County, Kenya. dissertation, Kenyatta University, 2022. [Google Scholar]
- Dudley, L.D. Continuity of care for tuberculosis patients between hospital and primary health care services in South Africa. dissertation, Stellenbosch University, 2020. [Google Scholar]
- Oyetunji, I.O. Nutritional and health status of HIV-positive adults stable on HAART attending a healthcare facility in Cape Town, South Africa. dissertation, 2022. [Google Scholar]
- Hopkins, K.L. Curbing the non-communicable disease epidemic: an evaluation of integrating rapid testing for NCD risk factors and navigated linkage to care into a standard HIV testing service platform for adults in Soweto, South Africa. dissertation, University of the Witwatersrand, 2021. [Google Scholar]
- Chilufya, M.M. The burden of non-communicable diseases among people living with HIV and the extent, cost and framework development of integrated HIV and non-communicable diseases care at primary health care facilities in Southern Africa. dissertation, University of Pretoria, 2024. [Google Scholar]
- World Health Organization. Operations manual for delivery of HIV prevention, care and treatment at primary health centres in high-prevalence, resource-constrained settings; WHO: Geneva, 2008. [Google Scholar]
- Mashao, M.M. The effect of highly active antiretroviral treatment on glucose and lipid metabolism in human immunodeficiency virus positive patients at clinics in the Polokwane Local Municipality, Limpopo Province, South Africa. dissertation, University of Limpopo, 2016. [Google Scholar]
- Mugisha, N.; Ghanem, L.; Komi, O.A.; Noureddine, R.; Shariff, S.; Wojtara, M.; Nanehkeran, M.M.; Uwishema, O. Addressing cardiometabolic challenges in HIV: insights, impact, and best practices for optimal management—a narrative review. Health Sci Rep. 2025, 8(4), e70727. [Google Scholar] [CrossRef] [PubMed]
- Songtaweesin, W.N.; Saisaengjan, C.; Soponphan, J.; Deeklum, P.; Nadsasarn, R.; Pitikawinwong, L.; Phasomsap, C.; Puthanakit, T. Outcomes of dolutegravir-based antiretroviral therapy in Thai youth aged 18 to 24 years living with HIV in Bangkok: a retrospective cohort study. J Med Assoc Thai 2023, 106(10). [Google Scholar]
- Nonterah, E.A. The effects of HIV and ART on serum lipids among adults in Agincourt in 2015. master’s thesis, University of the Witwatersrand, 2017. [Google Scholar]
- Hamada, Y. Research to understand multimorbidity in households affected by tuberculosis [dissertation]. University College London, 2025. [Google Scholar]
- Hopkins, K.L. Curbing the non-communicable disease epidemic: an evaluation of integrating rapid testing for NCD risk factors and navigated linkage to care into a standard HIV testing service platform for adults in Soweto, South Africa. dissertation, University of the Witwatersrand, 2021. [Google Scholar]
- Kiyimba, T.; Matthys, C.; Van Der Schueren, B.; Ogwok, P. Optimizing the cardiometabolic health of Ugandans living with HIV: unravelling the role of polyphenol-rich indigenous fruits and vegetables. 2024. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



