Submitted:
10 April 2026
Posted:
10 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Plasma Glucose with a Postprandial Time of Less Than 4 h
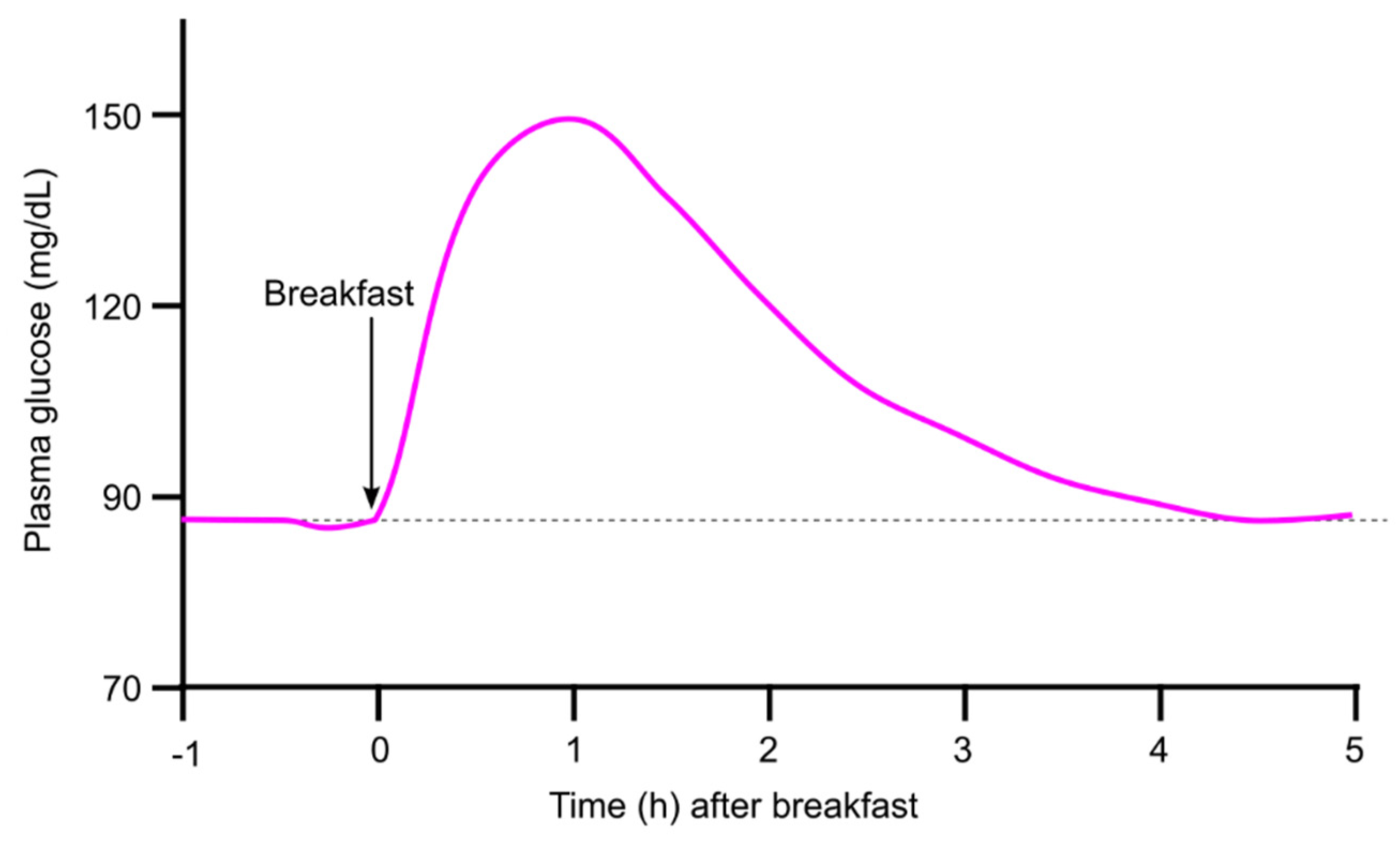
2.1. Plasma Glucose Returns to Baseline Within 4 h After a Meal
2.2. PPG Within 4 h After a Meal for Risk Prediction
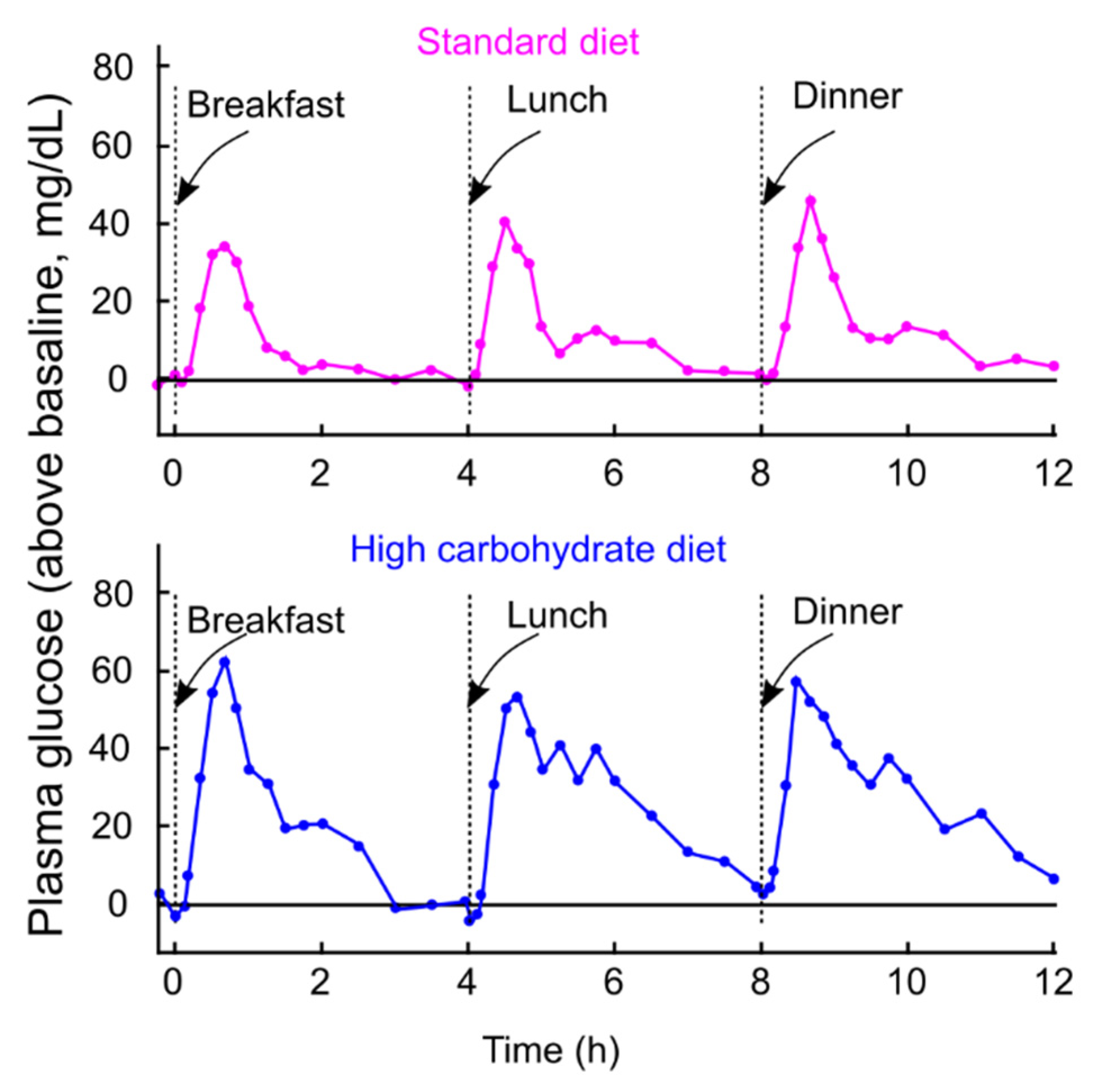
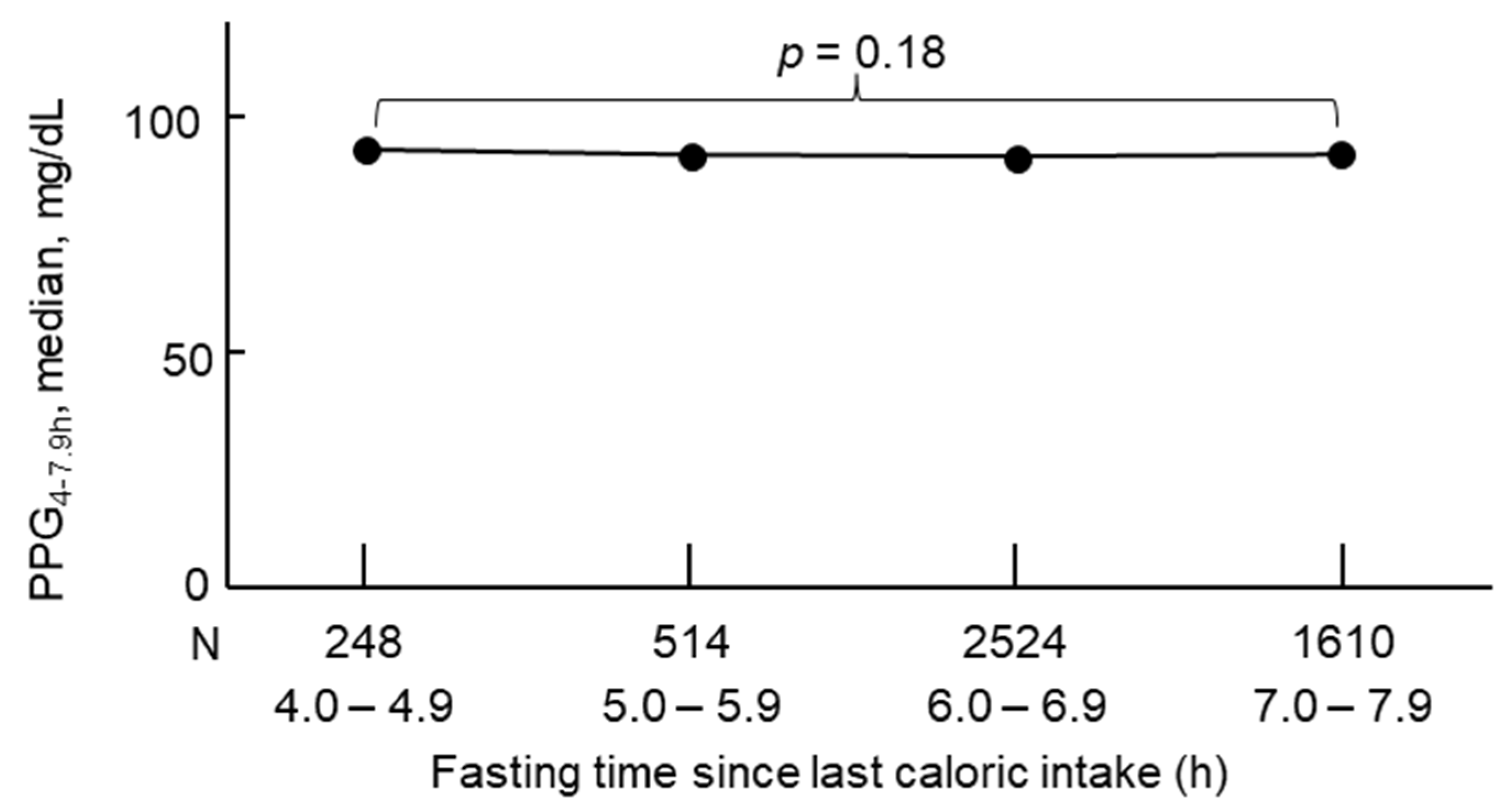
3. PPG Levels Are Stable Between 4 and 7.9 h After a Meal
4. Clinical Significance of PPG4-7.9h
4.1. PPG4-7.9h for Diabetes Diagnosis
4.2. PPG4–7.9h for Mortality Risk Prediction From CVD and Diabetes
4.3. PPG4-7.9h for Mortality Risk Prediction From Cancer
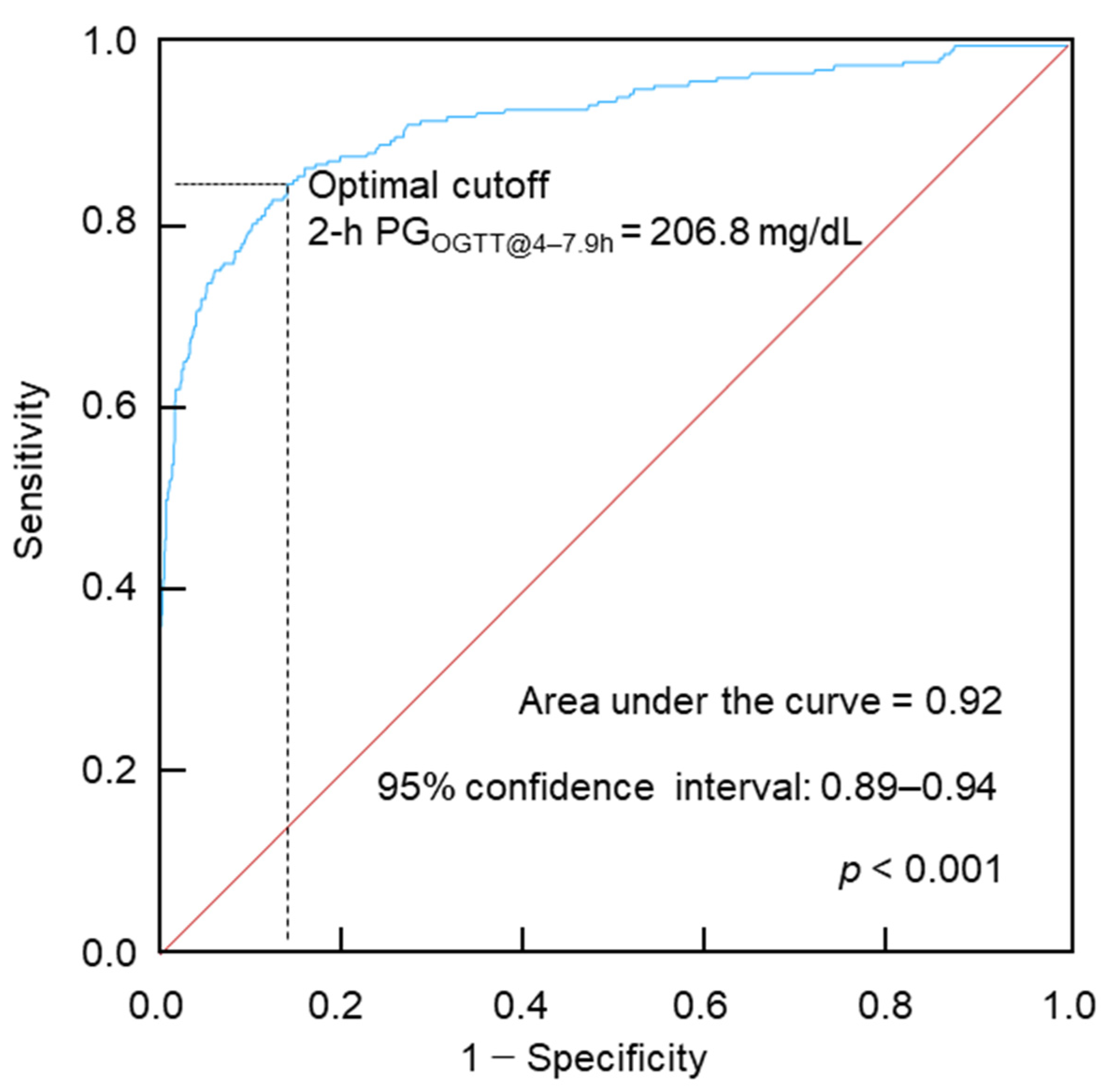
5. Two-hour Plasma Glucose During OGTT Performed Between 4 and 7.9 h After a Meal (2-h PGOGTT@4–7.9h)
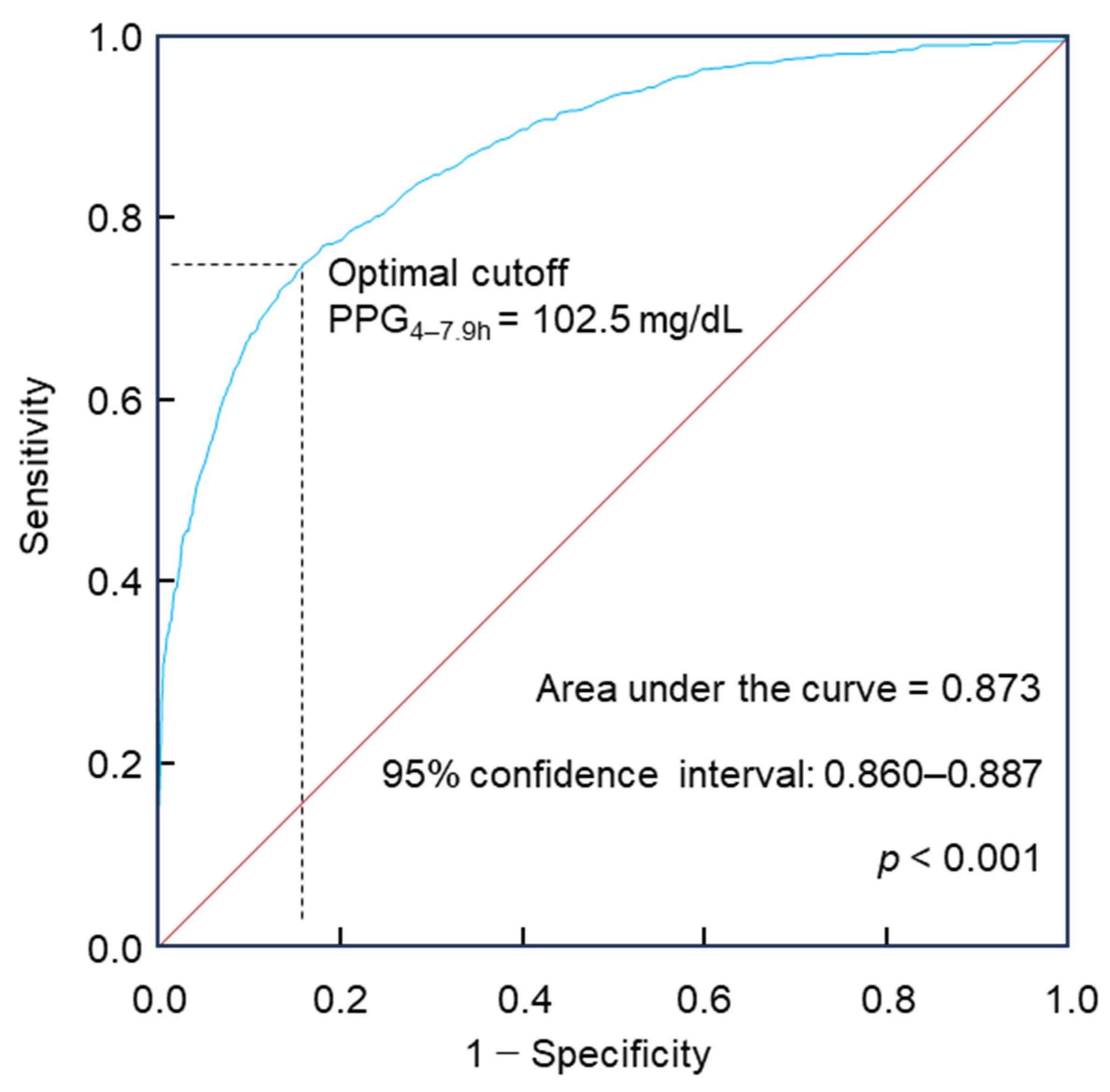
5.1. 2-h PGOGTT@4–7.9h for Diabetes Diagnosis
5.2. 2-h PGOGTT@4–7.9h for Prediabetes Risk
5.3. 2-h PGOGTT@4– 7.9h for Mortality Risk Prediction
6. Mechanisms Linking Postprandial Hyperglycemia to Cardiovascular Disease
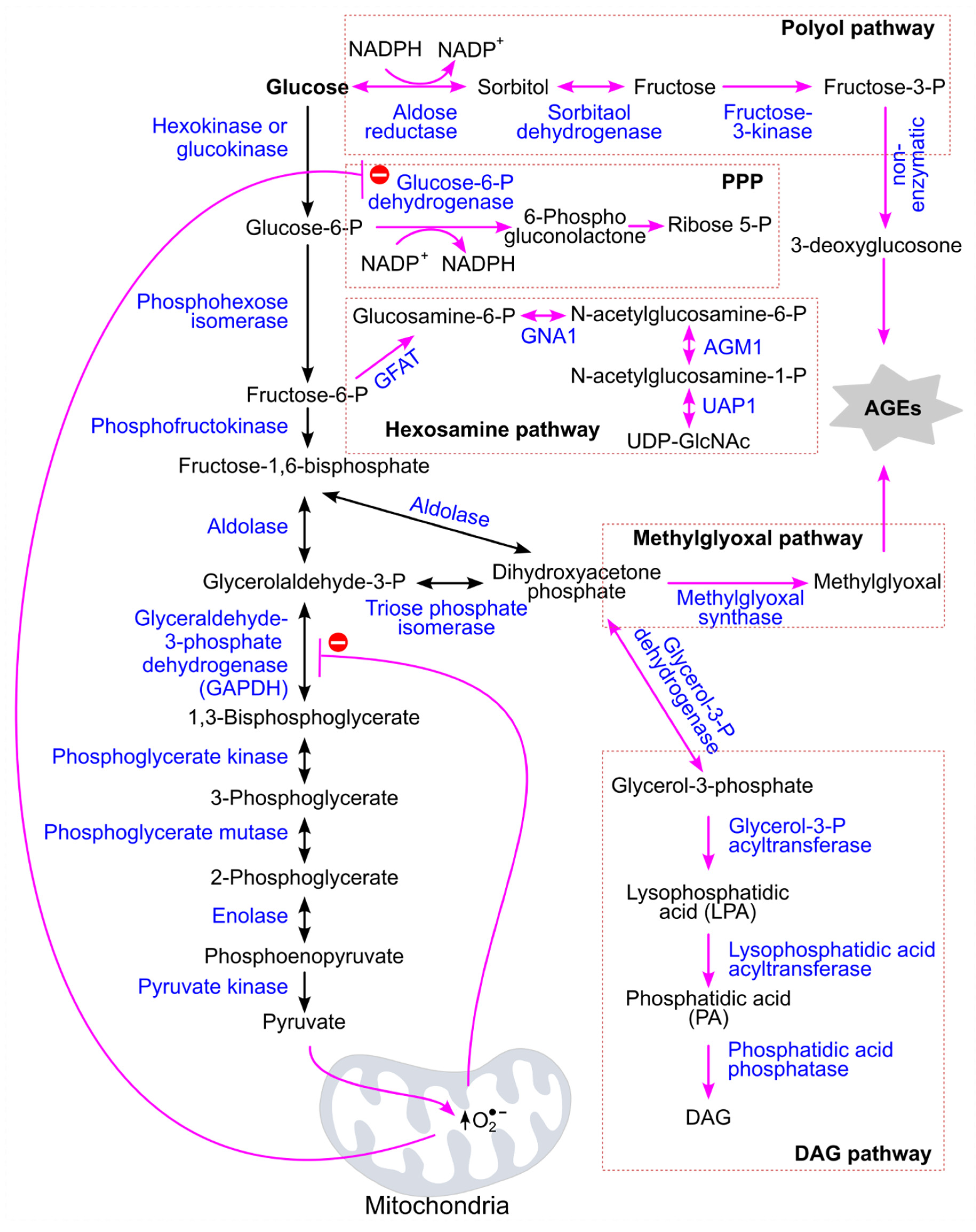
6.1. Postprandial Hyperglycemia Induces Changes in Glucose Metabolism
6.2. Mechanisms Underlying Postprandial Hyperglycemia-Induced Increase in CVD Risks
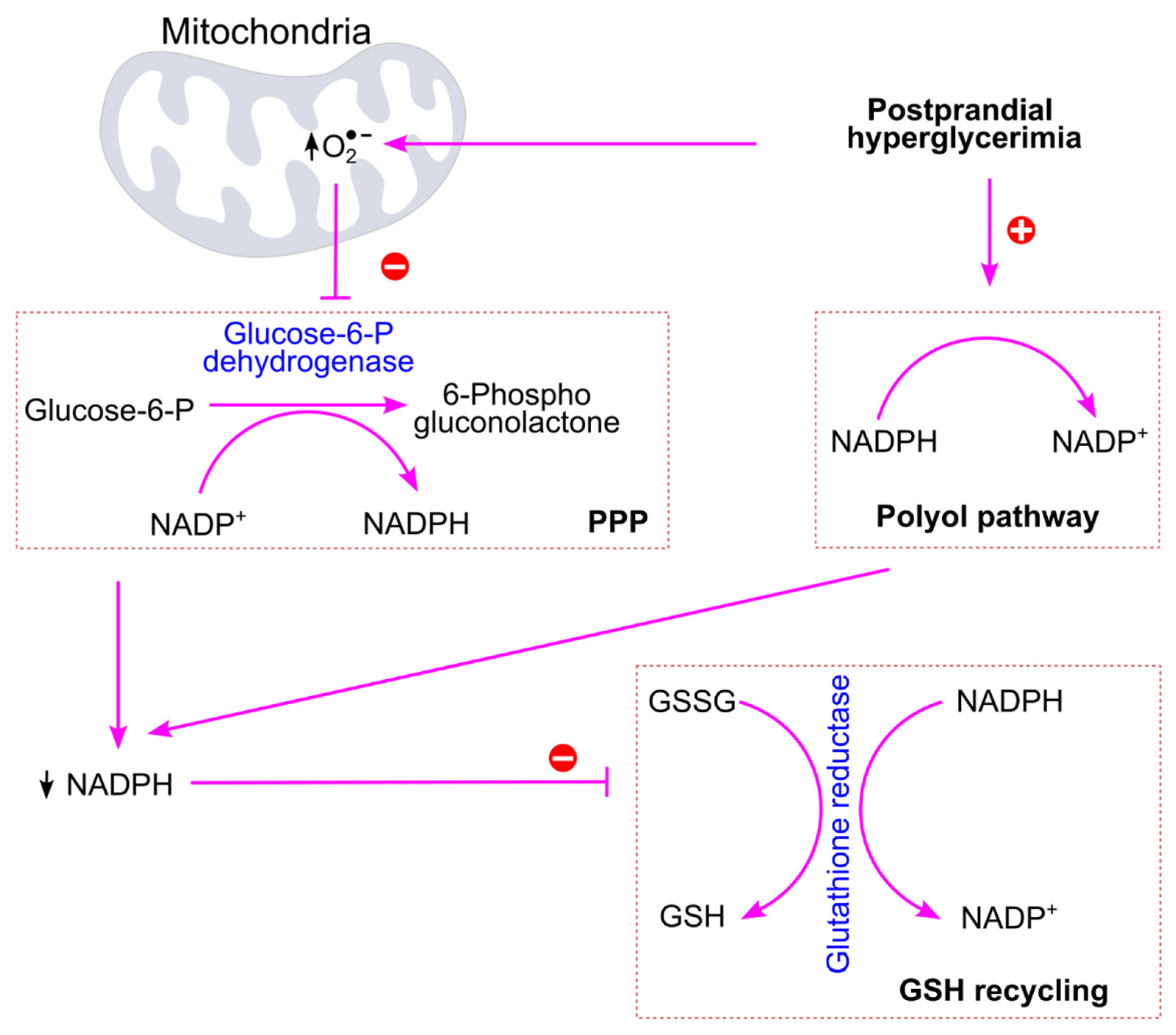
6.2.1. Postprandial Hyperglycemia Increases Oxidative Stress
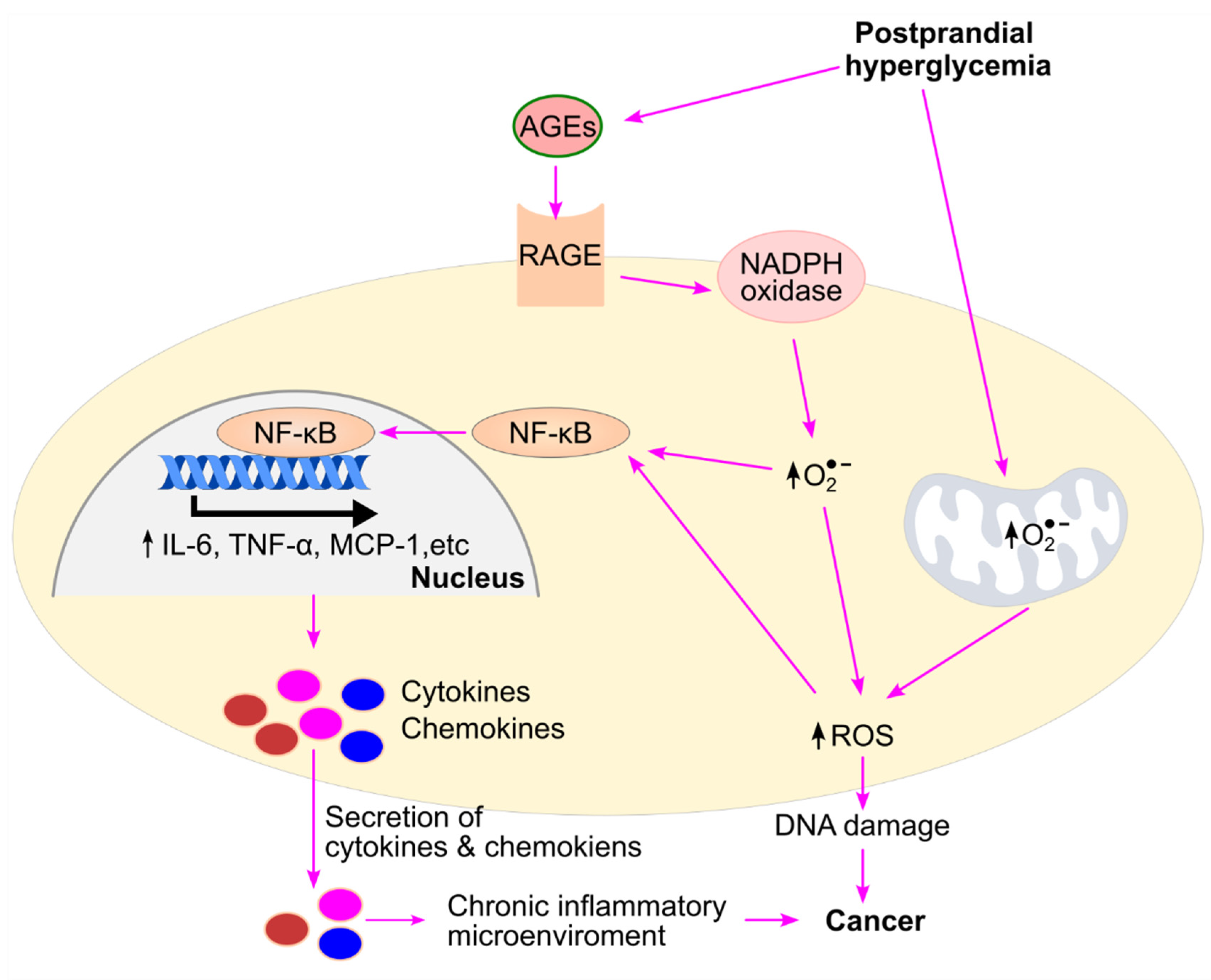
6.2.2. Postprandial Hyperglycemia Leads to Activation of Inflammatory Pathways
6.2.3. Postprandial Hyperglycemia Leads to Endothelial Dysfunction
6.2.4. Postprandial Hyperglycemia Promotes Atherogenesis
6.2.5. Postprandial Hyperglycemia Enhances Thrombotic Events
7. Mechanisms Linking Postprandial Hyperglycemia to Cancer
7.1. Postprandial Hyperglycemia Increases ROS Production and Inflammation
7.2. Postprandial Hyperglycemia Promotes Cancer Proliferation Through Increased Circulating Insulin
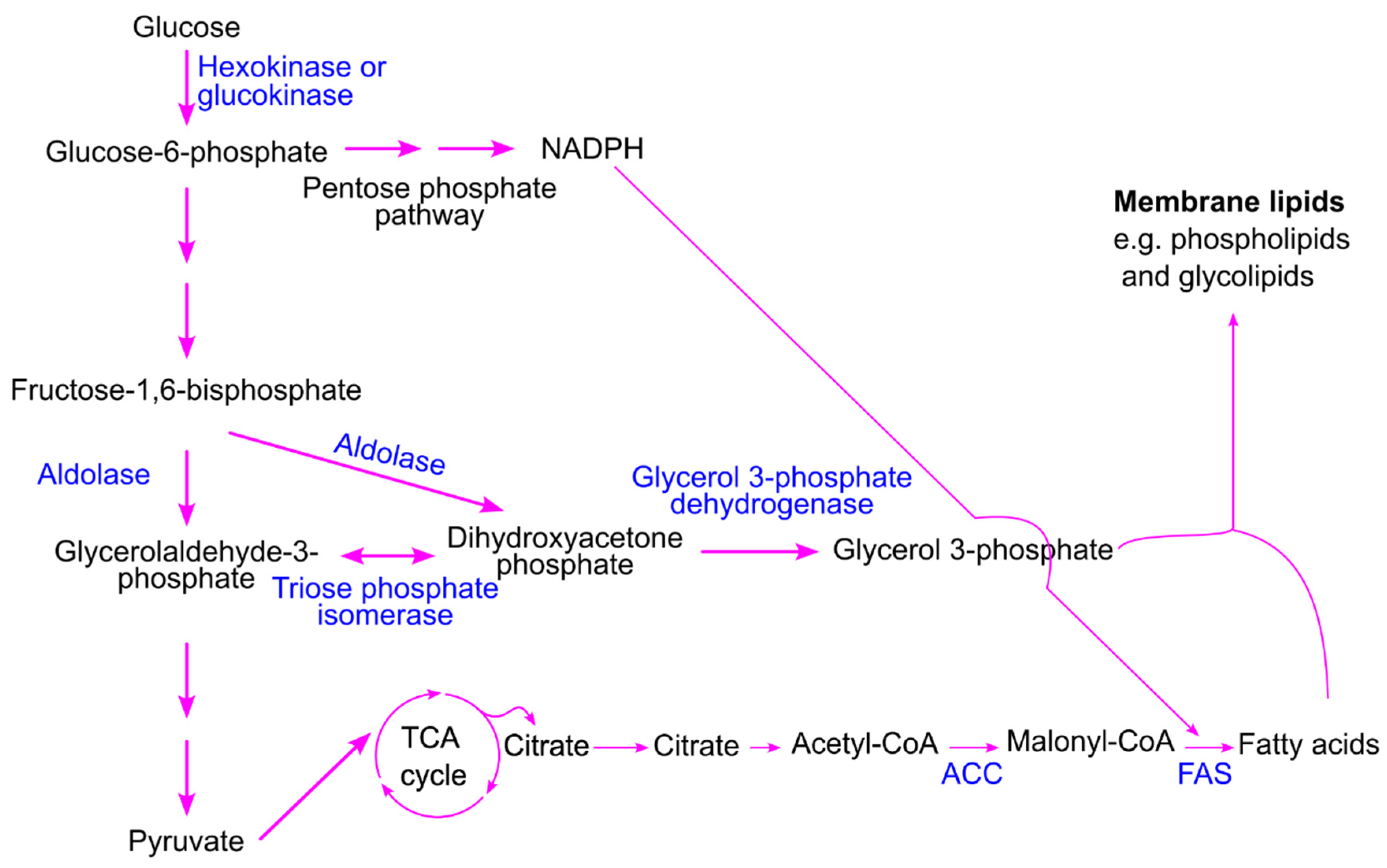
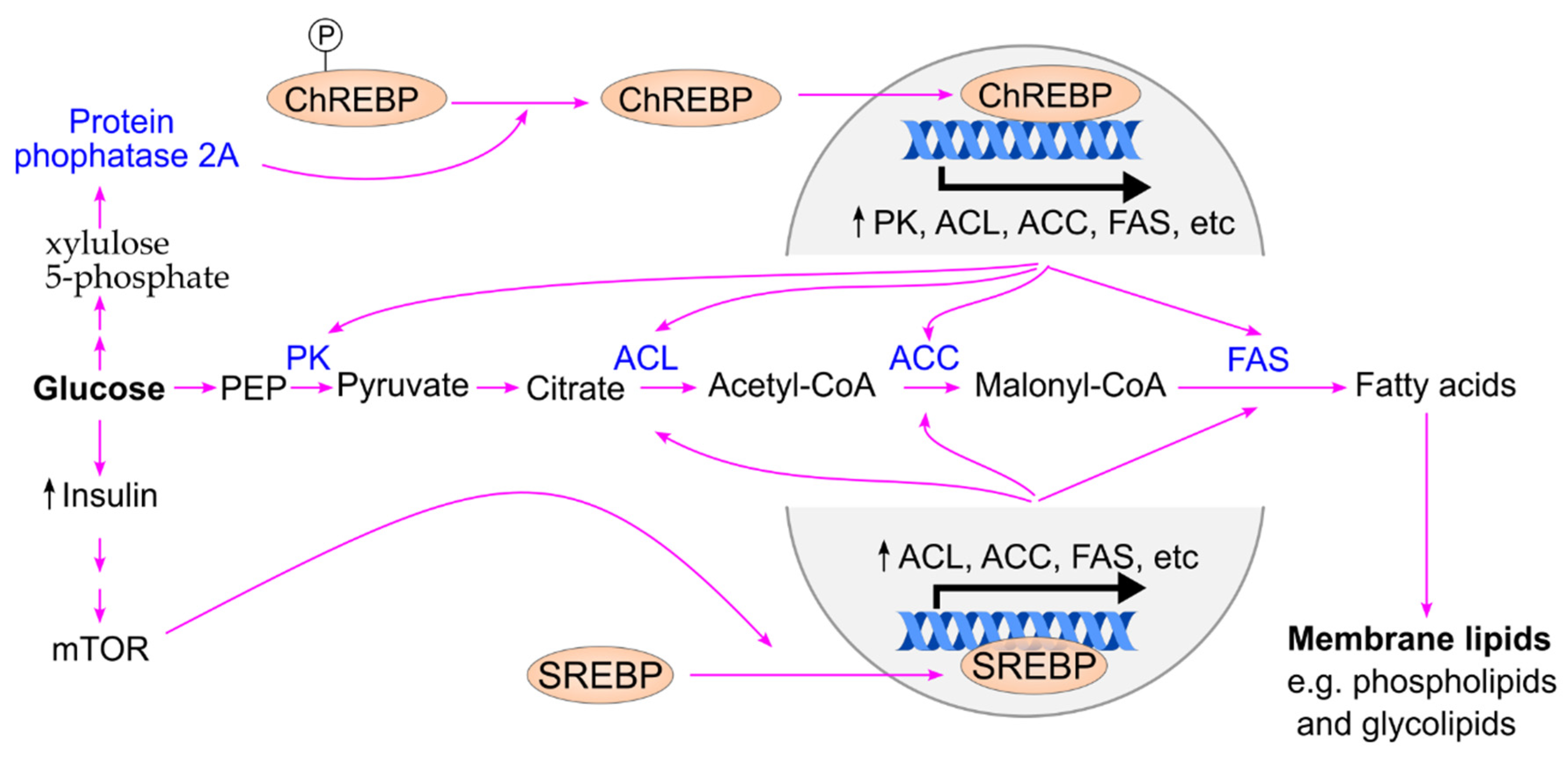
7.3. Postprandial Hyperglycemia Drives Lipogenesis to Support Cancer Cell Proliferation
8. Future Research
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 2-h PGOGTT@4–7.9h | 2-hour plasma glucose during an OGTT performed between 4 and 7.9 hours after a meal |
| ACC | acetyl-CoA carboxylase |
| ACL | ATP citrate lyase |
| AGE | advanced glycation end product |
| AGM1 | phosphoacetylglucosamine mutase |
| CI | confidence interval |
| CVD | cardiovascular disease |
| DAG | diacyl glycerol |
| FAS | fatty acid synthase |
| GSH | reduced glutathione |
| GSSG | glutathione disulfide |
| GAPDH | glyceraldehyde-3-phosphate dehydrogenase |
| GFAT | glutamine:fructose-6-phosphate aminotransferase |
| GNA1 | GlcN-6P acetyltransferase |
| h | hour |
| HbA1c | hemoglobin A1c |
| HDL | high-density lipoprotein |
| HR | hazard ratio |
| ICAM-1 | intercellular adhesion molecule 1 |
| IL | interleukin |
| LDL | low-density lipoprotein |
| LPA | lysophosphatidic acid |
| MAPK | mitogen-activated protein kinase |
| MCP-1 | monocyte chemoattractant protein-1 |
| mTOR | mammalian target of rapamycin |
| NADP+ | oxidized nicotinamide adenine dinucleotide phosphate |
| NADPH | reduced nicotinamide adenine dinucleotide phosphate |
| NF-κB | nuclear factor-κB |
| O2● | superoxide |
| NO | nitric oxide |
| OGTT | oral glucose tolerance test |
| P | phosphate |
| PA | phosphatidic acid |
| PEP | phosphoenopyruvate |
| PK | pyruvate kinase |
| PPG | postprandial plasma glucose |
| PPG4-7.9h | postprandial plasma glucose measured 4–7.9 hours after a meal |
| PPP | pentose phosphate pathway |
| RAGE | receptor for advanced glycation end-product |
| ROS | reactive oxygen species |
| T2DM | type 2 diabetes mellites |
| TCA | tricarboxylic acid |
| TNF-α | tumor necrosis factor-alpha |
| UAP1 | UDP-GlcNAc pyrophosphorylase |
| UDP-GlcNAc | uridine diphosphate N-acetylglucosamine |
| VCAM-1 | vascular cell adhesion protein 1 |
References
- Galicia-Garcia, U.; Benito-Vicente, A.; Jebari, S.; Larrea-Sebal, A.; Siddiqi, H.; Uribe, K.B.; Ostolaza, H.; Martín, C. Pathophysiology of Type 2 Diabetes Mellitus. Int. J. Mol. Sci. 2020, 21, 6275. [CrossRef]
- Hameed, I.; Masoodi, S.R.; Mir, S.A.; Nabi, M.; Ghazanfar, K.; Ganai, B.A. Type 2 diabetes mellitus: From a metabolic disorder to an inflammatory condition. World J. Diabetes 2015, 6, 598-612. [CrossRef]
- ElSayed, N.A.; Aleppo, G.; Aroda, V.R.; Bannuru, R.R.; Brown, F.M.; Bruemmer, D.; Collins, B.S.; Gaglia, J.L.; Hilliard, M.E.; Isaacs, D.; et al. 2. Classification and Diagnosis of Diabetes: Standards of Care in Diabetes—2023. Diabetes Care 2023, 46, S19-S40. [CrossRef]
- American Diabetes Association Professional Practice Committee for Diabetes. 2. Diagnosis and Classification of Diabetes: Standards of Care in Diabetes—2026. Diabetes Care 2026, 49, S27-S49. [CrossRef]
- Duncan, B.B.; Magliano, D.J.; Boyko, E.J. IDF diabetes atlas 11th edition 2025: global prevalence and projections for 2050. 2026, 41, 7-9.
- NCD Risk Factor Collaboration. Worldwide trends in diabetes since 1980: a pooled analysis of 751 population-based studies with 4.4 million participants. Lancet 2016, 387, 1513-1530. [CrossRef]
- Hu, F.B. Globalization of diabetes: the role of diet, lifestyle, and genes. Diabetes Care 2011, 34, 1249-1257. [CrossRef]
- Singhal, A.; Saxena, D.; Ahada, R.S.; Kurian, N.M.; Singh, A.P.; Garg, N.; Bhatia, E.; Yadav, S.C. Micro-and macrovascular complications in early-onset type 2 diabetes in Indians: prevalence and predictors in a prospective cross-sectional study. Endocrine Connections 2025, 14, e250226. [CrossRef]
- Dunn, T.J.; Tan, X.; Harton, J.; Kim, S.; Xie, L.; Gamble, C.; Rotroff, D. Macrovascular and microvascular complications in US Medicare enrollees with type 2 diabetes with and without atherosclerotic cardiovascular disease. Diabetes, Obesity and Metabolism 2025, 27, 4137-4147.
- Low Wang, C.C.; Hess, C.N.; Hiatt, W.R.; Goldfine, A.B. Clinical update: cardiovascular disease in diabetes mellitus: atherosclerotic cardiovascular disease and heart failure in type 2 diabetes mellitus–mechanisms, management, and clinical considerations. Circulation 2016, 133, 2459-2502.
- Qian, T.; Sun, H.; Xu, Q.; Hou, X.; Hu, W.; Zhang, G.; Drummond, G.R.; Sobey, C.G.; Charchar, F.J.; Golledge, J.; et al. Hyperuricemia is independently associated with hypertension in men under 60 years in a general Chinese population. J. Hum. Hypertens. 2021, 35, 1020-1028, doi:doi:10.1038/s41371-020-00455-7.
- Jackman, K.A.; Brait, V.H.; Wang, Y.; Maghzal, G.J.; Ball, H.J.; McKenzie, G.; De Silva, T.M.; Stocker, R.; Sobey, C.G. Vascular expression, activity and function of indoleamine 2,3-dioxygenase-1 following cerebral ischaemia-reperfusion in mice. Naunyn Schmiedebergs Arch. Pharmacol. 2011, 383, 471-481. [CrossRef]
- Zaccardi, F.; Ling, S.; Gillies, C.; Brown, K.; Davies, M.J.; Khunti, K. Trajectories of type 2 diabetes and cancer in 330 000 individuals with prediabetes: 20-year observational study in England. The Lancet Diabetes & Endocrinology 2026, 14, 41-49. [CrossRef]
- Safadi, H.; Balogh, Á.; Lám, J.; Nagy, A.; Belicza, É. Associations between diabetes and cancer: A 10-year national population-based retrospective cohort study. Diabetes Res. Clin. Pract. 2024, 211, 111665. [CrossRef]
- Rey-Reñones, C.; Baena-Díez, J.M.; Aguilar-Palacio, I.; Miquel, C.; Grau, M. Type 2 Diabetes Mellitus and Cancer: Epidemiology, Physiopathology and Prevention. Biomedicines 2021, 9, 1429. [CrossRef]
- Erickson, K.; Patterson, R.E.; Flatt, S.W.; Natarajan, L.; Parker, B.A.; Heath, D.D.; Laughlin, G.A.; Saquib, N.; Rock, C.L.; Pierce, J.P. Clinically defined type 2 diabetes mellitus and prognosis in early-stage breast cancer. J. Clin. Oncol. 2011, 29, 54-60. [CrossRef]
- Hosokawa, T.; Kurosaki, M.; Tsuchiya, K.; Matsuda, S.; Muraoka, M.; Suzuki, Y.; Tamaki, N.; Yasui, Y.; Nakata, T.; Nishimura, T.; et al. Hyperglycemia is a significant prognostic factor of hepatocellular carcinoma after curative therapy. World J. Gastroenterol. 2013, 19, 249-257. [CrossRef]
- Hu, D.; Peng, F.; Lin, X.; Chen, G.; Zhang, H.; Liang, B.; Ji, K.; Lin, J.; Chen, L.F.; Zheng, X.; et al. Preoperative Metabolic Syndrome Is Predictive of Significant Gastric Cancer Mortality after Gastrectomy: The Fujian Prospective Investigation of Cancer (FIESTA) Study. EBioMedicine 2017, 15, 73-80. [CrossRef]
- Okamura, A.; Watanabe, M.; Imamura, Y.; Hayami, M.; Yamashita, K.; Kurogochi, T.; Mine, S. Glycemic Status and Prognosis of Patients with Squamous Cell Carcinoma of the Esophagus. World J. Surg. 2017, 41, 2591-2597. [CrossRef]
- Ogurtsova, K.; Guariguata, L.; Barengo, N.C.; Ruiz, P.L.-D.; Sacre, J.W.; Karuranga, S.; Sun, H.; Boyko, E.J.; Magliano, D.J. IDF diabetes Atlas: Global estimates of undiagnosed diabetes in adults for 2021. Diabetes Res. Clin. Pract. 2022, 183, 109118. [CrossRef]
- Bergman, M.; Manco, M.; Satman, I.; Chan, J.; Schmidt, M.I.; Sesti, G.; Vanessa Fiorentino, T.; Abdul-Ghani, M.; Jagannathan, R.; Kumar Thyparambil Aravindakshan, P.; et al. International Diabetes Federation Position Statement on the 1-hour post-load plasma glucose for the diagnosis of intermediate hyperglycaemia and type 2 diabetes. Diabetes Res. Clin. Pract. 2024, 209, 111589. [CrossRef]
- The Royal Australian College of General Practitioners. General practice management of type 2 diabetes: 2016–18. East Melbourne, Vic: RACGP, 2016. Available from https://www.diabetesaustralia.com.au/wp-content/uploads/General-Practice-Management-of-Type-2-Diabetes-2016-18.pdf.
- Cowie, C.C.; Rust, K.F.; Byrd-Holt, D.D.; Gregg, E.W.; Ford, E.S.; Geiss, L.S.; Bainbridge, K.E.; Fradkin, J.E. Prevalence of diabetes and high risk for diabetes using A1C criteria in the U.S. population in 1988-2006. Diabetes Care 2010, 33, 562-568. [CrossRef]
- Darras, P.; Mattman, A.; Francis, G.A. Nonfasting lipid testing: the new standard for cardiovascular risk assessment. CMAJ 2018, 190, E1317-E1318. [CrossRef]
- Mach, F.; Baigent, C.; Catapano, A.L.; Koskinas, K.C.; Casula, M.; Badimon, L.; Chapman, M.J.; De Backer, G.G.; Delgado, V.; Ference, B.A.; et al. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk: The Task Force for the management of dyslipidaemias of the European Society of Cardiology (ESC) and European Atherosclerosis Society (EAS). Eur. Heart J. 2019, 41, 111-188. [CrossRef]
- Pearson, G.J.; Thanassoulis, G.; Anderson, T.J.; Barry, A.R.; Couture, P.; Dayan, N.; Francis, G.A.; Genest, J.; Grégoire, J.; Grover, S.A.; et al. 2021 Canadian Cardiovascular Society Guidelines for the Management of Dyslipidemia for the Prevention of Cardiovascular Disease in the Adult. Can. J. Cardiol. 2021, 37, 1129-1150. [CrossRef]
- Grundy, S.M.; Stone, N.J.; Bailey, A.L.; Beam, C.; Birtcher, K.K.; Blumenthal, R.S.; Braun, L.T.; de Ferranti, S.; Faiella-Tommasino, J.; Forman, D.E.; et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol. J. Am. Coll. Cardiol. 2019, 73, e285-e350. [CrossRef]
- Fang, Y.; Wang, Y. Fasting status modifies the association between triglyceride and all-cause mortality: A cohort study. Health Sci Rep 2022, 5, e642. [CrossRef]
- Fiamoncini, J.; Newman, J.; Brennan, L. Editorial: Postprandial physiology. Front Nutr 2022, 9, 1107480. [CrossRef]
- Smith, J.A.B.; Murach, K.A.; Dyar, K.A.; Zierath, J.R. Exercise metabolism and adaptation in skeletal muscle. Nat. Rev. Mol. Cell Biol. 2023, 24, 607-632. [CrossRef]
- Lewis, G.F.; Carpentier, A.C.; Pereira, S.; Hahn, M.; Giacca, A. Direct and indirect control of hepatic glucose production by insulin. Cell Metab. 2021, 33, 709-720. [CrossRef]
- Jalleh, R.J.; Jones, K.L.; Rayner, C.K.; Marathe, C.S.; Wu, T.; Horowitz, M. Normal and disordered gastric emptying in diabetes: recent insights into (patho)physiology, management and impact on glycaemic control. Diabetologia 2022, 65, 1981-1993. [CrossRef]
- Xiang, C.; Sun, Y.; Luo, Y.; Xie, C.; Huang, W.; Sun, Z.; Jones, K.L.; Horowitz, M.; Rayner, C.K.; Ma, J.; et al. Gastric emptying of a glucose drink is predictive of the glycaemic response to oral glucose and mixed meals, but unrelated to antecedent glycaemic control, in type 2 diabetes. Nutr. Diabetes 2024, 14, 13. [CrossRef]
- Woerle, H.J.; Szoke, E.; Meyer, C.; Dostou, J.M.; Wittlin, S.D.; Gosmanov, N.R.; Welle, S.L.; Gerich, J.E. Mechanisms for abnormal postprandial glucose metabolism in type 2 diabetes. Am. J. Physiol. Endocrinol. Metab. 2006, 290, E67-e77. [CrossRef]
- Eichenlaub, M.M.; Khovanova, N.A.; Gannon, M.C.; Nuttall, F.Q.; Hattersley, J.G. A Glucose-Only Model to Extract Physiological Information from Postprandial Glucose Profiles in Subjects with Normal Glucose Tolerance. J. Diabetes Sci. Technol. 2022, 16, 1532-1540. [CrossRef]
- Wang, Y.; Fang, Y. Late non-fasting plasma glucose predicts cardiovascular mortality independent of hemoglobin A1c. Sci. Rep. 2022, 12, 7778. [CrossRef]
- Knuiman, M.W.; Welborn, T.A.; Whittall, D.E. An analysis of excess mortality rates for persons with non-insulin-dependent diabetes mellitus in Western Australia using the Cox proportional hazards regression model. Am. J. Epidemiol. 1992, 135, 638-648. [CrossRef]
- Hanefeld, M.; Fischer, S.; Julius, U.; Schulze, J.; Schwanebeck, U.; Schmechel, H.; Ziegelasch, H.J.; Lindner, J. Risk factors for myocardial infarction and death in newly detected NIDDM: the Diabetes Intervention Study, 11-year follow-up. Diabetologia 1996, 39, 1577-1583. [CrossRef]
- Takao, T.; Suka, M.; Yanagisawa, H.; Iwamoto, Y. Impact of postprandial hyperglycemia at clinic visits on the incidence of cardiovascular events and all-cause mortality in patients with type 2 diabetes. J Diabetes Investig 2017, 8, 600-608. [CrossRef]
- Takao, T.; Suka, M.; Yanagisawa, H.; Kasuga, M. Thresholds for postprandial hyperglycemia and hypertriglyceridemia associated with increased mortality risk in type 2 diabetes patients: A real-world longitudinal study. J Diabetes Investig 2021, 12, 886-893. [CrossRef]
- Takao, T.; Takahashi, K.; Suka, M.; Suzuki, N.; Yanagisawa, H. Association between postprandial hyperglycemia at clinic visits and all-cause and cancer mortality in patients with type 2 diabetes: A long-term historical cohort study in Japan. Diabetes Res. Clin. Pract. 2019, 148, 152-159. [CrossRef]
- Cavalot, F.; Pagliarino, A.; Valle, M.; Di Martino, L.; Bonomo, K.; Massucco, P.; Anfossi, G.; Trovati, M. Postprandial blood glucose predicts cardiovascular events and all-cause mortality in type 2 diabetes in a 14-year follow-up: lessons from the San Luigi Gonzaga Diabetes Study. Diabetes Care 2011, 34, 2237-2243. [CrossRef]
- Cavalot, F.; Petrelli, A.; Traversa, M.; Bonomo, K.; Fiora, E.; Conti, M.; Anfossi, G.; Costa, G.; Trovati, M. Postprandial blood glucose is a stronger predictor of cardiovascular events than fasting blood glucose in type 2 diabetes mellitus, particularly in women: lessons from the San Luigi Gonzaga Diabetes Study. J. Clin. Endocrinol. Metab. 2006, 91, 813-819. [CrossRef]
- American Diabetes Association Professional Practice Committee. 6. Glycemic Goals and Hypoglycemia: Standards of Care in Diabetes-2025. Diabetes Care 2025, 48, S128-s145. [CrossRef]
- Wang, Y.; Fang, Y.; Habenicht, A.J.R.; Golledge, J.; Giovannucci, E.L.; Ceriello, A. Postprandial Plasma Glucose With a Fasting Time of 4-7.9 h Is Positively Associated With Cancer Mortality in US Adults. Diabetes Metab. Res. Rev. 2024, 40, e70008. [CrossRef]
- Jones, K.L.; Horowitz, M.; Wishart, M.J.; Maddox, A.F.; Harding, P.E.; Chatterton, B.E. Relationships between gastric emptying, intragastric meal distribution and blood glucose concentrations in diabetes mellitus. J. Nucl. Med. 1995, 36, 2220-2228.
- Wang, Y.; Fang, Y.; Aberson, C.L.; Charchar, F.J.; Ceriello, A. Postprandial Plasma Glucose between 4 and 7.9 h May Be a Potential Diagnostic Marker for Diabetes. Biomedicines 2024, 12, 1313.
- Wang, Y. Postprandial Plasma Glucose Measured from Blood Taken between 4 and 7.9 h Is Positively Associated with Mortality from Hypertension and Cardiovascular Disease. J. Cardiovasc. Dev. Dis. 2024, 11, 53. [CrossRef]
- Mandrekar, J.N. Receiver Operating Characteristic Curve in Diagnostic Test Assessment. J. Thorac. Oncol. 2010, 5, 1315-1316. [CrossRef]
- Wang, Y.; Fang, Y. Postabsorptive homeostasis model assessment for insulin resistance is a reliable biomarker for cardiovascular disease mortality and all-cause mortality. Diabetes Epidemiology and Management 2021, 6, 100045. [CrossRef]
- Zhou, X.; Pang, Z.; Gao, W.; Wang, S.; Zhang, L.; Ning, F.; Qiao, Q. Performance of an A1C and fasting capillary blood glucose test for screening newly diagnosed diabetes and pre-diabetes defined by an oral glucose tolerance test in Qingdao, China. Diabetes Care 2010, 33, 545-550. [CrossRef]
- Pajunen, P.; Peltonen, M.; Eriksson, J.G.; Ilanne-Parikka, P.; Aunola, S.; Keinänen-Kiukaanniemi, S.; Uusitupa, M.; Tuomilehto, J.; Lindström, J. HbA(1c) in diagnosing and predicting Type 2 diabetes in impaired glucose tolerance: the Finnish Diabetes Prevention Study. Diabet. Med. 2011, 28, 36-42. [CrossRef]
- Christensen, D.L.; Witte, D.R.; Kaduka, L.; Jørgensen, M.E.; Borch-Johnsen, K.; Mohan, V.; Shaw, J.E.; Tabák, A.G.; Vistisen, D. Moving to an A1C-based diagnosis of diabetes has a different impact on prevalence in different ethnic groups. Diabetes Care 2010, 33, 580-582. [CrossRef]
- Sato, Y.; Ohfusa, H.; Katakura, M.; Komatsu, M.; Yamada, S.; Yamauchi, K.; Ichikawa, K.; Aizawa, T.; Hashizume, K. A problem with the diagnosis of diabetes mellitus based on fasting plasma glucose. Diabet. Med. 2002, 19, 82-83. [CrossRef]
- Gatling, W.; Begley, J.P. Diagnosing diabetes mellitus in clinical practice: is fasting plasma glucose a good initial test? Practical Diabetes International 2001, 18, 89-93. [CrossRef]
- Huang, J.; Ou, H.Y.; Karnchanasorn, R.; Samoa, R.; Chuang, L.M.; Chiu, K.C.; Feng, W. Clinical implication of fasting and post-challenged plasma glucose in diagnosis of diabetes mellitus. Endocrine 2015, 48, 511-518. [CrossRef]
- Tonelli, M.; Pottie, K. Diabetes guidelines. CMAJ 2013, 185, 238. [CrossRef]
- Robinson, C.A.; Sohal, P. Diabetes guidelines. CMAJ 2013, 185, 237-238. [CrossRef]
- American Diabetes Association. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes—2021. Diabetes Care 2021, 44, S15-S33. [CrossRef]
- Peter, R.; Dunseath, G.; Luzio, S.D.; Chudleigh, R.; Roy Choudhury, S.; Owens, D.R. Daytime variability of postprandial glucose tolerance and pancreatic B-cell function using 12-h profiles in persons with Type 2 diabetes. Diabet. Med. 2010, 27, 266-273. [CrossRef]
- Avignon, A.; Radauceanu, A.; Monnier, L. Nonfasting plasma glucose is a better marker of diabetic control than fasting plasma glucose in type 2 diabetes. Diabetes Care 1997, 20, 1822-1826. [CrossRef]
- Monnier, L.; Colette, C.; Dejager, S.; Owens, D. The dawn phenomenon in type 2 diabetes: how to assess it in clinical practice? Diabetes Metab. 2015, 41, 132-137. [CrossRef]
- O’Neal, T.B.; Luther, E.E. Dawn Phenomenon. In StatPearls; StatPearls Publishing: Treasure Island (FL), 2022.
- Wang, Y.; Fang, Y.; Yang, G.; Prattichizzo, F.; Ceriello, A. Postprandial 2-h glucose tolerance is associated with diabetes diagnosis, diabetes mortality, and cardiovascular mortality. Sci. Rep. 2025, 15, 43853. [CrossRef]
- Du, X.L.; Edelstein, D.; Rossetti, L.; Fantus, I.G.; Goldberg, H.; Ziyadeh, F.; Wu, J.; Brownlee, M. Hyperglycemia-induced mitochondrial superoxide overproduction activates the hexosamine pathway and induces plasminogen activator inhibitor-1 expression by increasing Sp1 glycosylation. Proc. Natl. Acad. Sci. U. S. A. 2000, 97, 12222-12226. [CrossRef]
- Brownlee, M. Biochemistry and molecular cell biology of diabetic complications. Nature 2001, 414, 813-820. [CrossRef]
- Zhou, H.; Huo, Y.; Yang, N.; Taotao, W. Phosphatidic acid: from biophysical properties to diverse functions. The FEBS Journal 2023, 291, 1870-1885. [CrossRef]
- Meier, M.; King, G. Protein kinase C activation its pharmacological inhibition in vascular disease. Vascular medicine (London, England) 2000, 5, 173-185. [CrossRef]
- Wang, L.; Yang, T.; Pan, Y.; Shi, L.; Jin, Y.; Huang, X. The Metabolism of Reactive Oxygen Species and Their Effects on Lipid Biosynthesis of Microalgae. Int. J. Mol. Sci. 2023, 24, 11041.
- Gomez-Larrauri, A.; Benito-Vicente, A.; Uribe, K.B.; Martin, C.; Gomez-Muñoz, A. Regulation of Cell Proliferation and Migration by Extracellular Phosphatidic Acid. Biomedicines 2026, 14, 616.
- Bond, P. Phosphatidic acid: biosynthesis, pharmacokinetics, mechanisms of action and effect on strength and body composition in resistance-trained individuals. Nutr. Metab. (Lond.) 2017, 14, 12. [CrossRef]
- Khare, K.; Mendonca, T.; Rodrigues, G.; Kamath, M.; Hegde, A.; Nayak, S.; Kamath, A.; Kamath, S. Aldose reductase and glutathione in senile cataract nucleus of diabetics and non-diabetics. Int. Ophthalmol. 2023, 43, 3673-3680. [CrossRef]
- Santos, H.O.; Penha-Silva, N. Translating the advanced glycation end products (AGEs) knowledge into real-world nutrition strategies. Eur. J. Clin. Nutr. 2022, 76, 922-928. [CrossRef]
- Ramasamy, R.; Goldberg, I.J. Aldose Reductase and Cardiovascular Diseases, Creating Human-Like Diabetic Complications in an Experimental Model. Circ. Res. 2010, 106, 1449-1458. [CrossRef]
- Thornalley, P.J. The glyoxalase system: new developments towards functional characterization of a metabolic pathway fundamental to biological life. Biochem. J. 1990, 269, 1-11. [CrossRef]
- Li, Y.; Peng, Y.; Shen, Y.; Zhang, Y.; Liu, L.; Yang, X. Dietary polyphenols: regulate the advanced glycation end products-RAGE axis and the microbiota-gut-brain axis to prevent neurodegenerative diseases. Crit. Rev. Food Sci. Nutr. 2022, 63, 1-27. [CrossRef]
- Vašková, J.; Kováčová, G.; Pudelský, J.; Palenčár, D.; Mičková, H. Methylglyoxal Formation—Metabolic Routes and Consequences. Antioxidants 2025, 14, 212.
- Yanagi, K.; Komatsu, T.; Fujikawa, Y.; Kojima, H.; Okabe, T.; Nagano, T.; Ueno, T.; Hanaoka, K.; Urano, Y. Development of pathway-oriented screening to identify compounds to control 2-methylglyoxal metabolism in tumor cells. Communications Chemistry 2023, 6, 68. [CrossRef]
- Raimi, O.; Hurtado-Guerrero, R.; Borodkin, V.; Ferenbach, A.; Urbaniak, M.; Ferguson, M.; van Aalten, D. A mechanism-inspired UDP- N -acetylglucosamine pyrophosphorylase inhibitor. RSC Chemical Biology 2020, 1. [CrossRef]
- Dong, H.; Liu, Z.; Wen, H. Protein O-GlcNAcylation Regulates Innate Immune Cell Function. Front. Immunol. 2022, 13, 805018. [CrossRef]
- Caturano, A.; D’Angelo, M.; Mormone, A.; Russo, V.; Mollica, M.P.; Salvatore, T.; Galiero, R.; Rinaldi, L.; Vetrano, E.; Marfella, R.; et al. Oxidative Stress in Type 2 Diabetes: Impacts from Pathogenesis to Lifestyle Modifications. Curr. Issues Mol. Biol. 2023, 45, 6651-6666. [CrossRef]
- Zhang, Z.; Liew, C.W.; Handy, D.E.; Zhang, Y.; Leopold, J.A.; Hu, J.; Guo, L.; Kulkarni, R.N.; Loscalzo, J.; Stanton, R.C. High glucose inhibits glucose-6-phosphate dehydrogenase, leading to increased oxidative stress and beta-cell apoptosis. FASEB J. 2010, 24, 1497-1505. [CrossRef]
- Wang, Y. Triglycerides, Glucose Metabolism, and Type 2 Diabetes. Int. J. Mol. Sci. 2025, 26, 9910. [CrossRef]
- Goycheva, P.; Petkova-Parlapanska, K.; Georgieva, E.; Karamalakova, Y.; Nikolova, G. Biomarkers of Oxidative Stress in Diabetes Mellitus with Diabetic Nephropathy Complications. Int. J. Mol. Sci. 2023, 24, 13541. [CrossRef]
- Wells-Knecht, K.J.; Zyzak, D.V.; Litchfield, J.E.; Thorpe, S.R.; Baynes, J.W. Mechanism of autoxidative glycosylation: identification of glyoxal and arabinose as intermediates in the autoxidative modification of proteins by glucose. Biochemistry 1995, 34, 3702-3709. [CrossRef]
- Aoyama, K. Glutathione in the Brain. Int. J. Mol. Sci. 2021, 22, 5010. [CrossRef]
- Labarrere, C.A.; Kassab, G.S. Glutathione: A Samsonian life-sustaining small molecule that protects against oxidative stress, ageing and damaging inflammation. Frontiers in Nutrition 2022, 9, 1007816. [CrossRef]
- Ge, T.; Yang, J.; Zhou, S.; Wang, Y.; Li, Y.; Tong, X. The Role of the Pentose Phosphate Pathway in Diabetes and Cancer. Front. Endocrinol. (Lausanne) 2020, 11, 365. [CrossRef]
- Gansemer, E.R.; McCommis, K.S.; Martino, M.; King-McAlpin, A.Q.; Potthoff, M.J.; Finck, B.N.; Taylor, E.B.; Rutkowski, D.T. NADPH and Glutathione Redox Link TCA Cycle Activity to Endoplasmic Reticulum Homeostasis. iScience 2020, 23, 101116. [CrossRef]
- Silvestrini, A.; Meucci, E.; Ricerca, B.M.; Mancini, A. Total Antioxidant Capacity: Biochemical Aspects and Clinical Significance. Int. J. Mol. Sci. 2023, 24. [CrossRef]
- Beisswenger, P.J.; Brown, W.V.; Ceriello, A.; Le, N.A.; Goldberg, R.B.; Cooke, J.P.; Robbins, D.C.; Sarwat, S.; Yuan, H.; Jones, C.A.; et al. Meal-induced increases in C-reactive protein, interleukin-6 and tumour necrosis factor α are attenuated by prandial + basal insulin in patients with Type 2 diabetes. Diabet. Med. 2011, 28, 1088-1095. [CrossRef]
- Ridker, P.M. High-sensitivity C-reactive protein, inflammation, and cardiovascular risk: from concept to clinical practice to clinical benefit. Am. Heart J. 2004, 148, S19-26. [CrossRef]
- Clark, R.A.; Valente, A.J. Nuclear factor kappa B activation by NADPH oxidases. Mech. Ageing Dev. 2004, 125, 799-810. [CrossRef]
- El-Osta, A.; Brasacchio, D.; Yao, D.; Pocai, A.; Jones, P.L.; Roeder, R.G.; Cooper, M.E.; Brownlee, M. Transient high glucose causes persistent epigenetic changes and altered gene expression during subsequent normoglycemia. J. Exp. Med. 2008, 205, 2409-2417. [CrossRef]
- Ceriello, A.; Motz, E. Is oxidative stress the pathogenic mechanism underlying insulin resistance, diabetes, and cardiovascular disease? The common soil hypothesis revisited. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 816-823. [CrossRef]
- Node, K.; Inoue, T. Postprandial hyperglycemia as an etiological factor in vascular failure. Cardiovasc. Diabetol. 2009, 8, 23. [CrossRef]
- Turina, M.; Fry, D.E.; Polk, H.C., Jr. Acute hyperglycemia and the innate immune system: Clinical, cellular, and molecular aspects. Crit. Care Med. 2005, 33, 1624-1633. [CrossRef]
- Sasso, E.; Baticic, L.; Sotosek, V. Postprandial Dysmetabolism and Its Medical Implications. Life (Basel) 2023, 13, 2317. [CrossRef]
- Esposito, K.; Nappo, F.; Marfella, R.; Giugliano, G.; Giugliano, F.; Ciotola, M.; Quagliaro, L.; Ceriello, A.; Giugliano, D. Inflammatory cytokine concentrations are acutely increased by hyperglycemia in humans: role of oxidative stress. Circulation 2002, 106, 2067-2072. [CrossRef]
- Noh, H.; King, G.L. The role of protein kinase C activation in diabetic nephropathy. Kidney Int. 2007, 72, S49-S53. [CrossRef]
- Zhang, M.; Zhou, M.; Cai, X.; Zhou, Y.; Jiang, X.; Luo, Y.; Hu, Y.; Qiu, R.; Wu, Y.; Zhang, Y.; et al. VEGF promotes diabetic retinopathy by upregulating the PKC/ET/NF-κB/ICAM-1 signaling pathway. Eur. J. Histochem. 2022, 66, 3522. [CrossRef]
- Huang, X.; He, D.; Ming, J.; He, Y.; Zhou, C.; Ren, H.; He, X.; Wang, C.; Jin, J.; Ji, L.; et al. High-density lipoprotein of patients with breast cancer complicated with type 2 diabetes mellitus promotes cancer cells adhesion to vascular endothelium via ICAM-1 and VCAM-1 upregulation. Breast Cancer Res. Treat. 2016, 155, 441-455. [CrossRef]
- Chuah, Y.K.; Basir, R.; Talib, H.; Tie, T.H.; Nordin, N. Receptor for advanced glycation end products and its involvement in inflammatory diseases. Int J Inflam 2013, 2013, 403460. [CrossRef]
- Diwadkar, V.A.; Anderson, J.W.; Bridges, S.R.; Gowri, M.S.; Oelgten, P.R. Postprandial low-density lipoproteins in type 2 diabetes are oxidized more extensively than fasting diabetes and control samples. Proc. Soc. Exp. Biol. Med. 1999, 222, 178-184. [CrossRef]
- Williams, S.B.; Goldfine, A.B.; Timimi, F.K.; Ting, H.H.; Roddy, M.-A.; Simonson, D.C.; Creager, M.A. Acute Hyperglycemia Attenuates Endothelium-Dependent Vasodilation in Humans In Vivo. Circulation 1998, 97, 1695-1701. [CrossRef]
- Kawano, H.; Motoyama, T.; Hirashima, O.; Hirai, N.; Miyao, Y.; Sakamoto, T.; Kugiyama, K.; Ogawa, H.; Yasue, H. Hyperglycemia rapidly suppresses flow-mediated endothelium-dependent vasodilation of brachial artery. J. Am. Coll. Cardiol. 1999, 34, 146-154. [CrossRef]
- Tesfamariam, B. Free radicals in diabetic endothelial cell dysfunction. Free Radic. Biol. Med. 1994, 16, 383-391. [CrossRef]
- Tesfamariam, B.; Cohen, R.A. Free radicals mediate endothelial cell dysfunction caused by elevated glucose. Am. J. Physiol. 1992, 263, H321-326. [CrossRef]
- Giugliano, D.; Marfella, R.; Coppola, L.; Verrazzo, G.; Acampora, R.; Giunta, R.; Nappo, F.; Lucarelli, C.; D’Onofrio, F. Vascular effects of acute hyperglycemia in humans are reversed by L-arginine. Evidence for reduced availability of nitric oxide during hyperglycemia. Circulation 1997, 95, 1783-1790. [CrossRef]
- Liu, Z.; Lu, J.; Sha, W.; Lei, T. Comprehensive treatment of diabetic endothelial dysfunction based on pathophysiological mechanism. Front Med (Lausanne) 2025, 12, 1509884. [CrossRef]
- Ceriello, A.; Bortolotti, N.; Motz, E.; Pieri, C.; Marra, M.; Tonutti, L.; Lizzio, S.; Feletto, F.; Catone, B.; Taboga, C. Meal-induced oxidative stress and low-density lipoprotein oxidation in diabetes: the possible role of hyperglycemia. Metabolism 1999, 48, 1503-1508. [CrossRef]
- Ceriello, A. Postprandial hyperglycemia and diabetes complications: is it time to treat? Diabetes 2005, 54, 1-7. [CrossRef]
- Hong, C.G.; Florida, E.; Li, H.; Parel, P.M.; Mehta, N.N.; Sorokin, A.V. Oxidized low-density lipoprotein associates with cardiovascular disease by a vicious cycle of atherosclerosis and inflammation: A systematic review and meta-analysis. Frontiers in Cardiovascular Medicine 2023, 9, 1023651. [CrossRef]
- Goldin, A.; Beckman, J.A.; Schmidt, A.M.; Creager, M.A. Advanced Glycation End Products. Circulation 2006, 114, 597-605. [CrossRef]
- Temelkova-Kurktschiev, T.S.; Koehler, C.; Henkel, E.; Leonhardt, W.; Fuecker, K.; Hanefeld, M. Postchallenge plasma glucose and glycemic spikes are more strongly associated with atherosclerosis than fasting glucose or HbA1c level. Diabetes Care 2000, 23, 1830-1834. [CrossRef]
- Li, X.; Weber, N.C.; Cohn, D.M.; Hollmann, M.W.; DeVries, J.H.; Hermanides, J.; Preckel, B. Effects of Hyperglycemia and Diabetes Mellitus on Coagulation and Hemostasis. J Clin Med 2021, 10, 2419. [CrossRef]
- Cignarelli, A.; Genchi, V.A.; Caruso, I.; Natalicchio, A.; Perrini, S.; Laviola, L.; Giorgino, F. Diabetes and cancer: Pathophysiological fundamentals of a ‘dangerous affair’. Diabetes Res. Clin. Pract. 2018, 143, 378-388. [CrossRef]
- Duan, W.; Shen, X.; Lei, J.; Xu, Q.; Yu, Y.; Li, R.; Wu, E.; Ma, Q. Hyperglycemia, a neglected factor during cancer progression. Biomed Res Int 2014, 2014, 461917. [CrossRef]
- Pascual-Morena, C.; Garrido-Miguel, M.; Martínez-García, I.; Lucerón-Lucas-Torres, M.; Rodríguez-Gutiérrez, E.; Berlanga-Macías, C.; Fernández-Bravo-Rodrigo, J.; Patiño-Cardona, S. Association of Dietary Advanced Glycation End Products with Overall and Site-Specific Cancer Risk and Mortality: A Systematic Review and Meta-Analysis. Nutrients 2025, 17, 1638.
- Wautier, M.P.; Chappey, O.; Corda, S.; Stern, D.M.; Schmidt, A.M.; Wautier, J.L. Activation of NADPH oxidase by AGE links oxidant stress to altered gene expression via RAGE. Am. J. Physiol. Endocrinol. Metab. 2001, 280, E685-694. [CrossRef]
- Schröter, D.; Höhn, A. Role of Advanced Glycation End Products in Carcinogenesis and their Therapeutic Implications. Curr. Pharm. Des. 2018, 24, 5245-5251. [CrossRef]
- Turner, D.P. The Role of Advanced Glycation End-Products in Cancer Disparity. Adv. Cancer Res. 2017, 133, 1-22. [CrossRef]
- Wautier, J.L.; Wautier, M.P. Cellular and Molecular Aspects of Blood Cell-Endothelium Interactions in Vascular Disorders. Int. J. Mol. Sci. 2020, 21, 5315. [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.-C. NF-κB signaling in inflammation. Signal Transduction and Targeted Therapy 2017, 2, 17023. [CrossRef]
- Zhang, Y.-Y.; Li, Y.-J.; Xue, C.-D.; Li, S.; Gao, Z.-N.; Qin, K.-R. Effects of T2DM on cancer progression: pivotal precipitating factors and underlying mechanisms. Front. Endocrinol. (Lausanne) 2024, 15, 1396022. [CrossRef]
- Jiang, M.; Zhang, K.; Zhang, Z.; Zeng, X.; Huang, Z.; Qin, P.; Xie, Z.; Cai, X.; Ashrafizadeh, M.; Tian, Y.; et al. PI3K/AKT/mTOR Axis in Cancer: From Pathogenesis to Treatment. MedComm (2020) 2025, 6, e70295. [CrossRef]
- Ray, A.; Alalem, M.; Ray, B.K. Insulin signaling network in cancer. Indian J. Biochem. Biophys. 2014, 51, 493-498.
- Peng, Y.; Wang, Y.; Zhou, C.; Mei, W.; Zeng, C. PI3K/Akt/mTOR Pathway and Its Role in Cancer Therapeutics: Are We Making Headway? Front. Oncol. 2022, 12, 819128. [CrossRef]
- Sarkar, P.L.; Lee, W.; Williams, E.D.; Lubik, A.A.; Stylianou, N.; Shokoohmand, A.; Lehman, M.L.; Hollier, B.G.; Gunter, J.H.; Nelson, C.C. Insulin Enhances Migration and Invasion in Prostate Cancer Cells by Up-Regulation of FOXC2. Front. Endocrinol. (Lausanne) 2019, 10, 481. [CrossRef]
- Cai, W.; Ma, Y.; Song, L.; Cao, N.; Gao, J.; Zhou, S.; Tang, X. IGF-1R down regulates the sensitivity of hepatocellular carcinoma to sorafenib through the PI3K / akt and RAS / raf / ERK signaling pathways. BMC Cancer 2023, 23, 87. [CrossRef]
- Wang, Y.; Liu, H.; Ma, S.; Qian, T.; Sun, H.; Xu, Q.; Hou, X.; Hu, W.; Zhang, G.; Jelinic, M.; et al. Normal triglyceride levels are positively associated with plasma glucose levels and type 2 diabetes diagnosis in Chinese adults. Explor Endocr Metab Dis 2026, 3, 101459. [CrossRef]
- Wang, Y. Higher fasting triglyceride predicts higher risks of diabetes mortality in US adults. Lipids Health Dis. 2021, 20, 181. [CrossRef]
- Wang, Y.; Fang, Y.; Magliano, D.J.; Charchar, F.J.; Sobey, C.G.; Drummond, G.R.; Golledge, J. Fasting triglycerides are positively associated with cardiovascular mortality risk in people with diabetes. Cardiovasc. Res. 2023, 119, 826–834. [CrossRef]
- Wang, Y.; Magliano, D.J. Special Issue: “New Trends in Diabetes, Hypertension, and Cardiovascular Diseases”. Int. J. Mol. Sci. 2024, 25, 2711.
- Wang, Y.; Magliano, D.J. Special Issue: “New Trends in Diabetes, Hypertension, and Cardiovascular Diseases—2nd Edition”. Int. J. Mol. Sci. 2025, 26, 449.
- Wang, Y.; Magliano, D.J. Special Issue “New Trends in Diabetes, Hypertension and Cardiovascular Diseases: 3rd Edition”. Int. J. Mol. Sci. 2025, 26, 10536.
- Wang, Y.; Fang, Y.; Zhang, X.; Wu, N.-Q. Non-Fasting Plasma Triglycerides Are Positively Associated with Diabetes Mortality in a Representative US Adult Population. Targets 2024, 2, 93-103. [CrossRef]

| Mortality | Multivariate adjusted, without adjustment for HbA1c |
Multivariate adjusted, with further adjustment for HbA1c |
||||
| Hazard ratio | 95% CI | p | Hazard ratio | 95% CI | p | |
| 2-h PGOGTT@4–7.9h | ||||||
| All-cause mortality | 1.06 | 1.04–1.08 | <0.001 | 1.04 | 1.02–1.07 | 0.003 |
| Diabetes mortality | 1.46 | 1.33–1.61 | <0.001 | 1.39 | 1.17–1.65 | <0.001 |
| CVD mortality | 1.07 | 1.03–1.11 | <0.001 | 1.03 | 0.98–1.08 | 0.26 |
| Cancer mortality | 1.00 | 0.95–1.05 | 1.00 | 1.02 | 0.96–1.07 | 0.58 |
| 2-h PG during OGTT after fasting | ||||||
| All-cause mortality | 1.06 | 1.04–1.07 | <0.001 | 1.04 | 1.02–1.06 | <0.001 |
| Diabetes mortality | 1.29 | 1.21–1.38 | <0.001 | 1.26 | 1.12–1.43 | <0.001 |
| CVD mortality | 1.06 | 1.03–1.09 | <0.001 | 1.01 | 0.97–1.04 | 0.77 |
| Cancer mortality | 1.00 | 0.97–1.04 | 0.80 | 1.00 | 0.96–1.05 | 0.85 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




