Submitted:
09 April 2026
Posted:
10 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
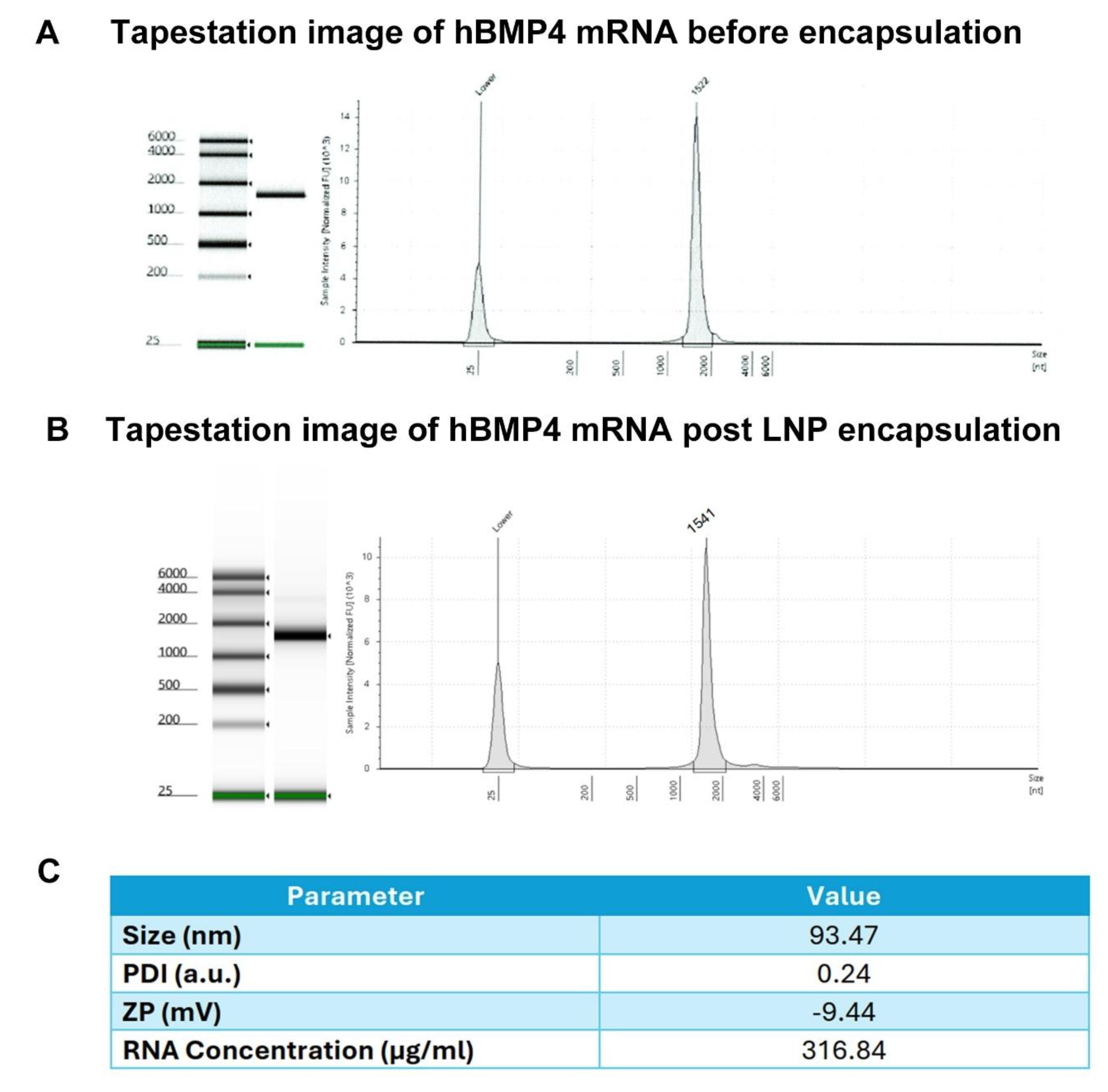
2.1. Human BMP4 (hBMP4) mRNA Preparation and Encapsulation
2.2. Human BMP4 mRNA for Critical-Sized Calvarial Defect Healing Using Fibrin Sealant as Scaffold
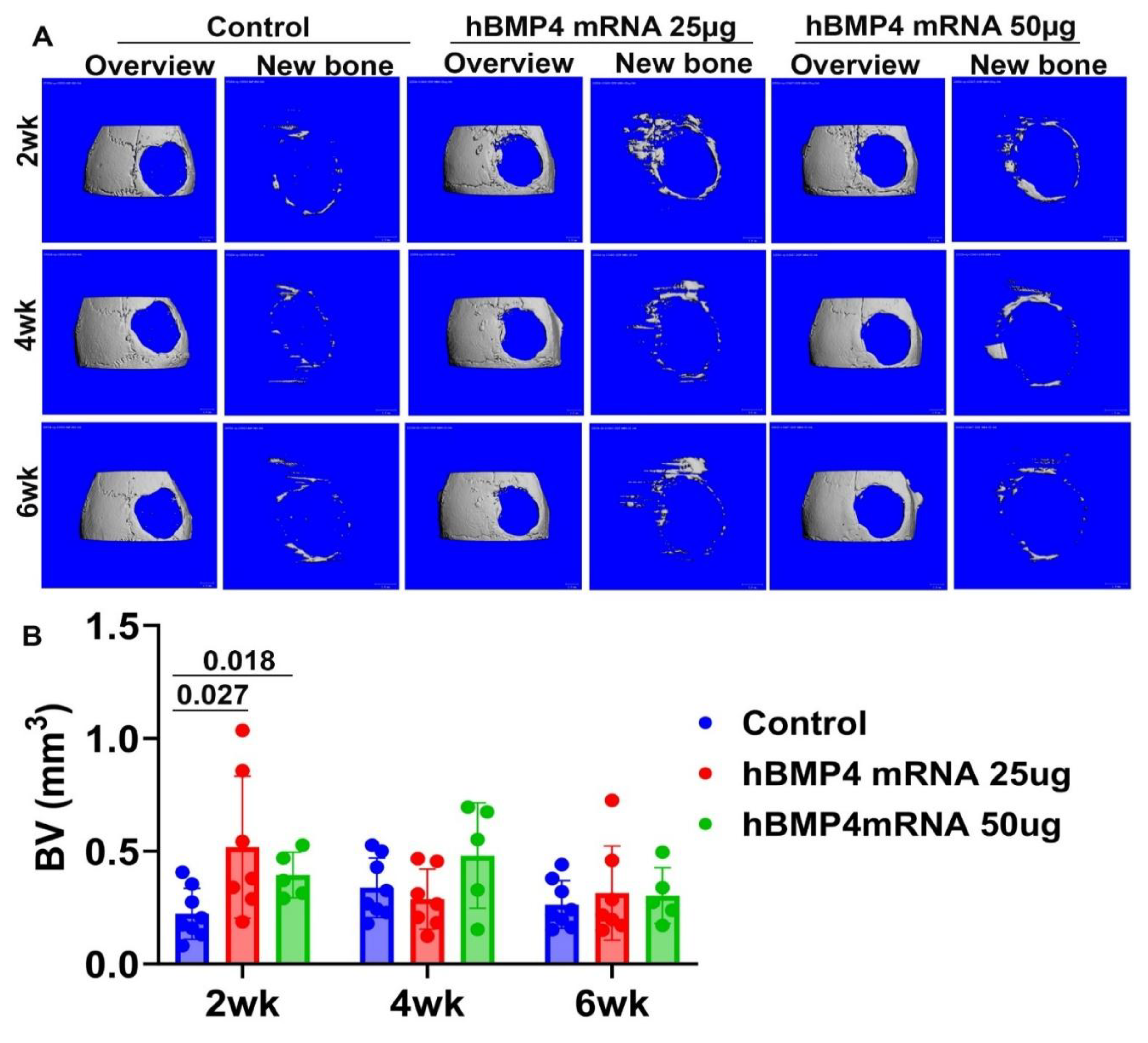
2.2.1. Live Micro-CT Scan to Monitor Bone Regeneration
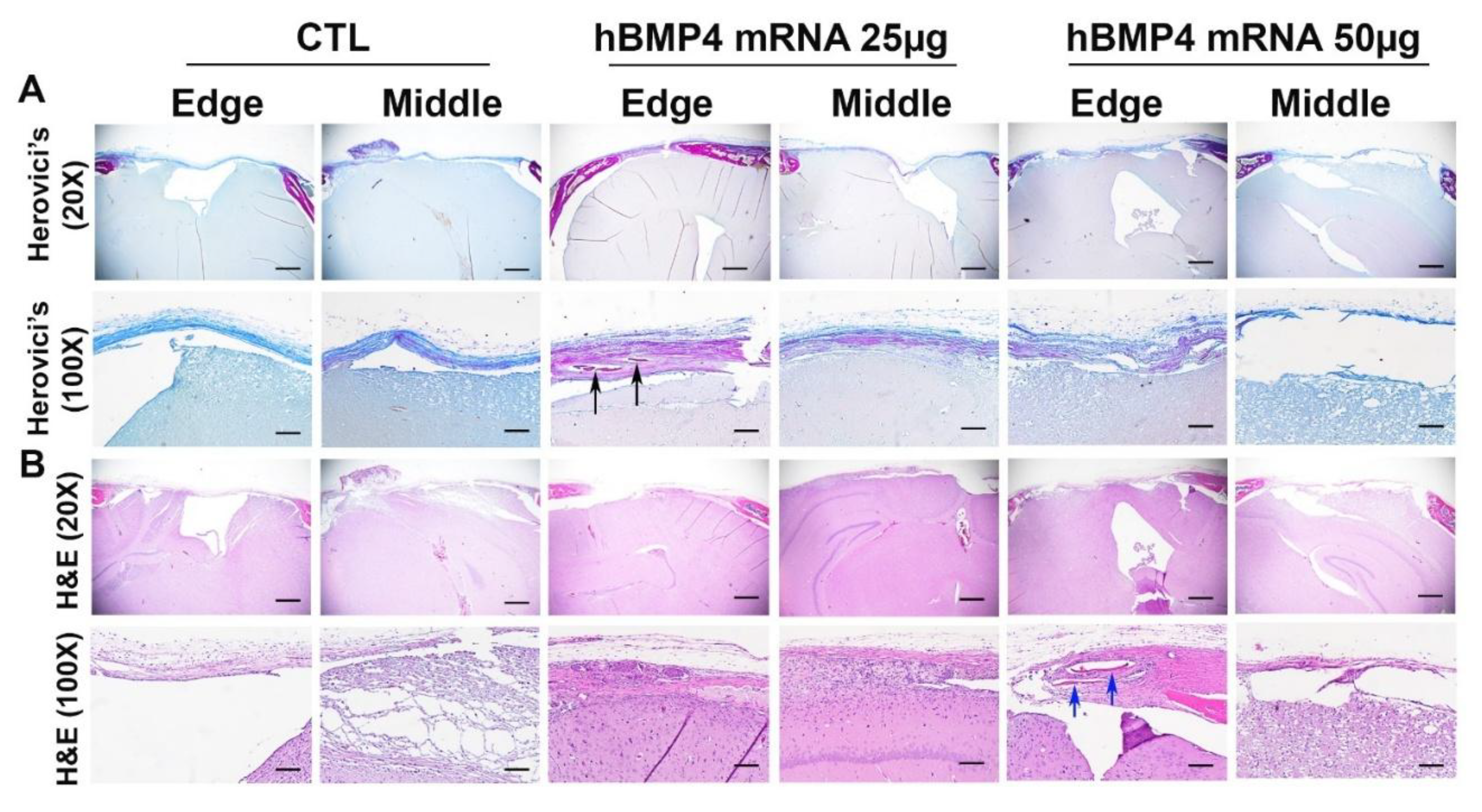
2.2.2. Bone Histology
2.3. Human BMP4 mRNA for the Treatment of Natural Aged Mice Osteoarthritis
2.3.1. Human BMP4 mRNA/LNP Intra-Articular Injection
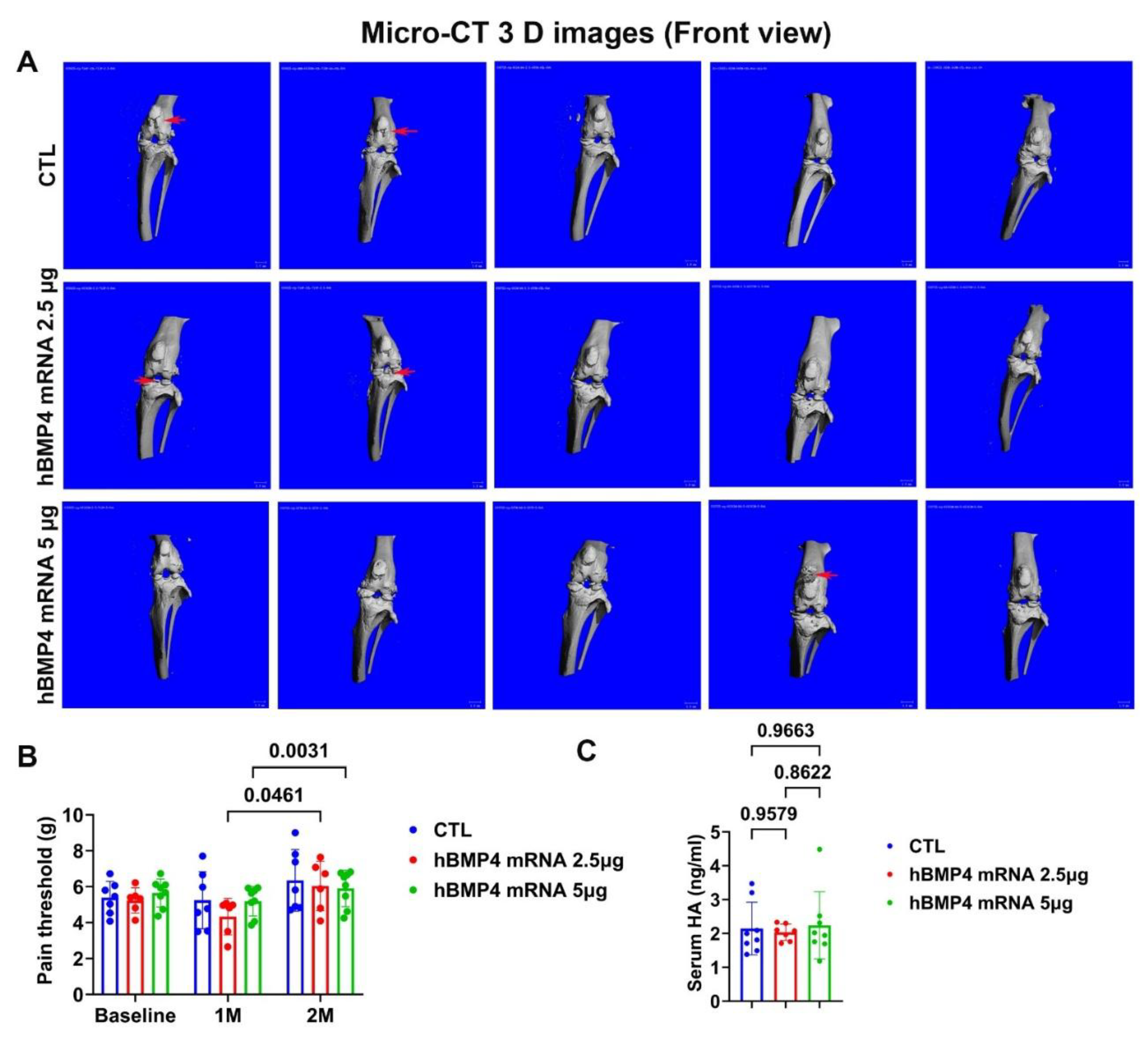
2.3.2. Pain Measurement
2.3.4. Micro-CT Scan and Analysis for Knee Joint
2.3.5. Histology
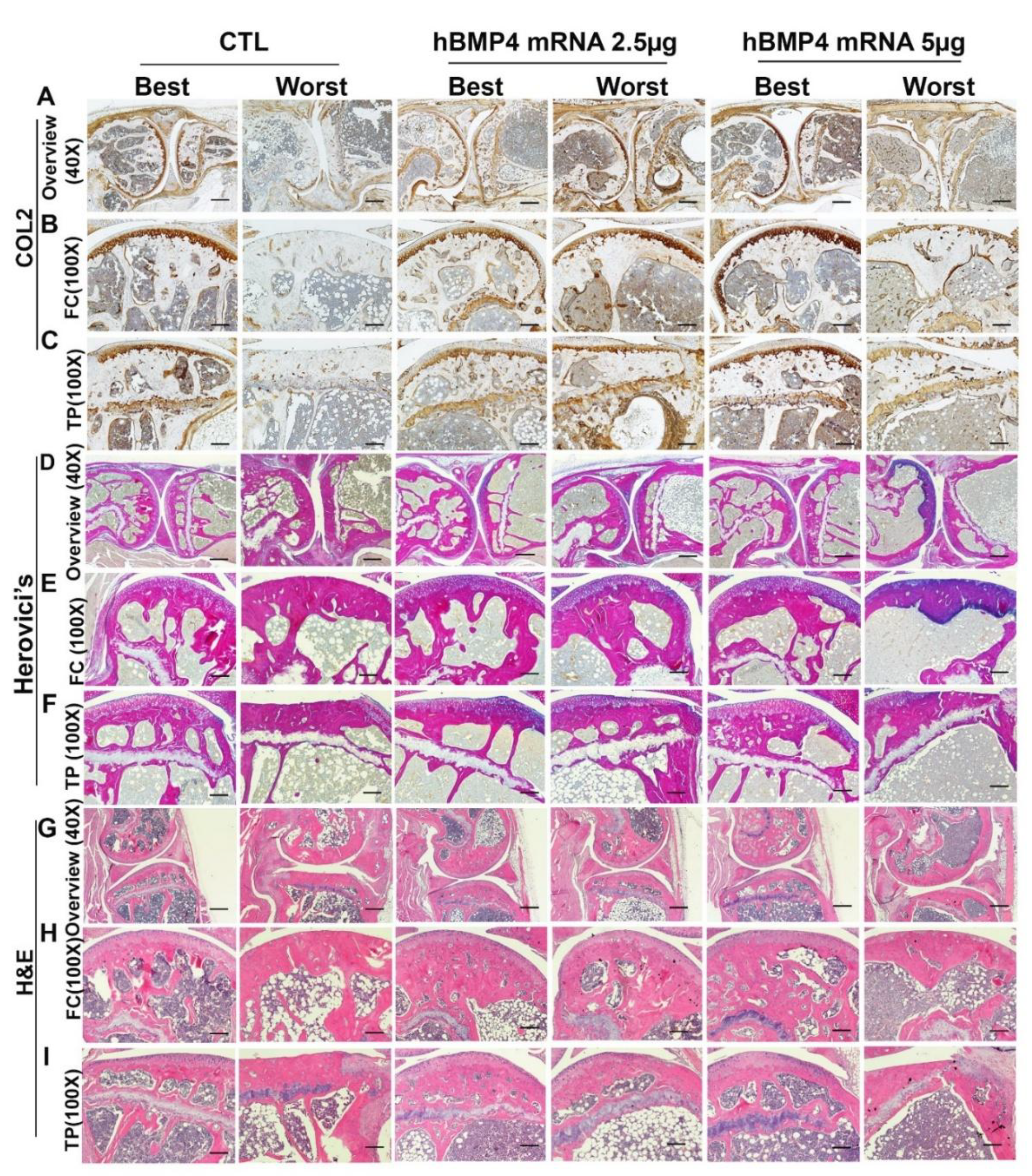
2.3.6. Immunohistochemistry Staining of COL2
2.3.7. Serum Cartilage Damage Marker Enzyme-Linked Immunosorbent Assay (ELISA)
2.4. Statistical Analysis
3. Results
3.1. HBMP4 mRNA In Vitro Transcription and Encapsulation Verification
3.2. HBMP4 mRNA Promoted Limited Bone Regeneration in a Critical-Sized Calvarial Bone Defect in Aged Mice
3.3. Histology Revealed Minimal New Bone Formation Mediated by hBMP4 mRNA/LNP in Critical-Sized Calvarial Bone Defect When Delivered with Fibrin Sealant
3.4. Micro-CT Results Revealed No Increase in Heterotopic Bone Formation in the Knee Joint at 8 weeks After Intra-Articular Injection of BMP4 mRNA
3.5. hBMP4 mRNA Intra-Articular Injection Appeared to Alleviate Pain Threshold Measured by Von-Frey Device
3.6. Intra-Articular Injection of hBMP4 mRNA Did not Significantly Change Serum Cartilage Damage Marker Hyaluronic Acid (HA)
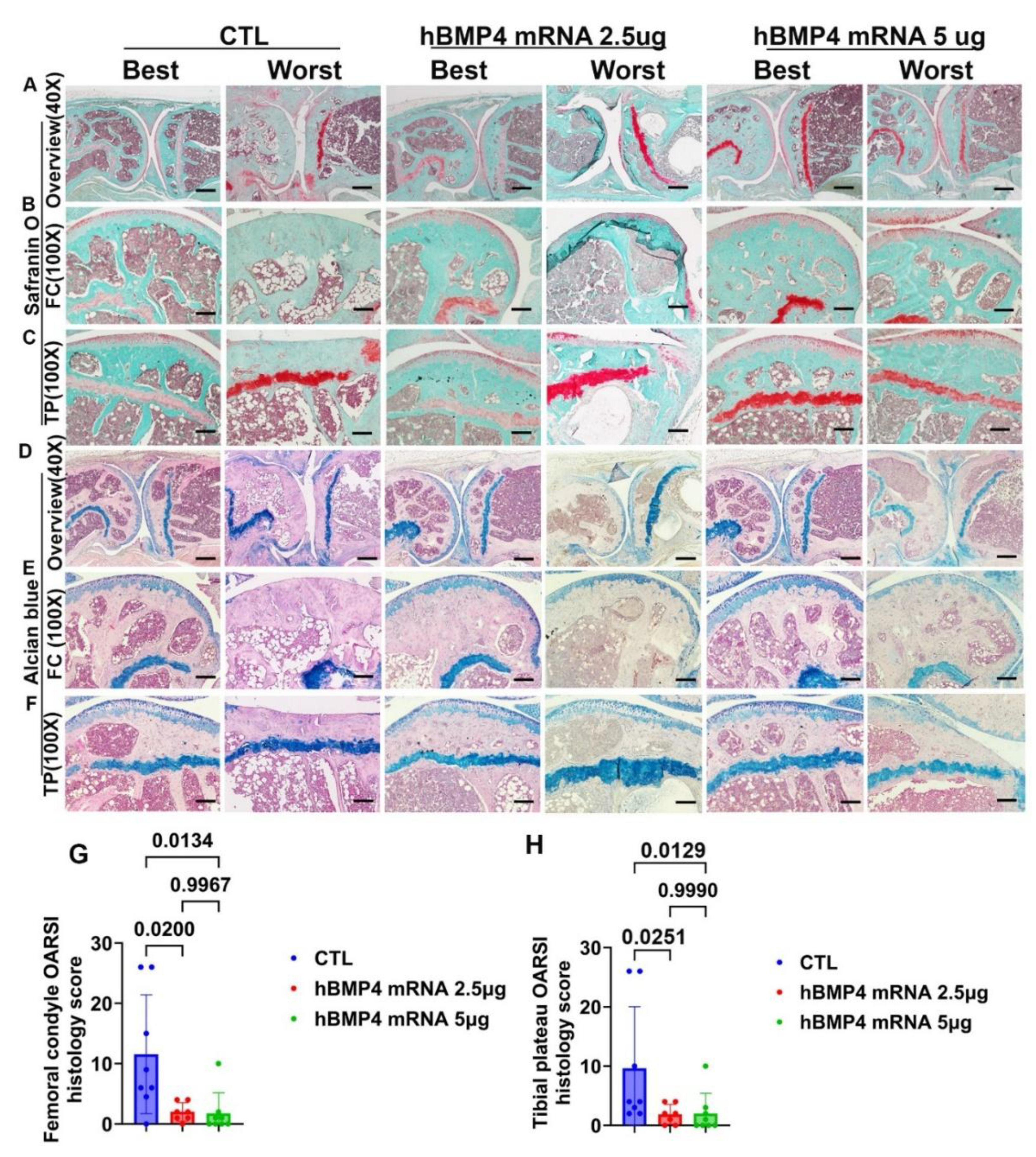
3.7. hBMP4mRNA/LNP Intra-Articular Injection Prevented Age-related Cartilage Loss and Improved Histology Score
3.8. hBMP4mRNA/LNP Intra-Articular Injection Maintained Cartilage Matrix During Aging
4. Discussion
5. Conclusions
Author Contributions
Funding
Data availability statement
Acknowledgments
Conflicts of Interest
References
- Hunter, D. J.; Bierma-Zeinstra, S. Osteoarthritis. Lancet 2019, 393((10182)), 1745–1759. [Google Scholar] [CrossRef]
- Rashidi, A.; Bastan, M. M.; Golestani, A.; Azadnajafabad, S.; Rezaei, N.; Zeinodinimeymand, M.; Busehail, M.; Yaqoob, M.; Heidari-Foroozan, M.; Rashidi, M. M. Burden of osteoarthritis in North Africa and Middle East from 1990 to 2021: findings from the Global Burden of Disease 2021 study. Clin Rheumatol 2025. [Google Scholar] [CrossRef]
- Kempny, T.; Holoubek, J.; Polovko, J.; Sedivy, O.; Votruba, T.; Kachlik, D.; Pilny, J. Reconstruction of large post-traumatic segmental femoral defects using vascularised bone flaps: a retrospective case series. BMC Musculoskelet Disord 2024, 25((1)), 919. [Google Scholar] [CrossRef]
- Tsang, S. J.; van Rensburg, A. J.; van Heerden, J.; Epstein, G. Z.; Venter, R.; Ferreira, N. The management of critical bone defects: outcomes of a systematic approach. Eur J Orthop Surg Traumatol 2024, 34((6)), 3225–3231. [Google Scholar] [CrossRef]
- Shen, J.; Wei, Z.; Wu, H.; Wang, X.; Wang, S.; Wang, G.; Luo, F.; Xie, Z. The induced membrane technique for the management of infected segmental bone defects. Bone Joint J 2024, 106-B((6)), 613–622. [Google Scholar] [CrossRef] [PubMed]
- Ying, Q. L.; Nichols, J.; Chambers, I.; Smith, A. BMP induction of Id proteins suppresses differentiation and sustains embryonic stem cell self-renewal in collaboration with STAT3. Cell 2003, 115((3)), 281–92. [Google Scholar] [CrossRef] [PubMed]
- Kirilly, D.; Spana, E. P.; Perrimon, N.; Padgett, R. W.; Xie, T. BMP signaling is required for controlling somatic stem cell self-renewal in the Drosophila ovary. Dev Cell 2005, 9((5)), 651–62. [Google Scholar] [CrossRef] [PubMed]
- Samavarchi-Tehrani, P.; Golipour, A.; David, L.; Sung, H. K.; Beyer, T. A.; Datti, A.; Woltjen, K.; Nagy, A.; Wrana, J. L. Functional genomics reveals a BMP-driven mesenchymal-to-epithelial transition in the initiation of somatic cell reprogramming. Cell Stem Cell 2010, 7((1)), 64–77. [Google Scholar] [CrossRef]
- Chen, J.; Liu, J.; Yang, J.; Chen, Y.; Chen, J.; Ni, S.; Song, H.; Zeng, L.; Ding, K.; Pei, D. BMPs functionally replace Klf4 and support efficient reprogramming of mouse fibroblasts by Oct4 alone. Cell Res 2011, 21((1)), 205–12. [Google Scholar] [CrossRef]
- Hayashi, Y.; Hsiao, E. C.; Sami, S.; Lancero, M.; Schlieve, C. R.; Nguyen, T.; Yano, K.; Nagahashi, A.; Ikeya, M.; Matsumoto, Y.; Nishimura, K.; Fukuda, A.; Hisatake, K.; Tomoda, K.; Asaka, I.; Toguchida, J.; Conklin, B. R.; Yamanaka, S. BMP-SMAD-ID promotes reprogramming to pluripotency by inhibiting p16/INK4A-dependent senescence. Proc Natl Acad Sci U S A 2016, 113((46)), 13057–13062. [Google Scholar] [CrossRef]
- Baboota, R. K.; Rawshani, A.; Bonnet, L.; Li, X.; Yang, H.; Mardinoglu, A.; Tchkonia, T.; Kirkland, J. L.; Hoffmann, A.; Dietrich, A.; Boucher, J.; Bluher, M.; Smith, U. BMP4 and Gremlin 1 regulate hepatic cell senescence during clinical progression of NAFLD/NASH. Nat Metab 2022, 4((8)), 1007–1021. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Cooper, G. M.; Gharaibeh, B.; Meszaros, L. B.; Li, G.; Usas, A.; Fu, F. H.; Huard, J. Cartilage repair in a rat model of osteoarthritis through intraarticular transplantation of muscle-derived stem cells expressing bone morphogenetic protein 4 and soluble Flt-1. Arthritis Rheum 2009, 60((5)), 1390–405. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Wright, N.; Huard, M.; Tan, J.; Ruzbarsky, J.; Lu, A.; Chubb, L.; Tuan, R.; Philippon, M. J.; Wang, Y.; Huard, J. Comparison of 5 BMPs for their chondrogenic potentials and microfracture-mediated cartilage repair using heparin/PEAD coacervate sustained release polymer. Bioact Mater 2025, 52, 588–603. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.; Gao, X.; Huard, M.; Lu, A.; Ruzbarsky, J. J.; Amra, S.; Wang, B.; Huard, J. Bone morphogenetic protein 4 rescues the bone regenerative potential of old muscle-derived stem cells via regulation of cell cycle inhibitors. Stem Cell Res Ther 2022, 13((1)), 385. [Google Scholar] [CrossRef]
- De La Vega, R. E.; van Griensven, M.; Zhang, W.; Coenen, M. J.; Nagelli, C. V.; Panos, J. A.; Peniche Silva, C. J.; Geiger, J.; Plank, C.; Evans, C. H.; Balmayor, E. R. Efficient healing of large osseous segmental defects using optimized chemically modified messenger RNA encoding BMP-2. Sci Adv 2022, 8((7)), eabl6242. [Google Scholar] [CrossRef]
- Khorsand, B.; Elangovan, S.; Hong, L.; Kormann, M. S. D.; Salem, A. K. A bioactive collagen membrane that enhances bone regeneration. J Biomed Mater Res B Appl Biomater 2019, 107((6)), 1824–1832. [Google Scholar] [CrossRef]
- Nelson, A. L.; Mancino, C.; Gao, X.; Choe, J. A.; Chubb, L.; Williams, K.; Czachor, M.; Marcucio, R.; Taraballi, F.; Cooke, J. P.; Huard, J.; Bahney, C.; Ehrhart, N. beta-catenin mRNA encapsulated in SM-102 lipid nanoparticles enhances bone formation in a murine tibia fracture repair model. Bioact Mater 2024, 39, 273–286. [Google Scholar]
- Gao, X.; Hwang, M. P.; Wright, N.; Lu, A.; Ruzbarsky, J. J.; Huard, M.; Cheng, H.; Mullen, M.; Ravuri, S.; Wang, B.; Wang, Y.; Huard, J. The use of heparin/polycation coacervate sustain release system to compare the bone regenerative potentials of 5 BMPs using a critical sized calvarial bone defect model. Biomaterials 2022, 288, 121708. [Google Scholar] [CrossRef]
- Gao, X.; Lu, A.; Tang, Y.; Schneppendahl, J.; Liebowitz, A. B.; Scibetta, A. C.; Morris, E. R.; Cheng, H.; Huard, C.; Amra, S.; Wang, B.; Hall, M. A.; Lowe, W. R.; Huard, J. Influences of donor and host age on human muscle-derived stem cell-mediated bone regeneration. Stem Cell Res Ther 2018, 9((1)), 316. [Google Scholar] [CrossRef]
- Turner, N. J.; Pezzone, M. A.; Brown, B. N.; Badylak, S. F. Quantitative multispectral imaging of Herovici’s polychrome for the assessment of collagen content and tissue remodelling. J Tissue Eng Regen Med 2013, 7((2)), 139–48. [Google Scholar] [CrossRef]
- Gao, X.; Usas, A.; Tang, Y.; Lu, A.; Tan, J.; Schneppendahl, J.; Kozemchak, A. M.; Wang, B.; Cummins, J. H.; Tuan, R. S.; Huard, J. A comparison of bone regeneration with human mesenchymal stem cells and muscle-derived stem cells and the critical role of BMP. Biomaterials 2014, 35((25)), 6859–70. [Google Scholar] [CrossRef]
- Mannarino, M.; Cherif, H.; Ghazizadeh, S.; Martinez, O. W.; Sheng, K.; Cousineau, E.; Lee, S.; Millecamps, M.; Gao, C.; Gilbert, A.; Peirs, C.; Naeini, R. S.; Ouellet, J. A.; L, S. S.; Haglund, L. Senolytic treatment for low back pain. Sci Adv 2025, 11((11)), eadr1719. [Google Scholar] [CrossRef]
- Deng, Z.; Gao, X.; Sun, X.; Amra, S.; Lu, A.; Cui, Y.; Eltzschig, H. K.; Lei, G.; Huard, J. Characterization of articular cartilage homeostasis and the mechanism of superior cartilage regeneration of MRL/MpJ mice. FASEB J 2019, 33((8)), 8809–8821. [Google Scholar] [CrossRef]
- Utsunomiya, H.; Gao, X.; Cheng, H.; Deng, Z.; Nakama, G.; Mascarenhas, R.; Goldman, J. L.; Ravuri, S. K.; Arner, J. W.; Ruzbarsky, J. J.; Lowe, W. R.; Philippon, M. J.; Huard, J. Intra-articular Injection of Bevacizumab Enhances Bone Marrow Stimulation-Mediated Cartilage Repair in a Rabbit Osteochondral Defect Model. Am J Sports Med 2021, 49((7)), 1871–1882. [Google Scholar] [CrossRef] [PubMed]
- Pritzker, K. P.; Gay, S.; Jimenez, S. A.; Ostergaard, K.; Pelletier, J. P.; Revell, P. A.; Salter, D.; van den Berg, W. B. Osteoarthritis cartilage histopathology: grading and staging. Osteoarthritis Cartilage 2006, 14((1)), 13–29. [Google Scholar] [CrossRef]
- Stake, I. K.; Gao, X.; Huard, M.; Fukase, N.; Ruzbarsky, J. J.; Ravuri, S.; Layne, J. E.; Philippon, M. J.; Clanton, T. O.; Huard, J. Effects of Losartan and Fisetin on Microfracture-Mediated Cartilage Repair of Ankle Cartilage in a Rabbit Model. Am J Sports Med 2024, 52((14)), 3625–3640. [Google Scholar] [CrossRef] [PubMed]
- Gillman, C. E.; Jayasuriya, A. C. FDA-approved bone grafts and bone graft substitute devices in bone regeneration. Mater Sci Eng C Mater Biol Appl 2021, 130, 112466. [Google Scholar] [CrossRef] [PubMed]
- Elangovan, S.; Khorsand, B.; Do, A. V.; Hong, L.; Dewerth, A.; Kormann, M.; Ross, R. D.; Sumner, D. R.; Allamargot, C.; Salem, A. K. Chemically modified RNA activated matrices enhance bone regeneration. J Control Release 2015, 218, 22–8. [Google Scholar] [CrossRef]
- Surisaeng, T.; Wisitrasameewong, W.; Champaiboon, C.; Sa-Ard-Iam, N.; Chanamuangkon, T.; Thongnuek, P.; Tam, Y. K.; Muramatsu, H.; Weissman, D.; Pardi, N.; Pichyangkul, S.; Mahanonda, R. BMP-2 mRNA-transfected BMSCs promote superior calvarial bone regeneration. Sci Rep 2025, 15((1)), 15022. [Google Scholar] [CrossRef]
- Khorsand, B.; Elangovan, S.; Hong, L.; Dewerth, A.; Kormann, M. S.; Salem, A. K. A Comparative Study of the Bone Regenerative Effect of Chemically Modified RNA Encoding BMP-2 or BMP-9. AAPS J 2017, 19((2)), 438–446. [Google Scholar] [CrossRef]
- Balmayor, E. R.; Geiger, J. P.; Aneja, M. K.; Berezhanskyy, T.; Utzinger, M.; Mykhaylyk, O.; Rudolph, C.; Plank, C. Chemically modified RNA induces osteogenesis of stem cells and human tissue explants as well as accelerates bone healing in rats. Biomaterials 2016, 87, 131–146. [Google Scholar] [CrossRef] [PubMed]
- Badieyan, Z. S.; Berezhanskyy, T.; Utzinger, M.; Aneja, M. K.; Emrich, D.; Erben, R.; Schuler, C.; Altpeter, P.; Ferizi, M.; Hasenpusch, G.; Rudolph, C.; Plank, C. Transcript-activated collagen matrix as sustained mRNA delivery system for bone regeneration. J Control Release 2016, 239, 137–48. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Usas, A.; Proto, J. D.; Lu, A.; Cummins, J. H.; Proctor, A.; Chen, C. W.; Huard, J. Role of donor and host cells in muscle-derived stem cell-mediated bone repair: differentiation vs. paracrine effects. FASEB J 2014, 28((8)), 3792–809. [Google Scholar] [CrossRef] [PubMed]
- Aini, H.; Itaka, K.; Fujisawa, A.; Uchida, H.; Uchida, S.; Fukushima, S.; Kataoka, K.; Saito, T.; Chung, U. I.; Ohba, S. Messenger RNA delivery of a cartilage-anabolic transcription factor as a disease-modifying strategy for osteoarthritis treatment. Sci Rep 2016, 6, 18743. [Google Scholar] [CrossRef]
- Pezzotti, G.; Zhu, W.; Terai, Y.; Marin, E.; Boschetto, F.; Kawamoto, K.; Itaka, K. Raman spectroscopic insight into osteoarthritic cartilage regeneration by mRNA therapeutics encoding cartilage-anabolic transcription factor Runx1. Mater Today Bio 2022, 13, 100210. [Google Scholar] [CrossRef]
- Wu, H.; Peng, Z.; Xu, Y.; Sheng, Z.; Liu, Y.; Liao, Y.; Wang, Y.; Wen, Y.; Yi, J.; Xie, C.; Chen, X.; Hu, J.; Yan, B.; Wang, H.; Yao, X.; Fu, W.; Ouyang, H. Engineered adipose-derived stem cells with IGF-1-modified mRNA ameliorates osteoarthritis development. Stem Cell Res Ther 2022, 13((1)), 19. [Google Scholar] [CrossRef]
- Huang, K.; Liu, X.; Qin, H.; Li, Y.; Zhu, J.; Yin, B.; Zheng, Q.; Zuo, C.; Cao, H.; Tong, Z.; Sun, Z. FGF18 encoding circular mRNA-LNP based on glycerolipid engineering of mesenchymal stem cells for efficient amelioration of osteoarthritis. Biomater Sci 2024, 12((17)), 4427–4439. [Google Scholar] [CrossRef]
- Sun, M.; Ma, B.; Pan, Z.; Zhao, Y.; Tian, L.; Fan, Y.; Kong, W.; Wang, J.; Xu, B.; Ao, Y.; Guo, Q.; Wang, X.; Peng, X.; Li, X.; Cheng, J.; Miao, L.; Wang, K.; Hu, X. Targeted Therapy of Osteoarthritis via Intra-Articular Delivery of Lipid-Nanoparticle-Encapsulated Recombinant Human FGF18 mRNA. Adv Healthc Mater 2024, 13((29)), e2400804. [Google Scholar] [CrossRef]
- Kong, K.; Li, B.; Chang, Y.; Zhao, C.; Qiao, H.; Jin, M.; Wu, X.; Fan, W.; Wang, L.; Qi, Y.; Xu, Y.; Zhai, Z.; Ma, P.; Li, H. Delivery of FGF18 using mRNA-LNP protects the cartilage against degeneration via alleviating chondrocyte senescence. J Nanobiotechnology 2025, 23((1)), 34. [Google Scholar] [CrossRef]
- Fontana, G.; Nemke, B.; Lu, Y.; Chamberlain, C.; Lee, J. S.; Choe, J. A.; Jiao, H.; Nelson, M.; Amitrano, M.; Li, W. J.; Markel, M.; Murphy, W. L. Local delivery of TGF-beta1-mRNA decreases fibrosis in osteochondral defects. Bioact Mater 2025, 45, 509–519. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



