Submitted:
09 April 2026
Posted:
10 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
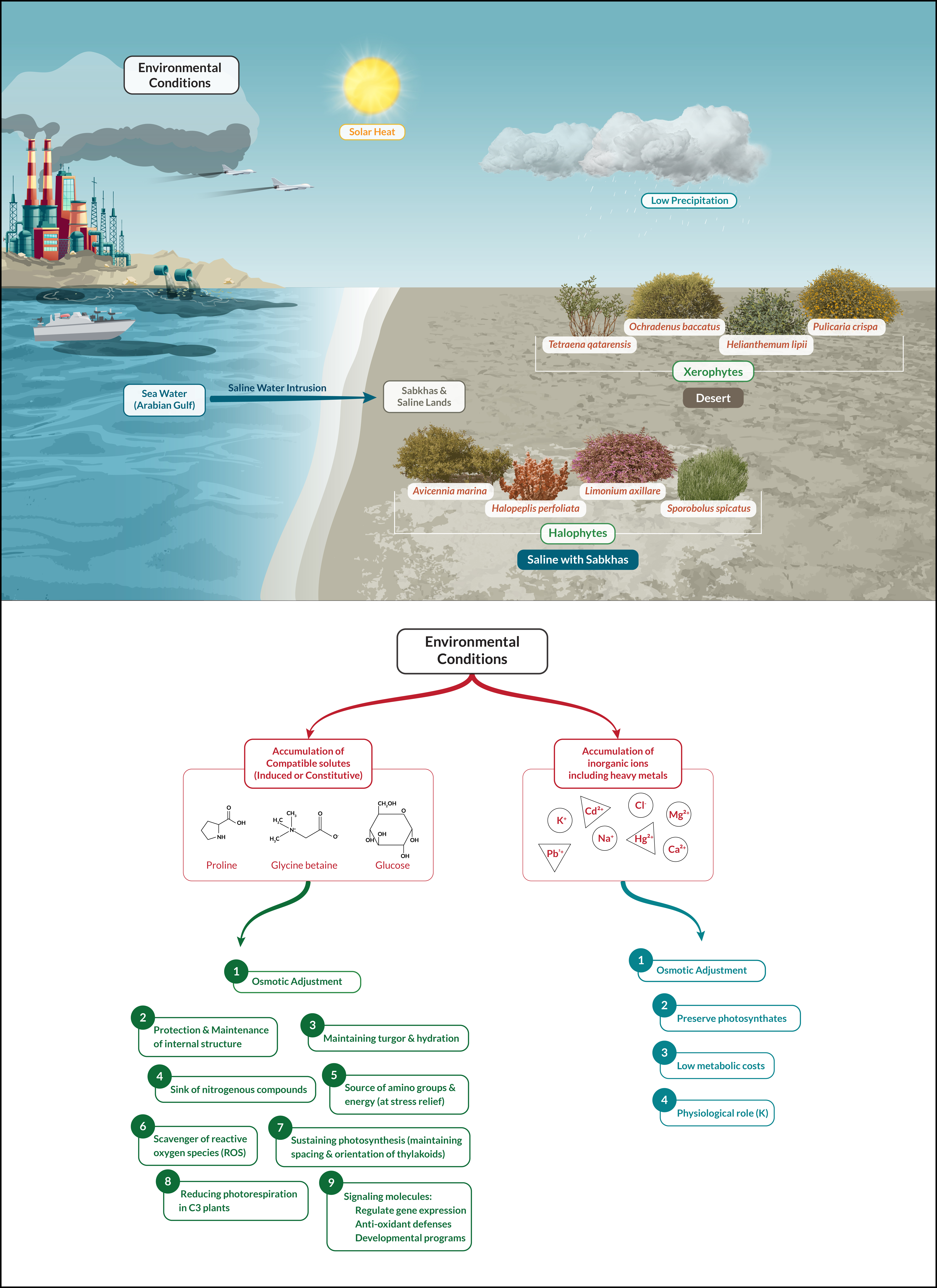
2. Adaptive Roles of Compatible Solutes in Native Plants
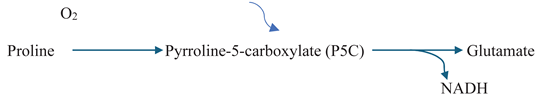
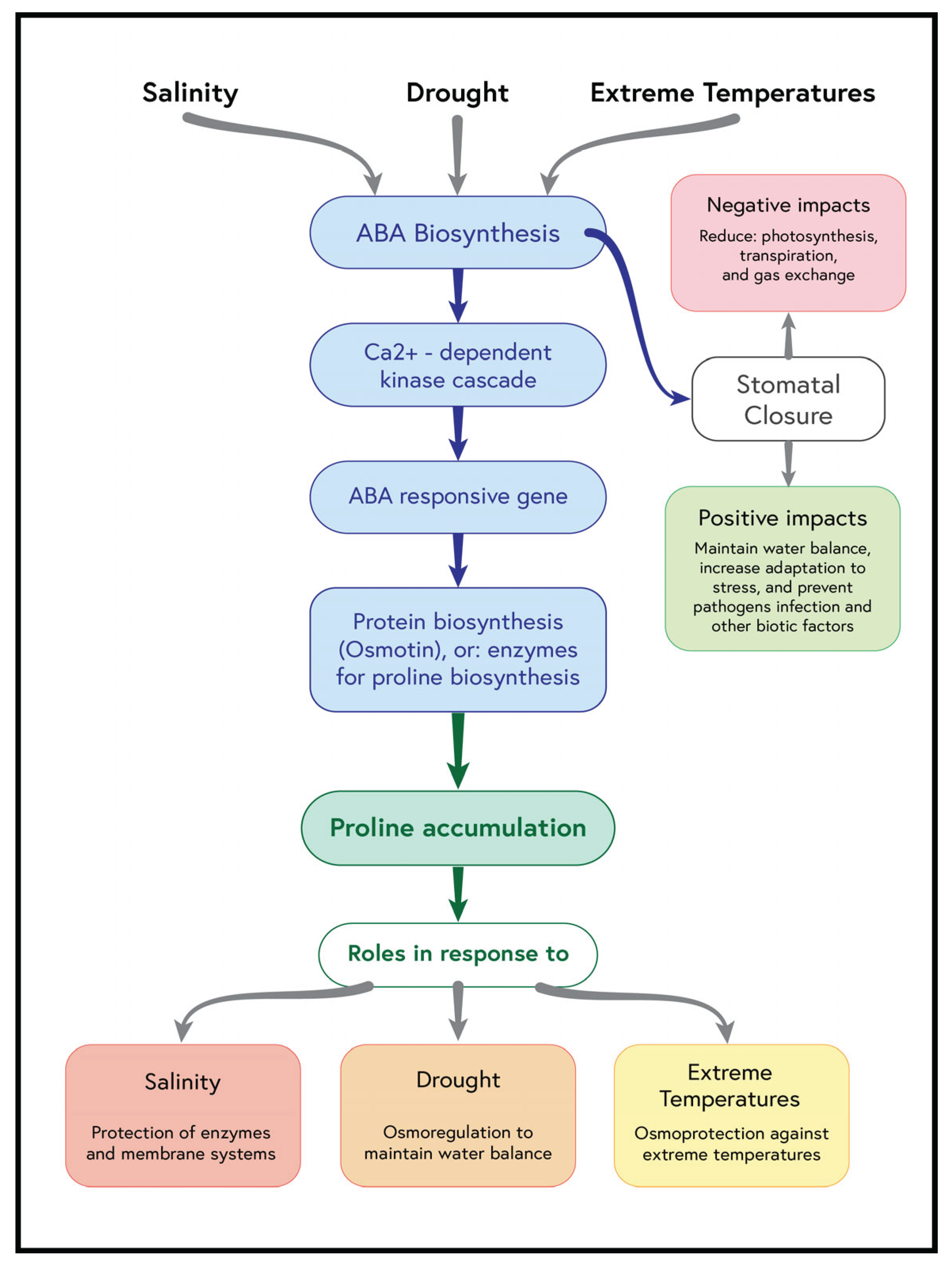
2.1. Compatible Solutes as Indicators of Osmotic Stress Response
3. Case Studies on Compatible Solutes in Native Plants in Qatar
3.1. Glycine Betaine Accumulation in Native Qatari Plants
4. Concluding Remarks and Future Perspectives
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Al-Thani, R.F.; Yasseen, B.T. Solutes in Native Plants in the Arabian Gulf Region and the Role of Microorganisms: Future Research. J. Plant Ecology 2018, 11, 671–684. [Google Scholar] [CrossRef]
- Basit, F.; Alyafei, M.; Hayat, F.; Al-Zayadneh, W.; El-Keblawy, A.; Sulieman, S.; Sheteiwy, M.S. Deciphering the role of glycine betaine in enhancing plant performance and defense mechanisms against environmental stresses. Front. Plant Sci. 2025, 16, 1582332. [Google Scholar] [CrossRef]
- Yasseen, B.T.; Al-Thani, R.F.; Alhadi, F.A.; Abbas, R.A.A. Soluble sugars in plants under stress at the Arabian Gulf region: Possible roles of microorganisms. J. Plant Biochem. Physiol. 2018, 6, 224. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, J.; Li, R.; Ge, Y.; Li, Y.; Li, R. Plants’ response to abiotic stress: Mechanisms and strategies. Int. J. Mol. Sci. 2023, 24, 10915. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Maurya, V.; Gupta, K.; Sharma, I.; Sharma, A.; Kumar, R. Salt stress and its eco-friendly management using biostimulants in grain legumes: a review. Discov. Agric. 2025, 3, 13. [Google Scholar] [CrossRef]
- Krasensky, J.; Jonak, C. Drought, salt, and temperature stress-induced metabolic rearrangements and regulatory networks. Journal of Experimental Botany 2012, 63, 1593–1608. [Google Scholar] [CrossRef]
- Raza, A.; Anas, M.; Bhardwaj, S.; Mir, R.A.; Charagh, S.; Elahi, M.; Zhang, X.; Mir, R.R.; Weckwerth, W.; Fernie, A.R.; Siddique, K.H.M.; Hu, Z.; Varshney, R.K. Harnessing metabolomics for enhanced crop drought tolerance. The Crop Journal 2025, 13, 311–327. [Google Scholar] [CrossRef]
- Al-Thani, R.F.; Yasseen, B.T. Phytoremediation of Polluted Soils and Waters by Native Qatari Plants: Future Perspectives. Environmental Pollution 2020, 259, 113694. [Google Scholar] [CrossRef]
- Al-Thani, R.F.; Yasseen, B.T. Possible Future Risks of Pollution Consequent to the Expansion of Oil and Gas Operations in Qatar. Environment and Pollution 2023, 12, 12–52. [Google Scholar] [CrossRef]
- Yasseen, B.T. Urban Development Threatening Wild Plants in Doha City-Qatar: Ecophysiology is a Prerequisite for Ecological Restoration. Journal of Plant Sciences 2011, 6, 113–123. [Google Scholar] [CrossRef]
- Kumar, V.; Khare, T.; Shaikh, S.; Wani, S.H. Compatible Solutes and Abiotic Stress Tolerance in Plants. In Metabolic Adaptations in Plants During Abiotic Stress; pp. 221–228. [CrossRef]
- Yasseen, B.T.; Al-Thani, R.F. Endophytes and Halophytes to Remediate Industrial Wastewater and Saline Soils: Perspectives from Qatar. PLANTS 2022, 11, 1497. [Google Scholar] [CrossRef]
- Ghosh, U.K.; Islam, M.N.; Siddiqui, M.N.; Khan, M.A.R. Understanding the roles of osmolytes for acclimatizing plants to changing environment: a review of potential mechanism. Plant Signal. Behav. 2021, 16, 1913306. [Google Scholar] [CrossRef]
- Silveira, J.A.G.; Araujo, S.A.M.; Lima, J.P.M.S.; Viegas, R.A. Roots and leaves display contrasting osmotic adjustment mechanisms in response to NaCl-salinity in Atriplex nummularia. Environmental and Experimental Botany 2009, 66, 1–8. [Google Scholar] [CrossRef]
- Hayat, S.; Hayat, Q.; Alyemeni, M.N.; Wani, A.S.; Pichtel, J.; Ahmad, A. Role of proline under changing environments: a review. Plant Signal Behav. 2012, 7, 1456–66. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, Y.K. Proline as a key player in heat stress tolerance: insights from maize. Discov. Agric. 2024, 2, 121. [Google Scholar] [CrossRef]
- Khan, P.; Abdelbacki, A.M.M.; Albaqami, M.; Jan, R.; Kim, K.-M. Proline promotes drought tolerance in Maize. Biology 2025, 14, 41. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.; Abbas, Z.; Seleiman, M.F.; Rizwan, M.; Yavas, İ.; Alhammad, B.A.; Shami, A.; Hasanuzzaman, M.; Kalderis, D. Glycine betaine accumulation, significance and interests for heavy metal tolerance in plants. Plants (Basel) 2020, 9, 896. [Google Scholar] [CrossRef]
- Zhu, Z.; Zhang, R.; Liu, T.; Zheng, H-L. Solute accumulation and osmotic adjustment characteristics of the mangrove Avicennia marina under NaCl-induced salinity stress. Botanica Marina 2011, 54, 335–341. [Google Scholar] [CrossRef]
- Mehta, D.; Vyas, S. Comparative bio-accumulation of osmo-protectants in saline stress tolerating plants: A review. Plant Stress 2023, 9, 100177. [Google Scholar] [CrossRef]
- Munns, R.; Passioura, J.B.; Colmer, T.D.; Byrt, C.S. Osmotic adjustment and energy limitations to plant growth in saline soil. New Phytologist 2019, 225, 1091–1096. [Google Scholar] [CrossRef]
- Chen, H.; Jiang, J-G. Osmotic adjustment and plant adaptation to environmental changes related to drought and salinity. Environmental Reviews 2025a, 18, 309–319. [Google Scholar] [CrossRef]
- Xu, C.; Tang, X.; Shao, H.; Wang, H. Salinity tolerance mechanism of economic halophytes from physiological to molecular hierarchy for improving food quality. Curr. Genomics 2016, 17, 207–14. [Google Scholar] [CrossRef]
- Sun, L.; Wang, J.; Lian, L.; Song, J.; Du, X.; Liu, W.; Zhao, W.; Yang, L.; Li, C.; Qin, Y.; Yang, R. Systematic analysis of the sugar accumulation mechanism in sucrose- and hexose- accumulating cherry tomato fruits. BMC Plant Biol. 2022, 22, 303. [Google Scholar] [CrossRef]
- Hosseinifard, M.; Stefaniak, S.; Javid, M.G.; Soltani, E.; Wojtyla, L.; Małgorzata Garnczarska, M. Contribution of Exogenous Proline to Abiotic Stresses Tolerance in Plants: A Review. Int. J. Mol. Sci. 2022, 23, 5186. [Google Scholar] [CrossRef]
- Wani, S.H.; Singh, N.B.; Haribhushan, A.; Mir, J.I. Compatible solute engineering in plants for abiotic stress tolerance—role of glycine betaine. Curr. Genomics 2013, 14, 157–65. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, S.; Devi, P.; Bhardwaj, A.; Jha, U.C.; Sharma, K.D.; Prasad, P.V.V.; Siddique, K.H.M.; Bindumadhava, H.; Kumar, S.; Nayyar, H. Identification and characterization of contrasting genotypes/cultivars for developing heat tolerance in agricultural crops: Current status and prospects. Front. Plant Sci. 2020, 11, 587264. [Google Scholar] [CrossRef] [PubMed]
- Meena, M.; Divyanshu, K.; Kumar, S.; Swapnil, P.; Zehra, A.; Shukla, V.; Yadav, M.; Upadhyay, R.S. Regulation of L-proline biosynthesis, signal transduction, transport, accumulation and its vital role in plants during variable environmental conditions. Heliyon 2019, 5, e02952. [Google Scholar] [CrossRef] [PubMed]
- Kavi Kishor, P.B.; Sangam, S.; Amrutha, R.N.; Sri Laxmi, P.; Naidu, K.R.; Rao, K.R.S.S.; Rao, S.; Reddy, P.; Theriappan, P.; Sreenivasulu, N. Regulation of proline biosynthesis, degradation, uptake and transport in higher plants: Its implications in plant growth and abiotic stress tolerance. Curr. Science 2005, 88, 424–438. [Google Scholar]
- Mushtaq, N.U.; Saleem, S.; Rasool, A.; Shah, W.H.; Tahir, I.; Seth, C.S.; Rehman, R.U. Proline tagging for stress tolerance in plants. Int. J. Genomics 2025, 2025, 9348557. [Google Scholar] [CrossRef]
- Yu, J.; Li, Y.; Tang, W.; Liu, J.; Lu, B-R.; Liu, Y. The Accumulation of Glycine Betaine Is Dependent on Choline Monooxygenase (OsCMO), Not on Phosphoethanolamine N-Methyltransferase (OsPEAMT1), in Rice (Oryza sativa L. ssp. japonica). Plant Molecular Biology Reporter 2014, 32, 916–922. [Google Scholar] [CrossRef]
- Gu, J.; Ma, X.; Liu, J.; Ma, M. Physiological and molecular mechanisms of glycine betaine in alleviating Na2SO4 stress in Glycyrrhiza uralensis. Front Plant Sci. 2025, 16, 1667006. [Google Scholar] [CrossRef]
- Taiz, L.; Zeiger, E. Plant Physiology, 5th ed.; Sinauer Associates, Inc.: Sunderland, MA, USA, 2010. [Google Scholar]
- Paleg, L.G.; Aspinall, D. The Physiology and Biochemistry of Drought Resistance in Plants; Academic Press: Sydney, New York, London, 1981. [Google Scholar]
- Abdel Bari, E.M.; Yasseen, B.T.; Al-Thani, R.F. Halophytes in the State of Qatar (Sponsored by Environmental Studies Center); Qatar University: Qatar, 2007. [Google Scholar]
- Siddique, A.; Kandpal, G.; Kumar, P. Proline accumulation and its defensive role under diverse stress condition in plants: An overview. J. Pure Appl. Microbiol. 2018, 12, 1655–1659. [Google Scholar] [CrossRef]
- Pallag, G.; Nazarian, S.; Ravasz, D.; Bui, D.; Komlódi, T.; Doerrier, C.; Gnaiger, E.; Seyfried, T.N.; Chinopoulos, C. Proline Oxidation Supports Mitochondrial ATP Production When Complex I Is Inhibited. Int. J. Mol. Sci. 2022, 23, 5111. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Zhang, L.; Natarajan, S.K.; Becker, D.F. Proline mechanisms of stress survival. Antioxid. Redox Signal. 2013, 19, 998–1011. [Google Scholar] [CrossRef] [PubMed]
- Trovato, M.; Forlani, G.; Signorelli, S.; Funk, D. Proline metabolism and its functions in development and stress tolerance. In Osmoprotectant-Mediated Abiotic Stress Tolerance in Plants; Springer Nature: Switzerland AG, 2019; pp. 41–72. [Google Scholar] [CrossRef]
- Renzetti, M.; Funck, D.; Trovato, M. Proline and ROS: A unified mechanism in plant development and stress response? Plants (Basel) 2024, 14, 2. [Google Scholar] [CrossRef]
- Krishnan, N.; Dickman, M.B.; Becker, D.F. Proline modulates the intracellular redox environment and protects mammalian cells against oxidative stress. Free Radic. Biol. Med. 2008, 44, 671–81. [Google Scholar] [CrossRef]
- Dutta, T.; Neelapu, N.R.R.; Wani, S.H.; Challa, S. Chapter 11—Compatible Solute Engineering of Crop Plants for Improved Tolerance Toward Abiotic Stresses. In Biochemical; Wani, S.H., Ed.; Physiological and Molecular Avenues for Combating Abiotic Stress Tolerance in Plants, 2018; pp. 221–254. [Google Scholar] [CrossRef]
- Rajasekaran, L.R.; Kriedemann, P.E.; Aspinall, D.; Paleg, L.R. Physiological significance of proline and Glycinebetaine: Maintaining photosynthesis during NaCL stress in wheat. Photosynthetica 1997, 34, 357–366. [Google Scholar] [CrossRef]
- Makela, P.; Kontturi, M.; Pehu, E.; Somersalo, S. Photosynthetic response of drought- and salt-stressed tomato and turnip rape plants to foliar-applied glycinebetaine. Physiol. Plant 1999, 105, 45–50. [Google Scholar] [CrossRef]
- Annunziata, M.G.; Ciarmiello, L.F.; Woodrow, P.; Dell’Aversana, E.; Carillo, P. Spatial and temporal profile of glycine betaine accumulation in plants under abiotic stresses. Front. Plant Sci. 2019, 10, 230. [Google Scholar] [CrossRef]
- Singhal, R.K.; Saha, D.; Skalicky, M.; Mishra, U.N.; Chauhan, J.; Behera, L.P.; Lenka, D.; Chand, S.; Kumar, V.; Dey, P.; Indu; Pandey, S.; Vachova, P.; Gupta, A.; Brestic, M.; El-Sabagh, A. Crucial signalling compounds crosstalk and integrative multi-omics techniques for salinity stress tolerance in plants. Front. Plant Sci. 2021, 12, 670369. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Cuin, T.A.; Zhou, M.; Twomey, A.; Naidu, B.P.; Shabala, S. Compatible solute accumulation and stress-mitigating effects in barley genotypes contrasting in their salt tolerance. Journal of Experimental Botany 2007, 58, 4245–4255. [Google Scholar] [CrossRef] [PubMed]
- Adzigbe, J.; Frimpong, F.; Danquah, A.; Danquah, E.Y.; Asante, I.K.; Abebrese, S.O.; Dormatey, R.; Afriyie-Debrah, C.; Ribeiro, P.F.; Danquah, E.O.; Agyeman, K.; Ralph Kwame Bam, R.K.; Asante, M.D. The responses and adaptations of rice (Oryza sativa L.) to drought stress: A review. Climate Smart Agriculture 2025, 2, 100080. [Google Scholar] [CrossRef]
- Yasseen, B.T. Further follow-up on proline accumulation in plants. Seminar presented at the Department of Biological & Environmental Sciences, Qatar University, Doha, Qatar, 2004. [Google Scholar]
- Verbruggen, N.; Hermans, C. Proline accumulation in plants: a review. Amino Acids 2008, 35, 753–759. [Google Scholar] [CrossRef]
- Stewart, G.R.; Lee, J.A.T. The role of proline accumulation in halophytes. Planta 1974, 120, 279–289. [Google Scholar] [CrossRef]
- Trovato, M.; Mattioli, R.; Costantino, P. Multiple roles of proline in plant stress tolerance and development. Rend. Fis. Acc. Lincei 2008, 19, 325–346. [Google Scholar] [CrossRef]
- Dar, M.I.; Naikoo, M.I.; Rehman, F.; Naushin, F.; Khan, F.A. Proline Accumulation in Plants: Roles in Stress Tolerance and Plant Development. In Osmolytes and Plants Acclimation to Changing Environment: Emerging Omics Technologies; Iqbal, N., Nazar, R., Khan, A.N., Eds.; Springer: New Delhi, 2016. [Google Scholar] [CrossRef]
- El-Moukhtari, A.; Cabassa-Hourton, C.; Farissi, M.; Savoure, A. How does proline treatment promote salt stress tolerance during crop plant development? Front. Plant Sci. 2020, 11, 1127. [Google Scholar] [CrossRef]
- Nguyen, H.T.T.; Bhowmik, S.D.; Long, H.; Cheng, Y.; Mundree, S.; Hoang, L.T.M. Rapid accumulation of proline enhances salinity tolerance Australian wild rice Oryza australiensis Domin. Plants 2021, 10, 2044. [Google Scholar] [CrossRef]
- Cebeci, E.; Boyaci, H.F.; Kiran, S.; Elliaitioglu, S.S. Comprehensive assessment to reveal that salt tolerance potential of cultivated eggplants and their wild relatives. Fron. Plant. Sci. 2025, 16, 1483409. [Google Scholar] [CrossRef]
- Yasseen, B.T. An analysis of the effects of salinity on leaf growth in Mexican wheats. Ph.D. thesis, The University of Leeds, United Kingdom, 1983. [Google Scholar]
- Moftah, A.E.; Michel, B.E. The effect of sodium chloride on solute potential and proline accumulation in soybean leaves. Plant Physiol. 1987, 83, 238–40. [Google Scholar] [CrossRef]
- Sundaresan, S.; Sudhakaran, P.R. Water stress-induced alterations in the proline metabolism of drought-susceptible and -tolerant cassava (Manihot esculenta) cultivars. Physiol. Plant 1995, 94, 635–642. [Google Scholar] [CrossRef]
- Liu, J.; Zhu, J.K. Proline accumulation and salt-stress-induced gene expression in a salt-hypersensitive mutant of Arabidopsis. Plant Physiol. 1997, 114, 591–596. [Google Scholar] [CrossRef]
- Lutts, S.; Majerus, V.; Kinet, J.M. NaCl effects on proline metabolism in rice (Oryza sativa) seedlings. Physiologia Plantarum 1999, 105, 450–458. [Google Scholar] [CrossRef]
- Yaish, M. Proline accumulation is a general response to abiotic stress in the date palm tree (Phoenix dactylifera L.). Genetics and Molecular Research 2015, 14, 9943–9950. [Google Scholar] [CrossRef]
- Kavi Kishor, P.B.; Sreenivasulu, N. Is proline accumulation per se correlated with stress tolerance or is proline homeostasis a more critical issue? Plant Cell Environ. 2015, 37, 300–11. [Google Scholar] [CrossRef] [PubMed]
- Spormann, S.; Nadais, P.; Sousa, F.; Pinto, M.; Martins, M.; Sousa, B.; Fidalgo, F.; Soares, C. Accumulation of proline in plants under contaminated soils-are we on the same page? Antioxidants (Basel) 2023, 12, 666. [Google Scholar] [CrossRef]
- Masheva, V.; Spasova-Apostolova, V.; Aziz, S.; Tomlekova, N. Variations in proline accumulation and relative water content under water stress characterize bean mutant lines (P. vulgaris L.). Bulg. J. Agric. Sci. 2022, 28, 430–436. [Google Scholar]
- Raza, A.; Charagh, S.; Abbas, S.; Hassan, M.U.; Saeed, F.; Haider, S.; Sharif, R.; Anand, A.; Corpas, F.J.; Jin, W.; Varshney, R.K. Assessment of proline function in higher plants under extreme temperatures. Plant Biol. (Stuttg) 2023, 25, 379–395. [Google Scholar] [CrossRef]
- Rai, G.K.; Khanday, D.M.; Choudhary, S.M.; Kumar, P.; Kumari, S.; Martinez-Andújar, C.; Martinez-Melgarejo, A.; Rai, P.K. Unlocking nature’s stress buster: Abscisic acid’s crucial role in defending plants against abiotic stress. Plant Stress 2024, 11, 100359. [Google Scholar] [CrossRef]
- Bharath, P.; Gahir, S.; Raghavendra, A.S. Abscisic acid-induced stomatal closure: an important component of plant defense against abiotic and biotic stress. Front. Plant Sci. 2021, 12, 615114. [Google Scholar] [CrossRef] [PubMed]
- Hussain, S.; Khalid, M.F.; Muhammad, S.; Anjum, M.A. Role of transporters in accumulating salt ions by halophytes. In Approaches of the Remediation of Inorganic Pollutants; Hasanuzzaman, M., Ed.; Springer: Singapore, 2021; pp. 11–40. [Google Scholar] [CrossRef]
- Rahman, M.; Mostofa, M.G.; Keya, S.S.; Siddiqui, N.; Ansary, M.U.; Das, A.K.; Rahman, A.; Tran, L.S.-P. Adaptive mechanisms of halophytes and their potential in improving salinity tolerance in plants. Int. J. Mol. Sci. 2021, 22, 10733. [Google Scholar] [CrossRef] [PubMed]
- Nilsen, E.T.; Orcutt, D.M. Physiology of Plants under Stress, Abiotic Factors; John Wiley & Sons, Inc.: New York, USA, 1996. [Google Scholar]
- Larcher, W. Physiological Plant Ecology. Eco-physiology and Stress Physiology of Functional Groups, 4th ed.; Springer: Berlin, 2003. [Google Scholar]
- Epstein, E. Crops tolerant of salinity and other mineral stresses. Ciba Foundation Symposium 97. Better crops for food; Pitman: London, 1983; pp. 61–82. [Google Scholar]
- Yasseen, B.T.; Abu-Al-Basal, M.A. Ecophysiology of Chenopodiaceae at the Coastline of Arabian Gulf-Qatar: Possible Destruction and Conservation Perspective. European Journal of Scientific Research 2010, 39, 90–104. [Google Scholar]
- Balasubramaniam, T.; Shen, G.; Esmaeili, N.; Zhang, H. Plants’ response mechanisms to salinity stress. Plants (Basel) 2023, 12, 2253. [Google Scholar] [CrossRef] [PubMed]
- Gul, B.; Hameed, A.; Ahmed, M.Z.; Hussain, T.; Rasool, S.G.; Nielsen, B.L. Thriving under salinity: Growth, ecophysiology and proteomic insights into the tolerance mechanisms of obligate halophyte Suaeda fruticosa. Plants (Basel) 2024, 13, 1529. [Google Scholar] [CrossRef]
- Al-Thani, R.F.; Yasseen, B.T. Cyanoremediation of polluted seawater in the Arabian Gulf: Risks and benefits to human health. Processes 2024, 12, 2733. [Google Scholar] [CrossRef]
- Al-Thani, R.F.; Yasseen, B.T. The Role of Phytoplankton in Phycoremediation of Polluted Seawater: Risks, Benefits to Human Health, and a Focus on Diatoms in the Arabian Gulf. Water 2025, 17, 920. [Google Scholar] [CrossRef]
- Atzori, G.; Garcia-Caparros, P.; Castagna, A.; Custodio, L. Salt-induced nutritional and metabolic shifts in halophytes: implications for food security. Journal of the Science of Food and Agriculture 2025. [Google Scholar] [CrossRef]
- Yasseen, B.T.; Al-Thani, R.F. Wild Plants in the Qatari Peninsula are Hidden Gene Bank for Future Research: Perspectives of Desirable Traits, Chapter 6. In Cutting Edge Research in Biology; 2023; Vol. 8. B P International pp, pp. 207–252. [Google Scholar] [CrossRef]
- Gurrieri, L.; Merico, M.; Trost, P.; Forlani, G.; Sparla, F. Impact of Drought on Soluble Sugars and Free Proline Content in Selected Arabidopsis Mutants. Biology (Basel) 2020, 9, 367. [Google Scholar] [CrossRef]
- Yu, J.; Zhang, R.; Li, X.; Dong, D.; Wang, S. Sugar metabolism and transport in response to drought-rehydration in Poa pratensis. Agronomy 2025, 15, 320. [Google Scholar] [CrossRef]
- Alhadi, F.A.; Yasseen, B.T.; Al-Dubaie, A.S. Changes in carbohydrate and nitrogen fractions during germination of fenugreek (Trigonella foenum-graecum L.) seeds presoaked in GA3, growing under different Osmotic potentials. Qatar Univ. Sci. J. 1997, 17, 271–279. [Google Scholar]
- Gill, P.K.; Sharma, A.D.; Singh, P.; Bhullar, S.S. Changes in germination, growth and soluble sugar contents of Sorghum bicolor (L.) Moench seeds under various abiotic stresses. Plant Growth Regulation 2003, 40, 157–162. [Google Scholar] [CrossRef]
- Yasseen, B.T.; Almuhannady, A.; Al-Marri, F.R.; Al-Hemiary, H. Changes in soluble sugars and proline in seedlings of a local wheat cultivar (Doha) due to the salt stress and temperature. Qatar Univ. Sci. J. 2006, 26, 71–82. [Google Scholar]
- Gupta, B.; Huang, B. Mechanism of salinity tolerance in plants: physiological, biochemical, and molecular characterization. Int. J. Genomics 2014, 2014, 701596. [Google Scholar] [CrossRef]
- Alhadi, F.A.; Yasseen, B.T.; Gabr, M.M. Water stress and gibberellic acid effects on growth of fenugreek plants. Irrig. Sci. 1999, 18, 185–190. [Google Scholar] [CrossRef]
- Jarin, A.; Ghosh, U.K.; Hossain, M.S.; Mahmud, A. Glycine betaine in plant responses and tolerance to abiotic stresses. Discover Agriculture 2024, 2, 127. [Google Scholar] [CrossRef]
- Park, E-J.; Jeknic, Z.; Sakamoto, A.; DeNoma, J. Genetic engineering of glycine betaine synthesis in tomato protects seeds, plants, and flowers from chilling damage. The Plant Journal 2004, 40, 474–487. [Google Scholar] [CrossRef]
- Khan, M.S.; Yu, X.; Kikuchi, A.; Asahina, M.; Watanabe, K.N. Genetic engineering of glycine betaine biosynthesis to enhance abiotic stress tolerance in plants. Plant Biotechnology 2009, 26, 125–134. [Google Scholar] [CrossRef]
- Ashraf, M.; Foolad, M.R. Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environmental & Experimental Botany 2007, 59, 206–216. [Google Scholar] [CrossRef]
- Dong, X.; Liu, Y.; Ma, X.; Wang, S.; Yang, H.; Gao, X.; Wang, G.; Wang, H. Disclosing the effect of exogenous betaine on growth of Suaeda salsa (L.) Pall in the Liaohe coastal wetland, North China. Marine Pollution Bulletin 2024, 198, 115852. [Google Scholar] [CrossRef]
- Dikilitas, M.; Simsek, E.; Roychoudhury, A. Role of proline and glycine betaine in overcoming abiotic stresses. In Protective Chemical Agents in the Amelioration of Plant Abiotic Stress; Roychoudhury, A., Tripathi, D.K., Eds.; 2020. [Google Scholar] [CrossRef]
- Dhansu, P.; Kaushik, S.; Kumar, R.; Krishnapriya, V.; Appunu, C.; Shariff, A.; Kaur, G.; Singh, P.; Kumari, A.; Kumar, A. Intricate relationship of physiological, biochemical and molecular mechanisms in deciphering abiotic stress tolerance in sugarcane. South African Journal of Botany 2026, 188, 135–150. [Google Scholar] [CrossRef]
- Abulfatih, H.A.; Abdel-Bari, E.M.; Alsubaey, A.; Ibrahim, Y.M. Vegetation of Qatar; Scientific and Applied Research Centre (SARC), University of Qatar: Doha, Qatar, 2001. [Google Scholar]
- Yasseen, B.T.; Al-Thani, R.F. Ecophysiology of Wild Plants and Conservation Perspectives in the State of Qatar, Chapter 3. In Agricultural Chemistry; Stoytcheva, M., Zlatev, R., Eds.; 2013; pp. 37–70. [Google Scholar] [CrossRef]
- Al-Thani, R.F.; Yasseen, B.T. Microbial ecology of Qatar, the Arabian Gulf: Possible roles of microorganisms. Front. Mar. Sci. 2021, 8, 697269. [Google Scholar] [CrossRef]
- Abdel-Bari, E.M.M. The Floraof Qatar. Environmental Studies Center; University of Qatar: Doha, Qatar, 2012; Volume 1. [Google Scholar]
- Youssef, A.M.; Hassanein, R.A.; Hassanein, A.A.; Morsy, A.A. Changes in quaternary ammonium compounds, proline and protein profile of certain halophytic plants under different habitat conditions. Pak. J. Biol. Sci. 2003, 6, 867–882. [Google Scholar] [CrossRef]
- Yasseen, B.T.; Al-Thani, R.F. Halophytes and associated properties of natural soils in the Doha area, Qatar. AEHMS 2007, 10, 320–326. [Google Scholar] [CrossRef]
- Sharma, A.; Shahzad, B.; Kumar, V.; Kohli, S.K.; Sidhu, G.P.S.; Bali, A.S.; Handa, N.; Kapoor, D.; Bhardwaj, R.; Zheng, B. Phytohormones Regulate Accumulation of Osmolytes Under Abiotic Stress. Biomolecules 2019, 9, 285. [Google Scholar] [CrossRef]
- Rindyastuti, R.; Hapsari, L.; Byun, C. Comparison of ecophysiological and leaf anatomical traits of native and invasive plant species. J. Ecology & Environ. 2021, 45. [Google Scholar] [CrossRef]
- Leotta, L.; Toscano, S.; Ferrante, A.; Romano, D. The use of Mediterranean native shrubs for improving the sustainability of urban environments. Front. Hortic. 2025, 4, 1652517. [Google Scholar] [CrossRef]
- Lohani, N.; Singh, M.B.; Bhalla, P.L. Biological parts for engineering abiotic stress tolerance in plants. BioDesign Research 2022, 2022, 9819314. [Google Scholar] [CrossRef] [PubMed]
- Kebert, M.; Stojnić, S.; Rašeta, M.; Kostić, S.; Vuksanović, V.; Ivanković, M.; Lanšćak, M.; Markić, A.G. Variations in proline content, polyamine profiles, and antioxidant capacities among different provenances of European beech (Fagus sylvatica L.). Antioxidants (Basel) 2024, 13, 227. [Google Scholar] [CrossRef]
- Jiang, Z.; van Zanten, M.; Sasidharan, R. Mechanisms of plant acclimation to multiple abiotic stresses. Commun. Biol. 2025, 8, 655. [Google Scholar] [CrossRef]
- Verslues, P.E.; Sharma, S. Proline metabolism and its implications for plant-environment interaction. The Arabidopsis Book 2010, 2010, e0140. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Dias, M.C.; Freitas, H. Drought and salinity stress responses and microbe-induced tolerance in plants. Front. Plant Sci. 2020, 11, 591911. [Google Scholar] [CrossRef]
- Yeo, A.R. Salinity resistance: physiologies and prices. Physiol. Plant 1983, 58, 214–222. [Google Scholar] [CrossRef]
- Grigore, M-N.; Boscaiu, M.; Vicente, O. Assessment of the relevance of osmolyte biosynthesis for salt tolerance of halophytes under natural conditions. The European Journal of Plant Science and Biotechnology 2011, 5, 12–19. [Google Scholar]
- Khan, F.; Adnan, M.Y.; Aziz, I. Metabolic implications of salt induced osmolyte accumulation in Avicennia marina. Pak. J. Bot. 2016, 48, 29–36. [Google Scholar]
- Mattioli, R.; Costantino, P.; Trovato, M. Proline accumulation in plants: not only stress. Plant Signal Behav. 2009, 4, 1016–8. [Google Scholar] [CrossRef]
- Hmidi, D.; Abdelly, C.; Athar, H.U.; Ashraf, M.; Messedi, D. Effect of salinity on osmotic adjustment, proline accumulation and possible role of ornithine-δ-aminotransferase in proline biosynthesis in Cakile maritima. Physiol. Mol. Biol. Plants 2018, 24, 1017–1033. [Google Scholar] [CrossRef]
- Hualpa-Ramirez, E.; Carrasco-Lozano, E.C.; Madrid-Espinoza, J.; Tejos, R.; Ruiz-Lara, S.; Stange, C.; Norambuena, L. Stress salinity in plants: New strategies to cope with in the foreseeable scenario. Plant Physiology and Biochemistry 2024, 208, 108507. [Google Scholar] [CrossRef] [PubMed]
- Yasseen, B.T.; Abu-Al–Basal, M.A. Ecophysiology of Limonium axillare and Avicennia marina from the Coastline of Arabian Gulf-Qatar. Journal of Coastal Conservation: Planning and Management 2008, 12, 35–42. [Google Scholar] [CrossRef]
- Alikhani, F.; Saboora, A.; Razavi, K. Changes in osmolites contents, lipid peroxidation, and photosynthetic pigment of Aeluropus lagopoides under potassium deficiency and salinity. Journal of Stress Physiology & Biochemistry 2011, 7, 5–19. [Google Scholar]
- Moinuddin, M.; Gulzar, S.; Ahmed, M.Z.; Gul, B.; Koyro, H-W.; Ajmal Khan, M. Excreting and non-excreting grasses exhibit different salt resistance strategies. AoB Plants 2014, 6, plu038. [Google Scholar] [CrossRef]
- Ajmal Khan, M.; Ungar, I.A.; Showalter, A.M.; Dewald, H.D. NaCl-induced accumulation of glycinebetaine in four subtropical halophytes from Pakistan. Physiol. Plant 1998, 102, 487–492. [Google Scholar] [CrossRef]
- Alam, H.; Zamin, M.; Adnan, M.; Ahmad, N.; Nawaz, T.; Saud, S.; Basi, A.; Liu, L.; Harrison, M.T.; Hassan, S.; Alharby, H.F.; Alzahrani, Y.M.; Alghamdi, S.A.; Majrashi, A.; Alharbi, B.M.; Alabdallah, N.M.; Fahad, S. Evaluating the resistance mechanism of Atriplex leucoclada (Orache) to salt and water stress; A potential crop for bio-saline agriculture. Front. Plant Sci. 2022, 13, 948736. [Google Scholar] [CrossRef]
- Hayes, M.A.; Chapman, S.; Jesse, A.; O’Brien, E.; Langley, J.A.; Bardou, R.; Devaney, J.; Parker, J.D.; Cavanaugh, K.C. Foliar water uptake by coastal wetland plants: A novel water acquisition mechanism in arid and humid subtropical mangroves. J. Ecol. 2020, 108, 2625–2637. [Google Scholar] [CrossRef]
- Khatib, M. Quaternary ammonium compounds in roots and leaves of Capparis spinosa L. from Saudi Arabia and Italy: investigation by HPLC-MS and 1H NMR. Natural Product Research 2018, 33. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, R.; AL-Khateeb, A.M.H.; Ashrafi, F.; Hafiz, M.I.A. Accumulation of compatible solutes (proline and glycinebetaine) as part of osmoregulation in Cleome species. Journal of Stress Physiology & Biochemistry 2012, 8. [Google Scholar]
- Bhattacharya, E.; Biswas, S.M. First report of the hyperaccumulating potential of cadmium and lead by Cleome rutidosperma DC. With brief insight into the chemical vocabulary of its roots. Fron. Environ. Sci. 2022, 10, 830087. [Google Scholar] [CrossRef]
- Masrahi, Y.S. Ecological significance of wood anatomy in two lianas from arid southwestern Saudi Arabia. Saudi J. Biol. Sci. 2014, 21, 334–41. [Google Scholar] [CrossRef]
- Sivasankaramoorthy, S.; Balasubramanian, T.; Amuthavalli, P.; Sivaraman, P. Effect of sodium chloride on the seedlings of Cressa cretica L. Journal of Ecobiotechnology 2010, 2, 01–03. Available online: http://journal-ecobiotechnology.com/.
- Ghafar, M.A.; Akram, N.A.; Saleem, M.H.; Wang, J.; Wijaya, L.; Alyemeni, M.N. Ecotypic morphological and physio-biochemical responses of two differentially adapted forage grasses, Cenchrus ciliaris L.; Cyperus arenarius Retz. to drought stress. Sustainability 2021, 13, 8069. [Google Scholar] [CrossRef]
- Chagah, S.; Chehrazi, M.; Albaji, M. Effects of drought stress on growth and development Frankenia plant (Frankenia leavis). Bulg. J. Agric. Sci. 2013, 19, 659–665. [Google Scholar]
- Kaur, G.; Sanwal, S.K.; Kumar, A.; Pundir, R.K.; Yadav, M.; Sehrawat, N. Role of osmolytes dynamics in plant metabolism to cope with salinity induced osmotic stress. Discov. Agric. 2024, 2, 59. [Google Scholar] [CrossRef]
- Zengin, F.K.; Munzuroğlu, Ö. Compatible solutes accumulation in Halocnemum strobilaceum (Pall.) MBieb. leaves in response to salinity. Journal of Soil Science and Plant Nutrition 2007, 13, 481–494. [Google Scholar]
- Briens, M.; Larher, F. Osmoregulation in halophytic higher plants: A comparative study of soluble carbohydrates, polyols, betaines and free proline. Plant, Cell & Environment 2006, 5, 287–292. [Google Scholar] [CrossRef]
- Al-Taisan, W. The effect of soil properties on characteristics of Halopeplis perfoliata community in the coastal environment in eastern Saudi Arabia. Fresenius Environmental Bulletin 2016, 25(12a), 5819–5829. [Google Scholar]
- Panda, A.; Rangani, J.; Parida, A.K. Unravelling salt responsive metabolites and metabolic pathways using non-targeted metabolomics approach and elucidation of salt tolerance mechanisms in the xero-halophyte Haloxylon salicornicum. Plant Physiology and Biochemistry 2021, 158, 284–296. [Google Scholar] [CrossRef] [PubMed]
- Yasseen, B.T. Traits of Wild Plants in Qatar Peninsula and Research Perspectives. J. Biology & Nature 2016, 5, 52–66. [Google Scholar]
- Bhatt, A.; Bhat, N.R.; Khalil, M.; Al-Mansour, H. Prioritization of potential native plants from Arabian Peninsula based on economic and ecological values: Implication for restoration. Sustainability 2023, 15, 6139. [Google Scholar] [CrossRef]
- Muslu, A.S.; Kadıoğlu, A. Heliotropium hirsutissimum from geothermal areas: evidence of thermal adaptation. Protoplasma 2025, 262, 1543–1562. [Google Scholar] [CrossRef]
- Pardo-Domenech, L.L.; Tifrea, A.; Grigore, M.N.; Boscaiu, M. Proline and glycine betaine accumulation in two succulent halophytes under natural and experimental conditions. Plant Biosystem 2016, 150, 904–915. [Google Scholar] [CrossRef]
- Syranidou, E.; Christofilopoulos, S.; Kalogerakis, N. Juncus spp.—The helophyte for all (phyto) remediation purposes? New Biotech. 2017, 38, 43–55. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Orenga, S.; Grigore, M-N.; Boscaiu, M.; Vicente, O. Constitutive and induced salt tolerance mechanisms and potential uses of Limonium Mill species. Agronomy 2021, 11, 413. [Google Scholar] [CrossRef]
- Loconsole, D.; Murillo-Amador, B.; Cristiano, G.; De Lucia, B. Halophytes common ice plants: A future solution to arable land salinization. Sustainability 2019, 11, 6076. [Google Scholar] [CrossRef]
- Kataoka, R.; Akashi, M.; Taniguchi, T.; Kinose, Y.; Yaprak, A.E.; Turgay, O.C. Metabolomics analyses reveal metabolites affected by plant growth-promoting endophytic bacteria in roots of the halophyte Mesembryanthemum crystallinum. Int. J. Mol. Sci. 2021, 22, 11813. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, A.; Smaoui, A.; Zouhaier, B.; Abdelly, C. Differential response to salinity and water deficit stress in Polypogon monspeliensis (L.) Desf. provenances during germination. Plant Biology 2011, 13, 541–545. [Google Scholar] [CrossRef]
- Farzamisepehr, M.; Nourozi, F. Physiological responses of Polypogon monspeliensis L. in petroleum-contaminated soils. Iranian J. Plant Physiol. 2018, 8, 2391–2401. [Google Scholar]
- García-Mercado, H.D.; Villagómez, G.F.; Garzón-Zúñiga, M.A.; Duran, M.C. Fate of mercury in a terrestrial biological lab process using Polypogon monspeliensis and Cyperus odoratus. Int. J. Phytoremediat. 2019, 21, 1–9. [Google Scholar] [CrossRef]
- Zeng, Y.; Wang, S.; Huang, F.; Luo, Q.; Ren, B.; Abo El-Maati, MF.; El-Sappah, A.H. Fate of polycyclic aromatic hydrocarbons in the phytoremediation of different hydrocarbon contaminated soils with cotton, ryegrass, tall fescue, and wheat. Front Plant Sci. 2025, 16, 1550234. [Google Scholar] [CrossRef] [PubMed]
- Moghaieb, R.E.A.; Saneoka, H.; Fujita, K. Effect of salinity on osmotic adjustment, glycine betaine accumulation and betaine aldehyde dehydrogenase expression in Salicornia europaea. Plant Science 2004, 166, 1345–1349. [Google Scholar] [CrossRef]
- Duan, H.; Tiika, R.J.; Tian, F.; Lu, Y.; Zhang, Q.; Hu, Y.; Cui, G.; Yang, H. Metabolomics analysis unveils important changes involved in the salt tolerance of Salicornia europaea. Front Plant Sci. 2023, 13, 1097076. [Google Scholar] [CrossRef]
- Homayouni, H.; Razi, H.; Izadi, M.; Alemzadeh, A.; Kazemeini, S.A.; Niazi, A.; Vicente, O. Temporal changes in biochemical responses to salt stress in three Salicornia. Plants 2024, 13, 979. [Google Scholar] [CrossRef]
- Mendis, C.L.; Padmathilake, R.E.; Attanayake, R.N.; Perera, D. Learning from Salicornia: Physiological, biochemical, molecular mechanisms of salinity tolerance. Int. J. Mol. Sci. 2025, 26, 5936. [Google Scholar] [CrossRef] [PubMed]
- Hadi, M.R. Biotechnological potentials of Seidlitzia rosmarinus: A mini review. Afr. J. Biotechnol. 2009, 8, 2429–2431. Available online: http://www.academicjournals.org/AJB.
- Dragovic, R.; Zlatkovic, B.; Dragovic, S.; Petrovic, J.; Mandic, L.T. Accumulation of heavy metals in different parts of Russian thistle (Salsola tragus, Chenopodiaceae), a potential hyperaccumulator plant species. Biol. Nyssana 2014, 5, 83–90. [Google Scholar]
- Centofanti, T.; Bañuelos, G. Evaluation of the halophyte Salsola soda as an alternative crop for saline soils high in selenium and boron. J. Environ. Mang. 2015, 157, 96–102. [Google Scholar] [CrossRef]
- Karakas, S.; Cullu, M.A.; Dikilitas, S. Comparison of two halophyte species (Salsola soda and Portulaca oleracea) for salt removal potential under different soil salinity conditions. Turkish Journal of Agriculture and Forestry 2017, 41, 183–190. [Google Scholar] [CrossRef]
- Amin, M.A.; Shahhat, I.; Ismail, M.A.; Ibrahim, A. Vicia faba overcomes drought stress by spraying with xerophytic Anabasis setifera extract. Edelweiss Applied Science and Technology 2024, 8, 8008–8022. [Google Scholar] [CrossRef]
- Hamilton, E.W.; McNaughton, S.J.; Coleman, J.S. Molecular, physiological, and growth responses to sodium stress in C4 grasses from a soil salinity gradient in the Serengeti ecosystem. Amer. J. Botany 2001, 88, 1258–1265. [Google Scholar] [CrossRef]
- Jesus, J.M.; Danko, A.S.; Fiúza, A.; Borges, M.T. Phytoremediation of salt-affected soils: a review of processes, applicability, and the impact of climate change. Environ. Sci. Pollut. Res. Int. 2015, 22, 6511–25. [Google Scholar] [CrossRef]
- Dawalibi, V.; Monteverdi, M.C.; Moscatello, S.; Battistelli, A.; Valentini, R. Effect of salt and drought on growth, physiological and biochemical responses of two Tamarix species. iForest—Biogeosciences and Forestry 2015, 8, 772–779. [Google Scholar] [CrossRef]
- Bali, A.S.; Singh Sidhu, G.P.; Grover, D.; Dahiya, B. Biological trace elements confer abiotic stress tolerance in plants. In Biostimulants for Crop Production and Sustainable Agriculture; Hasanuzzaman, M., Hawrylak-Nowak, B., Fujita, M., Islam, M.T., Eds.; CABI, 2022; pp. 306–322. [Google Scholar] [CrossRef]
- Chen, J.; Zhang, J.; Liu, Y.; Zhang, K.; Zhu, F.; Xie, Y. Advances in the biosynthetic regulation and functional mechanisms of glycine betaine for enhancing plant stress resilience. Int. J. Mol. Sci. 2025, 26, 7971. [Google Scholar] [CrossRef]
- Yasseen, B.T.; Al-Thani, R.F. Adaptive Strategies to Biotic Stress in Qatar’s Native Flora. Life 2025, 15, 1645. [Google Scholar] [CrossRef]
- Lianopoulou, V.; Patakas, A.A.; Bosabalidis, A.M. Seasonal dimorphism and winter chilling stress in Thymus sibthorpii. Biologia Plantarum 2014, 58, 139–146. [Google Scholar] [CrossRef]
- Khater, A.K.; Eldanasoury, M.; El-lamey, T.; El-Khamissi, H.A.Z. Seasonal Variation in Photosynthetic Pigments, Phytohormones, and Phenols of Teucrium polium L. Growing in Wadi Halazien, Egypt. Al-Azhar Journal of Agricultural Research 2021, 47. [Google Scholar] [CrossRef]
- Rhodes, D.; Hanson, A.D. Quaternary Ammonium and Tertiary Sulfonium Compounds in Higher Plants. Annual Review of Plant Physiology and Plant Molecular Biology 2003, 44, 357–384. [Google Scholar] [CrossRef]
- Obłąk, E.; Futoma-Kołoch, B.; Wieczyńska, A. Biological activity of quaternary ammonium salts and resistance of microorganisms to these compounds. World J. Microbiol. Biotechnol. 2021, 37, 22. [Google Scholar] [CrossRef] [PubMed]
- Hassanuzzaman, M.; Banerjee, A.; Bhuyan, M.H.M.B.; Roychoudhury, A. Targeting glycine betaine for abiotic stress tolerance in crop plants: Physiological mechanism, molecular interaction and signaling. Phyton-International Journal of Experimental Botany 2019, 88, 185–221. [Google Scholar] [CrossRef]
- Agboma, P.C.; Jones, M.G.K.; Peltonen-Sainio, P.; Rita, H. Exogenous glycinebetaine enhances grain yield of maize, sorghum and wheat grown under two supplementary watering regimes. Journal of Agronomy and Crop Science 2008, 178, 29–37. [Google Scholar] [CrossRef]
- McNeil, S.D.; Nuccio, M.L.; Ziemak, M.J.; Hanson, A.D. Enhanced synthesis of choline and glycine betaine in transgenic tobacco plants that overexpress phosphoethanolamine N-methyltransferase. Proc. Natl. Acad. Sci. USA 2001, 98, 10001–10005. [Google Scholar] [CrossRef]
- Xu, Z.; Sun, M.; Jiang, X.; Sun, H.; Dang, X.; Cong, H.; Qiao, F. Glycinebetaine biosynthesis in response to osmotic stress depends on jasmonate signalling in watermelon suspension cells. Front. Plant Sci. 2018, 9, 1469. [Google Scholar] [CrossRef]
- Norton, J.; Abdul Majid, S.; Allan, D.; Al Safran, M.; Böer, B.; Richer, R. An Illustrated Checklist of the Flora of Qatar; United Nations Educational, Scientific and Cultural Organization, UNESCO Office in Doha: Doha; Qatar Foundation, MAERSK OIL QATAR AS, 2009. [Google Scholar]
- Eisenhut, M.; Roell, M-S.; Weber, A.P.M. Mechanistic understanding of photorespiration paves the way to a new green revolution. New Phytologist 2019, 223, 1762–1769. [Google Scholar] [CrossRef]
- Wingler, A.; Lea, P.J.; Quick, W.P.; Leegood, R.C. Photorespiration: metabolic pathways and their role in stress protection. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2000, 355, 1517–1529. [Google Scholar] [CrossRef]
- Chen, T.H.H.; Murata, N. Glycine betaine: An effective protectant against abiotic stress in plants. Trends in Plant Stress in Plant Science 2008, 13, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Niu, T.; Zhang, T.; Qiao, Y.; Wen, P.; Zhai, G.; Liu, E.; Al-Bakre, D.A.; Al-Harbi, M.S.; Gao, X.; Yang, X. Glycinebetaine mitigates drought stress-induced oxidative damage in pears. PLoS One 2021, 16, e0251389. [Google Scholar] [CrossRef] [PubMed]
- Alam, Kh. A.; Karim, S.; Sultana, S.; Das, A.K.; Mahmud, A.; Rahman, Md. A.; Hossain, Md. M.; Arafat, Y.; Parvin, S.; Lee, M.-S. Glycine betaine improves photosynthetic performance and antioxidant defense in barley under water deficit conditions. Biomolecules 2026, 16, 372. [Google Scholar] [CrossRef] [PubMed]
- Al-Thani, R.F.; Yasseen, B.T. Perspectives of Future Water Sources in Qatar by Phytoremediation: Biodiversity at Ponds and Modern Approach. International Journal of Phytoremediation 2021, 23, 866–889. [Google Scholar] [CrossRef]
- Tan, T.; Hu, W.; Fu, T.; Li, M.; Campbell, G.; Smith, P.; Yi, Z.; Xue, S.; Xu, Y. Long-term Miscanthus cultivation enhances soil quality in degraded acidic red soils: A multi-indicator assessment. Industrial Crops and Products 2026, 242, 123001. [Google Scholar] [CrossRef]
| NaCl treatment (dSm-1) | μmoles g-1 FW | |
| Cajeme (salt resistance) | Yecora (salt sensitive) | |
| 0 | 0.21 | 0.21 |
| 75 | 0.33 | 0.57 |
| 150 | 1.96 | 3.75 |
| Plant species | Ecological type | μgg-1 FW* |
| Avicennia marina | Halophyte: Mangroves | 11–37 |
| Halopeplis perfoliata | Halophyte: Sabkhas | 56–281 |
| Limonium axillare | Halophyte: Sabkhas & coastlines | 103–810 |
| Tetraena qatarensis | Xero-halophyte | 419–1136 |
| Ochradenus baccatus | Xerophyte | 1277–1347 |
| Plant species | Family | Ecological type | Soil water content (% FC) I,* | ECe of the soil (dSm-1)* |
| Aeluropus lagopoides (Monocot) | Poaceae | Xerophyte; a grass can live in saline habitats as well | 40–66 | 45–50 |
| Anabasis setifera | Chenopodiaceae | Facultative halophyteII | 12–43 | 71–198 |
| Arhrocenmum macrostachyum | Amaranthaceae | Halophyte; it grows in coastal and inland salt marshes, alkali flats, and other habitats with saline soils | 24–48 | 27–198 |
| Avicennia marina | Acanthaceae | Halophyte; mangrove, well adapted at Coastal intertidal zones | Saturated Soil | Seawater |
| Capparis spinosa | Capparaceae | Xerophyte; its morphological and physiological features reflect its ecological type | 9–12 | 4–5 |
| Caroxylon imbricatum (previously Salsola imbricata) | Amaranthaceae | Xerohalophyte; a plant adapted to both drought and salinity; found in disturbed areas, saline soils, and coastal areas | 11–16 | 150–195 |
| Cocculus pedulus | Menispermaceae | Xerophyte | 9–12 | 4–5 |
| Halocnemum strobilaceum | Amaranthaceae | Halophyte; lives in tidal zones and salt flats, grows in coastal and inland salt marshes, alkali flats, and other habitats with saline soils | 34–61 | 12–198 |
| Halopeplis perfoliata | Amaranthaceae | Halophyte; found in highly saline sabkhas with sandy shelly soil | 34–50 | 12–178 |
| Haloxylon salicornicum | Amaranthaceae | Xerophyte; found in stony soil overlain with wind-blown sand | 9–12 | 80–90 |
| Heliotropium bacciferum | Boraginaceae | Xerophyte with minor salt tolerance, not a halophyte | 9–10 | 5–72 |
| Limonium axillare | Plumbaginaceae | Halophyte; found on coastline with saline shelly soil | 32–43 | 12–198 |
| Ochradenus baccatus | Resedaceae | Xerophyte; found in disturbed areas | 4–12 | 7–8 |
| Pulicaria crispa | Asteraceae | Xerophyte; found in low depressions and water catchment areas in sandy slightly saline soil | 5–8 | 4–6 |
| Pulicaria gnaphalodes | Asteraceae | Xerophyte; found in shallow depressions and rain pools with sandy stony soil, extremely woolly with pleasant smell | 5–8 | 4–6 |
| Salsola soda | Amaranthaceae | Halophyte; it thrives in saline environments, found in salt marshes, coastal areas, and saline soils | 43–71 | > 200 |
| Salicornia spp.III | Amaranthaceae | Halophytes; found on flooded landward side of mangrove associated with Salsola soda, and intidal zone and sabkha depressions | 43–71 | > 200 |
| Salsola rosmarinus (Syn. Seidlitzia rosmarinus) | Amaranthaceae | Xerohalophyte; can be regarded as both a halophyte and a xerophyte because it thrives in harsh, arid–saline desert habitats | 14–30 | 52–81 |
| Sporobolus spicatus (Monocot) | Poaceae | Halophyte; commonly known as salt grass due to its ability to thrive in high-salinity environments, has salt gland structures to excrete extra salts, grows in arid regions and can tolerate drought conditions | 32–44 | 107–128 |
| Suaeda aegyptiaca | Amaranthaceae | Xerohalophyte; is highly adapted to environments with both high salinity and low water availability, found in depressions with high water table and coastal areas | 11–16 | 150–195 |
| Suaeda vermiculata | Amaranthaceae | Halophyte; a xerophyte as well (drought-tolerant plant), found in moist saline soil and sabkhas | 8–43 | 32–142 |
| Tetraena qatarensisIV | Zygophyllaceae | Xerohalophyte; adapted to environments that are both dry and salty; many features should be discussed | 10–46 | 12–187 |
| Ziziphus nummularia | Rhamnaceae | Xerophyte; is highly adapted to and primarily found in hot, arid, and dry regions (xeric habitats); found in rawdahs with depth and fine soil |
9–12 | 4–5 |
| Plant species | Plant water content (%)* | Range of proline (μgg-1 fresh weight)* | Range of TSS (mgg -1) DW* | Range of TSN (μgg-1) DW* |
| Aeluropus lagopoides | 70–72 | 241–253 | 1.5–1.7 | 84–126 |
| Anabasis setiferaI (Syn. Salsola setifera) | 72–76 | 21–54 | 1.6–2.0 | 81–92 |
| Arhrocenmum macrostachyum | 81–89 | 10–112 | 4.0–14.1 | 79–165 |
| Avicennia marina | 66–69 | 11–37 | 2.2–6.0 | 40–63 |
| Capparis spinosa | 62–68 | 131–302 | No data | No data |
| Caroxylon imbricatum previously: Salsola imbricata | 80–81 | 38–82 | 1.6–3.1 | 77–128 |
| Cocculus pedulusII | 60–69 | 34–51 | No data | No data |
| Halocnemum strobilaceum | 71–80 | 19–73 | 1.8–2.5 | 48–146 |
| Halopeplis perfoliata | 78–88 | 56–281 | 2.9–3.5 | 46–88 |
| Haloxylon salicornicum | 70–78 | 91–119 | No data | No data |
| Heliotropium bacciferum | 69–76 | 151–192 | 10.6–16.6 | 73–140 |
| Limonium axillare | 64–72 | 103–810 | 4.7–8.0 | 67–169 |
| Ochradenus baccatus | 68–71 | 1277–1347 | 2.9–3.5 | 92–109 |
| Pulicaria crispa | 59–66 | 25–32 | 8.8–9.9 | 127–144 |
| Pulicaria gnaphalodes | 59–66 | 32–119 | 6.3–7.2 | 63–92 |
| Salicornia spp.III | 80–87 | 55–172 | No data | No data |
| Salsola rosmarinus (Syn. Seidlitzia rosmarinus) | 84–88 | 161–327 | 2.8–4.7 | 77–119 |
| Salsola soda | 83–89 | 132–185 | No data | No data |
| Sporopolus spicatus (Monocot) | 58–66 | 40–147 | 3.3–3.4 | 195–236 |
| Suaeda aegyptiaca | 86–88 | 80–317 | 3.9–4.3 | 100–119 |
| Suaeda vermiculata | 77–82 | 96–409 | 6.7–19.8 | 104–161 |
| Tetraena qatarensis | 81–87 | 419–1136 | 2.9–4.3 | 85–190 |
| Ziziphus nummularia | 62–67 | 87–97 | No data | No data |
| Species/Ecotype/Family | Phytoremediation activity | Compatible solute accumulation# | Remarks and observations | References |
|
Aeluropus lagopoides* Fam.: Poaceae |
Petroleum hydrocarbons, heavy metals such as Cd, Pb | Possible accumulation | C4 grass, exhibits some xerophytic adaptations | [12,116,117] |
| Arhrocenmum spp.*, 4 species, Amaranthaceae | Remediates heavy metals; petroleum hydrocarbons require further investigation | Little proline was detected; higher levels of soluble sugars and nitrogen were observed, and GB accumulation is possible | C3 plant, some adaptations to desert habitats | [12,18] |
|
Atriplex spp.*** 2 species Amaranthaceae |
Heavy metals: Cd, Cu, Ni, Pb, Zn; petroleum hydrocarbons need further investigation | Accumulate proline, GB, and soluble sugars in their natural habitats, ABA increases under salt stress | C3, C4 plants, xerophytes and halophytes are recognized among this genus | [118,119] |
|
Avicennia marina* Acanthaceae |
Heavy metals: Cd, Co, Cr, Cu, Fe, Ni, Zn, and petroleum hydrocarbons |
Little proline was detected; higher levels of soluble sugars were observed; GB needs further confirmation | C3 plant, mangrove tree | [12,120] |
|
Capparis spinosa** Capparaceae |
Accumulates heavy metals and degrades petroleum hydrocarbon | Accumulates proline, and QAC; needs further investigation for GB and soluble sugars | C3 plant, might resist salinity to a certain level | [35,121] |
| Cleome spp.** Cleomaceae (formerly Capparaceae) | Remediate heavy metals such as Cd and Cu, needs confirmation | Accumulate proline, GB, and soluble sugars in their natural habitats | C3 or C4 plants, depending on species, accumulates some fatty acids for detoxification | [12,122,123] |
| Cocculus pendulus** Menispermaceae | No information, needs to be tested | Little proline was detected, might accumulate GB, needs further investigation for soluble sugars | C3 plant | [35,45,124] |
| Cressa cretica* Convolvulaceae | Remediates some heavy metals, possibly remediates petroleum hydrocarbons |
Accumulates proline, needs further investigation for GB and soluble sugars | C4 plant | [12,125] |
| Cyperus spp. *, **, $, 3 species Cyperaceae | Remediates many heavy metals such as Al, Cd, Co, Cr, Cu, Fe, Hg, Mn, Ni, Pb, and Zn (phyto-stabilization of Ni), petroleum hydrocarbons | Accumulates proline, possibly GB, and soluble sugars | Mainly C4, some C3, species in Qatar are salt tolerant, xerophytes, and mesophytes | [12,126] |
| Frankenia pulverulenta* Frankeniaceae | Remediates heavy metals such as Cd, Cr, Cu, Ni, Sr, and Zn, petroleum hydrocarbons | Accumulates compatible solutes: proline, GB, and soluble sugars | C3 plant, found in moist saline soil | [12,127,128] |
| Halocnemum strobilaceum* Amaranthaceae | Accumulates heavy metals, such as Cd, Cu, Fe, Mn, Ni, Pb, Zn; needs further studies for petroleum hydrocarbons | Accumulates compatible solutes such as proline and soluble sugars | C4 plant, obligate halophyte, might accumulate GB | [1,12,129] |
| Halodule uninervis* Hydrocharitaceae | Accumulates heavy metals such as Cu, Fe, Ni, Pb; phyto-remediates petroleum hydrocarbons | Possible accumulation of compatible solutes; needs to be tested | C3 plant, marine hydrophyte seagrass, halophyte | [12,77,130] |
| Halopeplis perfoliata* Amaranthaceae | Remediates some heavy metals and may help remediate petroleum hydrocarbons | Accumulates proline and possibly other compatible solutes such as GB and soluble sugars; needs confirmation | C4 plant, succulent | [1,35,131] |
| Halopyrum mucronatum* Poaceae | Some heavy metals are accumulated; considered as bioindicator for Cr, Fe, Pb, and Zn; needs to be tested for phytoremediation of petroleum hydrocarbons | Accumulates GB and possibly other compatible solutes | C4 plant, coastal halophytic grass | [1,3,117,118] |
| Haloxylon salicornicum** Amaranthaceae | Accumulates heavy metals such as Cu, Fe, Mn, Zn; possibly phyto-remediates petroleum hydrocarbons | Little proline was detected; accumulates GB | C4 plant, native to arid and semi-arid desert region | [12,100,118,132] |
| Heliotropium spp., **, 8 species, Boraginaceae | Possible role in phytoremdiation of organic pollutants and heavy metals | Little proline was detected; large amounts of soluble sugars accumulated, needs to be tested for GB | C3 plants, can be considered as semi-xerophyte, might be found in coastal saline soil | [3,35,133,134,135] |
| Juncus rigidus***, Juncaceae | Phyto-remediates organic compounds, heavy metals, and saline soil | Might accumulate compatible solutes, further investigation is needed | C3 plant, shows some xerophytic features | [136,137] |
| Limonium axillare* Plumbaginaceae | Remediates saline soil and heavy metals | Proline and soluble sugars accumulate, some relatively compatible solutes such as such as β-alanine betaine & choline-O-sulphate | C3 plant, found in coastal saline habitats, moist lands | [12,115,138] |
| Mesembryanthemum spp. ***, 2 species, Aizoaceae | Could be used for phytoremediation of contaminated soil, needs testing | Might accumulate compatible solutes under stress, needs testing | CAM plant | [139,140] |
| Ochradenus baccatus**, Resedaceae | Needs testing for petroleum hydrocarbon phytoremediation | Accumulates compatible solutes such as proline, soluble sugars, and possibly GB | C3 plant | [1,3,35] |
| Polypogon monspeliensis*, Poaceae | Accumulates heavy metals, phyto-remediates petroleum hydrocarbons and saline soil | Needs testing for compatible solutes | C3 plant, possible accumulator of GB | [1,12,14,142,143] |
| Pulicaria spp.** 4 species, Asteraceae | Possible candidates for petroleum hydrocarbons, need testing | Little proline was detected, accumulate significant amounts of soluble sugars, possible accumulation of GB; needs testing | C3 plants, tolerate salinity | [35,45,144] |
| Salicornia spp. *, 3 species, Amaranthaceae | Possible phytoremediation candidates for petroleum hydrocarbon and heavy metals, need further investigation | Very strong evidence that these species accumulate compatible solutes such as proline, soluble sugars and GB; further investigation is needed | C3 plants, obligate halophytes, biochemical adaptation; these species have several applications in the food, feed, pharmaceutical, cosmetics, and bioenergy sectors | [35,145,146,147,148] |
|
Salsola spp., *, **, 9 species, Amaranthaceae |
Possible role in phytoremediation, especially heavy metals and saline soils, some species remediate heavy metals such as B, Cu, Mn, and Se; further investigations are needed | Accumulates proline and soluble sugars and might accumulate other compatible solutes such as GB; needs testing | C4 plants, including halophytes and xerophytes, some bioactive uses were reported; industrial and nutritional values | [35,149,150,151,152,153] |
| Sporobolus spicatus* (Monocot), Poaceae | Phytoremediates petroleum hydrocarbons, heavy metals, and saline soils | Accumulates compatible solutes such as proline and soluble sugars; GB might have a role in salt tolerance | C4 plant; proven efficient in controlling polluted soils | [12,154,155] |
| Suaeda spp. * 7 species, Amaranthaceae | Phytoremediate heavy metals and possibly petroleum hydrocarbons; testing is needed | Accumulates proline, soluble sugars, and possibly GB, some species proved to accumulate GB | C3, C4 plants are found in the genus, application of GB might improve plant growth in saline conditions | [35,92] |
| Tamarix spp.***, 5 species, Tamaricaceae | Phytoremediate heavy metals and his polycyclic aromatic hydrocarbons |
Accumulate significant amounts of compatible solutes such as proline and soluble sugars; GB accumulation needs confirmation | C3 plants | [12,156,157] |
| Tetraena qatarensis***, Zygophyllaceae | Accumulates heavy metals, and possible phytoremediation of petroleum hydrocarbons | Accumulates significant amounts of proline and les soluble sugars; GB accumulation needs confirmation, | C4 plant | [35,45,158,159] |
| Teucrium polium**, Lamiaceae | Accumulates heavy metals such as Co, Ni; phytoremediates petroleum hydrocarbons | Accumulates compatible solutes such as proline and soluble sugars | C3 plant | [12,160,161] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




