Submitted:
09 April 2026
Posted:
10 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Proposed Approaches for Dynamic Channel Measurement
3. Dynamic Channel Characteristic Analysis of CIC
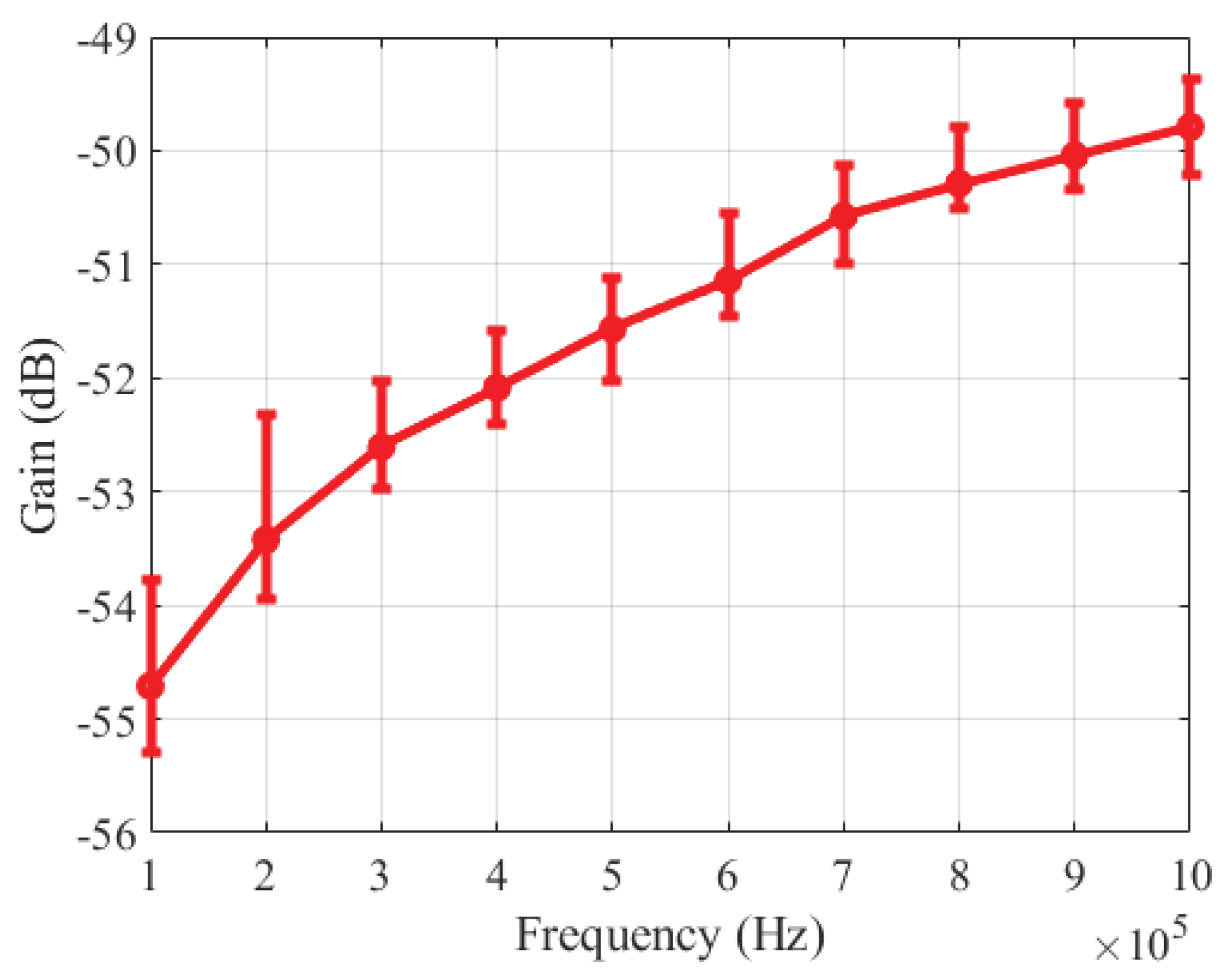
3.1. Amplitude-Frequency Characteristic
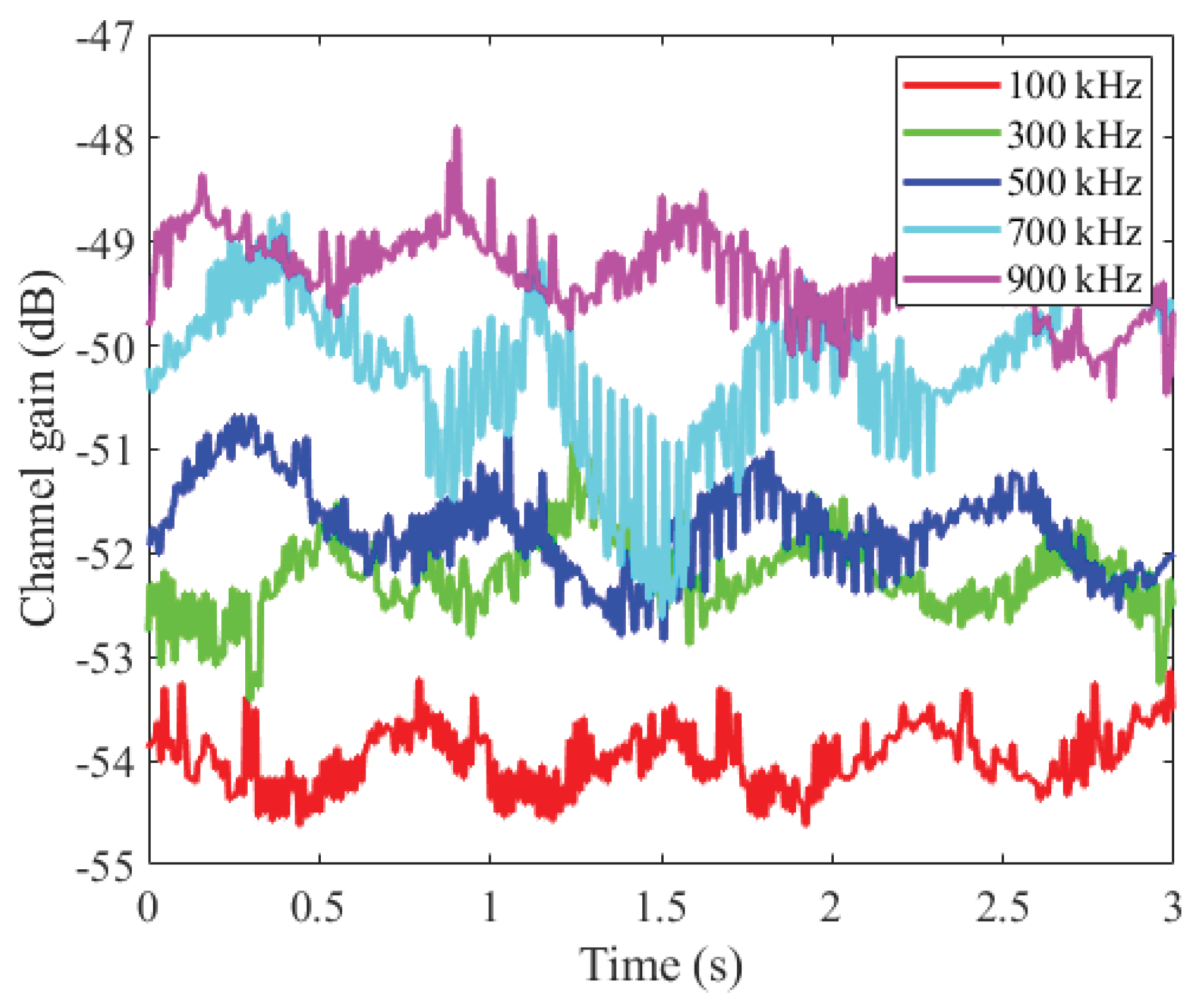
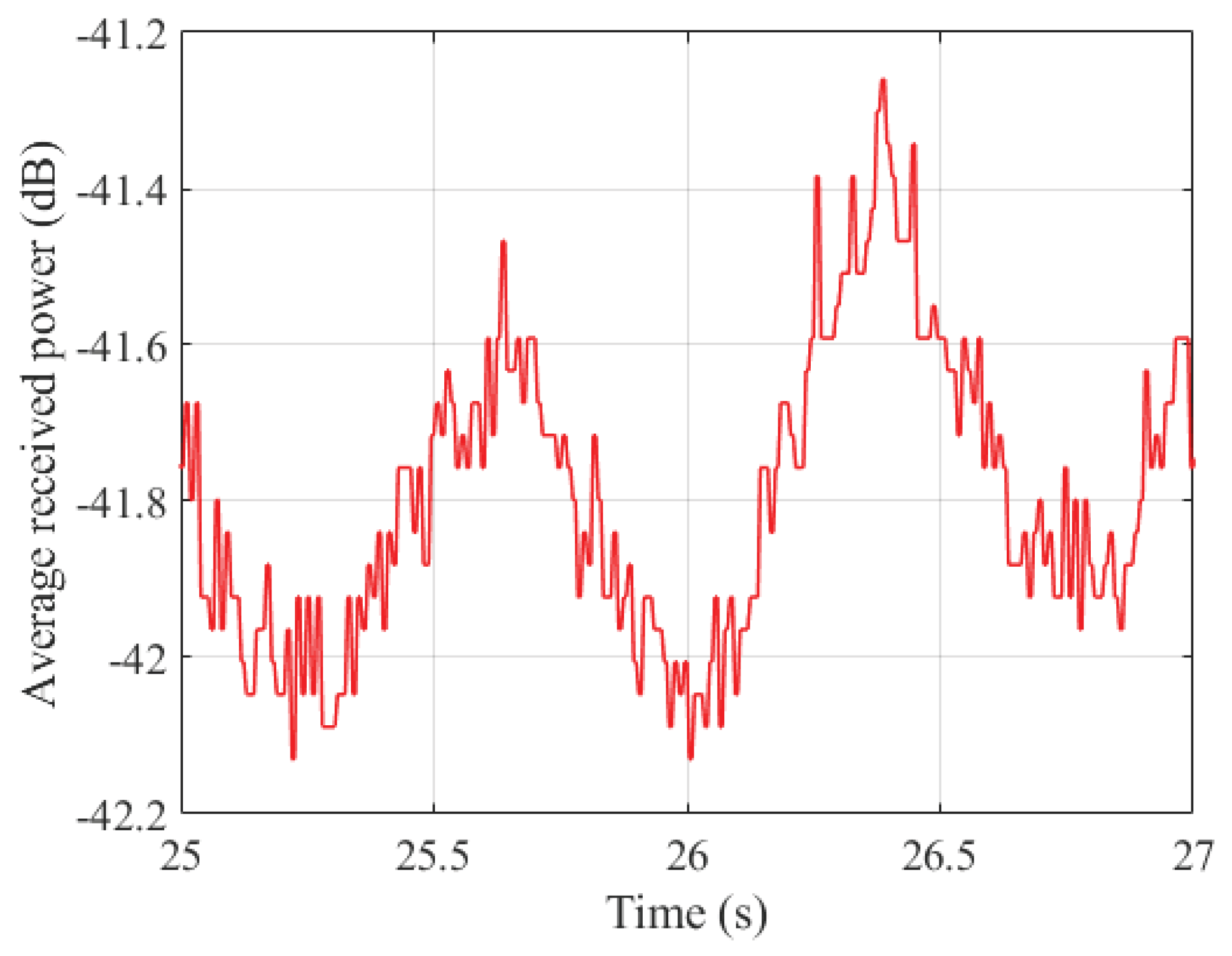
3.2. Amplitude-Time Characteristic
3.3. Analysis of Influencing Factors on Channel Characteristics
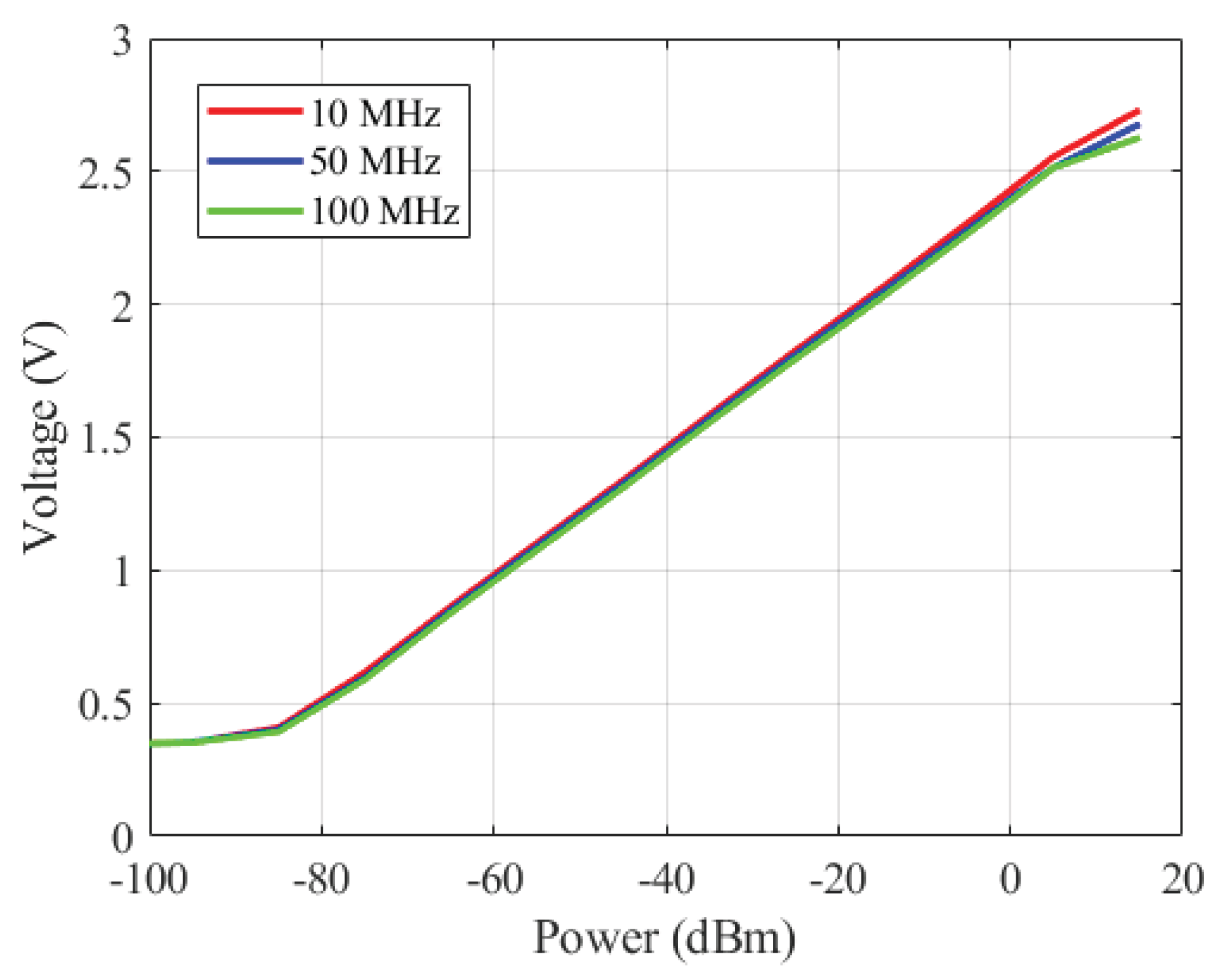
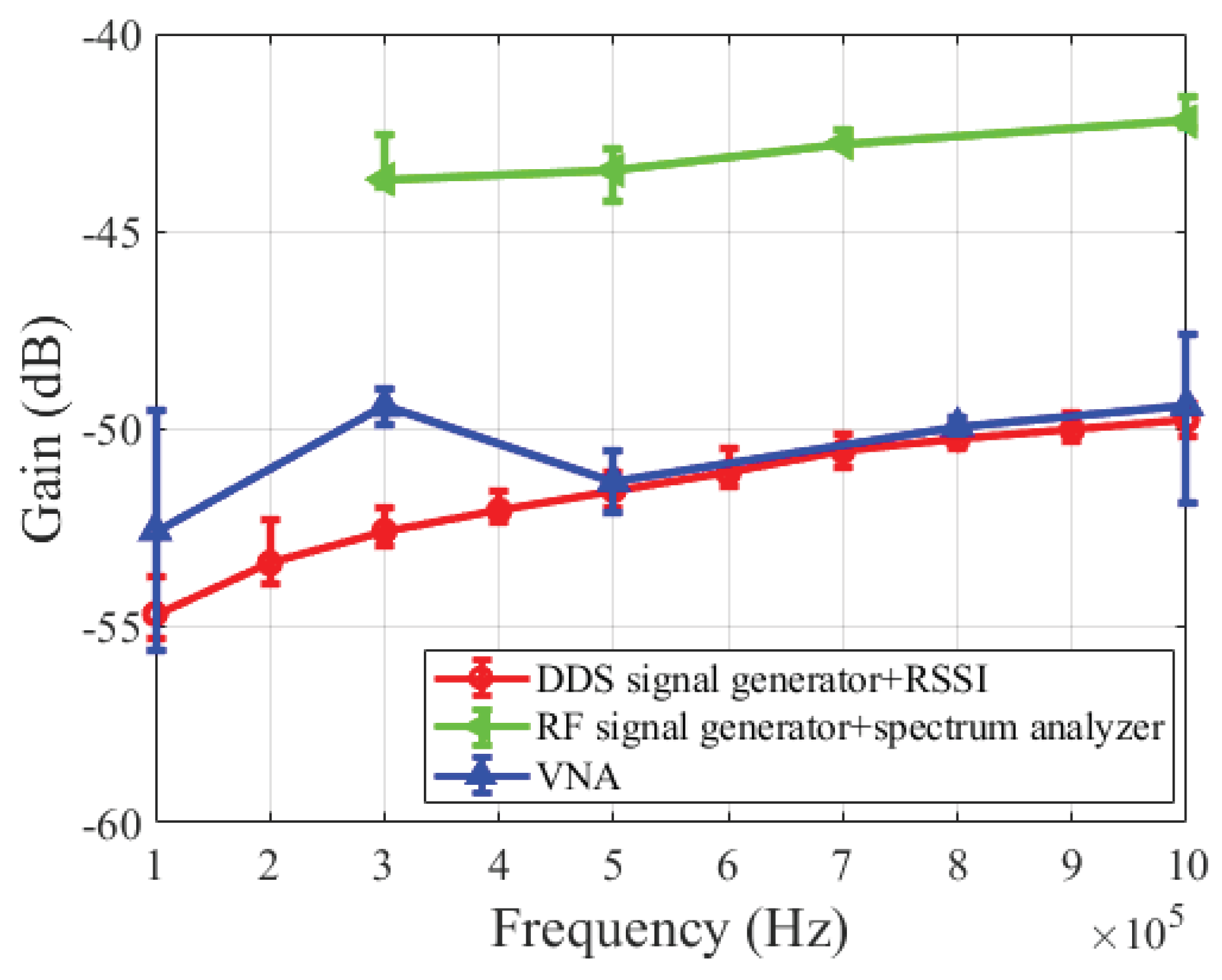
3.3.1. Instruments Difference
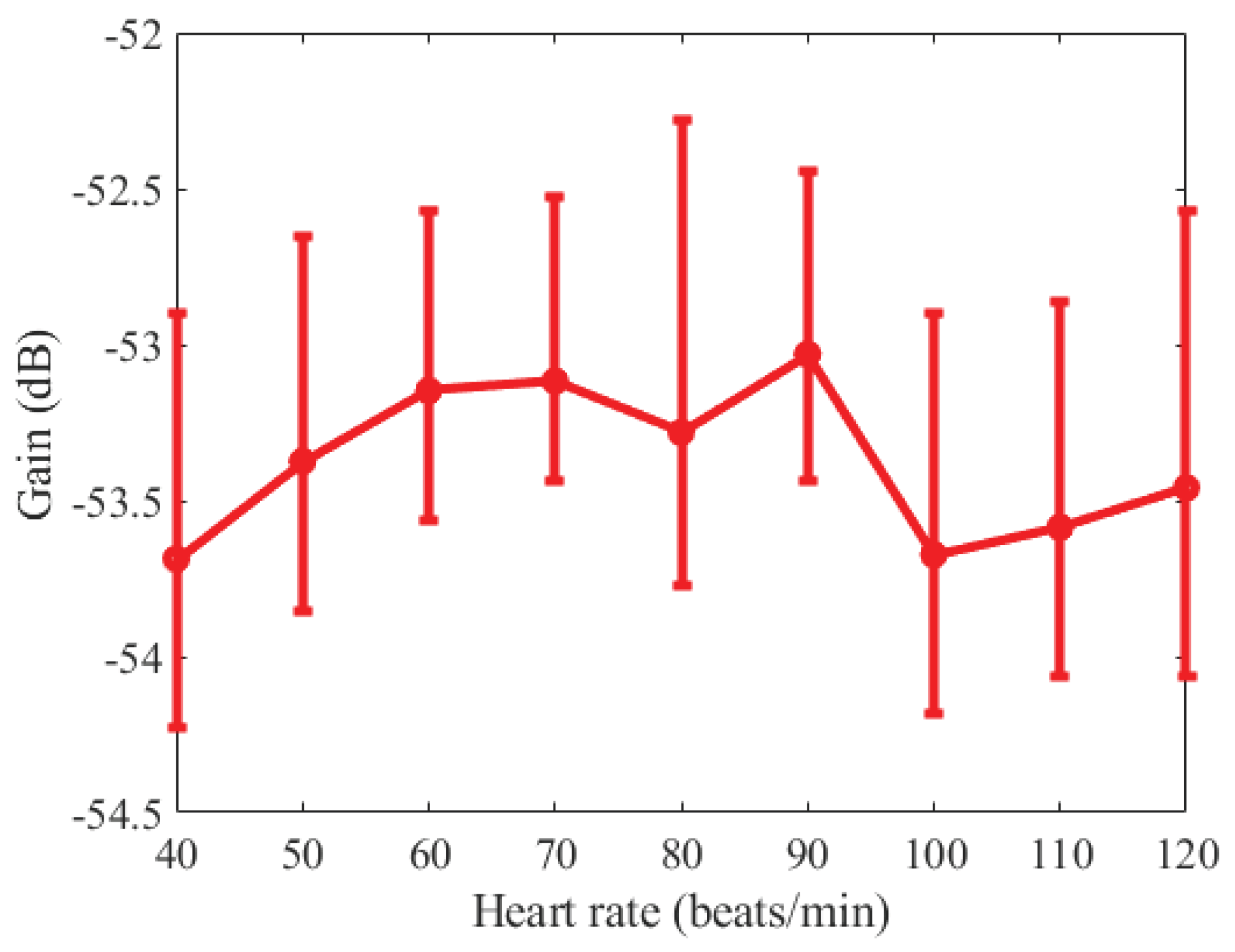
3.3.2. Heart Rate
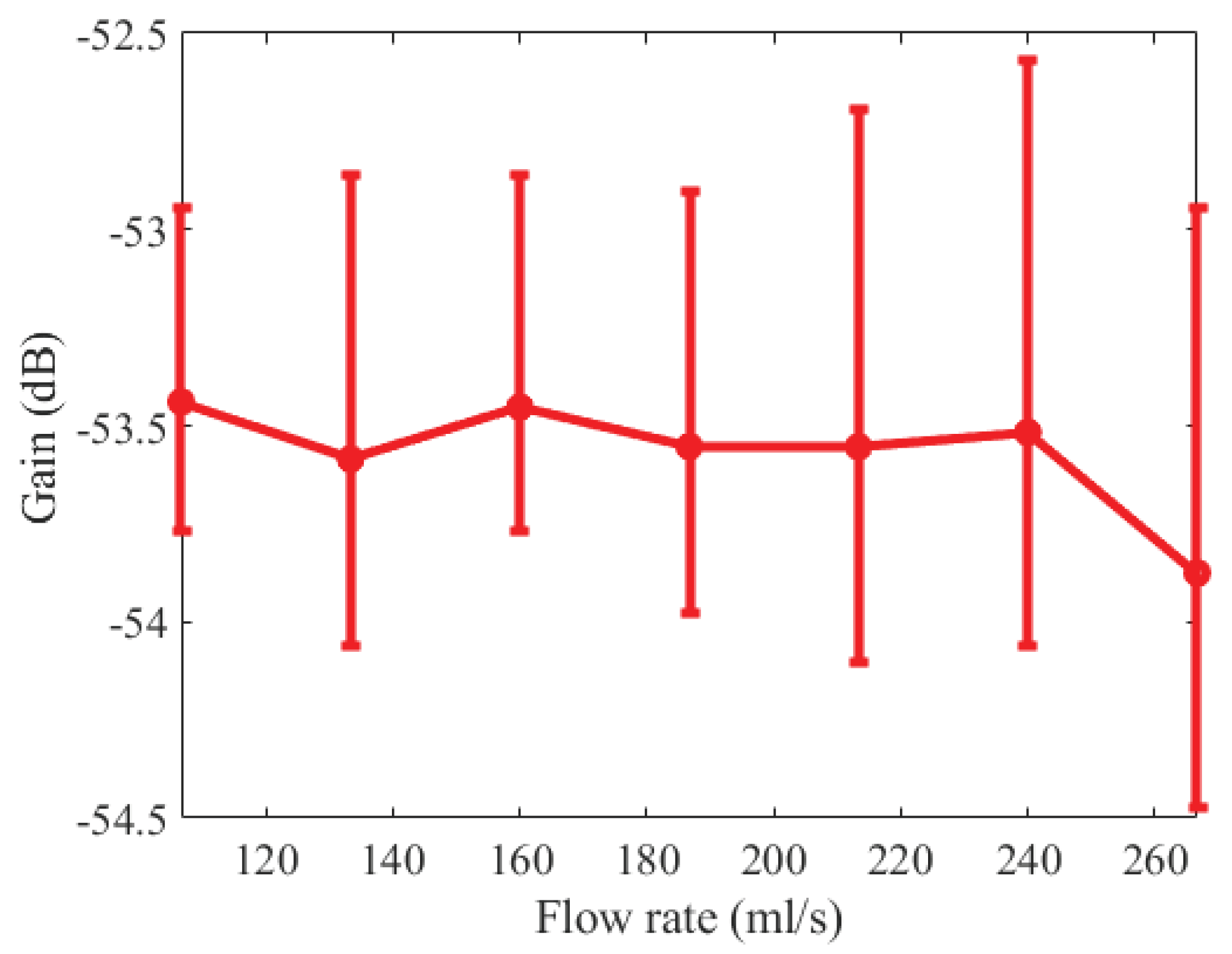
3.3.3. Flow Rate
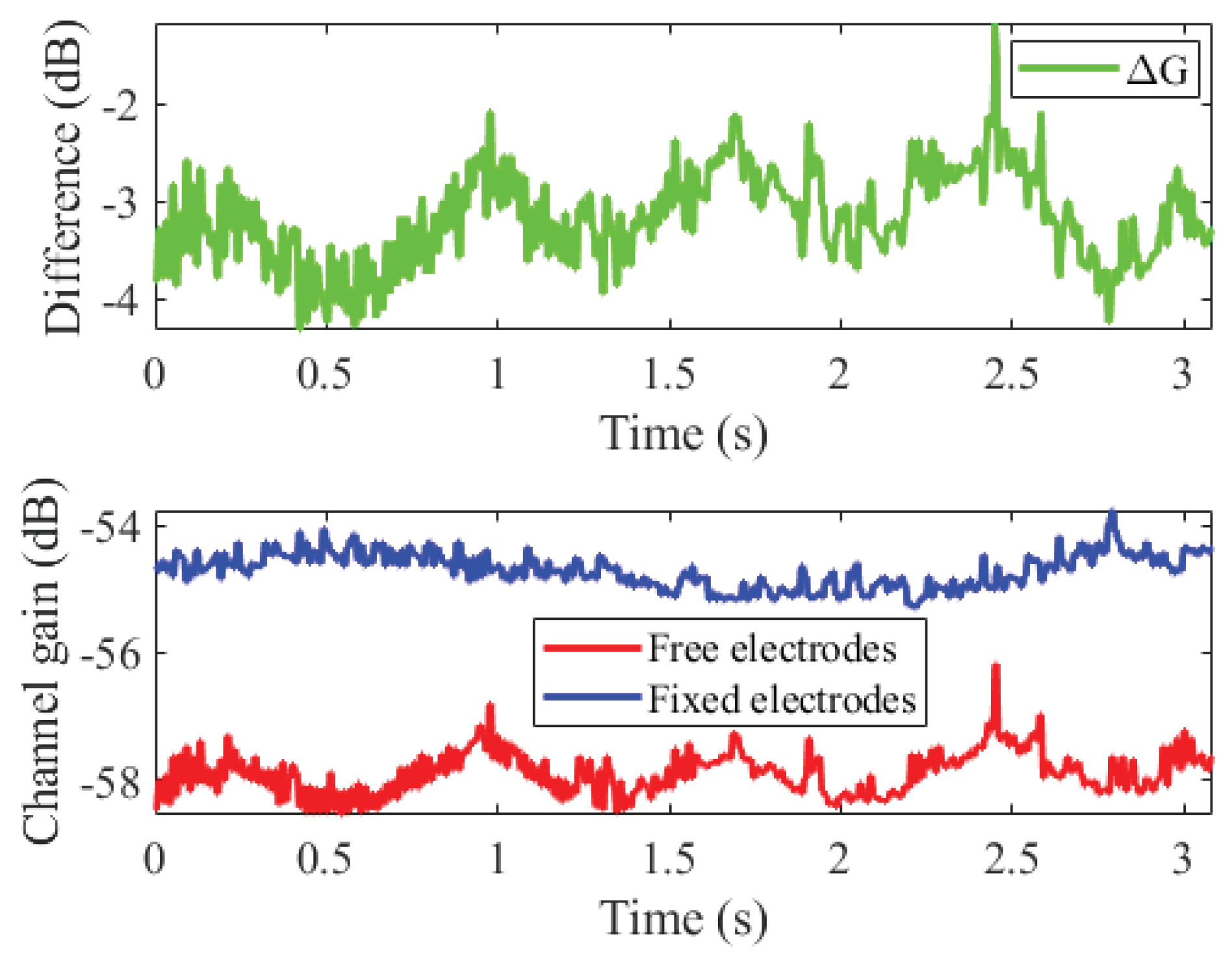
3.3.4. Fixed Electrodes vs Free Electrodes
4. Analysis of Channel Fading and Propagation Effects for CIC
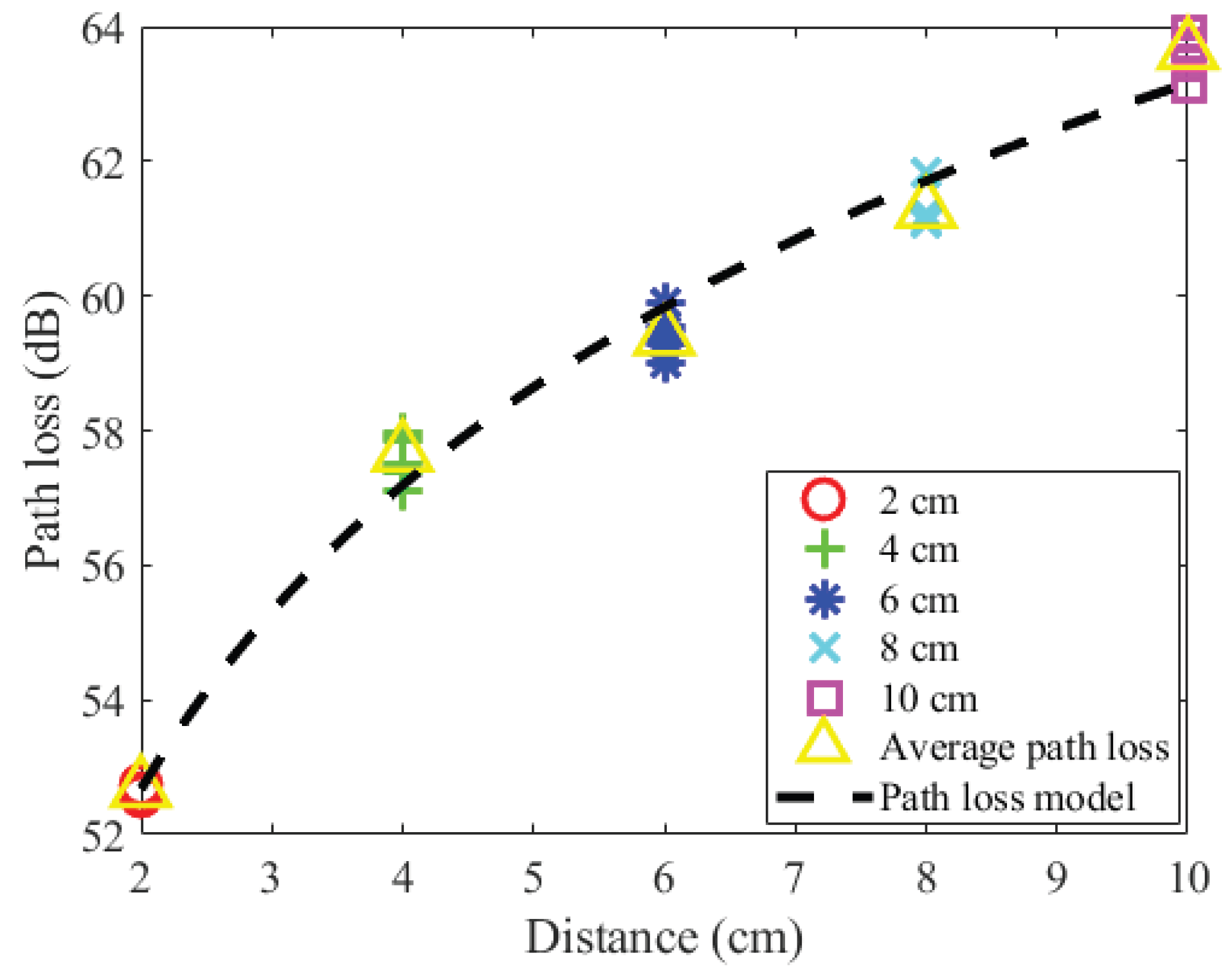
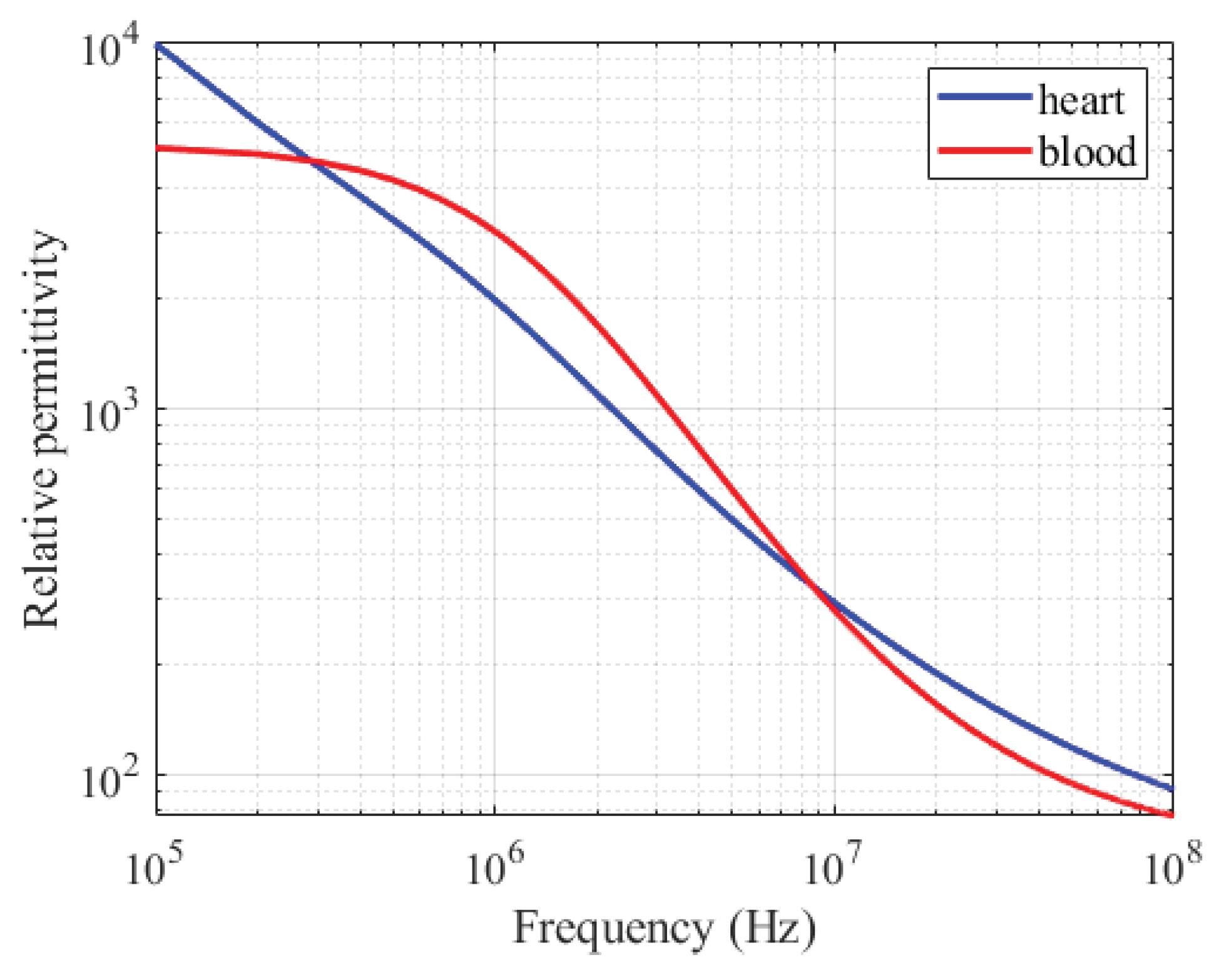
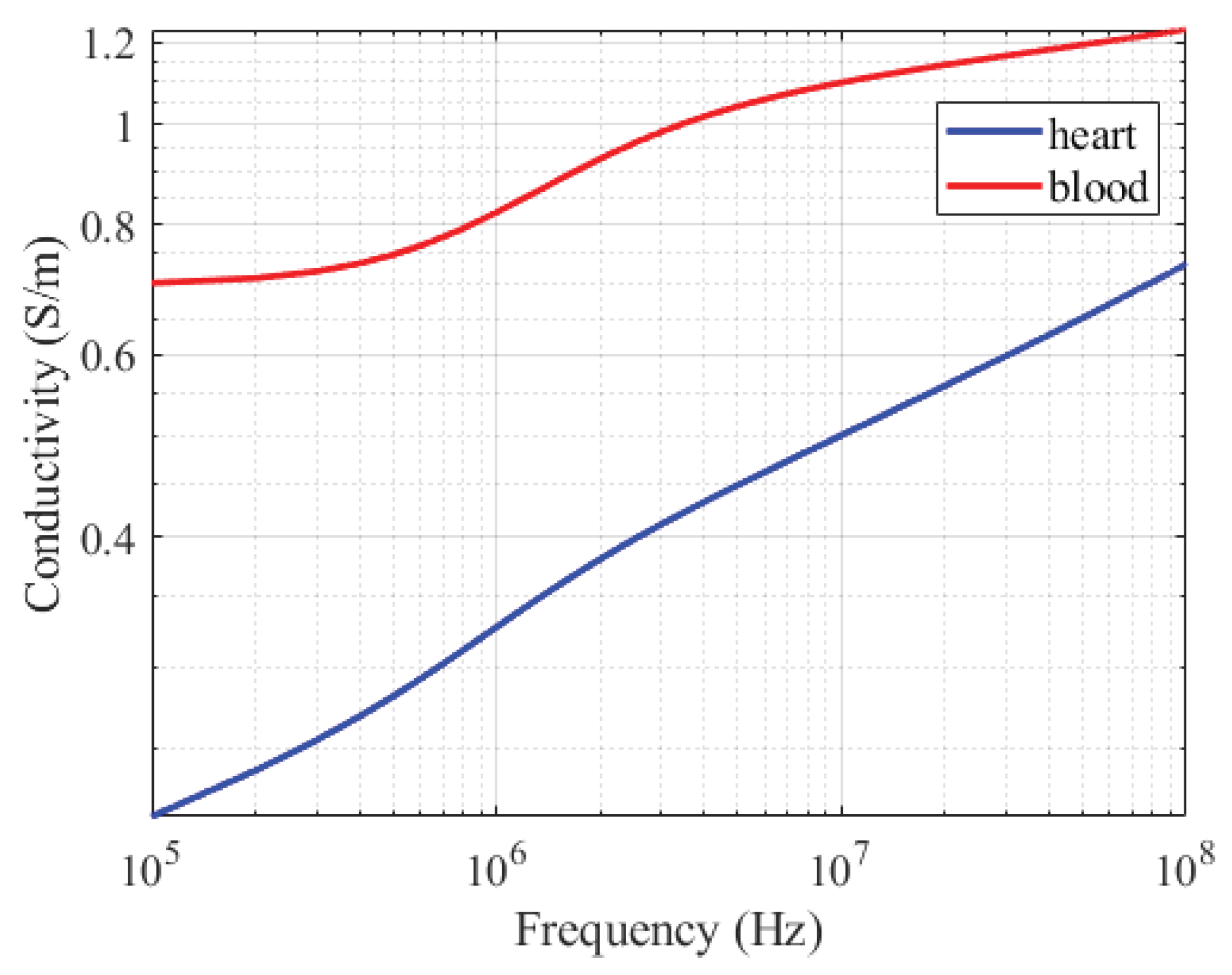
4.1. Path Loss
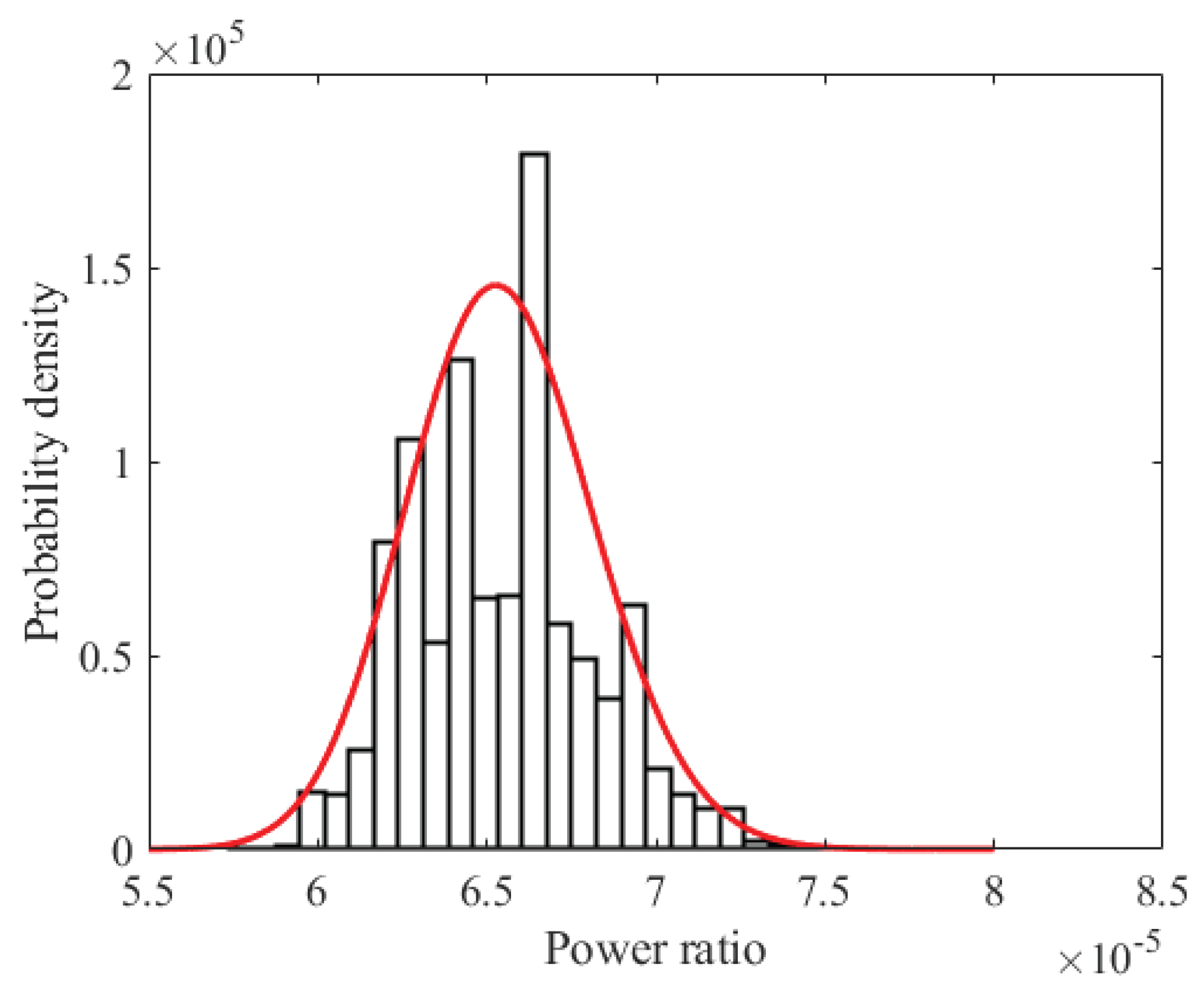
4.2. Shadow Fading
4.3. Multipath Fading
4.4. Doppler Fading
5. Analyze of Composite Fading Dynamic Channel Model for CIC
5.1. Proposed Composite Fading Dynamic Channel Model of CIC
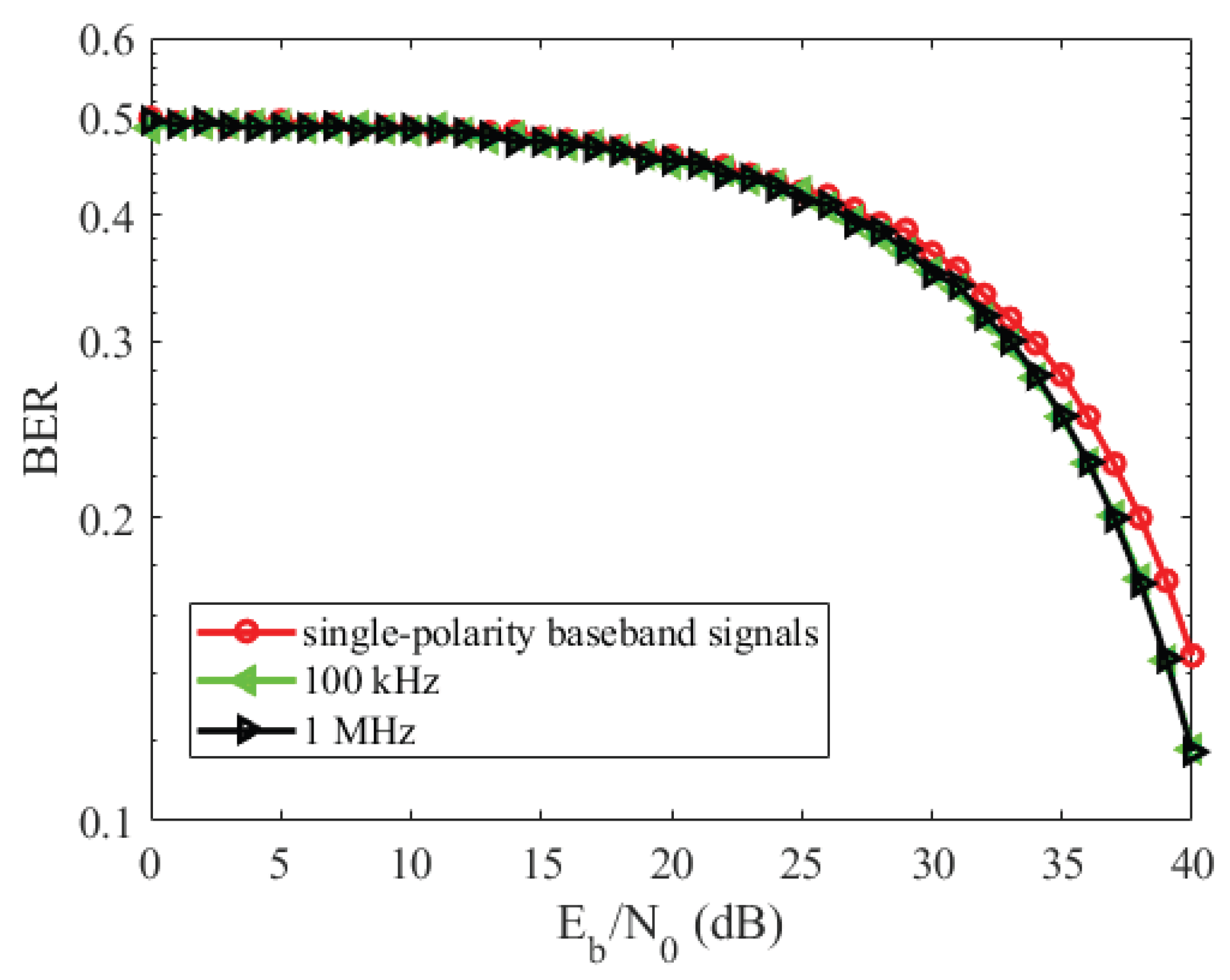
5.2. BER Analysis for Dynamic Channel Model of CIC
6. Limitations of Ex Vivo Experiment and Future Work
7. Conclusion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Saleem-Talib, S.; Hoevenaars, C.P.; Molitor, N.; van Driel, V.J.; van der Heijden, J.; Breitenstein, A.; van Wessel, H.; van Schie, M.S.; de Groot, N.M.; Ramanna, H. Leadless pacing: a comprehensive review. European Heart Journal 2025, 46, 1979–1990. [Google Scholar] [CrossRef] [PubMed]
- Maldari, M.; Albatat, M.; Bergsland, J.; Haddab, Y.; Jabbour, C.; Desgreys, P. Wide frequency characterization of intra-body communication for leadless pacemakers. IEEE Transactions on Biomedical Engineering 2020, 67, 3223–3233. [Google Scholar] [CrossRef] [PubMed]
- Bereuter, L.; Kuenzle, T.; Niederhauser, T.; Kucera, M.; Obrist, D.; Reichlin, T.; Tanner, H.; Haeberlin, A. Fundamental characterization of conductive intracardiac communication for leadless multisite pacemaker systems. IEEE transactions on biomedical circuits and systems 2018, 13, 237–247. [Google Scholar] [CrossRef] [PubMed]
- Khaleghi, A.; Noormohammadi, R.; Balasingham, I. Conductive impulse for wireless communication in dual-chamber leadless pacemakers. IEEE Transactions on Microwave Theory and Techniques 2020, 69, 443–451. [Google Scholar] [CrossRef]
- Noormohammadi, R.; Khaleghi, A.; Bergsland, J.; Balasingham, I. Conductive backscatter communication for dual-chamber leadless pacemakers. IEEE Transactions on Microwave Theory and Techniques 2022, 70, 2442–2450. [Google Scholar] [CrossRef]
- Liu, Y.; Gao, Y.; Chen, L.; Liu, T.; Yang, J.; Pun, S.; Vai, M.; Du, M. A variable-volume heart model for galvanic coupling-based conductive intracardiac communication. Sensors 2022, 22, 4455. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.; Wang, H.; Li, D.; Vai, M.I.; Pun, S.H.; Yang, J.; Du, M.; Gao, Y. A Time-Varying Equivalent Circuit Modeling and Measuring Approach for Intracardiac Communication in Leadless Pacemakers. IEEE Transactions on Biomedical Circuits and Systems 2024, 18, 872–884. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Wang, J.; Wang, H.; Huang, X.; Yang, J.; Gao, Y.; Li, H.C.; Vai, M.I.; Pun, S.H. A Dynamic High-Fidelity Equivalent Circuit Phantom for Intracardiac Communication in Pacemaker Indications. IEEE Transactions on Instrumentation and Measurement 2025, 74, 1–12. [Google Scholar] [CrossRef]
- Wang, H.; Li, D.; Vai, M.I.; Pun, S.H.; Yang, J.; Li, H.C.; Gao, Y. Analysis and Circuit Design of Imbalanced Impedance Channels for Conductive Intracardiac Communication. IEEE Transactions on Biomedical Circuits and Systems 2025, 19, 815–826. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Liu, Y.; Chen, Z.; Pun, S.H.; Vai, M.I.; Gao, Y. An investigation on conductive intracardiac communication dynamic channel gain during the cardiac cycle for leadless pacemakers. IEEE Journal of Electromagnetics, RF and Microwaves in Medicine and Biology 2022, 7, 82–89. [Google Scholar] [CrossRef]
- Li, D.; Wang, Z.; Wang, H.; Hang Pun, S.; Un Mak, P.; Zhang, A.; Liu, Y.; Li, H.C.; Gao, Y.; Du, M.; et al. Dynamic Path Gain Compensation for Enhancing Intracardiac Communication in Leadless Pacemakers. IEEE Transactions on Instrumentation and Measurement 2025, 74, 1–11. [Google Scholar] [CrossRef]
- Bose, P.; Khaleghi, A.; Balasingham, I. In-body and off-body channel modeling for future leadless cardiac pacemakers based on phantom and animal experiments. IEEE Antennas and Wireless Propagation Letters 2018, 17, 2484–2488. [Google Scholar] [CrossRef]
- Bose, P.; Khaleghi, A.; Mahmood, S.; Albatat, M.; Bergsland, J.; Balasingham, I. Evaluation of data telemetry for future leadless cardiac pacemaker. IEEE Access 2019, 7, 157933–157945. [Google Scholar] [CrossRef]
- Ryser, A.; Schmid, T.; Bereuter, L.; Burger, J.; Reichlin, T.; Niederhauser, T.; Haeberlin, A. Modulation scheme analysis for low-power leadless pacemaker synchronization based on conductive intracardiac communication. IEEE transactions on biomedical circuits and systems 2022, 16, 419–429. [Google Scholar] [CrossRef] [PubMed]
- Ryser, A.; Baeriswyl, C.; Moser, M.; Burger, J.; Reichlin, T.; Niederhauser, T.; Haeberlin, A. A Direct-Digital 40 μA 100 kb/s Intracardiac Communication Receiver With 250 μs Startup Time for Low Duty-Cycle Leadless Pacemaker Synchronization. IEEE Transactions on Biomedical Circuits and Systems 2024, 18, 1338–1353. [Google Scholar] [CrossRef] [PubMed]
- Bereuter, L.; Niederhauser, T.; Kucera, M.; Loosli, D.; Steib, I.; Schildknecht, M.; Zurbuchen, A.; Noti, F.; Tanner, H.; Reichlin, T.; et al. Leadless cardiac resynchronization therapy: An in vivo proof-of-concept study of wireless pacemaker synchronization. Heart rhythm 2019, 16, 936–942. [Google Scholar] [CrossRef] [PubMed]
- Bereuter, L.; Gysin, M.; Kueffer, T.; Kucera, M.; Niederhauser, T.; Fuhrer, J.; Heinisch, P.; Zurbuchen, A.; Obrist, D.; Tanner, H.; et al. Leadless dual-chamber pacing: a novel communication method for wireless pacemaker synchronization. JACC: Basic to Translational Science 2018, 3, 813–823. [Google Scholar] [PubMed]
- Gabriel, S.M.; Lau, R.W.; Gabriel, C. The dielectric properties of biological tissues: III. Parametric models for the dielectric spectrum of tissues. Physics in medicine and biology 1996, 41 11, 2271–93. [Google Scholar] [CrossRef] [PubMed]
- Ieee, B.E. IEEE Standard for Local and metropolitan area networksPart 15.6: Wireless Body Area Networks. IEEE 2012. [Google Scholar]
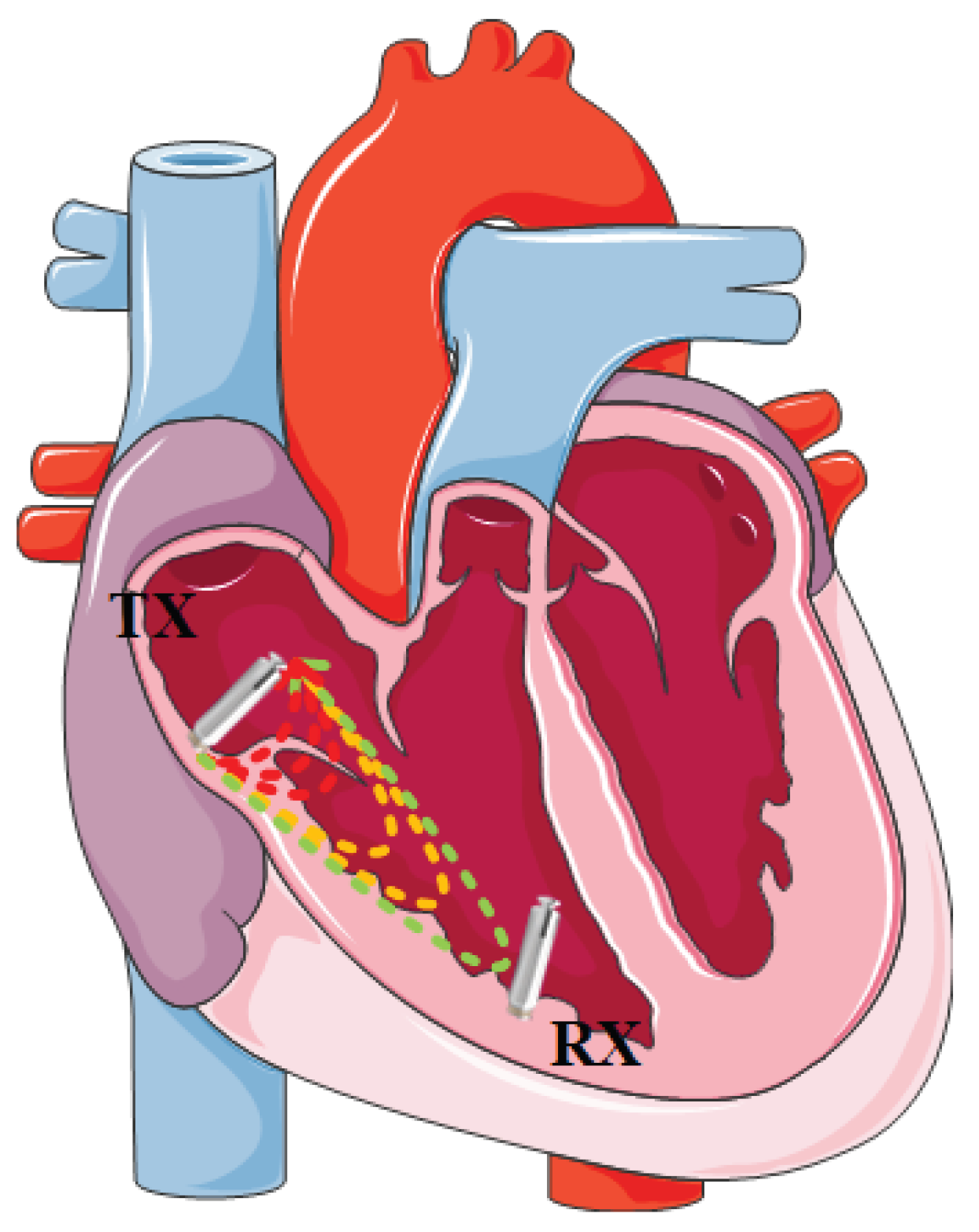
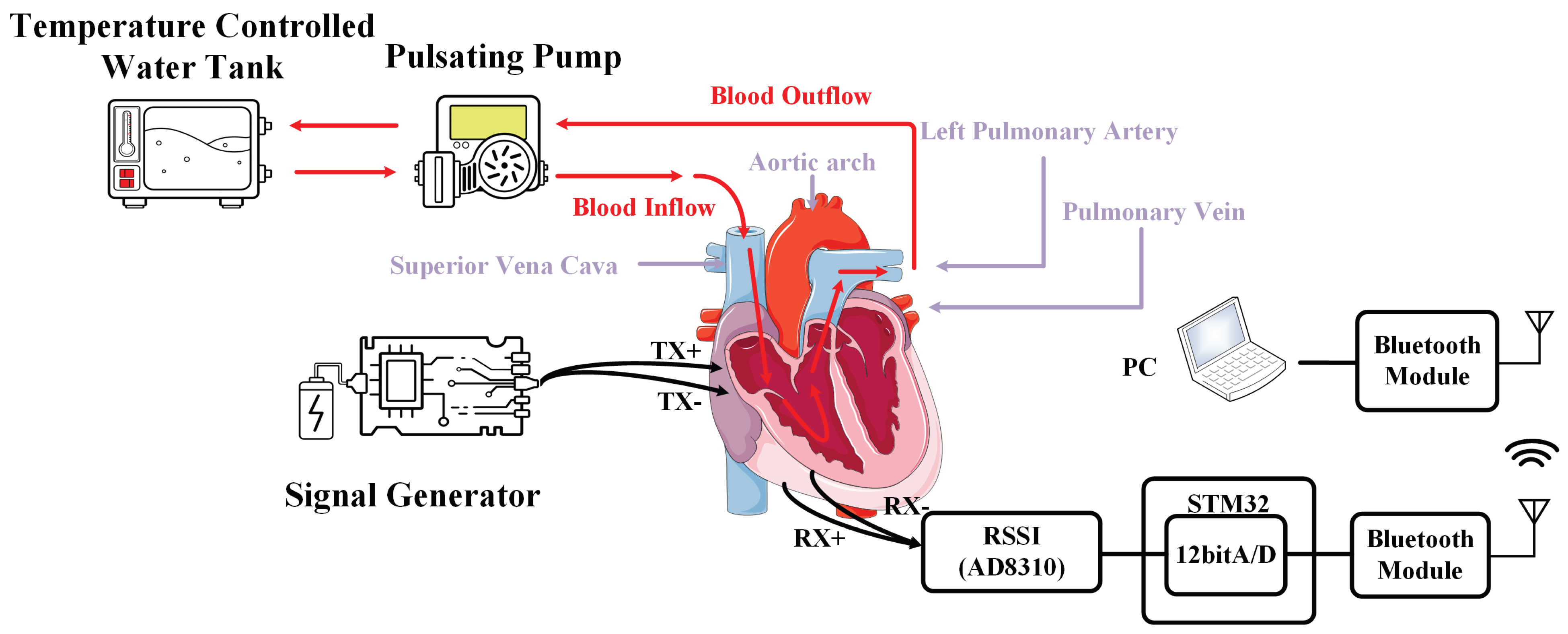
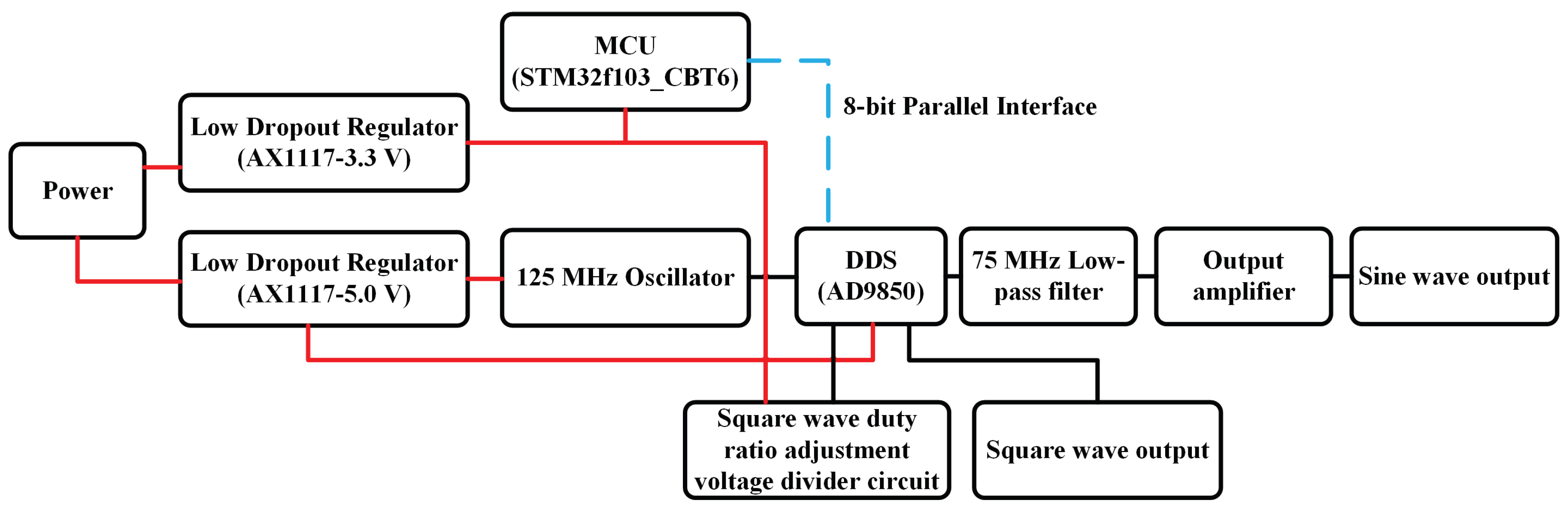

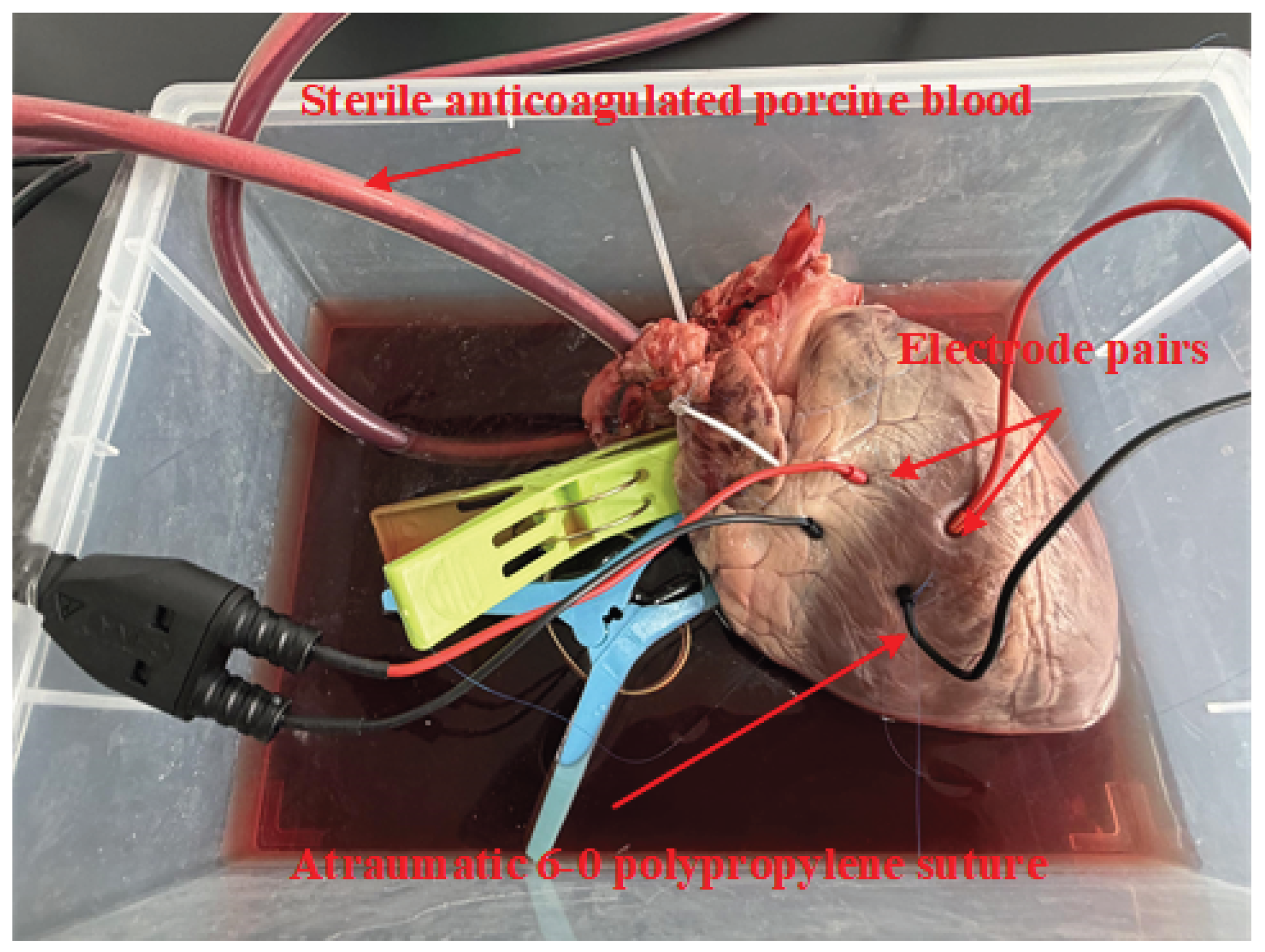
| Category | Contents | Work |
| Channel modeling and simulation approach | Finite element cardiac model and simulation | Mirko Maldari[2] |
| Finite element method (FEM) model for tissue | Lukas Bereuter[3] | |
| Finite-element method, frequency-domain, and finite-difference time-domain (FDTD) in CST | Ali Khaleghi[4] | |
| HUGO model in CST | Reza Noormohammadi[5] | |
| A variable-volume finite element cardiac model | Yiming Liu[6] | |
| Dynamic circuit model | A time-varying equivalent circuit model | Ziliang Wei[7] |
| A dynamic equivalent circuit model | Dongming Li[8] | |
| An equivalent circuit model simulating cardiac biomechanical impedance | Han Wang[9] | |
| Dynamic channel characteristics | Dynamic response of amplitude-frequency characteristics below 1 MHz | Lukas Bereuter[3] |
| Dynamic response of amplitude-frequency characteristics within 1 MHz-15 MHz | Ali Khaleghi[4] | |
| Channel gain variations during the cardiac cycle within 10 kHz-10 MHz | Liting Chen[10] | |
| Compensation method for dynamic channel gain | Dongming Li[11] | |
| CIC data transmission method | In-body and off-body channel path-loss models at 2.4 GHz | Pritam Bose[12] |
| Backscatter communication based on conductive coupling | Reza Noormohammadi[5] | |
| Modulation technology | Evaluation of different energy-efficient modulation schemes at 433 MHz | Pritam Bose[13] |
| Analysis for optimal communication parameters with On-Off-Keying and Manchester-encoded baseband transmission (BB-MAN) | Adrian Ryser[14] | |
| A synchronization algorithm to reduce the power consumption of wireless cardiac pacemakers | Adrian Ryser[15] | |
| Dynamic channel modeling approach | Amplitude-frequency characteristic within 100 kHz-1 MHz; Amplitude-time characteristic; Influencing factors: instruments difference, heart rate, flow rate and comparative experiments of free electrodes and fixed electrodes; Path loss; Shadowing effect; Multipath effect; Doppler spread; Dynamic channel modeling | This work |
| Distributions | Anderson-Darling statistic |
| Normal | 85.3607 |
| Exponential | 3517.8 |
| Weibull | 113.3626 |
| Extreme value | 117.6824 |
| Log-normal | 84.9720 |
| Rayleigh | 3369.06 |
| Nakagami-m | 549.7009 |
| Rician | 28537.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




