Submitted:
09 April 2026
Posted:
10 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
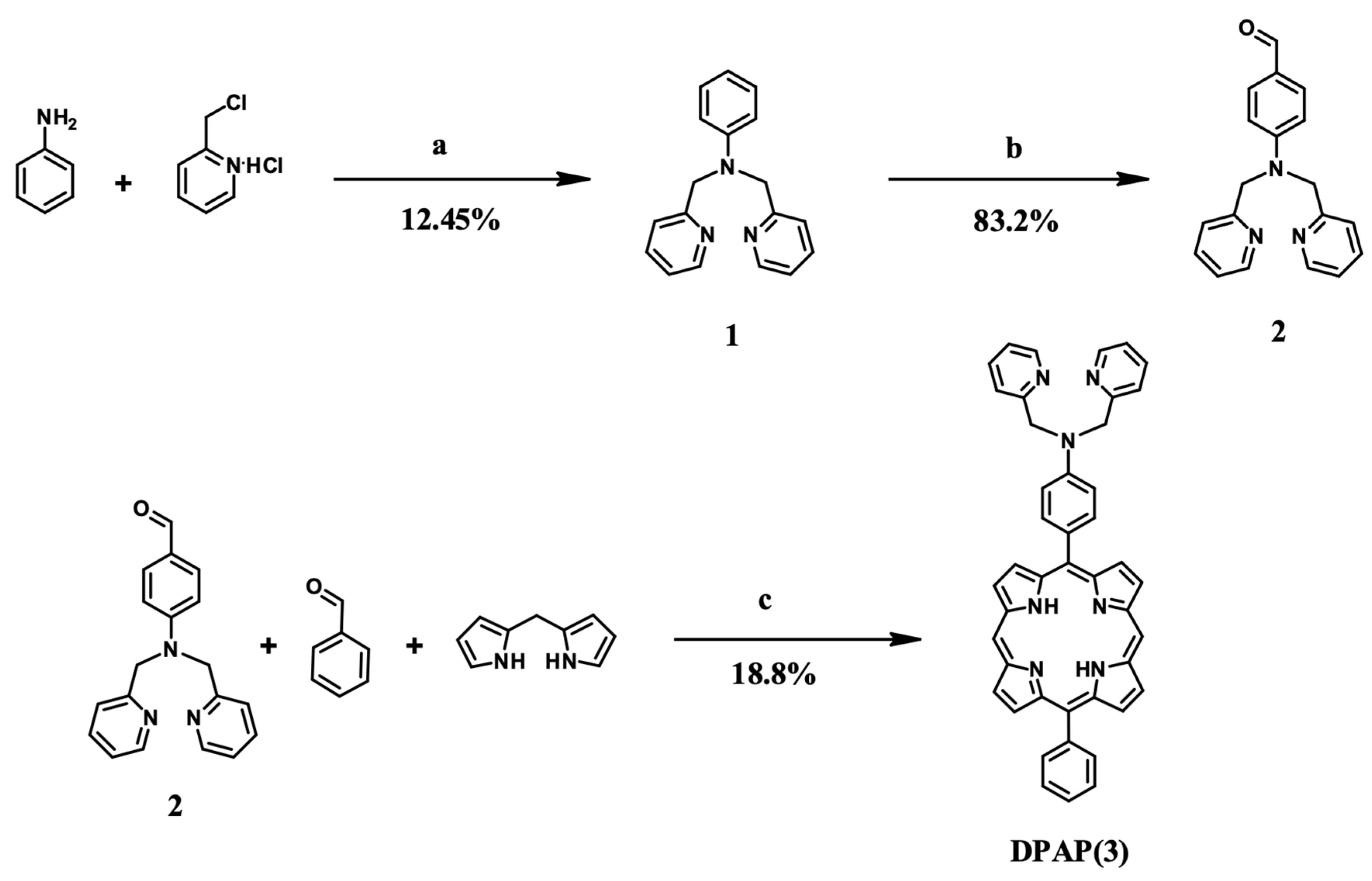
2.1. Synthesis and Characterization
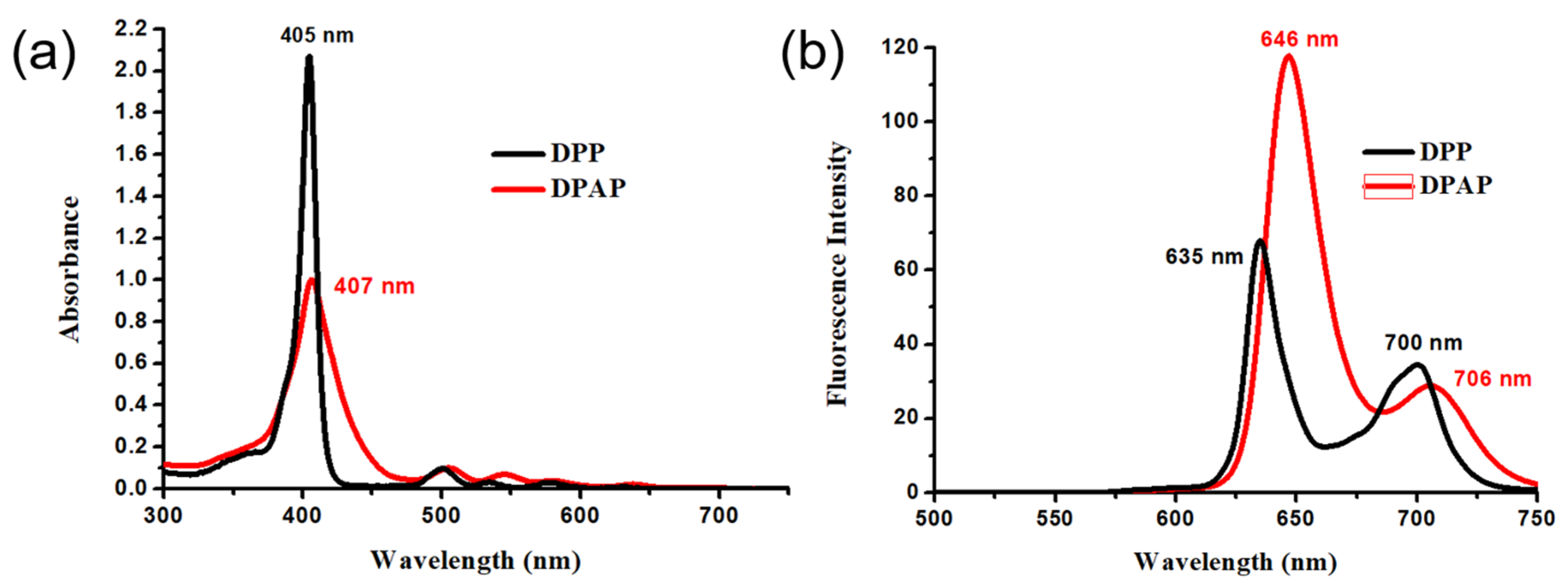
2.2. Optical Properties
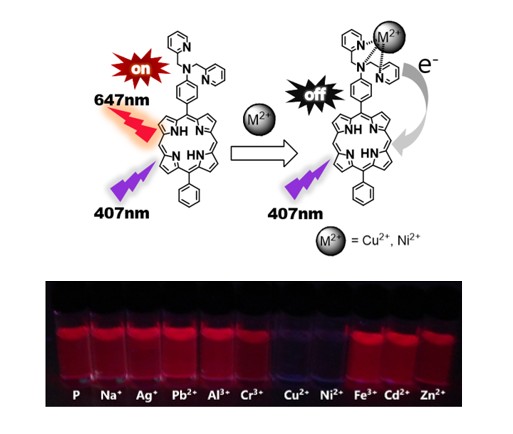
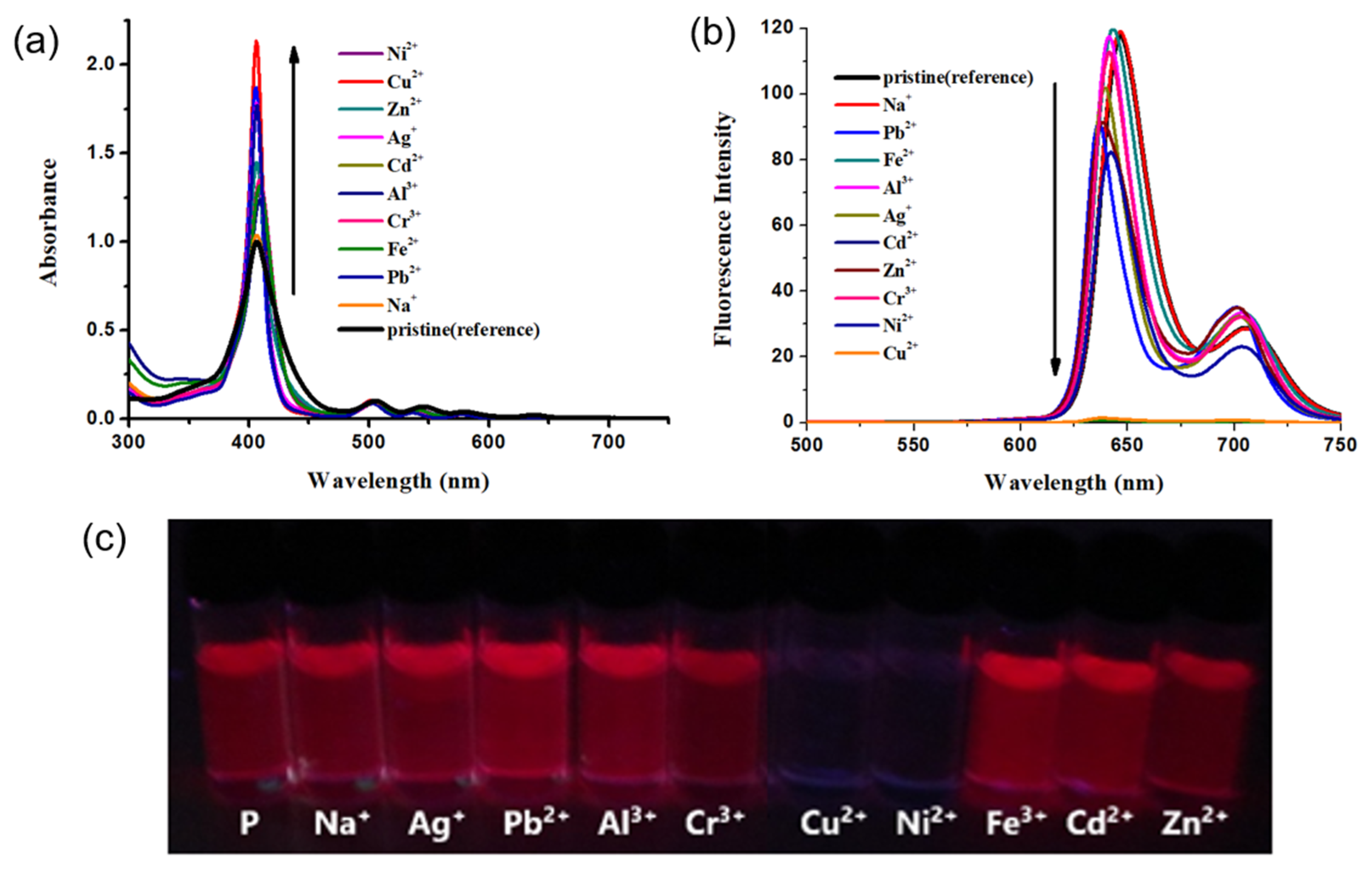
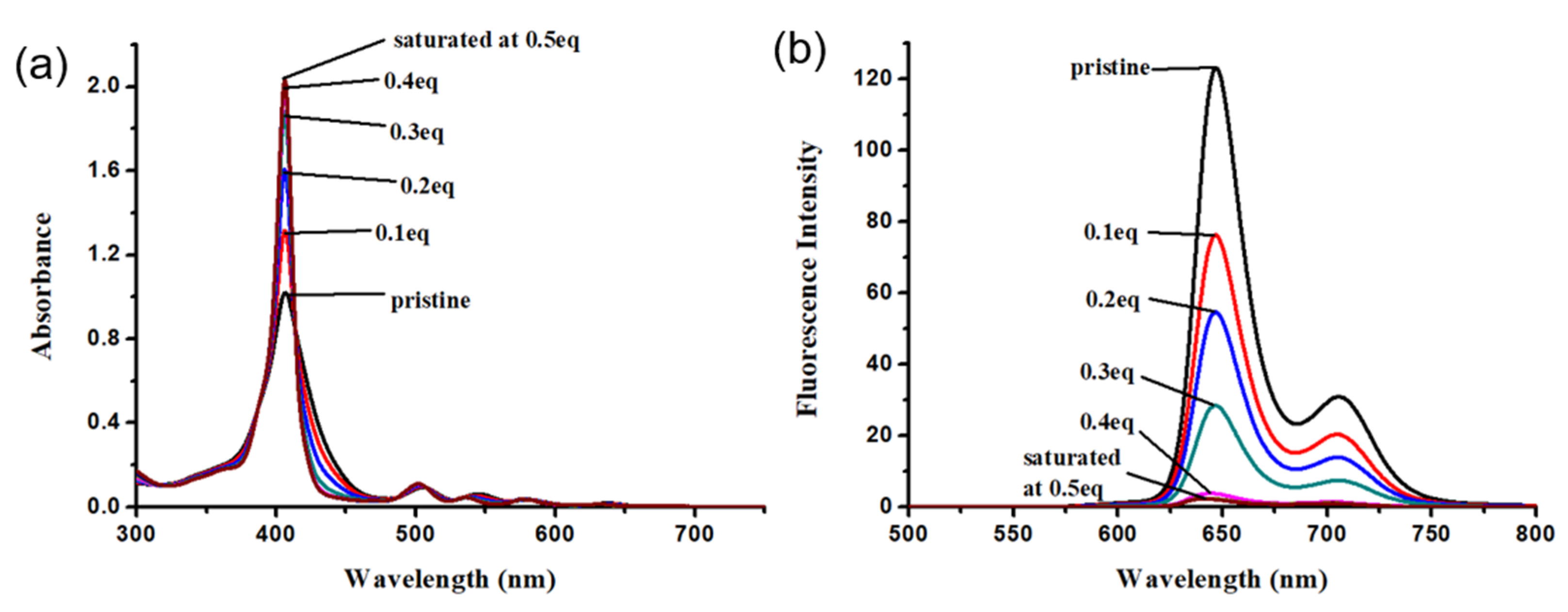
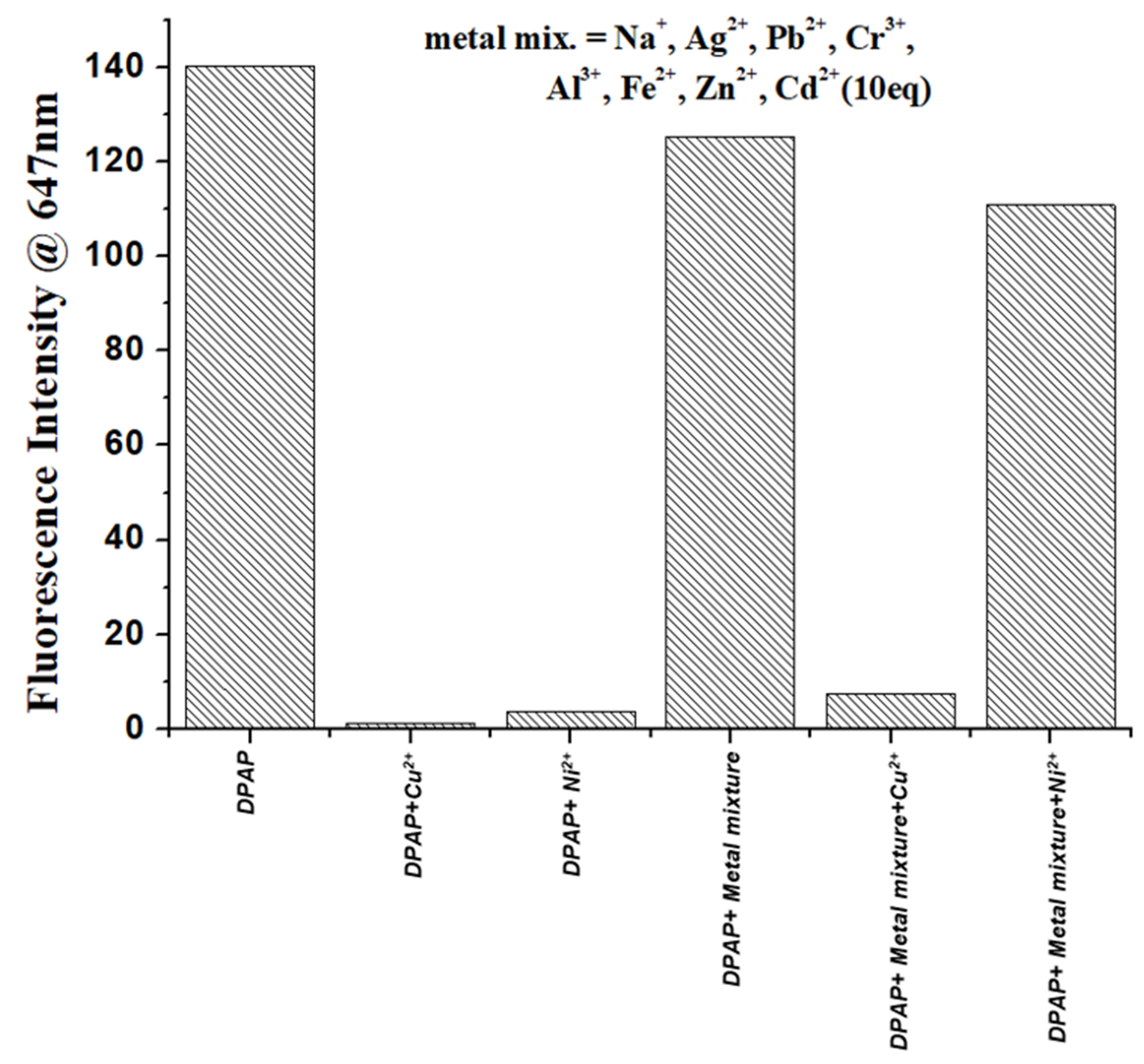
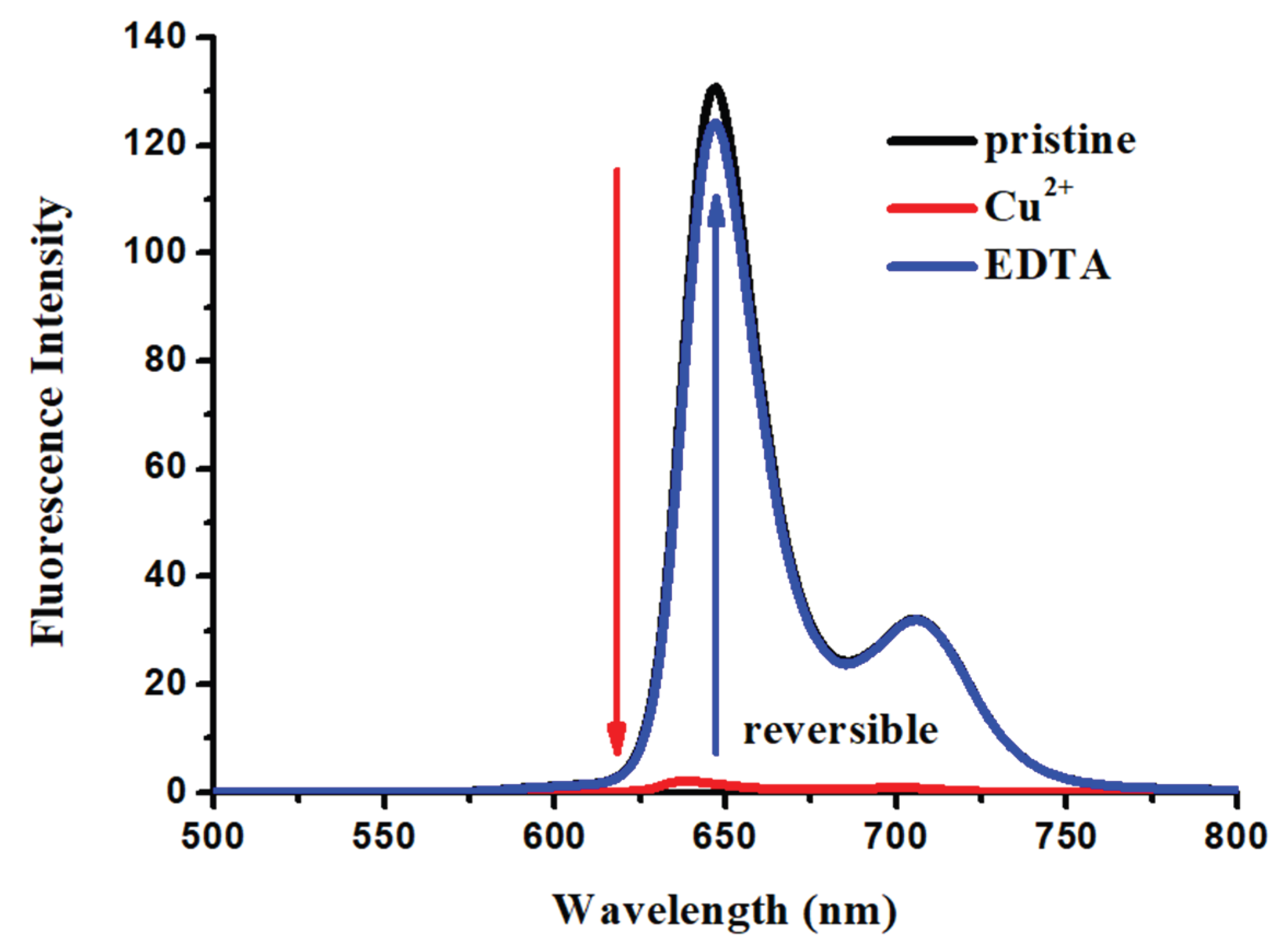
2.3. Effect of Metal Ions on the Absorption and Emission of DPAP
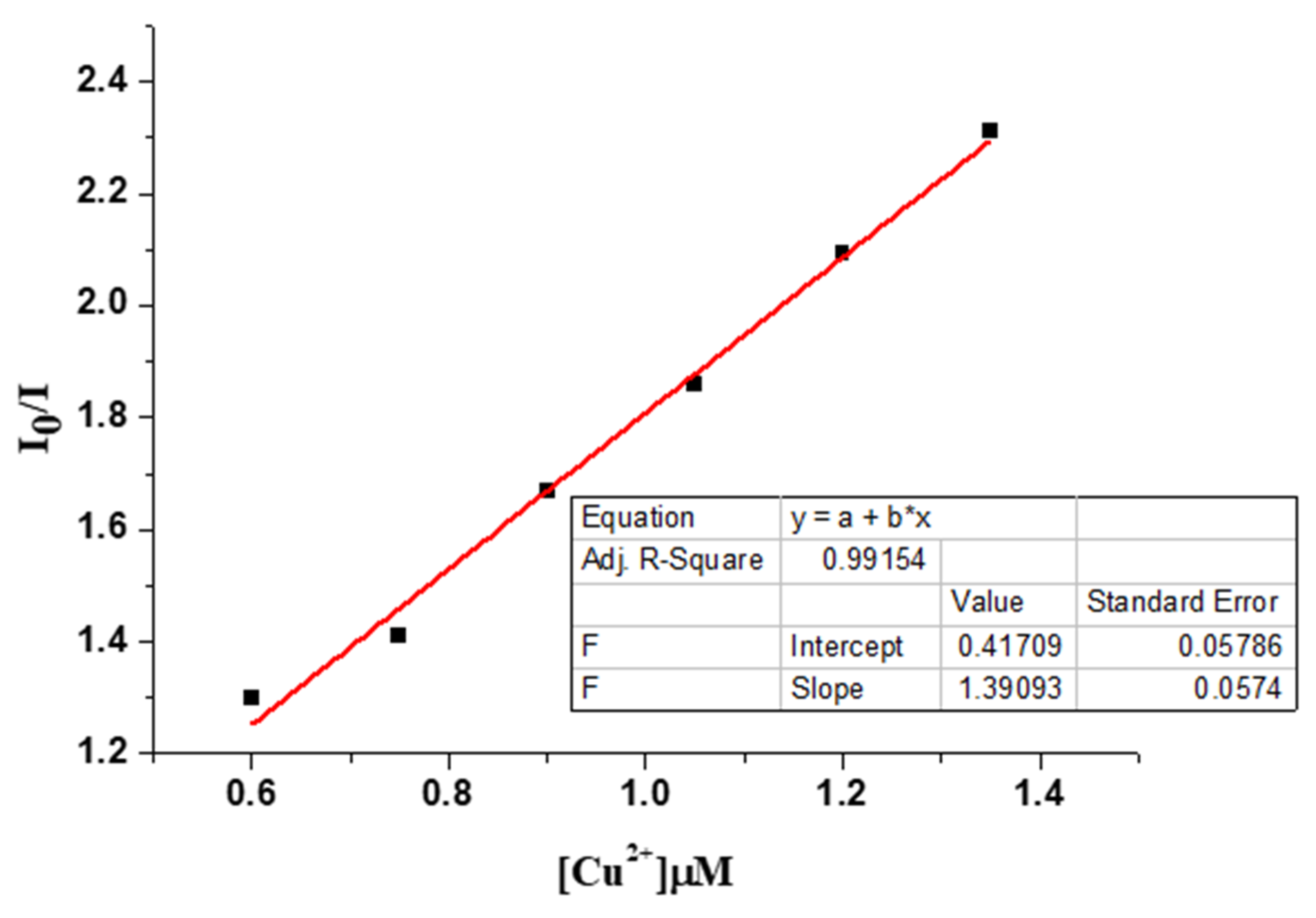
2.4. Binding Stoichiometry and Binding Affinity
3. Experimental
3.1. Materials
3.2. Measurements
3.3. DPAP Synthesis
3.4. Limit of Detection
3.5. Quantum Yield Measurements
3.6. Binding Constants and Stoichiometry
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sie, Y.W.; Li, C.L.; Wan, C.F.; Yan, H.; Wu, A.T. A novel fluorescence sensor for dual sensing of Hg2+ and Cu2+ ions. J. Photochem. Photobiol. A Chem. 2018, 353, 19–25. [Google Scholar] [CrossRef]
- Nekouei, F.; Nekouei, S. Determination of copper, nickel and cobalt in water and food samples by FAAS after separation and preconcentration using multiwalled carbon nanotubes modified by methyl-(2-pyridyl) ketone oxime. J. Anal. Chem. 2014, 8, 138–145. [Google Scholar]
- Neupane, L.N.; Oh, E.T.; Park, H.J.; Lee, K.H. Selective and sensitive detection of heavy metal ions in 100% aqueous solution and cells with a fluorescence chemosensor based on peptide using aggregation-induced emission. Anal. Chem. 2016, 88, 3333–3340. [Google Scholar] [CrossRef]
- Momidi, B.K.; Tekuri, V.; Trivedi, D.R. Multi-signaling thiocarbohydrazide-based colorimetric sensors for selective recognition of heavy metal ions in aqueous medium. Spectrochim. Acta A 2017, 180, 175–182. [Google Scholar] [CrossRef]
- Zhou, X. A highly selective fluorescent sensor for distinguishing cadmium from zinc ions based on a quinoline platform. Inorg. Chem. 2012, 51, 9226–9231. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, R.; Fan, C.; Liu, G.; Pu, S. A highly selective fluorescent sensor for Cd2+ based on a diarylethene with a 1,8-naphthyridine unit. Dyes Pigments 2017, 139, 208–217. [Google Scholar] [CrossRef]
- Cheng, T. Red-emission fluorescent probe sensing cadmium and pyrophosphate selectively in aqueous solution. Org. Lett. 2011, 13, 3656–3659. [Google Scholar] [CrossRef]
- Baslak, C.; Kursunlu, A.N. A naked-eye fluorescent sensor for copper(II) ions based on a naphthalene-conjugated BODIPY dye. Photochem. Photobiol. Sci. 2018, 17, 1091–1097. [Google Scholar] [CrossRef]
- Chae, J.B. Highly sensitive dansyl-based chemosensor for detection of Cu2+ in aqueous solution and zebrafish. ACS Omega 2019, 4, 12537–12543. [Google Scholar] [CrossRef]
- Tian, Z.; Cui, S.; Pu, S. A highly selective fluorescent sensor for dual detection of Zn2+ and F− based on a diarylethene. Tetrahedron Lett. 2016, 57, 2703–2707. [Google Scholar] [CrossRef]
- Chen, X. Aggregation-induced emission enhancement-based ratiometric fluorescent sensor for detecting trace uranyl ion and application in living-cell imaging. J. Lumin. 2017, 186, 301–306. [Google Scholar] [CrossRef]
- Huang, W.B. A porphyrin-based fluorescent probe for optical detection of toxic Cd2+ ion in aqueous solution and living cells. Dyes Pigments 2017, 143, 427–435. [Google Scholar] [CrossRef]
- Diana, R. Data on a real-time tripodal colorimetric/fluorescence sensor for multiple target metal ions. Data Brief 2018, 19, 2119–2125. [Google Scholar] [CrossRef]
- Zhao, Q. A highly selective on/off fluorescence sensor for cadmium(II). Inorg. Chem. 2011, 50, 10041–10046. [Google Scholar] [CrossRef] [PubMed]
- Surjeet, S. 2-(2,2-Bis-benzylamino-1-cyano-vinyl)-benzonitrile: A Selective Turn-off Fluorescent Cu2+ Sensor. Chemistry Select 2016, 1, 2576–2580. [Google Scholar]
- Li, Z.X. Fluoranthene-based pyridine as a fluorescent chemosensor for Fe3+. Inorg. Chem. Commun. 2011, 14, 1656–1658. [Google Scholar] [CrossRef]
- Han, A.; Liu, X.; Prestwich, G.D.; Zang, L. Fluorescent sensor for Hg2+ detection in aqueous solution. Sens. Actuators B 2014, 198, 274–277. [Google Scholar] [CrossRef]
- Narayanaswamy, N.; Govindaraju, T. Aldazine-based colorimetric sensors for Cu2+ and Fe3+. Sens. Actuators B 2012, 161, 304–310. [Google Scholar] [CrossRef]
- Bagheri, S.; Squitti, R.; Haertlé, T.; Siotto, M.; Saboury, A.A. Role of copper in the onset of Alzheimer’s disease compared to other metals. Front. Aging Neurosci. 2018, 9, 1–15. [Google Scholar] [CrossRef] [PubMed]
- He, X. A BODIPY-based colorimetric and fluorometric dual-mode chemosensor for Hg2+ and Cu2+. Sens. Actuators B 2014, 192, 29–35. [Google Scholar] [CrossRef]
- Kang, H.; Fan, C.; Xu, H.; Liu, G.; Pu, S. A highly selective fluorescence switch for Cu2+ and Fe3+ based on a diarylethene with a triazole-linked rhodamine 6G unit. Tetrahedron 2018, 74, 4390–4399. [Google Scholar] [CrossRef]
- Jin, X. Dual-functional probe based on rhodamine for sequential Cu2+ and ATP detection in vivo. Spectrochim. Acta A 2018, 204, 657–664. [Google Scholar] [CrossRef]
- Shinde, R.G. Fluorescence off–on signalling of esculetin in the presence of copper and thiol: implications in cellular thiol sensing. Photochem. Photobiol. Sci. 2018, 17, 1197–1205. [Google Scholar] [CrossRef]
- Tang, L. Rapid and highly selective relay recognition of Cu(II) and sulfide ions by a benzimidazole-based fluorescent sensor in water. Sens. Actuators B 2013, 185, 188–194. [Google Scholar] [CrossRef]
- Hu, S.; Zhang, S.; Hu, Y.; Tao, Q.; Wu, A. A selective pyrazoline-based fluorescent chemosensor for Cu2+ in aqueous solution. Dyes Pigments 2013, 96, 509–515. [Google Scholar] [CrossRef]
- Tang, L. Relay recognition of Cu2+ and S2− in water by a benzimidazole-derived fluorescent sensor via ESIPT modulation. Spectrochim. Acta A 2014, 122, 656–660. [Google Scholar] [CrossRef]
- Fan, J. A fluorescent ratiometric chemodosimeter for Cu2+ based on TBET and its application in living cells. Org. Lett. 2013, 15, 492–495. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Fan, C.; Liu, G.; Pu, S. A colorimetric and fluorescent sensor for Cu2+ and F− based on a diarylethene with a naphthalimide Schiff-base unit. Sens. Actuators B 2017, 239, 295–303. [Google Scholar] [CrossRef]
- Frangioni, J.V. In vivo near-infrared fluorescence imaging. Curr. Opin. Chem. Biol. 2003, 7, 626–634. [Google Scholar] [CrossRef] [PubMed]
- Sujatha, V. Use of tetra-ammonium tetrakis(4-sulphonatophenyl) porphyrin for Pseudomonas and Bacillus cell imaging. Int. J. Anal. Chem. 2010, 697528. [Google Scholar]
- Guo, B. Decoration of porphyrin with tetraphenylethene: converting aggregation-caused quenching to aggregation-induced emission enhancement. J. Mater. Chem. B 2016, 4, 4690–4698. [Google Scholar] [CrossRef] [PubMed]
- Shiraishi, Y.; Matsunaga, Y.; Hirai, T. Selective colorimetric sensing of Co(II) in aqueous media using a spiropyran-amide-dipicolylamine linkage. Chem. Commun. 2012, 48, 5485–5487. [Google Scholar] [CrossRef]
- Ahn, S.H. Novel cobalt(II) complexes containing N,N-di(2-picolyl)amine-based ligands: synthesis, characterization and application in methyl methacrylate polymerisation. J. Mol. Struct. 2016, 1113, 24–31. [Google Scholar] [CrossRef]
- Song, Y. Cadmium(II) complexes containing N′-substituted N,N-di(2-picolyl)amine ligands. J. Organomet. Chem. 2015, 783, 55–63. [Google Scholar] [CrossRef]
- Jeong, K. Diisopropyl fluorophosphate degradation activity using transition-metal dipicolylamine complexes. Appl. Organomet. Chem. 2018, 32, e4041. [Google Scholar] [CrossRef]
- Bussey, K.A. Synthesis, X-ray crystallography and catalytic activity of bis(2-pyridylmethyl)amine copper complexes in ATRA reactions. Polyhedron 2016, 114, 256–267. [Google Scholar] [CrossRef]
- Götzke, L. Nickel(II) and zinc(II) complexes of N-substituted di(2-picolyl)amine derivatives. Polyhedron 2011, 30, 708–714. [Google Scholar] [CrossRef]
- Milaeva, E.R. Redox-active metal complexes with dipicolylamine-ferrocenyl ligands. J. Organomet. Chem. 2017, 839, 60–70. [Google Scholar] [CrossRef]
- Azuma, Y. Dipicolylamine as a structural switching element for helical peptides. Org. Biomol. Chem. 2012, 10, 6062–6068. [Google Scholar] [CrossRef] [PubMed]
- Praktikum, P.; Quenching, F. Fluorescence quenching studies. Phys. Prakt. 2016, 1, 1–14. [Google Scholar]
- Kumari, N. Selective ppb-level detection of Cu2+ and Hg2+ using a micellar medium and application in cell imaging. ChemPlusChem 2014, 79, 1643–1652. [Google Scholar] [CrossRef]
- Zhang, L. Fluorescent binary ensemble based on pyrene derivative and SDS assemblies for discriminating metal ions. ACS Sens. 2017, 2, 1821–1830. [Google Scholar] [CrossRef]
- Ncube, P. Fluorescent sensing and determination of mercury(II) ions in water. Water SA 2014, 40, 175–182. [Google Scholar] [CrossRef]
- Milaeva, E.R. Metal complexes with functionalised dipicolylamine ligands containing antioxidant moieties. Dalton Trans. 2013, 42, 6817–6828. [Google Scholar] [CrossRef]
- Lindsey, J.S. Rothemund and Adler–Longo reactions revisited: synthesis of tetraphenylporphyrins under equilibrium conditions. J. Org. Chem. 1987, 52, 827–836. [Google Scholar] [CrossRef]
- Quantum Bioapplications, An introduction to fluorescence spectroscopy. Chem 2007, 312.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



