Submitted:
08 April 2026
Posted:
13 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Databases
2.2. Search Strategy, Inclusion and Exclusion Criteria
2.3. Literature Search
3. Results
3.1. Annual Publication Output
3.2. “Core” Journals Publishing on Leptin in Colorectal Malignancy
3.3. “Core” Journals Publishing on Adiponectin in Colorectal Malignancy
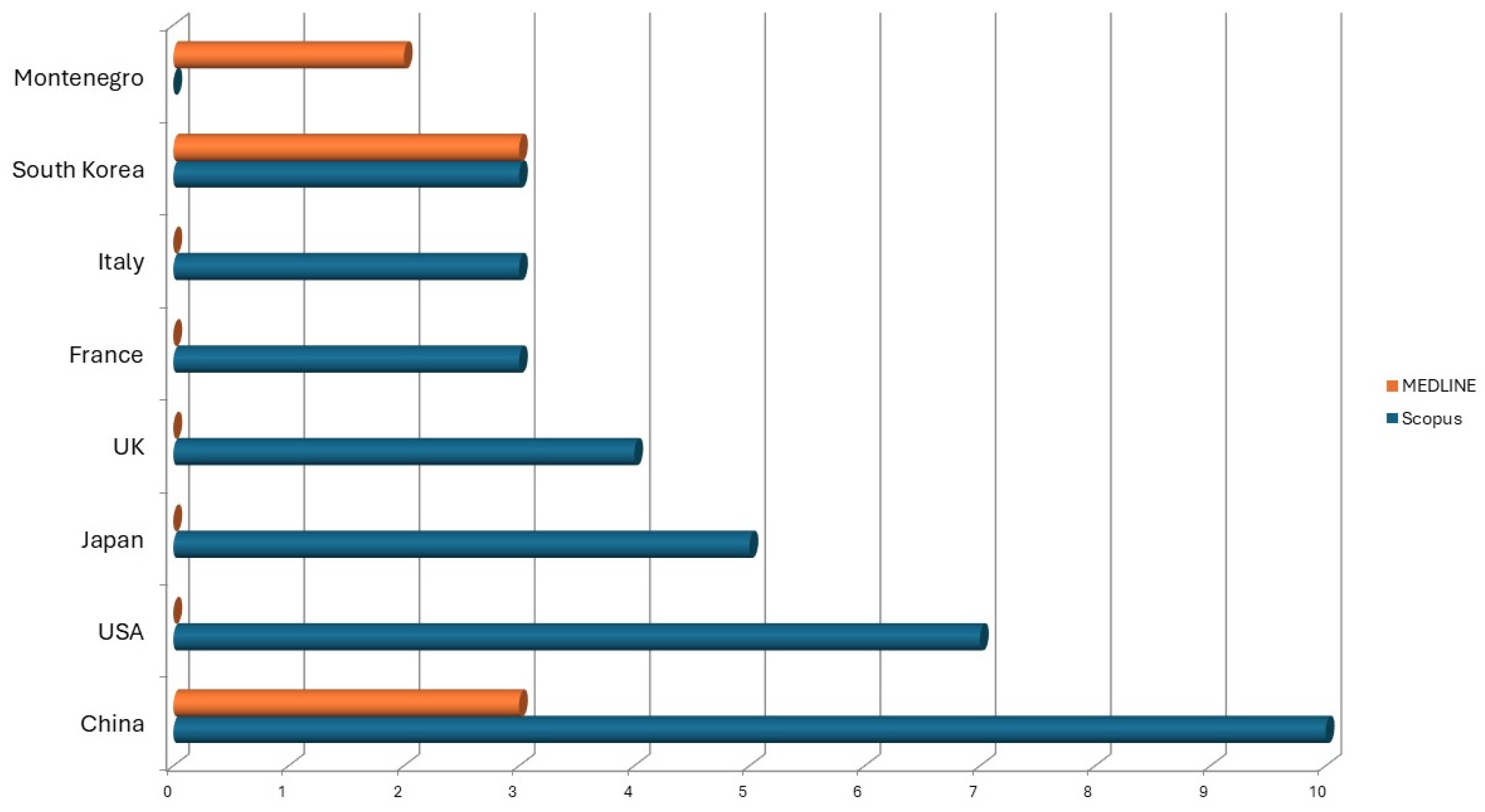
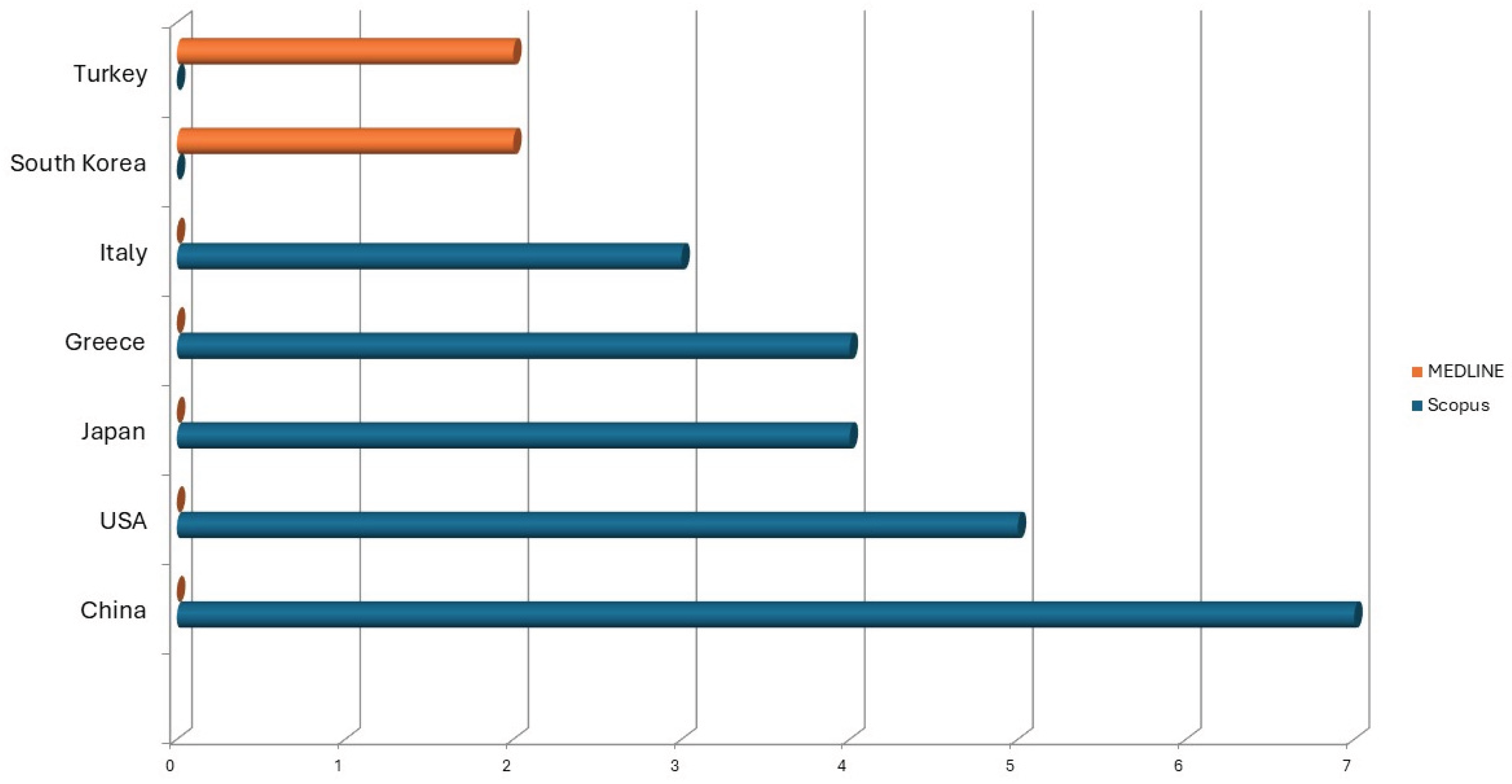
3.4. Geographic Distribution of Leptin and Adiponectin Related Publications
3.5. Literature Review on IHC Biomarkers in CRC with Focus on Leptin and Adiponectin
3.5.1. Mechanistic Role of Leptin in CRC
3.5.2. Mechanistic Role of Adiponectin in CRC
3.5.3. Immunohistochemical Expression Patterns of Leptin and Adiponectin in CRC
3.5.4. Comparison with Other IHC Biomarkers in CRC
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nguyen, M. L. T.; Bui, L. A.; Pham, C.; Nham, P. L. T.; Can, V. M.; Doan, T. D. H.; Bozko, P.; Bui, K. C.; Nguyen, L. T. Adiponectin receptor agonist suppresses human colorectal cancer. Anticancer Res. 2025, 45, 3059–3068. [Google Scholar] [CrossRef]
- Vahed, I. E.; Moshgelgosha, M.; Kor, A.; Minadi, M.; Ebrahimi, F.; Azhdarian, A.; Arjmandi, M.; Alamdar, A.; Zare, M.; Shabani, N.; Soltaninejad, H.; Rahmanian, M. The role of adiponectin and leptin in colorectal cancer and adenoma: a systematic review and meta-analysis. BMC Cancer 2025, 25, 968. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Han, Q. Y.; Zhang, J. G.; Lin, J.; Yan, W. H.; Lin, A. Human leukocyte antigen-G isoform HLA-G2/6, but not HLA-G1/4/5, is an independent indicator of poor survival in patients with colorectal cancer. Front. Immunol. 2025, 16, 1672144. [Google Scholar] [CrossRef] [PubMed]
- Yue, B.; Lu, Z.; Zong, D.; Hu, Y.; Yang, Z. Global, regional, and national burden of early-onset and late-onset colorectal cancer attributable to high body-mass index from 1990 to 2021: a trend analysis and forecasts up to 2040 based on the Global Burden of Disease Study 2021. BMC Gastroenterol. 2025, 25, 816. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Zhang, Y.; Lin, Z.; Wei, M. Global burden of colorectal cancer in 2022 and projections to 2050: incidence and mortality estimates from GLOBOCAN. BMC Cancer 2025, 25, 1770. [Google Scholar] [CrossRef]
- Guadagni, F.; Roselli, M.; Martini, F.; Spila, A.; Riondino, S.; D’Alessandro, R.; Del Monte, G.; Formica, V.; Laudisi, A.; Portarena, I.; Palmirotta, R.; Ferroni, P. Prognostic significance of serum adipokine levels in colorectal cancer patients. Anticancer Res. 2009, 29, 3321–3327. [Google Scholar]
- Pham, D. V.; Park, P. H. Tumor metabolic reprogramming by adipokines as a critical driver of obesity-associated cancer progression. Int. J. Mol. Sci. 2021, 22, 1444. [Google Scholar] [CrossRef]
- Endo, H.; Hosono, K.; Uchiyama, T.; Sakai, E.; Sugiyama, M.; Takahashi, H.; Nakajima, N.; Wada, K.; Takeda, K.; Nakagama, H.; Nakajima, A. Leptin acts as a growth factor for colorectal tumours at stages subsequent to tumour initiation in murine colon carcinogenesis. Gut 2011, 60, 1363–1371. [Google Scholar] [CrossRef]
- Otani, K.; Ishihara, S.; Yamaguchi, H.; Murono, K.; Yasuda, K.; Nishikawa, T.; Watanabe, T. Adiponectin and colorectal cancer. Surgery today 2017, 47, 151–158. [Google Scholar] [CrossRef]
- Chen, K.; Collins, G.; Wang, H.; Toh, J. W. T. Pathological features and prognostication in colorectal cancer. Current Oncology 2021, 28, 5356–5383. [Google Scholar] [CrossRef]
- Guinney, J.; Dienstmann, R.; Wang, X.; de Reyniès, A.; Schlicker, A.; Soneson, C.; Marisa, L.; Roepman, P.; Nyamundanda, G.; Angelino, P.; Bot, B. M.; Morris, J. S.; Simon, I. M.; Gerster, S.; Fessler, E.; De Sousa E Melo, F.; Missiaglia, E.; Ramay, H.; Barras, D.; Homicsko, K.; Maru, D.; Manyam, G. C.; Broom, B.; Boige, V.; Perez-Villamil, B.; Laderas, T.; Salazar, R.; Gray, J. W.; Hanahan, D.; Tabernero, J.; Bernards, R.; Friend, S. H.; Laurent-Puig, P.; Medema, J. P.; Sadanandam, A.; Wessels, L.; Delorenzi, M.; Kopetz, S.; Vermeulen, L.; Tejpar, S. The consensus molecular subtypes of colorectal cancer. Nat. Med. 2015, 21, 1350–1356. [Google Scholar] [CrossRef]
- Valdeolivas, A.; Amberg, B.; Giroud, N.; Richardson, M.; Gálvez, E. J. C.; Badillo, S.; Julien-Laferrière, A.; Túrós, D.; Voith von Voithenberg, L.; Wells, I.; Pesti, B.; Lo, A. A.; Yángüez, E.; Das Thakur, M.; Bscheider, M.; Sultan, M.; Kumpesa, N.; Jacobsen, B.; Bergauer, T.; Saez-Rodriguez, J.; Rottenberg, S.; Schwalie, P. C.; Hahn, K. Profiling the heterogeneity of colorectal cancer consensus molecular subtypes using spatial transcriptomics. NPJ Precis. Oncol. 2024, 8, 10. [Google Scholar] [CrossRef]
- Trinh, A.; Trumpi, K.; De Sousa E Melo, F.; Wang, X.; de Jong, J. H.; Fessler, E.; Kuppen, P. J.; Reimers, M. S.; Swets, M.; Koopman, M.; Nagtegaal, I. D.; Jansen, M.; Hooijer, G. K.; Offerhaus, G. J.; Kranenburg, O.; Punt, C. J.; Medema, J. P.; Markowetz, F.; Vermeulen, L. Practical and robust identification of molecular subtypes in colorectal cancer by immunohistochemistry. Clin. Cancer Res. 2017, 23, 387–398. [Google Scholar] [CrossRef]
- Li, X.; Larsson, P.; Ljuslinder, I.; Ling, A.; Löfgren-Burström, A.; Zingmark, C.; Edin, S.; Palmqvist, R. A modified protein marker panel to identify four consensus molecular subtypes in colorectal cancer using immunohistochemistry. Pathol. Res. Pract. 2021, 220, 153379. [Google Scholar] [CrossRef]
- Tomov, D. T. Adipobiology: a bibliometric view to its increasing significance. Biomed. Rev. 2006, 17, 113–118. [Google Scholar] [CrossRef]
- Yaneva, G.; Dimitrova, Ts.; Ivanov, D.; Ingilizova, G.; Slavov, S. Institutionalization patterns in breast cancer immunohistochemistry. J. IMAB 2022, 28, 4211–4216. [Google Scholar] [CrossRef]
- Tomov, D.; Ivanov, K.; Kolev, N.; Donev, I. Dynamic institutionalization of research in the field of colorectal tumour markers. J. Gastroenterol. Its Complications 2018, 2, 9. [Google Scholar]
- Uddin, S.; Hussain, A. R.; Khan, O. S.; Al-Kuraya, K. S. Role of Dysregulated Expression of Leptin and Leptin Receptors in Colorectal Carcinogenesis. Tumour Biol. 2014, 35, 871–879. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, A.; Saeidi, J.; Azimi-Nejad, M.; Hashemy, S. I. Leptin-Induced Signaling Pathways in Cancer Cell Migration and Invasion. Cell. Oncol. 2019, 42, 243–260. [Google Scholar] [CrossRef]
- Cao, D.; Luo, Y.; Qin, S.; et al. Metallopanstimulin-1 (MPS-1) Mediates the Promotion Effect of Leptin on Colorectal Cancer through Activation of JNK/c-Jun Signaling Pathway. Cell Death Dis. 2019, 10, 655. [Google Scholar] [CrossRef] [PubMed]
- Socol, C. T.; Chira, A.; Martinez-Sanchez, M. A.; Nuñez-Sanchez, M. A.; Maerescu, C. M.; Mierlita, D.; Rusu, A. V.; Ruiz-Alcaraz, A. J.; Trif, M.; Ramos-Molina, B. Leptin Signaling in Obesity and Colorectal Cancer. Int. J. Mol. Sci. 2022, 23, 4713. [Google Scholar] [CrossRef]
- Uchiyama, T.; et al. Role of the Long Form Leptin Receptor and of the STAT3 Signaling Pathway in Colorectal Cancer Progression. Int. J. Oncol. 2011, 39, 935–940. [Google Scholar] [CrossRef]
- Endo, H.; et al. Leptin Acts as a Growth Factor for Colorectal Tumours at Stages Subsequent to Tumour Initiation in Murine Colon Carcinogenesis. Gut 2011, 60, 1363–1371. [Google Scholar] [CrossRef] [PubMed]
- Taguri, M.; Kuchiba, A.; Yamaji, T.; Sawada, N.; Goto, A.; Iwasaki, M.; Tsugane, S. Importance of Circulating Leptin and Adiponectin in the Causal Pathways Between Obesity and the Development of Colorectal Cancer in Japanese Men. J. Epidemiol. 2024, 34, 563–569. [Google Scholar] [CrossRef]
- Kafeel, S.; et al. Unravelling the Adiponectin Hallmark and Exploring the Therapeutic Potential of Its Receptor Agonists in Cancer Metabolic Reprogramming. Biomolecules 2025, 15, 820. [Google Scholar] [CrossRef]
- Chakraborty, D.; Jin, W.; Wang, J. The Bifurcated Role of Adiponectin in Colorectal Cancer. Life Sci. 2021, 278, 119524. [Google Scholar] [CrossRef] [PubMed]
- Jones, A. N.; et al. Obesity and Inflammatory Factors in the Progression of Early-Onset Colorectal Cancer. Cancers 2024, 16, 1403. [Google Scholar] [CrossRef]
- Kounatidis, D.; Vallianou, N.G.; Karampela, I.; Rebelos, E.; Kouveletsou, M.; Dalopoulos, V.; Koufopoulos, P.; Diakoumopoulou, E.; Tentolouris, N.; Dalamaga, M. Anti-Diabetic Therapies and Cancer: From Bench to Bedside. Biomolecules 2024, 14, 1479. [Google Scholar] [CrossRef] [PubMed]
- Scheurlen, K. M. The Role of Obesity in Macrophage-Mediated Mechanisms Promoting Early-Onset Colon Cancer. 2022. [Google Scholar]
- Koda, M.; Sulkowska, M.; Koda, K.; Sobaniec-Lotowska, M.; Wolczynski, S.; Sulkowski, S. Overexpression of the obesity hormone leptin in human colorectal cancer. J. Clin. Pathol. 2007, 60, 902–906. [Google Scholar] [CrossRef]
- Paik, S. S.; Jang, S. M.; Jang, K. S.; Lee, K. H.; Choi, D.; Jang, S. J. Leptin expression correlates with favorable clinicopathologic phenotype and better prognosis in colorectal adenocarcinoma. Ann. Surg. Oncol. 2009, 16, 297–303. [Google Scholar] [CrossRef]
- Jeong, W. K.; Baek, S. K.; Kim, M. K.; Kwon, S. Y.; Kim, H. S. Prognostic significance of tissue leptin expression in colorectal cancer patients. Ann. Coloproctol. 2015, 31, 222–227. [Google Scholar] [CrossRef]
- Al-Maghrabi, J. A.; Qureshi, I. A.; Khabaz, M. N. Expression of leptin in colorectal adenocarcinoma showed significant different survival patterns associated with tumor size, lymphovascular invasion, distant metastasis, local recurrence, and relapse of disease in the western province of Saudi Arabia. Medicine (Baltimore) 2018, 97, e12052. [Google Scholar] [CrossRef]
- Li, C.; Quan, J.; Wei, R.; Zhao, Z.; Guan, X.; Liu, Z.; Zou, S.; Wang, X.; Jiang, Z. Leptin overexpression as a poor prognostic factor for colorectal cancer. Biomed. Res. Int. 2020, 2020, 7532514. [Google Scholar] [CrossRef] [PubMed]
- Al-Shibli, S. M.; Harun, N.; Ashour, A. E.; Mohd Kasmuri, M. H. B.; Mizan, S. Expression of leptin and leptin receptors in colorectal cancer — an immunohistochemical study. PeerJ 2019, 7, e7624. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi-Nesheli, M.; Alizadeh-Navaei, R.; Vahedi, L.; Amjadi, O.; Taghvaei, T.; Maleki, I.; Shekarriz, R.; Kazemi, A.; Omrani-Nava, V.; Alizadeh-Foroutan, M. Evaluation of circulating leptin and its receptor (Ob-R) tissue expression in colorectal cancer, a report from North of Iran. Iran. J. Pathol. 2023, 18, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Chludzińska-Kasperuk, S.; Lewko, J.; Sierżantowicz, R.; Krajewska-Kułak, E.; Reszeć-Giełażyn, J. The effect of serum leptin concentration and leptin receptor expression on colorectal cancer. Int. J. Environ. Res. Public Health 2023, 20, 4951. [Google Scholar] [CrossRef]
- Parmesh, P.; Dinesh, U. S.; Khandagale, A. S.; Bapu, A. B.; Sadashiv, R.; Reddy, P. Correlation of leptin and adiponectin receptor expression with clinicopathological parameters in colorectal carcinoma — a cross-sectional prospective study. Arq. Gastroenterol. 2024, 61, e24016. [Google Scholar] [CrossRef]
- Hanafy, S. M.; Hegazy, A. A.; Elmigdadi, F.; Eladl, I. M.; Embaby, A.; Zaitoun, M. A.; Gobran, M. A. Evaluation of the prognostic role of leptin and cyclin D1 expression in colorectal cancer. Middle East J. Cancer 2025, 16. [Google Scholar]
- Williams, C. J.; Mitsiades, N.; Sozopoulos, E.; Hsi, A.; Wolk, A.; Nifli, A. P.; Mantzoros, C. S. Adiponectin receptor expression is elevated in colorectal carcinomas but not in gastrointestinal stromal tumors. Endocr. Relat. Cancer 2008, 15, 289–299. [Google Scholar] [CrossRef]
- Barresi, V.; Tuccari, G.; Barresi, G. Adiponectin immunohistochemical expression in colorectal cancer and its correlation with histological grade and tumour microvessel density. Pathology 2009, 41, 533–538. [Google Scholar] [CrossRef]
- Byeon, J. S.; Jeong, J. Y.; Kim, M. J.; Lee, S. M.; Nam, W. H.; Myung, S. J.; Yang, S. K.; Kim, J. H.; Kim, K. J.; Ye, B. D.; Cho, Y. K.; Chung, Y. H.; Lee, Y. S.; Suh, D. J. Adiponectin and adiponectin receptor in relation to colorectal cancer progression. Int. J. Cancer 2010, 127, 2758–2767. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, T. E.; Yamada, Y.; Hamano, T.; Furuta, K.; Matsuda, T.; Fujita, S.; Kato, K.; Hamaguchi, T.; Shimada, Y. Adipocytokines as new promising markers of colorectal tumors: adiponectin for colorectal adenoma, and resistin and visfatin for colorectal cancer. Cancer Sci. 2010, 101, 1286–1291. [Google Scholar] [CrossRef]
- Gialamas, S. P.; Petridou, E. T.; Tseleni-Balafouta, S.; Spyridopoulos, T. N.; Matsoukis, I. L.; Kondi-Pafiti, A.; Mantzoros, C. S. Serum adiponectin levels and tissue expression of adiponectin receptors are associated with risk, stage, and grade of colorectal cancer. Metabolism 2011, 60, 1530–1538. [Google Scholar] [CrossRef] [PubMed]
- An, W.; Bai, Y.; Deng, S.-X.; Gao, J.; Ben, Q.-W.; Cai, Q.-C.; Zhang, H.-G.; Li, Z.-S. Adiponectin levels in patients with colorectal cancer and adenoma: a meta-analysis. Eur. J. Cancer Prev. 2012, 21, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Canhoroz, M.; Kanat, O.; Saraydaroglu, O.; Buluc, E.; Avci, N.; Cubukcu, E.; Olmez, O. F.; Manavoglu, O. Clinical significance of adiponectin expression in colon cancer patients. J. Cancer Res. Ther. 2014, 10, 347–353. [Google Scholar] [CrossRef]
- Vetvik, K. K.; Sonerud, T.; Lindeberg, M.; Lüders, T.; Størkson, R. H.; Jonsdottir, K.; Bukholm, I. Globular adiponectin and its downstream target genes are up-regulated locally in human colorectal tumors: ex vivo and in vitro studies. Metabolism 2014, 63, 672–681. [Google Scholar] [CrossRef]
- Wei, T.; Ye, P.; Peng, X.; Wu, L. L.; Yu, G. Y. Circulating adiponectin levels in various malignancies: an updated meta-analysis of 107 studies. Oncotarget 2016, 7, 48671–48691. [Google Scholar] [CrossRef]
- Zhou, L.; Zhang, H. F.; Ning, W.; Song, X.; Liu, X.; Liu, J. X. Associations of adiponectin receptor 2 (AdipoR2) gene polymorphisms and AdipoR2 protein expression levels with the risk of colorectal cancer: a case-control study. Mol. Med. Rep. 2017, 16, 2511–2518. [Google Scholar] [CrossRef]
- Lugli, A.; Tzankov, A.; Zlobec, I.; Terracciano, L. M. Differential Diagnostic and Functional Role of the Multi-Marker Phenotype CDX2/CK20/CK7 in Colorectal Cancer Stratified by Mismatch Repair Status. Mod. Pathol. 2008, 21, 1403–1412. [Google Scholar] [CrossRef]
- Ilieva, N.; Tashkova, D.; Staykov, D.; et al. Immunohistochemical Expression of CK20, CK7, and CDX2 in Colorectal Carcinoma in Correlation with Pathomorphological Characteristics. Folia Med. (Plovdiv) 2022, 64, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Werling, R. W.; Yaziji, H.; Bacchi, C. E.; Gown, A. M. CDX2, a Highly Sensitive and Specific Marker of Adenocarcinomas of Intestinal Origin: An Immunohistochemical Survey of 476 Primary and Metastatic Carcinomas. Am. J. Surg. Pathol. 2003, 27, 303–310. [Google Scholar] [CrossRef]
- Choi, H. B.; et al. Diagnostic and Prognostic Roles of CDX2 Immunohistochemical Expression in Colorectal Cancer: A Meta-Analysis. Diagnostics (Basel) 2022, 12, 757. [Google Scholar] [CrossRef]
- Loughrey, M. B.; McGrath, J.; Coleman, H. G.; Bankhead, P.; Maxwell, P.; McGready, C.; Bingham, V.; Humphries, M. P.; Craig, S. G.; McQuaid, S.; Salto-Tellez, M.; James, J. A. Identifying Mismatch Repair-Deficient Colon Cancer: Near-Perfect Concordance between Immunohistochemistry and Microsatellite Instability Testing in a Large, Population-Based Series. Histopathology 2021, 78, 401–413. [Google Scholar] [CrossRef]
- Wu, E. Y.; Resnick, M. Predictive and Prognostic Implications of Microsatellite Instability and Mismatch Repair Deficiency in Carcinomas of the Gastrointestinal Tract, Liver, and Pancreas. Precision Cancer Med. 2019, 2. [Google Scholar] [CrossRef]
- Tashkova, D.; Ilieva, N.; Serteva, D.; Feodorova, Y.; Mehterov, N.; Mollova, A.; Bachurska, S. Immunohistochemical Phenotype of Colorectal Carcinoma in Patients with KRAS Mutation and Mismatch Repair Status. Folia Med. (Plovdiv) 2023, 65, 378–383. [Google Scholar] [CrossRef]
- Sun, Q.; Li, Q.; Gao, F.; Wu, H.; Fu, Y.; Yang, J.; Fan, X.; Cui, X.; Pu, X. HER2 Overexpression/Amplification Status in Colorectal Cancer: A Comparison between Immunohistochemistry and Fluorescence In Situ Hybridization Using Five Different Immunohistochemical Scoring Criteria. J. Cancer Res. Clin. Oncol. 2023, 149, 579–592. [Google Scholar] [CrossRef] [PubMed]
- Zeng, D. T.; Li, M. J.; Lin, R.; Huang, W. J.; Li, S. D.; Huang, W. Y.; Li, B.; Li, Q.; Chen, G.; Jiang, J. S. Prognostic Role of Ki-67 in Colorectal Carcinoma: Development and Evaluation of Machine Learning Prediction Models. World J. Clin. Oncol. 2025, 16, 107306. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Wei, C.; Wang, S.; Shi, D.; Zhang, C.; Lin, X.; Dou, R.; Xiong, B. Elevated CD163+/CD68+ Ratio at Tumor Invasive Front Is Closely Associated with Aggressive Phenotype and Poor Prognosis in Colorectal Cancer. Int. J. Biol. Sci. 2019, 15, 984–998. [Google Scholar] [CrossRef]
- Balasubramanian, S.; Priyathersini, N.; Johnson, T. Expression of Vascular Endothelial Growth Factor (VEGF) in Colorectal Adenoma and Carcinoma in a Tertiary Care Center. Cureus 2022, 14, e31393. [Google Scholar] [CrossRef]
- Lee, S. H.; Pankaj, A.; Rickelt, S.; Ting, D.; Ferrone, C.; Patil, D. T.; Yilmaz, O.; Berger, D.; Deshpande, V.; Yilmaz, O. β2-Microglobulin Expression Is Associated with Aggressive Histology, Activated Tumor Immune Milieu, and Outcome in Colon Carcinoma. Am. J. Clin. Pathol. 2024, 162, 500–508. [Google Scholar] [CrossRef]
- Fosam, A.; Perry, R. J. Current mechanisms in obesity and tumor progression. Curr. Opin. Clin. Nutr. Metab. Care 2020, 23, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Riondino, S.; Roselli, M.; Palmirotta, R.; Della-Morte, D.; Ferroni, P.; Guadagni, F. Obesity and colorectal cancer: role of adipokines in tumor initiation and progression. World J. Gastroenterol. 2014, 20, 5177. [Google Scholar] [CrossRef]
- Fenton, J. I.; Birmingham, J. M.; Hursting, S. D.; Hord, N. G. Adiponectin Blocks Multiple Signaling Cascades Associated with Leptin-Induced Cell Proliferation in ApcMin/+ Colon Epithelial Cells. Int. J. Cancer 2008, 122, 2437–2445. [Google Scholar] [CrossRef] [PubMed]
- Yamaji, T.; Iwasaki, M.; Sasazuki, S.; Tsugane, S. Interaction between Adiponectin and Leptin Influences the Risk of Colorectal Adenoma. Cancer Res. 2010, 70, 5430–5437. [Google Scholar] [CrossRef] [PubMed]
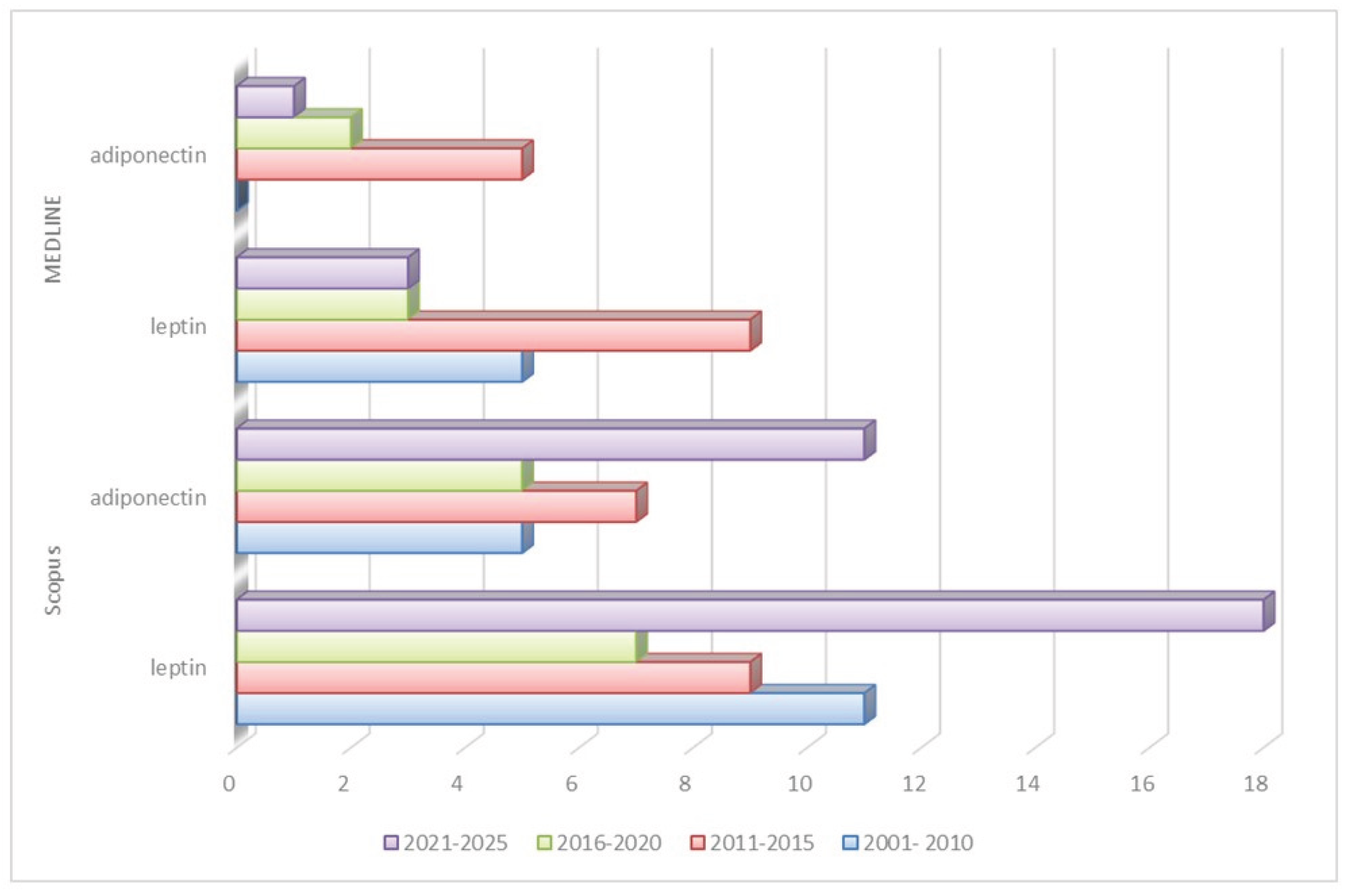
| Year span | Scopus | MEDLINE | ||
| n | % | n | % | |
| 2000- 2010 | 11 | 24,44 | 5 | 25,00 |
| 2011-2015 | 9 | 20,00 | 9 | 45,00 |
| 2016-2020 | 7 | 15,56 | 3 | 15,00 |
| 2021-2025 | 18 | 40,00 | 3 | 15,00 |
| total | 45 | 100,00 | 20 | 100,00 |
| Year span | Scopus | MEDLINE | ||
| n | % | n | % | |
| 2000-2010 | 5 | 17,86 | 0 | 0 |
| 2011-2015 | 7 | 25,00 | 5 | 62,50 |
| 2016-2020 | 5 | 17,86 | 2 | 25,00 |
| 2021-2025 | 11 | 39,28 | 1 | 12,50 |
| total | 28 | 100,00 | 8 | 100,00 |
| Journal title | Scopus | MEDLINE | ||
| n | % | n | % | |
| Cancer Research | 2 | 4,44 | 2 | 10,00 |
| Journal of BUON | 2 | 4,44 | 2 | 10,00 |
| Cells | 2 | 4,44 | - | - |
| Journal title | Scopus | MEDLINE | ||
| n | % | n | % | |
| BMC Cancer | 2 | 7,14 | - | - |
| Asian Pacific Journal of Cancer Prevention | - | - | 2 | 25,00 |
| Country | Scopus | MEDLINE | |||
| n | % | Country | n | % | |
| China | 10 | 22,22 | China | 3 | 15,00 |
| USA | 7 | 15,56 | South Korea | 3 | 15,00 |
| Japan | 5 | 11,11 | Montenegro | 2 | 10,00 |
| UK | 4 | 8,89 | |||
| France | 3 | 6,67 | |||
| Italy | 3 | 6,67 | |||
| South Korea | 3 | 6,67 | |||
| Country | Scopus | MEDLINE | |||
| n | % | Country | n | % | |
| China | 7 | 25,00 | South Korea | 2 | 25,00 |
| USA | 5 | 17,86 | Turkey | 2 | 25,00 |
| Japan | 4 | 14,29 | |||
| Greece | 4 | 14,29 | |||
| Italy | 3 | 10,71 | |||
| Reference |
N (Patients) |
Method/Scoring (IHC) | Main Findings |
| Koda et al., 2007 [30] |
166 | IHC for leptin; expression was scored as 0 (<10% positive cells), 1+ (10–50%), or 2+ (>50%) | Leptin overexpressed in CRC is associated with poorer differentiation and metastasis |
| Paik et al., 2009 [31] |
437 | IHC; expression graded neg/weak/moderate/strong | High leptin expression is associated with better survival of colorectal cancer patients. |
| Jeong et al., 2015 [32] |
146 | IHC; expression graded neg/weak/moderate/strong and grouped for analysis | Strong leptin expression observed in subset; inversely associated with nodal stage in this cohort |
| Al-Maghrabi et al., 2018 [33] |
155 | Tissue microarray; IHC positive/negative; intensity stratified | Leptin positive in most tumors (≈93.5%); higher expression associated with several adverse features (distant metastasis, recurrence) in subgroup analyses |
| Li et al., 2020 [34] |
407 | IHC; high vs low leptin expression; survival analyses, supported by TCGA data | Leptin overexpression correlated with metastasis stage and lymph node involvement |
| Al-Shibli et al., 2019 [35] |
44 | IHC; intensity scoring for leptin and LEPR | Tumors showed high intensity of LEP and LEPR versus adjacent normal mucosa |
| Mahmoudi-Nesheli et al., 2023 [36] |
90 | IHC for leptin and LEPR; ELISA for serum leptin | Reported tissue expression patterns for leptin and LEPR; no significant difference in leptin and LEPR expression between CRC patients and healthy controls. |
| Chludzińska-Kasperuk et al., 2023 [37] |
61 | IHC for leptin and leptin receptor (LEPR) in tissue; ELISA for serum leptin | Higher expression of leptin and LEPR are linked to obesity, suggesting they may influence obesity-associated cancer progression |
| Parmesh et al., 2024 [38] |
60 |
IHC for leptin and adiponectin receptor; expression was scored as 0 (no positive cells), 1 (<10% positive cells), 2 (10–50%), 3 (>51-80%) or 4(˃80% positive cells) | Leptin positivity significantly correlated with larger tumor size, lymph node and distant metastasis, distant metastasis and advanced TNM stage |
| Hanafy et al., 2024 [39] |
60 | IHC for leptin and cyclin D1; scoring by intensity | Significantly increased leptin and cyclin D1 expression in CRC; elevated leptin and cyclin D1 are associated with advanced stage, lymph node and distant metastasis. |
| Reference |
N (Patients) |
Method/Scoring (IHC) | Main Findings |
| Williams et al.,2008 [40] |
40 | Semiquantitative IHC intensity/percentage, categorized as negative, positive, strongly positive for AdipoR1/R2 | Marked increase of both receptors in carcinomas vs normal, suggesting CRC-specific up-regulation |
| Barresi et al., 2009 [41] |
45 | IHC using adiponectin antibody | Decreased adiponectin expression in tumor tissue compared to adjacent normal mucosa |
| Byeon et al., 2010 [42] |
100 | IHC graded by staining intensity/extent; cases grouped by grade of AdipoR1/R2 immunostaining | Expression of both receptors is inversely related to T stage; lowest expression in poorly differentiated adenocarcinoma; |
| Nakajima et al., 2010 [43] |
119 | IHC in CRC tissues; intensity & % scoring | Low adiponectin expression correlated with advanced TNM stage and poor differentiation |
| Gialamas et al., 2011 [44] |
104 | IHC staining | Low adiponectin expression correlated with lymph node metastasis |
| An et al., 2012 [45] |
92 | IHC staining in paraffin-embedded CRC tissue; H-score | Reduced adiponectin expression in tumor tissue compared to adjacent normal mucosa |
| Canhoroz et al., 2014 [46] |
53 | IHC; scoring system for staining intensity | Adiponectin expression is reduced in CRC |
| Vetvik et al., 2014 [47] |
60 | IHC for globular adiponectin (gAd) and ADIPOR1; relative staining compared between tumor and mucosa | Adiponectin (mRNA and globular protein) and AdipoR1, were significantly higher in tumors vs adjacent mucosa |
| Wei et al., 2015 [48] |
156 | IHC; semi-quantitative scoring (intensity and extent) | Adiponectin expression decreased in high-grade and advanced stage tumors |
| Zhou et al., 2017 [49] |
281 | IHC for AdipoR2; cases classified as positive vs negative expression | AdipoR2 protein positivity associated with degree of differentiation, tumor infiltration and lymphatic metastasis |
| Parmesh et al., 2024 [38] |
60 | IHC for adiponectin receptors AdipoR1,R2; semi-quantitative scoring; cases grouped as positive vs negative | Negative adiponectin receptor expression significantly associated with advanced tumor stage, lymph node and distant metastasis; combined pattern of positive leptin and negative adiponectin receptor predicted higher metastatic risk |
| Marker | Biological Role | Diagnostic Utility | Prognostic Value | Clinical Implications | References |
| CK20/CK7 | Cytokeratins | CK20+/CK7− supports colorectal origin | Limited | Differentiates CRC from other primary tumors | [50] [51] |
| CDX2 | Transcription factor | Sensitive/specific for colorectal origin | Loss leads to worse prognosis | Supports diagnosis | [50] [51] [52] [53] |
| MMR (MLH1, MSH2, MSH6, PMS2) | DNA repair | Loss → MSI/dMMR | Favorable early stage | Guides Lynch + immunotherapy | [54] [55] |
| BRAF V600E | MAPK mutation | Detects mutant CRC | Poor prognosis | Targeted therapy relevance | [56] |
| HER2 | Growth receptor | Identifies subset | Variable | HER2-targeted therapy | [57] |
| Ki-67 | Proliferation | Adjunct | High = aggressive | Supports grading | [58] |
| CD68/CD163 | Macrophage markers | Immune profiling | Emerging | Immunotherapy relevance | [59] |
| VEGF | Angiogenesis | Limited | Investigational | Anti-angiogenic relevance | [60] |
| β2-microglobulin | Immune-related | Refines profiling | Investigational | Immunotherapy context | [61] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




