4. Discussion
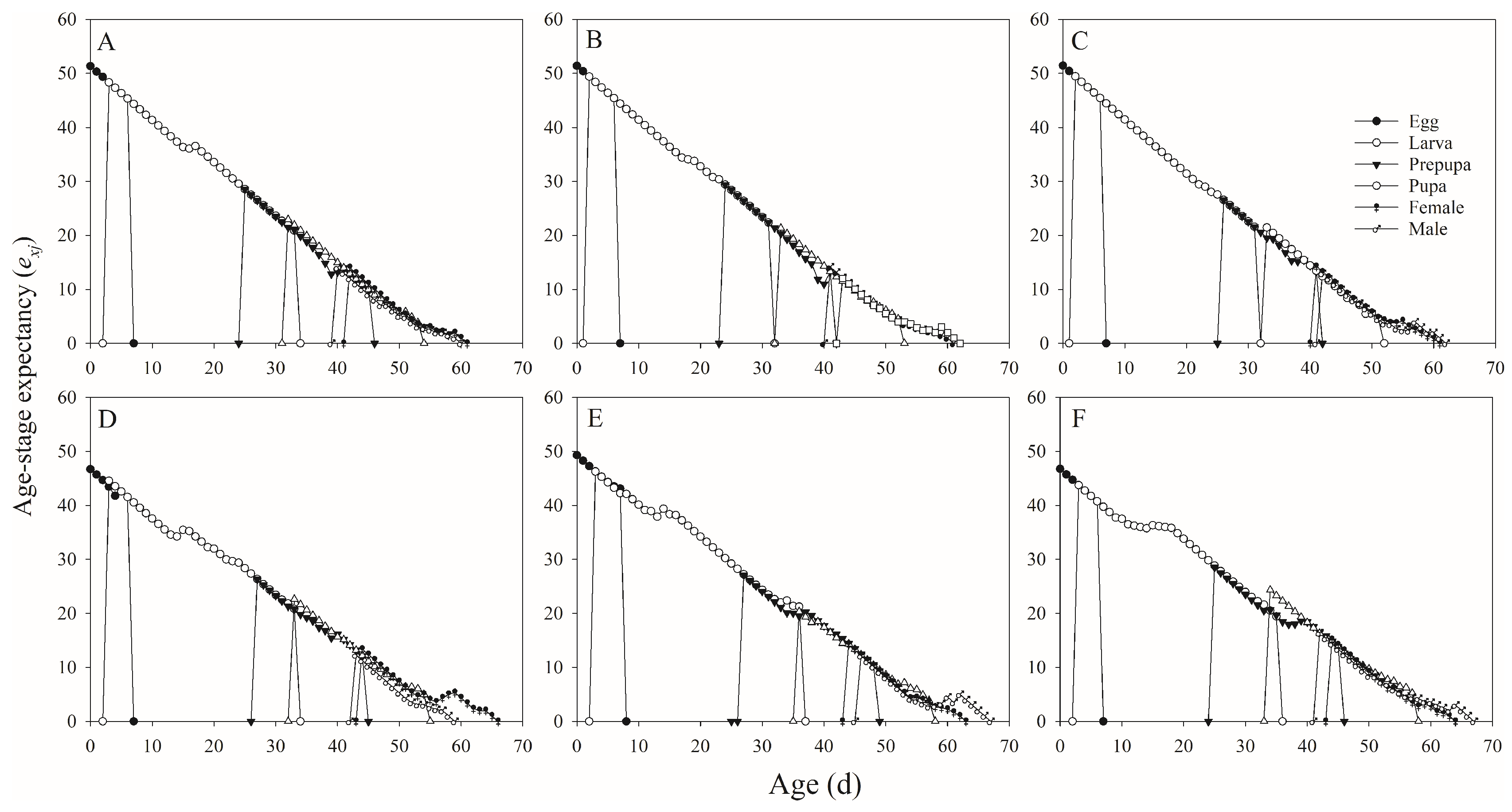
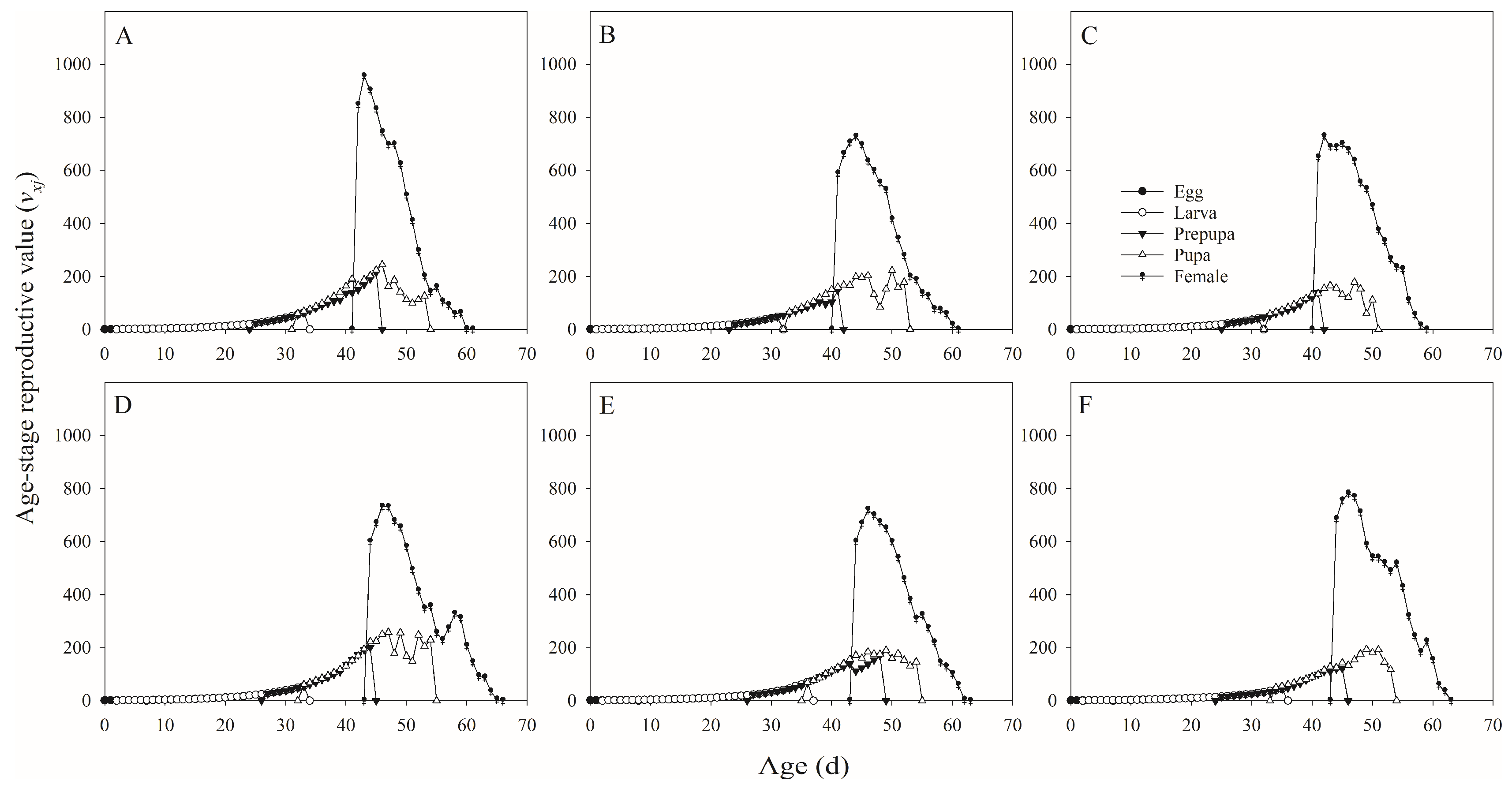
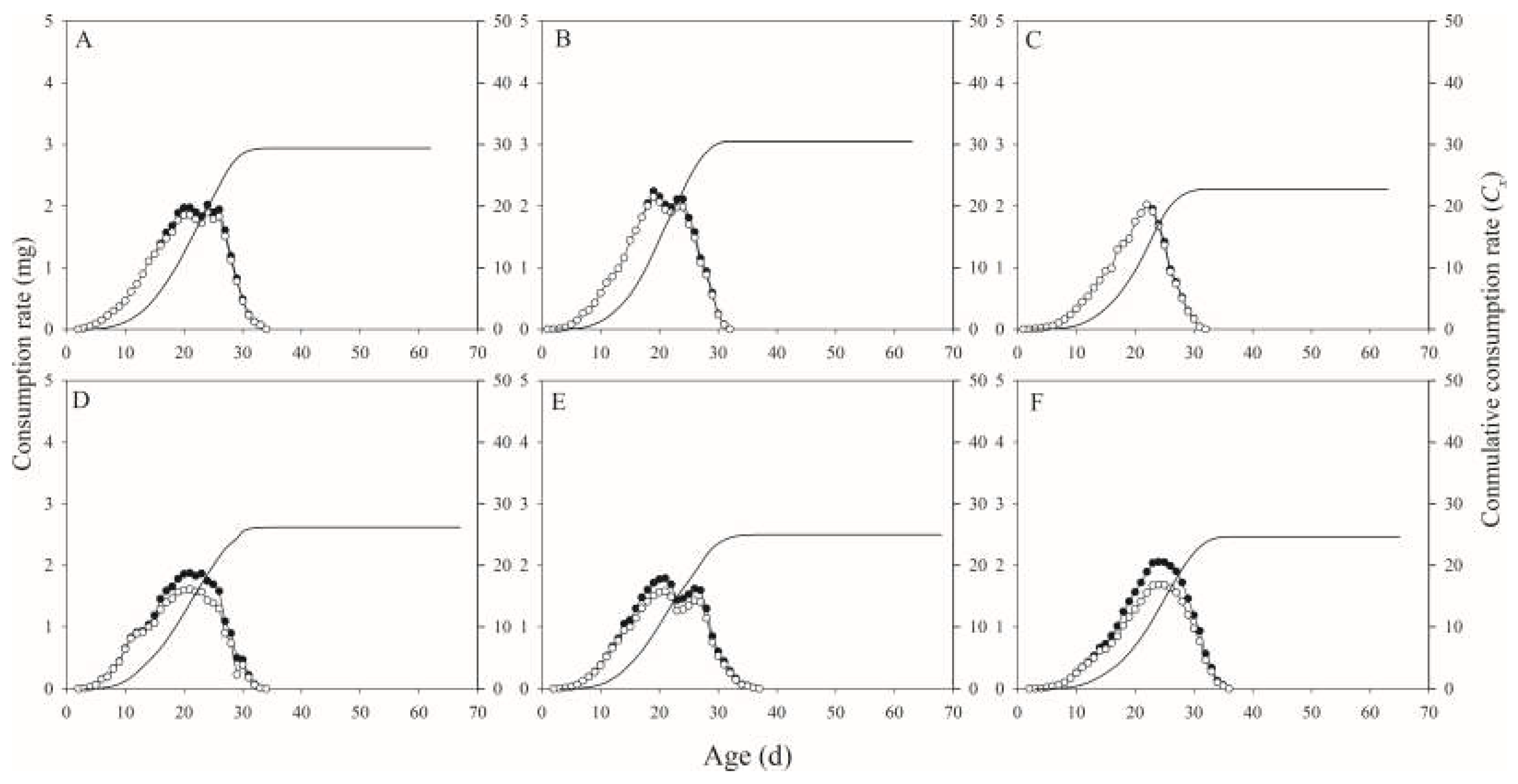
In this study, we found that several tested parameters were influenced by salt concentration levels, with negative effects on the developmental duration, reproductive potential, net consumption rate, crude fat and protein, fatty acids, amino acids, and mineral composition of H. illucens, and positive effect on the ash content.
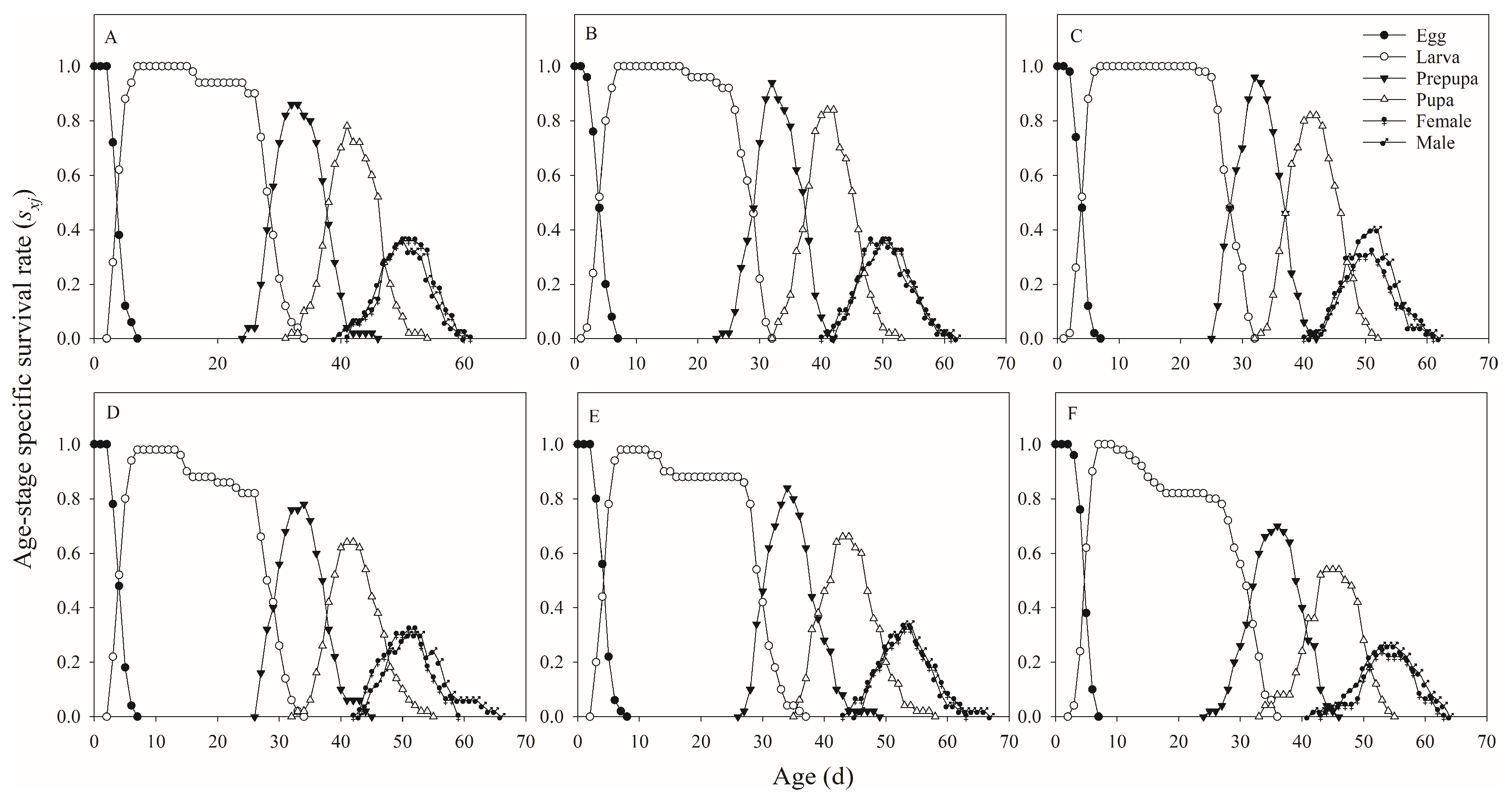
According to previous reports, the developmental rate and duration of
H. illucens were negatively affected by many factors, including food, suboptimal environmental temperatures, and geographical population [
8,
28]. In a recent study, sex-specific developmental responses to manure types were observed, with males delayed in cow dung and females accelerated in other substrates [
8]. The development duration of
H. illucens larva feeding on food waste (12 d) was longer those feeding on waste+bean sprouts (9 d) and food waste+wheat bran (7 d) treatments [
20]. We also found that the larva development duration of
H. illucens males and females was significantly affected by substrates salinity. In addition, when feeding on artificial diet (milk powder, yeast, and sugar), the development time was 23.03 d [
29]. Furthermore, the developing rate of
H. illucens was affected by temperature, the larval stage of
H. illucens was 22.5-24.1 d at 27 °C [
30]. In a previous study, the larval period varied from two weeks to four months in the adverse environment [
31].
Similarly, the reproductive capacity of
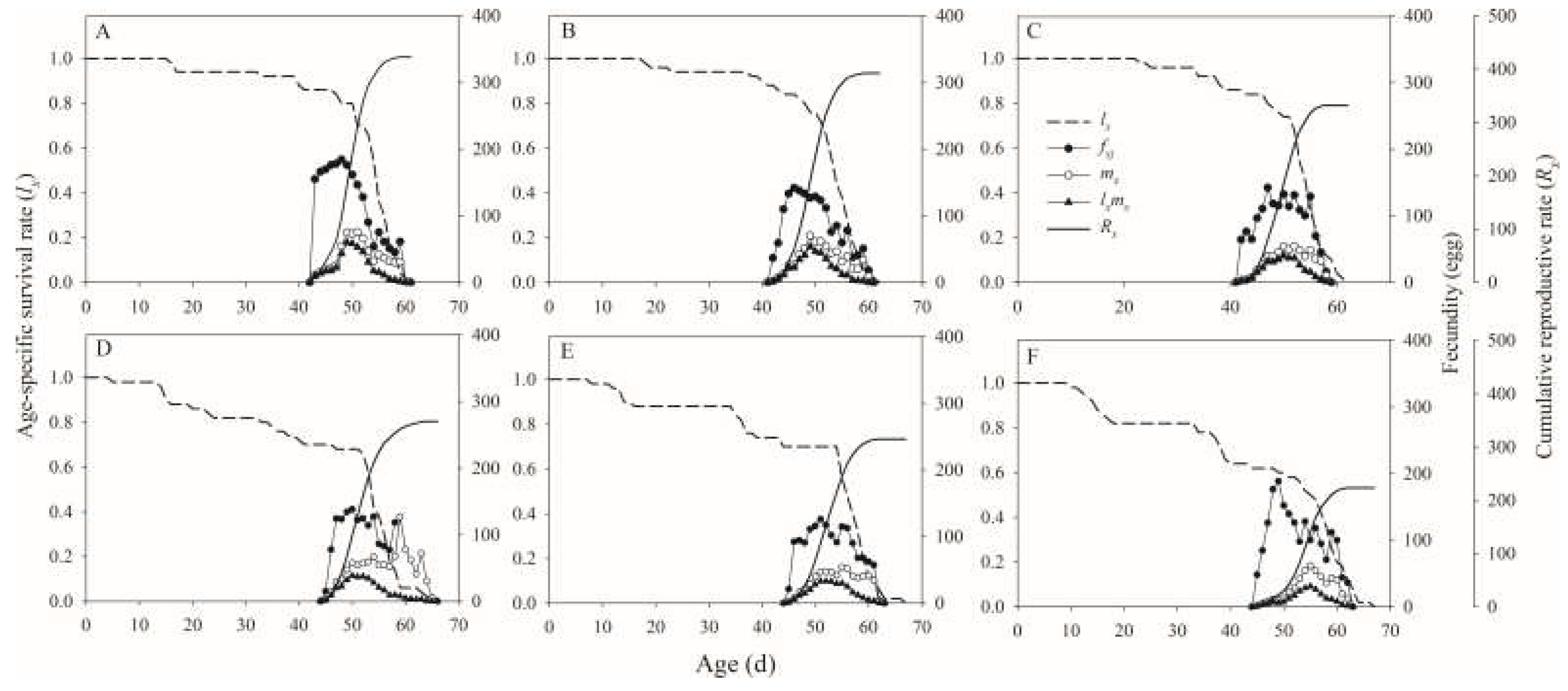
H. illucens was also influenced by foodstuff. In this study, the biological indicator intrinsic rate of increase (
rm) in control was significantly more than those in 3%, 4%, and 5% treatments, respectively. These results suggested that the population dynamics were influenced by the foodstuff salinity. Recently, Wang et al. reported that the
rm of
H. illucens in food waste, chicken manure, pig manure, and cow dung treatments were 0.1249 d
-1, 0.1167 d
-1, 0.1154 d
-1, and 0.1049 d
-1, respectively [
8]. Samayoa and Hwang showed that
rm of
H. illucens feeding on mixed organic waste including kitchen scraps, okara, and pineapple peel was 0.0498 d
-1 [
32]. Furthermore, Samayoa et al. reported that when feeding on an artificial blended diet based on milk powder, yeast, and sugar, the
rm of
H. illucens was 0.0756 d
-1 [
29]. These studies suggested that the fecundity of
H. illucens was influenced by the intake nutrients.
Additionally, we found that the net reproductive rate (
R0) of
H. illucens in control was significantly higher than those in 2%, 3%, 4%, and 5% salinity treatments, respectively. These findings suggested that salinity in foodstuffs affects the reproductive potential. Wang et al. showed that the
R0 of
H. illucens feeding on food waste and pig manure was significantly higher than that in cow dung treatment, and the value of
R0 was 445.4, 399.3, and 320.6 eggs, respectively [
8]. The
R0 value in the current study is consistent with Wang et al. [
8]. In addition, the
R0 of
H. illucens feeding on artificial mixed food including wheat bran and chicken feed was only 68.2 eggs [
29], and when feeding on mixed organic waste including kitchen scraps, okara, and pineapple peel, the the
R0 was 118.3 eggs [
32]. The differences in the fecundity among these studies suggested that the intake of nutrient affected the reproductive potential of black soldier flies.
According to Makkar et al., the essential components of insects include ash, crude protein, and fat, and the main nutritional substances are protein and fat [
33]. In this study, we found that the ash content in 3%, 4% and 5% treatments was significantly less than that in control. Spranghers et al. indicated that the ash of
H. illucens feeding on digestate, chicken feed, vegetable waste, and restaurant waste were 197, 100, 96, and 27 g·kg
-1 LPM, respectively [
34]. Wang et al. showed that the ash of dry
H. illucens prepupae power in food waste, chicken manure, and cow dung were 30.8, 37.6, and 49.5 g·kg
-1 LPM, respectively [
8]. These studies demonstrated that the ash content is influenced by the type of food consumed.
In addition, we found that the content of crude fat in control were significantly more than those in 2%, 3%, 4%, and 5% treatments. Wang et al. found that the content of crude fat in
H. illucens prepupae feeding on kitchen waste and poultry cultivation wastes was 35.7-37.2% and less than that in the current study [
8]. In some previous reports, the percentage of fat were 35%, 15-25%, and 33.7% in cattle manure [
35], poultry manure [
36], swine manure [
37] treatments, respectively. Those studies indicated that the content of fat in
H. illucens was affected by the ingested nutrients. The crude protein content of
H. illucens prepupa in control (446.3 g·kg
-1) was significantly more than those in 1% (430.6 g·kg
-1), 2% (423.3 g·kg
-1), 3% (415.4 g·kg
-1), 4% (410.8 g·kg
-1) and 5% (409.8 g·kg
-1) treatments, respectively. These results are consistent with Spranghers et al. [
34] and Wang et al. [
8], who reported the content of crude protein was 412.0-431.0 g·kg
-1 LPM, and 412.5-436.9 g·kg
-1 LPM.
Furthermore, we also found that the types of fatty acid in the current study were consistent with previous reports [
8,
34,
38,
39]. These studies suggested that the type of substrate negatively affected the fatty acid content but did not alter the fatty acid profile of the prepupae. Except for decanoic acid, stearic acid, and linoleic acid, the fatty acids content of
H. illucens prepupae in control were significantly more than those in 3%, 4%, and 5% treatments, respectively. Furthermore, except for lauric acid, when feeding on kitchen waste, the content of fatty acid was significantly more than those in cow dung treatment [
8]. Spranghers et al. and Wang et al. found that the percentage of lauric acid (C12:0) in prepupa was higher than other fatty acids [
8,
34]. In addition, a large amount of branched-chain fatty acids (C16:1, C18:1n9c, and C18:2n6c) were found in the control and all of salinity treatments. Furthermore, the content of fatty acids were consistent with previous studies of Spranghers et al. and Wang et al. [
8,
34].
Additionally, in the current study we found most of the nonessential and essential amino acids which are consistent with that reported by Newton et al. [
35,
40], St-Hilaire et al. [
41], Sealey et al. [
38], and Spranghers et al. [
34]. Previous studies reported that five amino acids including threonine, lysine, valine, methionine, and isoleucine, were commonly used in the production of livestock and poultry feed [
34,
35,
38,
40,
41]. The content of these five amino acids in
H. illucens prepupae seems to be sufficient for the needs of raising pigs and poultry [
42,
43]. Similar results are also found by Spranghers et al. and Wang et al. [
8,
34]. In contrast, Makkar et al. found that there is a lack of cysteine, methionine, and threonine in the pupa [
33]. The differences in the types and contents of amino acids may be caused by food salinity, food types, or geographic populations. For tryptophan, limited information is available because of the complicated method of determination [
44]. The content range of tryptophan was 2.0-5.9 g·kg
-1 LPM when
H. illucens prepupae fed with various foodstuffs [
35,
40]. In the recent two studies, the content of tryptophan was 5.4-6.7 g·kg
-1 LPM [
34] and 7.1-7.8 g·kg
-1 LPM [
8], which was similar to that in this study. The high differences in the content of tryptophan might be caused by various reasons such as experimental population, measurement techniques, or food intake. In the recent research of Wang et al. [
8], only the content of serine was significantly affected by foodstuff. However, we found that non-essential amino acids such as aspartate, alanine, proline, perine, histidine, and essential amino acids including leucine, lysine, threonine, and isoleucine were significantly affected by foodstuff salinity. The content of amino acids was higher than those reported by Makkar et al. and Spranghers et al. [
33,
34]. In summary, the amino acid content in the prepupae of
H. illucens may be influenced by various factors, such as feed type, salt content, nutrient intake, and geographic population.
The content of mineral in
H. illucens prepupae were influenced by various factors, especially by foodstuff. In this study, we found that the content of Ca changed with a smaller range of 20.52-27.10 g·kg
-1 LPM, which was less than that in the report of Makkar et al. [
33]. The range of Ca content in
H. illucens prepupae feeding food waste, pig manure, chicken manure, and cow dung was 19.48-34.71 g·kg
-1 LPM [
8]. However, Spranghers et al. (2017) reported that there was a greater range of Ca among all treatments, and the range value was 1.23-66.2 g·kg
-1 LPM [
34]. For three principal metallic elements such as K, Mg, and P, that play an important role in animal feed, the weight proportion was significantly higher than that of other minerals. This may be the primary reason the ash content in the 3%, 4%, and 5% treatments was significantly greater than that in the control group. According to the feeding suggestions for livestock (pigs) and poultry (layer hens) [
42,
43], excessive ash content in feed formulations was detrimental to the growth of livestock and poultry, which suggested that the
H. illucens prepupae produced from feed with higher salinity level may have certain drawbacks when used as animal feed. In industrial production, the crude fat should be separated before using
H. illucen pupae to produce feed, which might further increase the proportion of minerals and ash. However, the excessive content of Ca in foodstuffs may increase the gastric juice pH value of livestock and poultry, further increasing the risk of bacterial infection [
45]. Therefore, the
H. illucen prepupae with low ash content produced from the feed with lower salinity may be the more suitable feed for processing.
In particular, a decade of scientific research has already highlighted the huge potential of using insects to improve sustainability of poultry supply chain. So far, the industry can rely on two different ways: 1) larvae can be processed into meals and fats to be used as protein and energy sources, respectively, in poultry diets; or 2) larvae can be used as live by exploiting their potential as environmental enrichment [
46]. Research has already been conducted on the use of
H. illucen meal [
47,
48,
49,
50],
H. illucen dried larvae and
H. illucen fat with positive results in terms of animal health and performance, as well as eggs quality [
51,
52,
53]. Live
H. illucen larva supplementation on laying hens represents a valuable environmental enrichment [
54,
55,
56]: birds are historically foragers of live insects, being part of their traditional diet and allowing them to express a natural behavior [
57,
58,
59]. The animals performance in relations to live insect’s supplementation, as well as the economically feasibility of introducing live
H. illucen larva into the entire production cycle of laying hens, should be evaluated in future.
The composition of gut microbiota develops with host from birth and regulates its health, mainly influenced by dietary factors [
60]. The composition of the gut microbiota in mice was significantly changed by high salt diet, especially the inhibition of lactic acid bacteria, and the similar inhibition from high salt diet was also found in human [
61]. The immune system of
Drosophila melanogaster larvae without symbiotic bacteria could be quickly activated by salt stress, and the pathogen infection was effectively resisted by higher immune activity, which protected
D. melanogaster larvae and extended their lifespan [
62]. The number of gut microbial communities in
D. melanogaster larvae was increased by high salt feed, and the salt stress response could be exacerbated by gut symbiotic bacteria, both of which would inhibit the innate immune system activity and increase the gut microbiota. The survival rate of
D. melanogaster larvae was ultimately reduced by intestinal barrier dysfunction [
63]. In this study, the innate immune activity of
H. illucens larvae with gut microbiota might be inhibited by high salt feed, leading to the dysbiosis of the gut microbiota and the loss of intestinal barrier function, ultimately decreasing the survival rate of
H. illucens larvae. In addition, the high-salt diet could damaged and aged the skeletal muscle cells in
D. melanogaster larvae, and lead to certain negative effects on the larvae heart [
64]. Furthermore, the levels of neurotransmitter and its secretion or transport was significantly inhibited by higher salt diet [
65]. Due to that both
D. melanogaster and
H. illucens belonged to the
Diptera, the negative effects of high-salt feed on
D. melanogaster larvae might occurred for
H. illucens larvae, which might decrease the survival rate of
H. illucens larvae when feeding on the high-salt feed.
In conclusion, the reproductive potential of H. illucen was significantly affected by foodstuff salinity. There was no significant difference in fecundity among the control, 1%, and 2% treatments, which suggested that decreasing the salinity in the foodstuff might be implemented in industrial production. In addition, as the salinity increased, the ash content significantly enhanced while the crude protein decreased. At the same time, both amino acids and mineral contents suggested that dry H. illucens prepupae feeding on low-salinity foodstuffs might be more suitable for use as a feed additive. Therefore, the foodstuff salinity should be controlled in the breeding process of H. illucens prepupae used as livestock and poultry feed. In addition, H. illucens prepupae has great potential in the production of biodiesel due to the higher level of crude fat, and more research should be done in the future.