Submitted:
07 April 2026
Posted:
09 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
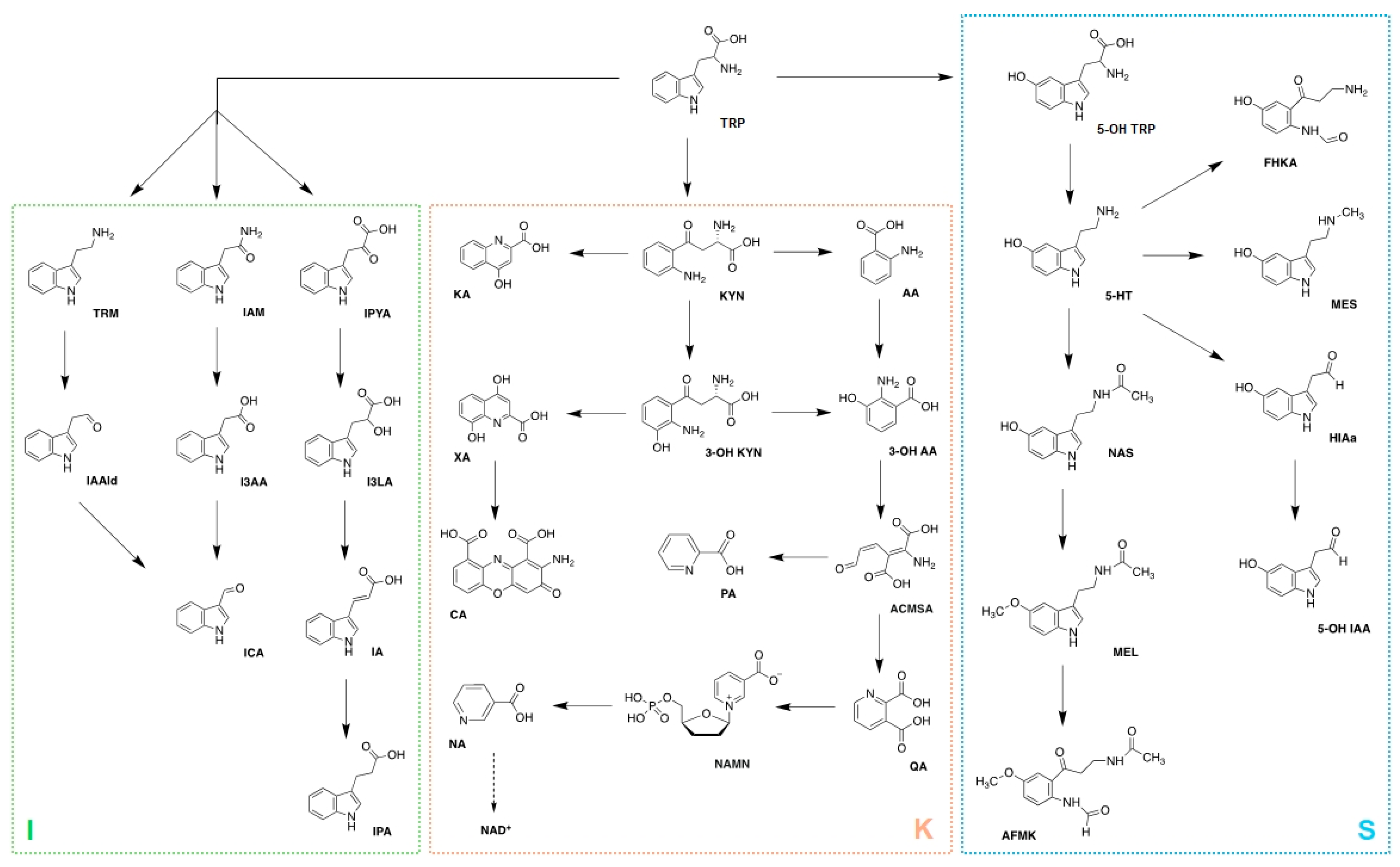
2. Tryptophan Metabolism
2.1. Kynurenine Pathway
2.2. Serotonin Pathway
2.3. Indole Pathway
3. The Neuromodulatory Roles of Kynurenines
3.1. Quinolinic Acid
3.2. Kynurenic Acid
3.3. 3-Hydroxykynurenine
3.4. Xanthurenic and Picolinic Acid
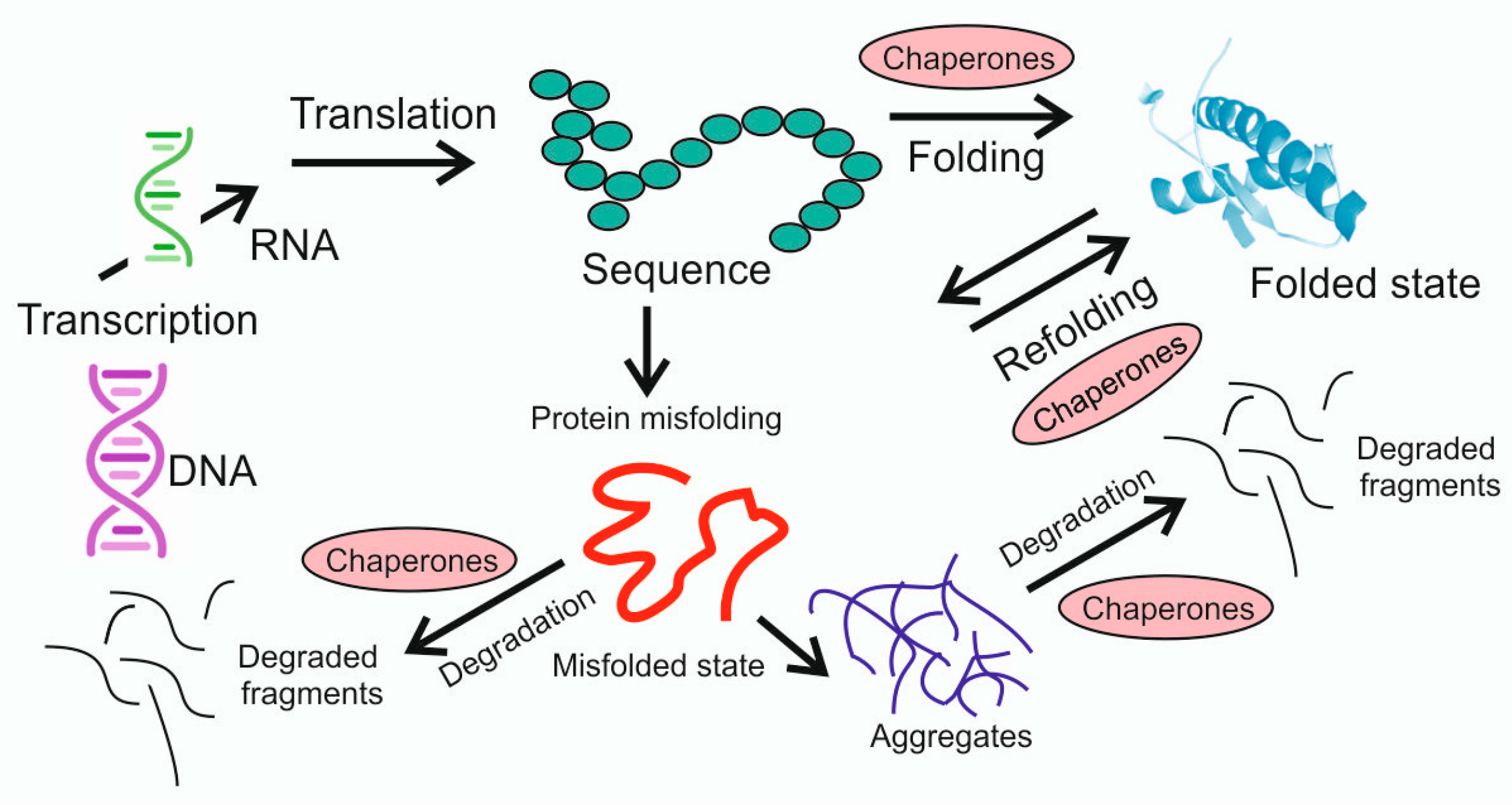
4. Proteinopathies
4.1. Tauopathies
4.2. Amyloidoses
4.3. Synucleinopathies
5. Changes of the Kynurenine Pathway in Selected Neurological Conditions
5.1. Parkinson’s Disease
5.2. Alzheimer’s Disease
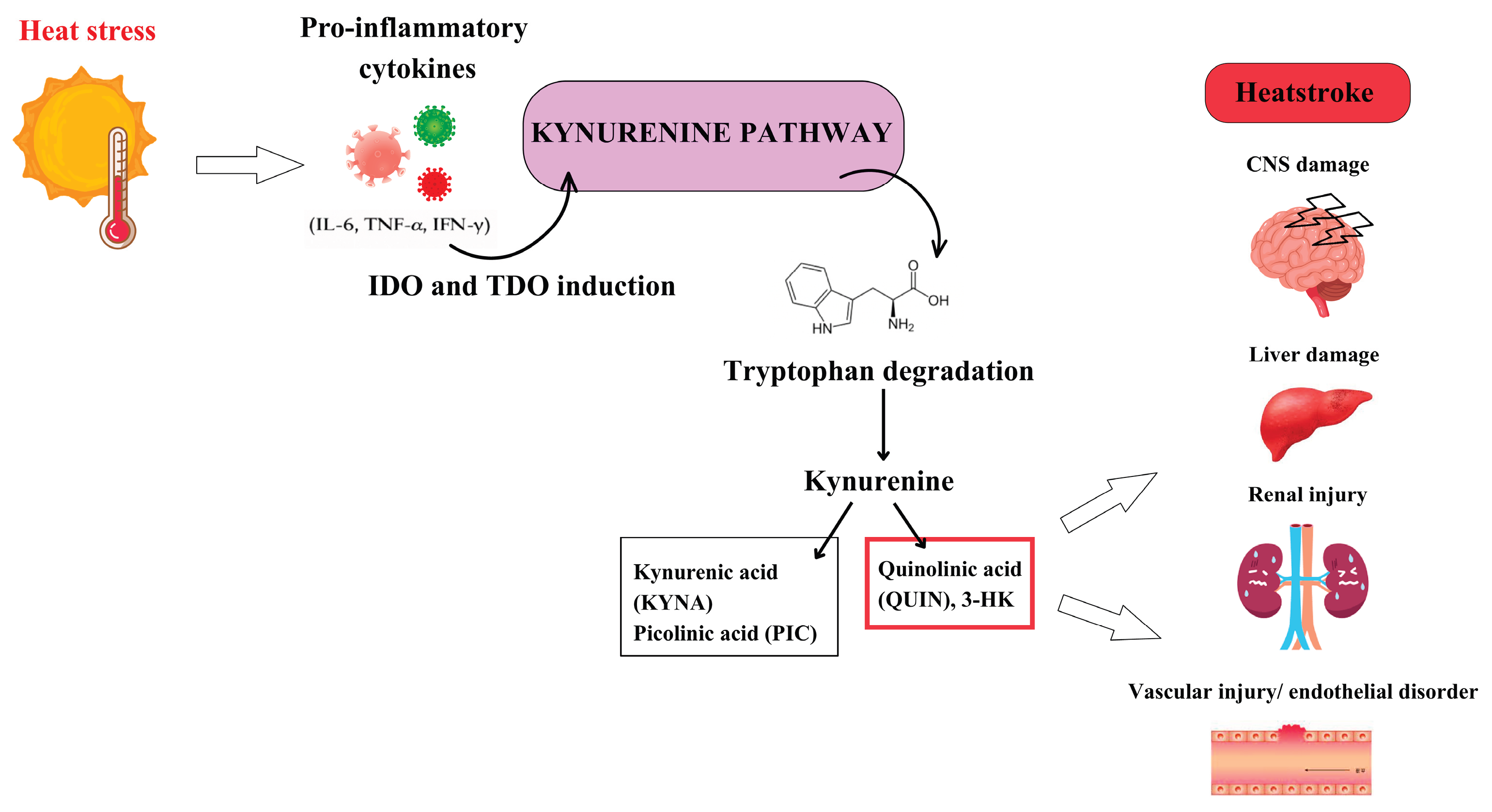
5.3. Heatstroke
6. Conclusions
Acknowledgments
Conflicts of Interest
Abbreviations
| AA | Anthralinic acid |
| Aβ | Amyloid Beta |
| AD | Alzheimer’s disease |
| AhR | Aryl hydrocarbon receptor |
| ALS | Amyotrophic lateral sclerosis |
| AFMK | N-acetyl-N-formyl-5-methoxykynurenamine |
| BBB | Blood-brain barrier |
| CCL1 | C-C motif chemokine ligand 1 |
| CCL5 | C-C motif chemokine ligand 5 |
| CCR3 | Chemokine receptor 3 |
| CCR5 | Chemokine receptor 5 |
| CNS | Central nervous system |
| CSF | Cerebralspinal fluid |
| CXCR4 | C-X-C chemokine receptors 4 |
| CXCR6 | C-X-C chemokine receptors 6 |
| DLB | Dementia with Lewy bodies |
| IDO (IDO1) | Indoleamine 2,3-dioxygenase |
| IFN-γ | Interferon-gamma |
| IL-1β | Interleukin-1β |
| IL-6 | Interleukin-6 |
| IL-8 | Interleukin-8 |
| KAT | Kynurenine aminotransferase |
| KMO | Kynurenine 3-monooxygenase |
| KP | Kynurenine pathway |
| KYN | Kynurenine |
| KYNA | Kynurenine acid |
| LAT1 | L-type amino acid transporter 1 |
| LNAA | Large neutral amino acid |
| MCP-1 | Monocyte chemoattractant protein-1 |
| MEL | Melatonin |
| MIP-1 | Macrophage inflammatory protein-1 |
| MSA | Multiple system atrophy |
| NA | Nicotinic acid |
| NAD+ | Nicotinamide adenine dinucleotide |
| NAS | N-acetylserotonin |
| NMDA | N-methyl-D-aspartate |
| NO | Nitric Oxide |
| nNOS | Neuronal nitric oxide synthase |
| iNOS | Inducible nitric oxide synthase |
| PA | Picolinic acid |
| PARP-1 | Poly(ADP-ribose) polymerase-1 |
| PD | Parkinson’s disease |
| QA | Quinolinic acid |
| ROS | Reactive oxygen species |
| SNpc | Substantia nigra pars compacta |
| SP | Serotonin pathway |
| TDO | Tryptophan 2,3-dioxygenase |
References
- Fallarino, F.; Grohmann, U.; Hwang, K.W.; Orabona, C.; Vacca, C.; Bianchi, R.; Belladonna, M.L.; Fioretti, M.C.; Alegre, M.L.; Puccetti, P. Modulation of tryptophan catabolism by regulatory T cells. Nat Immunol 2003, 4, 1206–1212. [Google Scholar] [CrossRef]
- Starhof, C.; Winge, K.; Heegaard, N.H.H.; Skogstrand, K.; Friis, S.; Hejl, A. Cerebrospinal fluid pro-inflammatory cytokines differentiate parkinsonian syndromes. J Neuroinflammation 2018, 15, 305. [Google Scholar] [CrossRef]
- Streit, W.J.; Mrak, R.E.; Griffin, W.S. Microglia and neuroinflammation: a pathological perspective. J Neuroinflammation 2004, 1, 14. [Google Scholar] [CrossRef]
- Perry, V.H.; Teeling, J. Microglia and macrophages of the central nervous system: the contribution of microglia priming and systemic inflammation to chronic neurodegeneration. Semin Immunopathol 2013, 35, 601–612. [Google Scholar] [CrossRef]
- Liu, Y.Z.; Wang, Y.X.; Jiang, C.L. Inflammation: The Common Pathway of Stress-Related Diseases. Front Hum Neurosci 2017, 11, 316. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Liu, X.; Luo, X.; Luo, Z.; He, G.; Yang, X. Activation of the liver X receptor alpha protects the blood-brain barrier against heatstroke-induced injury. Int J Hyperthermia 2025, 42, 2559022. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Deng, J.; He, Q.; Zhong, Y. ZBP1 and heatstroke. Front Immunol 2023, 14, 1091766. [Google Scholar] [CrossRef]
- Roth, W.; Zadeh, K.; Vekariya, R.; Ge, Y.; Mohamadzadeh, M. Tryptophan Metabolism and Gut-Brain Homeostasis. Int J Mol Sci 2021, 22. [Google Scholar] [CrossRef] [PubMed]
- Palego, L.; Betti, L.; Rossi, A.; Giannaccini, G. Tryptophan Biochemistry: Structural, Nutritional, Metabolic, and Medical Aspects in Humans. J Amino Acids 2016, 2016, 8952520. [Google Scholar] [CrossRef]
- Lovelace, M.D.; Varney, B.; Sundaram, G.; Lennon, M.J.; Lim, C.K.; Jacobs, K.; Guillemin, G.J.; Brew, B.J. Recent evidence for an expanded role of the kynurenine pathway of tryptophan metabolism in neurological diseases. Neuropharmacology 2017, 112, 373–388. [Google Scholar] [CrossRef]
- Savonije, K.; Weaver, D.F. The Role of Tryptophan Metabolism in Alzheimer’s Disease. Brain Sci 2023, 13. [Google Scholar] [CrossRef]
- Atsumi, M.; Mawatari, K.I.; Morooka, A.; Yasuda, M.; Fukuuchi, T.; Yamaoka, N.; Kaneko, K.; Nakagomi, K.; Oku, N. Simultaneous Determination of Kynurenine and Kynurenic Acid by High-Performance Liquid Chromatography Photoirradiation System Using a Mobile Phase Containing 18-crown-6. Int J Tryptophan Res 2019, 12, 1178646919834551. [Google Scholar] [CrossRef]
- Davidson, M.; Rashidi, N.; Nurgali, K.; Apostolopoulos, V. The Role of Tryptophan Metabolites in Neuropsychiatric Disorders. Int J Mol Sci 2022, 23. [Google Scholar] [CrossRef]
- Gal, E.M.; Sherman, A.D. L-kynurenine: its synthesis and possible regulatory function in brain. Neurochem Res 1980, 5, 223–239. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Wu, Q.; Li, K.; Khan, M.A.S.; Zhang, A.; Sinha, B.; Li, S.; Chang, S.L.; Brody, D.L.; Grinstaff, M.W.; et al. Tryptophan Metabolism in Alzheimer’s Disease with the Involvement of Microglia and Astrocyte Crosstalk and Gut-Brain Axis. Aging Dis 2024, 15, 2168–2190. [Google Scholar] [CrossRef]
- Routy, J.P.; Routy, B.; Graziani, G.M.; Mehraj, V. The Kynurenine Pathway Is a Double-Edged Sword in Immune-Privileged Sites and in Cancer: Implications for Immunotherapy. Int J Tryptophan Res 2016, 9, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Hajsl, M.; Hlavackova, A.; Broulikova, K.; Sramek, M.; Maly, M.; Dyr, J.E.; Suttnar, J. Tryptophan Metabolism, Inflammation, and Oxidative Stress in Patients with Neurovascular Disease. Metabolites 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, F.B.; Byrne, L.M.; Lowe, A.J.; Tortelli, R.; Heins, M.; Flik, G.; Johnson, E.B.; De Vita, E.; Scahill, R.I.; Giorgini, F.; et al. Kynurenine pathway metabolites in cerebrospinal fluid and blood as potential biomarkers in Huntington’s disease. J Neurochem 2021, 158, 539–553. [Google Scholar] [CrossRef]
- Zhang, J.; Zheng, M.; Shi, W. Parkinson’s disease: an integrative bioinformatics and machine learning analysis reveals tryptophan metabolism-associated gene biomarkers. BMC Neurol 2025, 25, 487. [Google Scholar] [CrossRef]
- Liang, Y.; Xie, S.; He, Y.; Xu, M.; Qiao, X.; Zhu, Y.; Wu, W. Kynurenine Pathway Metabolites as Biomarkers in Alzheimer’s Disease. Dis Markers 2022, 2022, 9484217. [Google Scholar] [CrossRef]
- Song, P.; Ramprasath, T.; Wang, H.; Zou, M.H. Abnormal kynurenine pathway of tryptophan catabolism in cardiovascular diseases. Cell Mol Life Sci 2017, 74, 2899–2916. [Google Scholar] [CrossRef]
- Platten, M.; Nollen, E.A.A.; Rohrig, U.F.; Fallarino, F.; Opitz, C.A. Tryptophan metabolism as a common therapeutic target in cancer, neurodegeneration and beyond. Nat Rev Drug Discov 2019, 18, 379–401. [Google Scholar] [CrossRef]
- Dehhaghi, M.; Panahi, H.K.S.; Kavyani, B.; Heng, B.; Tan, V.; Braidy, N.; Guillemin, G.J. The Role of Kynurenine Pathway and NAD(+) Metabolism in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Aging Dis 2022, 13, 698–711. [Google Scholar] [CrossRef]
- Jacobs, K.R.; Lim, C.K.; Blennow, K.; Zetterberg, H.; Chatterjee, P.; Martins, R.N.; Brew, B.J.; Guillemin, G.J.; Lovejoy, D.B. Correlation between plasma and CSF concentrations of kynurenine pathway metabolites in Alzheimer’s disease and relationship to amyloid-beta and tau. Neurobiol Aging 2019, 80, 11–20. [Google Scholar] [CrossRef]
- Kaleta, M.; Henykova, E.; Mensikova, K.; Friedecky, D.; Kvasnicka, A.; Klicova, K.; Konickova, D.; Strnad, M.; Kanovsky, P.; Novak, O. Patients with Neurodegenerative Proteinopathies Exhibit Altered Tryptophan Metabolism in the Serum and Cerebrospinal Fluid. ACS Chem Neurosci 2024, 15, 582–592. [Google Scholar] [CrossRef]
- Mondanelli, G.; Volpi, C. The double life of serotonin metabolites: in the mood for joining neuronal and immune systems. Curr Opin Immunol 2021, 70, 1–6. [Google Scholar] [CrossRef]
- Morimoto, S.; Takao, M.; Hatsuta, H.; Nishina, Y.; Komiya, T.; Sengoku, R.; Nakano, Y.; Uchino, A.; Sumikura, H.; Saito, Y.; et al. Homovanillic acid and 5-hydroxyindole acetic acid as biomarkers for dementia with Lewy bodies and coincident Alzheimer’s disease: An autopsy-confirmed study. PLoS One 2017, 12, e0171524. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, J.J.; Noristani, H.N.; Verkhratsky, A. The serotonergic system in ageing and Alzheimer’s disease. Prog Neurobiol 2012, 99, 15–41. [Google Scholar] [CrossRef] [PubMed]
- Lefevre, A.; Mavel, S.; Nadal-Desbarats, L.; Galineau, L.; Attucci, S.; Dufour, D.; Sokol, H.; Emond, P. Validation of a global quantitative analysis methodology of tryptophan metabolites in mice using LC-MS. Talanta 2019, 195, 593–598. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.B.; Tanaka, A.; Kuhara, T.; Xiao, J.Z. Potential Effects of Indole-3-Lactic Acid, a Metabolite of Human Bifidobacteria, on NGF-induced Neurite Outgrowth in PC12 Cells. Microorganisms 2020, 8. [Google Scholar] [CrossRef]
- Konopelski, P.; Mogilnicka, I. Biological Effects of Indole-3-Propionic Acid, a Gut Microbiota-Derived Metabolite, and Its Precursor Tryptophan in Mammals’ Health and Disease. Int J Mol Sci 2022, 23. [Google Scholar] [CrossRef]
- Zhou, Y.; Chen, Y.; He, H.; Peng, M.; Zeng, M.; Sun, H. The role of the indoles in microbiota-gut-brain axis and potential therapeutic targets: A focus on human neurological and neuropsychiatric diseases. Neuropharmacology 2023, 239, 109690. [Google Scholar] [CrossRef]
- Leng, F.; Edison, P. Neuroinflammation and microglial activation in Alzheimer disease: where do we go from here? Nat Rev Neurol 2021, 17, 157–172. [Google Scholar] [CrossRef]
- Jiang, X.; Xu, L.; Tang, L.; Liu, F.; Chen, Z.; Zhang, J.; Chen, L.; Pang, C.; Yu, X. Role of the indoleamine-2,3-dioxygenase/kynurenine pathway of tryptophan metabolism in behavioral alterations in a hepatic encephalopathy rat model. J Neuroinflammation 2018, 15, 3. [Google Scholar] [CrossRef]
- DiSabato, D.J.; Quan, N.; Godbout, J.P. Neuroinflammation: the devil is in the details. J Neurochem 2016, 139 Suppl 2, 136–153. [Google Scholar] [CrossRef]
- Taylor, M.W.; Feng, G.S. Relationship between interferon-gamma, indoleamine 2,3-dioxygenase, and tryptophan catabolism. FASEB J 1991, 5, 2516–2522. [Google Scholar] [CrossRef]
- Schwarcz, R.; Stone, T.W. The kynurenine pathway and the brain: Challenges, controversies and promises. Neuropharmacology 2017, 112, 237–247. [Google Scholar] [CrossRef] [PubMed]
- Maes, M. Depression is an inflammatory disease, but cell-mediated immune activation is the key component of depression. Prog Neuropsychopharmacol Biol Psychiatry 2011, 35, 664–675. [Google Scholar] [CrossRef] [PubMed]
- Braidy, N.; Grant, R.; Brew, B.J.; Adams, S.; Jayasena, T.; Guillemin, G.J. Effects of Kynurenine Pathway Metabolites on Intracellular NAD Synthesis and Cell Death in Human Primary Astrocytes and Neurons. Int J Tryptophan Res 2009, 2, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Stone, T.W.; Clanchy, F.I.L.; Huang, Y.S.; Chiang, N.Y.; Darlington, L.G.; Williams, R.O. An integrated cytokine and kynurenine network as the basis of neuroimmune communication. Front Neurosci 2022, 16, 1002004. [Google Scholar] [CrossRef]
- Giorgini, F.; Huang, S.Y.; Sathyasaikumar, K.V.; Notarangelo, F.M.; Thomas, M.A.; Tararina, M.; Wu, H.Q.; Schwarcz, R.; Muchowski, P.J. Targeted deletion of kynurenine 3-monooxygenase in mice: a new tool for studying kynurenine pathway metabolism in periphery and brain. J Biol Chem 2013, 288, 36554–36566. [Google Scholar] [CrossRef] [PubMed]
- Zwilling, D.; Huang, S.Y.; Sathyasaikumar, K.V.; Notarangelo, F.M.; Guidetti, P.; Wu, H.Q.; Lee, J.; Truong, J.; Andrews-Zwilling, Y.; Hsieh, E.W.; et al. Kynurenine 3-monooxygenase inhibition in blood ameliorates neurodegeneration. Cell 2011, 145, 863–874. [Google Scholar] [CrossRef]
- Campesan, S.; Green, E.W.; Breda, C.; Sathyasaikumar, K.V.; Muchowski, P.J.; Schwarcz, R.; Kyriacou, C.P.; Giorgini, F. The kynurenine pathway modulates neurodegeneration in a Drosophila model of Huntington’s disease. Curr Biol 2011, 21, 961–966. [Google Scholar] [CrossRef]
- de Carvalho, L.P.; Bochet, P.; Rossier, J. The endogenous agonist quinolinic acid and the non endogenous homoquinolinic acid discriminate between NMDAR2 receptor subunits. Neurochem Int 1996, 28, 445–452. [Google Scholar] [CrossRef]
- Guidetti, P.; Hoffman, G.E.; Melendez-Ferro, M.; Albuquerque, E.X.; Schwarcz, R. Astrocytic localization of kynurenine aminotransferase II in the rat brain visualized by immunocytochemistry. Glia 2007, 55, 78–92. [Google Scholar] [CrossRef]
- Chen, Y.; Brew, B.J.; Guillemin, G.J. Characterization of the kynurenine pathway in NSC-34 cell line: implications for amyotrophic lateral sclerosis. J Neurochem 2011, 118, 816–825. [Google Scholar] [CrossRef] [PubMed]
- Pierozan, P.; Ferreira, F.; de Lima, B.O.; Pessoa-Pureur, R. Quinolinic acid induces disrupts cytoskeletal homeostasis in striatal neurons. Protective role of astrocyte-neuron interaction. J Neurosci Res 2015, 93, 268–284. [Google Scholar] [CrossRef]
- Guillemin, G.J.; Croitoru-Lamoury, J.; Dormont, D.; Armati, P.J.; Brew, B.J. Quinolinic acid upregulates chemokine production and chemokine receptor expression in astrocytes. Glia 2003, 41, 371–381. [Google Scholar] [CrossRef]
- Rahman, A.; Ting, K.; Cullen, K.M.; Braidy, N.; Brew, B.J.; Guillemin, G.J. The excitotoxin quinolinic acid induces tau phosphorylation in human neurons. PLoS One 2009, 4, e6344. [Google Scholar] [CrossRef]
- Maddison, D.C.; Giorgini, F. The kynurenine pathway and neurodegenerative disease. Semin Cell Dev Biol 2015, 40, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Guillemin, G.J.; Wang, L.; Brew, B.J. Quinolinic acid selectively induces apoptosis of human astrocytes: potential role in AIDS dementia complex. J Neuroinflammation 2005, 2, 16. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.S.; Ogbechi, J.; Clanchy, F.I.; Williams, R.O.; Stone, T.W. IDO and Kynurenine Metabolites in Peripheral and CNS Disorders. Front Immunol 2020, 11, 388. [Google Scholar] [CrossRef]
- Kiss, C.; Ceresoli-Borroni, G.; Guidetti, P.; Zielke, C.L.; Zielke, H.R.; Schwarcz, R. Kynurenate production by cultured human astrocytes. J Neural Transm (Vienna) 2003, 110, 1–14. [Google Scholar] [CrossRef]
- Blanco Ayala, T.; Lugo Huitron, R.; Carmona Aparicio, L.; Ramirez Ortega, D.; Gonzalez Esquivel, D.; Pedraza Chaverri, J.; Perez de la Cruz, G.; Rios, C.; Schwarcz, R.; Perez de la Cruz, V. Alternative kynurenic acid synthesis routes studied in the rat cerebellum. Front Cell Neurosci 2015, 9, 178. [Google Scholar] [CrossRef]
- Flores-Barrera, E.; Thomases, D.R. Kynurenic acid and cognitive function. Neuropsychopharmacology 2017. [Google Scholar]
- Gonzalez-Sanchez, M.; Jimenez, J.; Narvaez, A.; Antequera, D.; Llamas-Velasco, S.; Martin, A.H.; Arjona, J.A.M.; Munain, A.L.; Bisa, A.L.; Marco, M.P.; et al. Kynurenic Acid Levels are Increased in the CSF of Alzheimer’s Disease Patients. Biomolecules 2020, 10. [Google Scholar] [CrossRef]
- Pierozan, P.; Biasibetti-Brendler, H. Kynurenic acid and oxidative stress modulation. Neurotoxicity Research 2018. [Google Scholar]
- Kozak, R.; Campbell, B.M.; Strick, C.A.; Horner, W.; Hoffmann, W.E.; Kiss, T.; Chapin, D.S.; McGinnis, D.; Abbott, A.L.; Roberts, B.M.; et al. Reduction of brain kynurenic acid improves cognitive function. J Neurosci 2014, 34, 10592–10602. [Google Scholar] [CrossRef] [PubMed]
- DiNatale, B.C.; Murray, I.A.; Schroeder, J.C.; Flaveny, C.A.; Lahoti, T.S.; Laurenzana, E.M.; Omiecinski, C.J.; Perdew, G.H. Kynurenic acid is a potent endogenous aryl hydrocarbon receptor ligand that synergistically induces interleukin-6 in the presence of inflammatory signaling. Toxicol Sci 2010, 115, 89–97. [Google Scholar] [CrossRef]
- Reyes-Ocampo, J.; Lugo-Huitron, R. Kynurenines and neurodegeneration. In Oxidative Medicine and Cellular Longevity; 2014. [Google Scholar]
- Leipnitz, G.; Schumacher, C. Protective effects of kynurenine metabolites. In Neurochemistry International; 2007. [Google Scholar]
- Sathyasaikumar, K.V.; Tararina, M.; Wu, H.Q.; Neale, S.A.; Weisz, F.; Salt, T.E.; Schwarcz, R. Xanthurenic Acid Formation from 3-Hydroxykynurenine in the Mammalian Brain: Neurochemical Characterization and Physiological Effects. Neuroscience 2017, 367, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Cioczek-Czuczwar, A.; Czuczwar, P.; Turski, W.A.; Parada-Turska, J. Influence of picolinic acid on seizure susceptibility in mice. Pharmacological Reports 2017, 69, 77–80. [Google Scholar] [CrossRef]
- Vrooman, B.M.; Jhamandas, K. Picolinic acid and glutamatergic neurotransmission. Brain Research 1993. [Google Scholar]
- Delacourte, A. Tauopathies: recent insights into old diseases. Folia Neuropathol 2005, 43, 244–257. [Google Scholar]
- Engelender, S.; Stefanis, L.; Oddo, S.; Bellucci, A. Can We Treat Neurodegenerative Proteinopathies by Enhancing Protein Degradation? Mov Disord 2022, 37, 1346–1359. [Google Scholar] [CrossRef]
- Biswas, P. Theoretical and computational advances in protein misfolding. Adv Protein Chem Struct Biol 2019, 118, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Kulichikhin, K.Y.; Malikova, O.A.; Zobnina, A.E.; Zalutskaya, N.M.; Rubel, A.A. Interaction of Proteins Involved in Neuronal Proteinopathies. Life (Basel) 2023, 13. [Google Scholar] [CrossRef]
- Lee, G.; Leugers, C.J. Tau and tauopathies. Prog Mol Biol Transl Sci 2012, 107, 263–293. [Google Scholar] [CrossRef] [PubMed]
- Ashrafian, H.; Zadeh, E.H.; Khan, R.H. Review on Alzheimer’s disease: Inhibition of amyloid beta and tau tangle formation. Int J Biol Macromol 2021, 167, 382–394. [Google Scholar] [CrossRef]
- Sexton, C.; Snyder, H.; Beher, D.; Boxer, A.L.; Brannelly, P.; Brion, J.P.; Buee, L.; Cacace, A.M.; Chetelat, G.; Citron, M.; et al. Current directions in tau research: Highlights from Tau 2020. Alzheimers Dement 2022, 18, 988–1007. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, G.G. Tauopathies. Handb Clin Neurol 2017, 145, 355–368. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, K.M.; Yang, L.; Dong, Q.; Yu, J.T. Tauopathies: new perspectives and challenges. Mol Neurodegener 2022, 17, 28. [Google Scholar] [CrossRef]
- Pandey, P.; Sliker, B.; Peters, H.L.; Tuli, A.; Herskovitz, J.; Smits, K.; Purohit, A.; Singh, R.K.; Dong, J.; Batra, S.K.; et al. Amyloid precursor protein and amyloid precursor-like protein 2 in cancer. Oncotarget 2016, 7, 19430–19444. [Google Scholar] [CrossRef]
- Chen, G.F.; Xu, T.H.; Yan, Y.; Zhou, Y.R.; Jiang, Y.; Melcher, K.; Xu, H.E. Amyloid beta: structure, biology and structure-based therapeutic development. Acta Pharmacol Sin 2017, 38, 1205–1235. [Google Scholar] [CrossRef]
- Jeremic, D.; Jimenez-Diaz, L.; Navarro-Lopez, J.D. Past, present and future of therapeutic strategies against amyloid-beta peptides in Alzheimer’s disease: a systematic review. Ageing Res Rev 2021, 72, 101496. [Google Scholar] [CrossRef] [PubMed]
- Yiannopoulou, K.G.; Papageorgiou, S.G. Current and Future Treatments in Alzheimer Disease: An Update. J Cent Nerv Syst Dis 2020, 12, 1179573520907397. [Google Scholar] [CrossRef] [PubMed]
- Abeysinghe, A.; Deshapriya, R.; Udawatte, C. Alzheimer’s disease; a review of the pathophysiological basis and therapeutic interventions. Life Sci 2020, 256, 117996. [Google Scholar] [CrossRef] [PubMed]
- Gireud-Goss, M.; Mack, A.F.; McCullough, L.D.; Urayama, A. Cerebral Amyloid Angiopathy and Blood-Brain Barrier Dysfunction. Neuroscientist 2021, 27, 668–684. [Google Scholar] [CrossRef]
- Yang, W.; Yu, S. Synucleinopathies: common features and hippocampal manifestations. Cell Mol Life Sci 2017, 74, 1485–1501. [Google Scholar] [CrossRef]
- Stoll, A.C.; Kemp, C.J.; Patterson, J.R.; Kubik, M.; Kuhn, N.; Benskey, M.; Duffy, M.F.; Luk, K.C.; Sortwell, C.E. Alpha-synuclein inclusion responsive microglia are resistant to CSF1R inhibition. J Neuroinflammation 2024, 21, 108. [Google Scholar] [CrossRef]
- Yang, Z.; Xie, Y.; Dou, K.; Yang, L.; Xie, A. Associations of striatal dopamine transporter binding with motor and non-motor symptoms in early Parkinson’s disease. Clin Transl Sci 2023, 16, 1021–1038. [Google Scholar] [CrossRef]
- Dexter, D.T.; Jenner, P. Parkinson disease: from pathology to molecular disease mechanisms. Free Radic Biol Med 2013, 62, 132–144. [Google Scholar] [CrossRef]
- Langston, J.W.; Ballard, P.; Tetrud, J.W.; Irwin, I. Chronic Parkinsonism in Humans Due to a Product of Meperidine-Analog Synthesis. Science 1983, 219, 979–980. [Google Scholar] [CrossRef]
- Langston, J.W. The MPTP Story. J Parkinsons Dis 2017, 7, S11–S19. [Google Scholar] [CrossRef] [PubMed]
- Schildknecht, S.; Di Monte, D.A.; Pape, R.; Tieu, K.; Leist, M. Tipping Points and Endogenous Determinants of Nigrostriatal Degeneration by MPTP. Trends Pharmacol Sci 2017, 38, 541–555. [Google Scholar] [CrossRef] [PubMed]
- Knyihár-Csillik, E.; Chadaide, Z.; Mihály, A.; Krisztin-Péva, B.; Fenyő, R.; Vécsei, L. Effect of 6-hydroxydopamine treatment on kynurenine aminotransferase-I immunoreactivity. Acta Neuropathologica 2006, 112, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.Y.; Lee, K.S.; Lee, H.J.; Noh, Y.H.; Kim, D.H.; Lee, J.Y.; Cho, S.H.; Yoon, O.J.; Lee, W.B.; Kim, K.Y.; et al. Kynurenic acid attenuates MPP(+)-induced dopaminergic neuronal cell death via a Bax-mediated mitochondrial pathway. Eur J Cell Biol 2008, 87, 389–397. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, S.; Fu, P.; Zhang, Z.; Lin, K.; Ko, J.K.; Yung, K.K. Roles of Glutamate Receptors in Parkinson’s Disease. Int J Mol Sci 2019, 20. [Google Scholar] [CrossRef]
- Silva-Adaya, D.; Perez-De La Cruz, V.; Villeda-Hernandez, J.; Carrillo-Mora, P.; Gonzalez-Herrera, I.G.; Garcia, E.; Colin-Barenque, L.; Pedraza-Chaverri, J.; Santamaria, A. Protective effect of L-kynurenine and probenecid on 6-hydroxydopamine-induced striatal toxicity in rats: implications of modulating kynurenate as a protective strategy. Neurotoxicol Teratol 2011, 33, 303–312. [Google Scholar] [CrossRef]
- Lim, C.K.; Fernandez-Gomez, F.J.; Braidy, N.; Estrada, C.; Costa, C.; Costa, S.; Bessede, A.; Fernandez-Villalba, E.; Zinger, A.; Herrero, M.T.; et al. Involvement of the kynurenine pathway in the pathogenesis of Parkinson’s disease. Prog Neurobiol 2017, 155, 76–95. [Google Scholar] [CrossRef]
- Beal, M.F.; Matson, W.R.; Storey, E.; Milbury, P.; Ryan, E.A.; Ogawa, T.; Bird, E.D. Kynurenic acid concentrations are reduced in Huntington’s disease cerebral cortex. J Neurol Sci 1992, 108, 80–87. [Google Scholar] [CrossRef]
- Okuda, S.; Nishiyama, N.; Saito, H.; Katsuki, H. 3-hydroxykynurenine, an endogenous oxidative stress generator, causes neuronal cell death with apoptotic features and region selectivity. Journal of Neurochemistry 1998, 70, 299–307. [Google Scholar] [CrossRef] [PubMed]
- Wilson, E.N.; Umans, J.; Swarovski, M.S.; Minhas, P.S.; Mendiola, J.H.; Midttun, O.; Ulvik, A.; Shahid-Besanti, M.; Linortner, P.; Mhatre, S.D.; et al. Parkinson’s disease is characterized by vitamin B6-dependent inflammatory kynurenine pathway dysfunction. NPJ Parkinsons Dis 2025, 11, 96. [Google Scholar] [CrossRef]
- Ogawa, T.; Matson, W.R.; Beal, M.F.; Myers, R.H.; Bird, E.D.; Milbury, P.; Saso, S. Kynurenine pathway abnormalities in Parkinson’s disease. Neurology 1992, 42, 1702–1706. [Google Scholar] [CrossRef]
- Chang, K.H.; Cheng, M.L.; Tang, H.Y.; Huang, C.Y.; Wu, Y.R.; Chen, C.M. Alternations of Metabolic Profile and Kynurenine Metabolism in the Plasma of Parkinson’s Disease. Mol Neurobiol 2018, 55, 6319–6328. [Google Scholar] [CrossRef]
- Havelund, J.F.; Andersen, A.D.; Binzer, M.; Blaabjerg, M.; Heegaard, N.H.H.; Stenager, E.; Faergeman, N.J.; Gramsbergen, J.B. Changes in kynurenine pathway metabolism in Parkinson patients with L-DOPA-induced dyskinesia. J Neurochem 2017, 142, 756–766. [Google Scholar] [CrossRef]
- Oxenkrug, G.; van der Hart, M.; Roeser, J.; Summergrad, P. Peripheral tryptophan-kynurenine metabolism in Parkinson’s disease. Endocrinology Diabetes and Metabolism Journal 2017, 1. [Google Scholar]
- Heilman, P.L.; Wang, E.W.; Lewis, M.M.; Krzyzanowski, S.; Capan, C.D.; Burmeister, A.R.; Du, G.; Escobar Galvis, M.L.; Brundin, P.; Huang, X.; et al. Tryptophan Metabolites Are Associated With Symptoms and Nigral Pathology in Parkinson’s Disease. Mov Disord 2020, 35, 2028–2037. [Google Scholar] [CrossRef] [PubMed]
- Fukuyama, K.; Tanahashi, S.; Hoshikawa, M.; Shinagawa, R.; Okada, M. Zonisamide regulates basal ganglia transmission via astroglial kynurenine pathway. Neuropharmacology 2014, 76 Pt A, 137–145. [Google Scholar] [CrossRef]
- Luchowski, P.; Luchowska, E.; Turski, W.A.; Urbanska, E.M. 1-Methyl-4-phenylpyridinium and 3-nitropropionic acid diminish cortical synthesis of kynurenic acid via interference with kynurenine aminotransferases in rats. Neurosci Lett 2002, 330, 49–52. [Google Scholar] [CrossRef]
- Liu, G.; Fan, G.; Guo, G.; Kang, W.; Wang, D.; Xu, B.; Zhao, J. FK506 Attenuates the Inflammation in Rat Spinal Cord Injury by Inhibiting the Activation of NF-kappaB in Microglia Cells. Cell Mol Neurobiol 2017, 37, 843–855. [Google Scholar] [CrossRef]
- Kou, Y.; Jin, Z.; Yuan, Y.; Ma, B.; Xie, W.; Han, N. FK506 contributes to peripheral nerve regeneration by inhibiting neuroinflammatory responses and promoting neuron survival. Neural Regen Res 2025, 20, 2108–2115. [Google Scholar] [CrossRef]
- Campbell, B.M.; Charych, E.; Lee, A.W.; Moller, T. Kynurenines in CNS disease: regulation by inflammatory cytokines. Front Neurosci 2014, 8, 12. [Google Scholar] [CrossRef] [PubMed]
- Duan, Z.; Zhang, S.; Liang, H.; Xing, Z.; Guo, L.; Shi, L.; Du, L.; Kuang, C.; Takikawa, O.; Yang, Q. Amyloid beta neurotoxicity is IDO1-Kyn-AhR dependent and blocked by IDO1 inhibitor. Signal Transduct Target Ther 2020, 5, 96. [Google Scholar] [CrossRef] [PubMed]
- Novoa, C.; Salazar, P.; Cisternas, P.; Gherardelli, C.; Vera-Salazar, R.; Zolezzi, J.M.; Inestrosa, N.C. Inflammation context in Alzheimer’s disease, a relationship intricate to define. Biol Res 2022, 55, 39. [Google Scholar] [CrossRef] [PubMed]
- Dugue, P.A.; Hodge, A.M.; Ulvik, A.; Ueland, P.M.; Midttun, O.; Rinaldi, S.; MacInnis, R.J.; Li, S.X.; Meyer, K.; Navionis, A.S.; et al. Association of Markers of Inflammation, the Kynurenine Pathway and B Vitamins with Age and Mortality, and a Signature of Inflammaging. J Gerontol A Biol Sci Med Sci 2022, 77, 826–836. [Google Scholar] [CrossRef]
- Huang, T.; Song, J.; Gao, J.; Cheng, J.; Xie, H.; Zhang, L.; Wang, Y.H.; Gao, Z.; Wang, Y.; Wang, X.; et al. Adipocyte-derived kynurenine promotes obesity and insulin resistance by activating the AhR/STAT3/IL-6 signaling. Nat Commun 2022, 13, 3489. [Google Scholar] [CrossRef]
- Sun, P.; Wang, M.; Liu, Y.X.; Li, L.; Chai, X.; Zheng, W.; Chen, S.; Zhu, X.; Zhao, S. High-fat diet-disturbed gut microbiota-colonocyte interactions contribute to dysregulating peripheral tryptophan-kynurenine metabolism. Microbiome 2023, 11, 154. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, T.; Lidberg, L. Decreased plasma ratio of tryptophan to competing large neutral amino acids in human immunodeficiency virus type 1 infected subjects: possible implications for development of neuro-psychiatric disorders. J Neural Transm (Vienna) 1996, 103, 157–164. [Google Scholar] [CrossRef]
- Gulaj, E.; Pawlak, K.; Bien, B.; Pawlak, D. Kynurenine and its metabolites in Alzheimer’s disease patients. Adv Med Sci 2010, 55, 204–211. [Google Scholar] [CrossRef]
- Fernandes, B.S.; Inam, M.E.; Enduru, N.; Quevedo, J.; Zhao, Z. The kynurenine pathway in Alzheimer’s disease: a meta-analysis of central and peripheral levels. Braz J Psychiatry 2023, 45, 286–297. [Google Scholar] [CrossRef]
- Bakker, L.; Kohler, S.; Eussen, S.; Choe, K.; van den Hove, D.L.A.; Kenis, G.; Rutten, B.P.F.; Ulvik, A.; Ueland, P.M.; Verhey, F.R.J.; et al. Correlations between kynurenines in plasma and CSF, and their relation to markers of Alzheimer’s disease pathology. Brain Behav Immun 2023, 111, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Willette, A.A.; Pappas, C.; Hoth, N.; Wang, Q.; Klinedinst, B.; Willette, S.A.; Larsen, B.; Pollpeter, A.; Li, T.; Le, S.; et al. Inflammation, negative affect, and amyloid burden in Alzheimer’s disease: Insights from the kynurenine pathway. Brain Behav Immun 2021, 95, 216–225. [Google Scholar] [CrossRef]
- Whiley, L.; Chappell, K.E.; D’Hondt, E.; Lewis, M.R.; Jimenez, B.; Snowden, S.G.; Soininen, H.; Kloszewska, I.; Mecocci, P.; Tsolaki, M.; et al. Metabolic phenotyping reveals a reduction in the bioavailability of serotonin and kynurenine pathway metabolites in both the urine and serum of individuals living with Alzheimer’s disease. Alzheimers Res Ther 2021, 13, 20. [Google Scholar] [CrossRef]
- Baran, H.; Jellinger, K.; Deecke, L. Kynurenine metabolism in Alzheimer’s disease. J Neural Transm (Vienna) 1999, 106, 165–181. [Google Scholar] [CrossRef]
- Sorgdrager, F.J.H.; Vermeiren, Y.; Van Faassen, M.; van der Ley, C.; Nollen, E.A.A.; Kema, I.P.; De Deyn, P.P. Age- and disease-specific changes of the kynurenine pathway in Parkinson’s and Alzheimer’s disease. J Neurochem 2019, 151, 656–668. [Google Scholar] [CrossRef]
- Majerova, P.; Olesova, D.; Golisova, G.; Buralova, M.; Michalicova, A.; Vegh, J.; Piestansky, J.; Bhide, M.; Hanes, J.; Kovac, A. Analog of kynurenic acid decreases tau pathology by modulating astrogliosis in rat model for tauopathy. Biomed Pharmacother 2022, 152, 113257. [Google Scholar] [CrossRef]
- Patel, W.; Rimmer, L.; Smith, M.; Moss, L.; Smith, M.A.; Snodgrass, H.R.; Pirmohamed, M.; Alfirevic, A.; Dickens, D. Probenecid Increases the Concentration of 7-Chlorokynurenic Acid Derived from the Prodrug 4-Chlorokynurenine within the Prefrontal Cortex. Mol Pharm 2021, 18, 113–123. [Google Scholar] [CrossRef]
- Hokari, M.; Wu, H.Q.; Schwarcz, R.; Smith, Q.R. Facilitated brain uptake of 4-chlorokynurenine and conversion to 7-chlorokynurenic acid. Neuroreport 1996, 8, 15–18. [Google Scholar] [CrossRef] [PubMed]
- Yoneda, K.; Hosomi, S.; Ito, H.; Togami, Y.; Oda, S.; Matsumoto, H.; Shimazaki, J.; Ogura, H.; Oda, J. How can heatstroke damage the brain? A mini review. Front Neurosci 2024, 18, 1437216. [Google Scholar] [CrossRef] [PubMed]
- Louvrou, V.; Eimantas, N.; Juskeviciute, E.; Solianik, R.; Brazaitis, M.; Engberg, G.; Erhardt, S. Effects of Heat Exposure on Kynurenine Pathway Metabolism-Comparison Between Exertional and Exogenous Heating of the Human Body. Acta Physiol (Oxf) 2025, 241, e70123. [Google Scholar] [CrossRef] [PubMed]
- Epstein, Y.; Yanovich, R. Heatstroke. N Engl J Med 2019, 380, 2449–2459. [Google Scholar] [CrossRef] [PubMed]
- Leon, L.R.; Helwig, B.G. Heat stroke: role of the systemic inflammatory response. J Appl Physiol (1985) 2010, 109, 1980–1988. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.Y.; Zhao, Y.J.; Wang, Y.Y.; Wei, W.; Hu, W.; Chang, Y. Involvement of the tryptophan-kynurenine pathway in the pathogenesis of autoimmune diseases. Exp Gerontol 2025, 209, 112837. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



