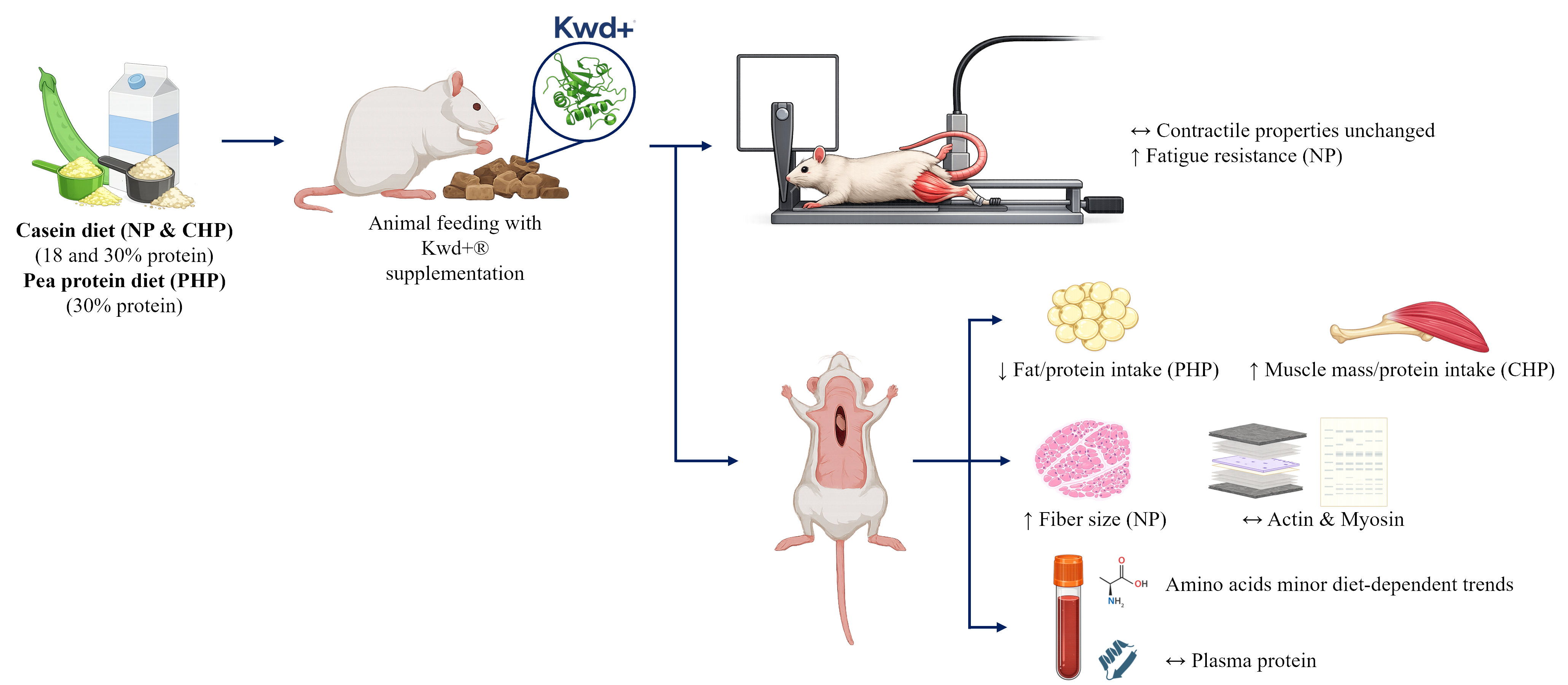
Protein utilization efficiency is a key determinant of metabolic health, body composition, and muscle function, particularly under high-protein dietary conditions and when using plant-based protein sources with lower digestibility. However, the extent to which en-hancing early gastric proteolysis translates into improved whole-body protein utilization remains unclear. The present in vivo study investigated whether supplementation with an actinidin-enriched kiwifruit concentrate (Kwd+®) modulates protein utilization efficiency and physiological outcomes under normoproteic and hyperproteic dietary conditions us-ing casein or pea protein as dietary sources. Rats were fed normoproteic casein (NP), high-protein casein (CHP), or high-protein pea (PHP) diets with or without Kwd+® sup-plementation for 8 weeks. Gastric protein hydrolysis patterns were evaluated by SDS–PAGE, while metabolic outcomes were assessed through body composition, plasma amino acid profiling, skeletal muscle morphology, and contractile function. Kwd+® supplemen-tation enhanced gastric proteolysis in a protein source-dependent manner. Despite the absence of significant changes in circulating amino acid concentrations after multiple comparison correction, Kwd+® improved markers of protein utilization efficiency de-pending on dietary protein source. In animals fed a high-protein pea diet, supplementa-tion significantly reduced fat mass relative to accumulated protein intake, indicating im-proved nutrient partitioning. In contrast, in animals fed a high-protein casein diet, Kwd+® increased muscle mass relative to protein intake, suggesting enhanced anabolic efficiency. Under normoproteic conditions, Kwd+® supplementation was associated with increased muscle fiber cross-sectional area and improved fatigue resistance without alterations in fiber type composition or contractile protein abundance. These findings demonstrate that modulation of early gastric proteolysis through actinidin produces protein source-dependent effects on protein utilization efficiency, nutrient partitioning, and mus-cle function. This work highlights a novel nutritional strategy to improve metabolic out-comes and muscle performance, particularly in the context of high-protein and plant-based diets.