Introduction
Shiga toxin-producing
Escherichia coli (STEC) are among the most important foodborne pathogens in Europe. In 2024, EU Member States reported 11,738 confirmed human STEC infections (3.5 cases per 100,000 population), making STEC the third most commonly reported zoonosis after campylobacteriosis and salmonellosis, with a significant increasing trend over the past five years [
1]. STEC infections can result in bloody diarrhoea, haemolytic uraemic syndrome (HUS) and death, with children under five years disproportionately affected.
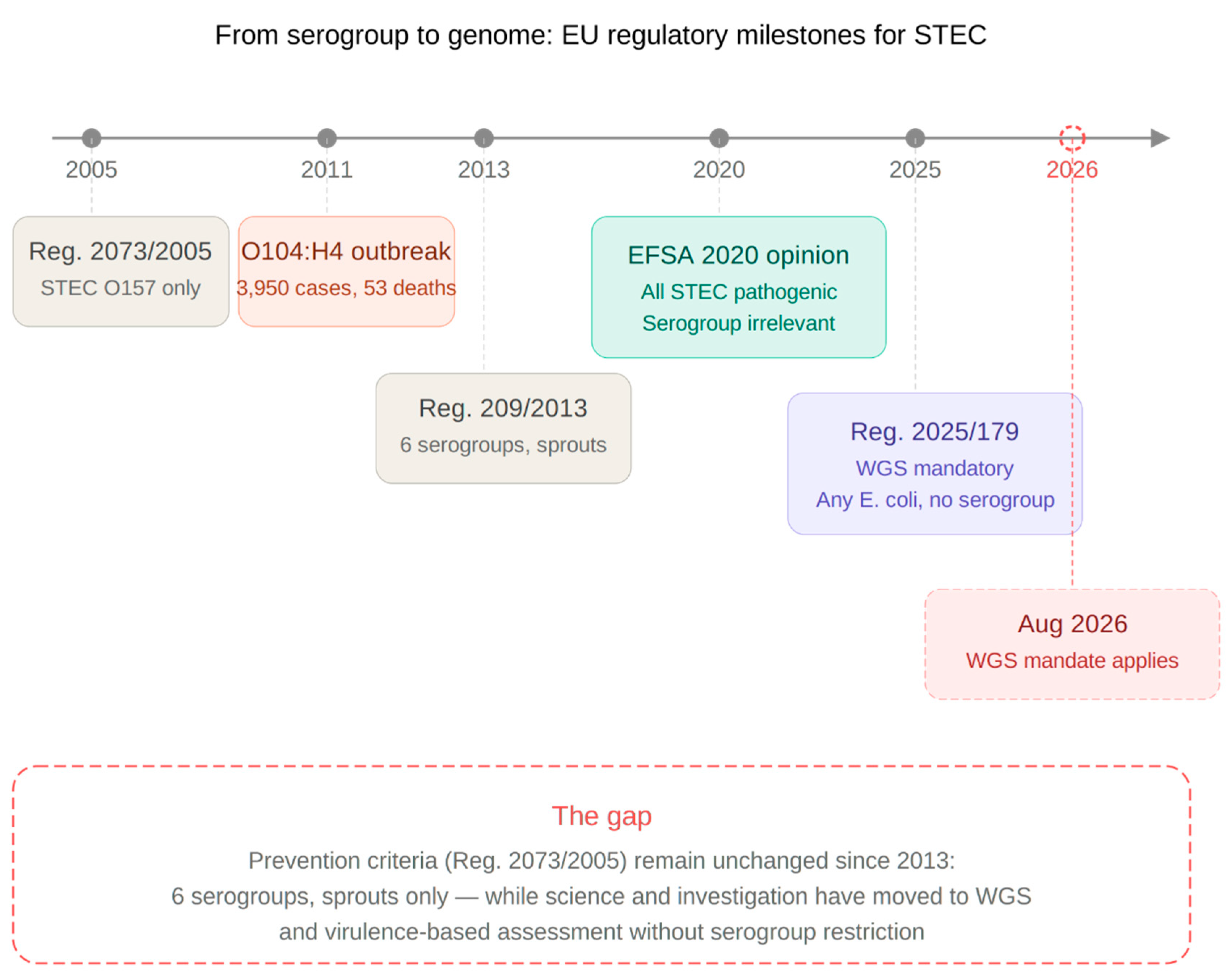
Commission Regulation (EC) No 2073/2005 on microbiological criteria for foodstuffs constitutes the regulatory cornerstone for STEC prevention in the EU food chain [
2]. Since the devastating
E. coli O104:H4 outbreak in Germany in 2011, which caused approximately 3,950 cases and 53 deaths [
3], the regulatory and scientific landscape surrounding STEC has evolved considerably. Three developments now converge to create an urgent need for reflection on the adequacy of current preventive criteria: the 2020 EFSA scientific opinion on STEC pathogenicity [
4], the adoption of Implementing Regulation (EU) 2025/179 mandating whole genome sequencing (WGS) in outbreak investigations [
5], and the ongoing revision of ISO/TS 13136 for STEC detection in food [
6].
This Perspective examines whether EU preventive food safety criteria for STEC remain aligned with the current scientific evidence and the Union’s own evolving regulatory framework.
The current preventive framework: serogroups in sprouts, absence of harmonised criteria elsewhere
The EU’s mandatory STEC food safety criteria are remarkably narrow in scope. Regulation 2073/2005, as amended by Regulation (EU) No 209/2013 following the 2011 outbreak, establishes a single food safety criterion for STEC (Annex I, row 1.29): absence of six serogroup designations — O157, O26, O111, O103, O145, and O104:H4 — in 25 grams of sprouts, using the CEN/ISO TS 13136 method [
7]. This criterion was introduced specifically in response to the O104:H4 outbreak, which was traced to fenugreek sprouts.
For all other food categories of recognised public health concern — including raw milk cheese, minced meat, fresh produce other than sprouts, and ready-to-eat foods — Regulation 2073/2005 does not establish any STEC-specific food safety criterion. It should be noted that Article 14 of Regulation (EC) No 178/2002 establishes a general food safety obligation under which any food containing STEC, regardless of serogroup, could be deemed unsafe. However, in the absence of a harmonised microbiological criterion with defined sampling plans and analytical methods, the detection of STEC in these matrices depends entirely on voluntary operator initiatives or ad hoc national guidelines. There is no mandatory sampling frequency, no standardised method, and no EU-wide enforcement baseline. The gap, therefore, is not legal but operational: without an explicit criterion, STEC in high-risk foods other than sprouts is unlikely to be systematically detected before reaching consumers. This stands in contrast to the United States, where the USDA Food Safety and Inspection Service has declared seven STEC serogroups as adulterants in a broader range of raw beef products [
8].
Table 1 summarises the current EU regulatory landscape for STEC across key food categories, illustrating the limited scope of mandatory criteria.
The scientific paradigm shift: all STEC are pathogenic
In January 2020, the EFSA Panel on Biological Hazards published a comprehensive reassessment of STEC pathogenicity that fundamentally challenged the rationale underlying serogroup-based food safety criteria [
4]. The Panel reviewed confirmed human STEC infections reported across the EU/EEA between 2012 and 2017 and reached three conclusions of direct regulatory significance.
First, all STEC strains are pathogenic to humans, capable of causing at least diarrhoea. Second, isolates positive for any of the reported Shiga toxin (
stx) gene subtypes may be associated with severe illness, defined as HUS, bloody diarrhoea, and hospitalisation. While strains carrying
stx2a exhibited the highest rates of severe outcomes, all other
stx subtypes for which sufficient data existed were also associated with at least one severe clinical outcome. Third, and most consequentially for the regulatory framework, serogroup cannot be used as a predictor of clinical outcome, and the presence of the intimin gene (
eae) is not essential for severe illness [
4].
The epidemiological data underpinning these conclusions are striking: between 2012 and 2017, 49 different O-serogroups were reported among HUS cases, 88 among hospitalised cases, and 95 among cases of bloody diarrhoea in the EU [
4]. The six serogroups tested under Regulation 2073/2005 represent a fraction of the STEC diversity associated with severe human disease.
These findings have been corroborated internationally. A recent analysis of STEC illness in New Zealand (2016–2022) confirmed that serogroup is a poor discriminator within STEC and should not be used as an epidemiological typing marker in isolation [
9]. In France, the Agence nationale de sécurité sanitaire (ANSES) has adopted a new classification in which serogroup is no longer used to categorise strain pathogenicity; instead, a four-group system based on
stx subtype profiles and
eae gene presence defines the risk level [
10]. While this framework represents a concrete step towards virulence-based classification, it remains subject to technical debate — particularly regarding which
stx subtypes should trigger immediate market withdrawal and which warrant less stringent action. Nevertheless, it illustrates that the transition away from serogroup-based assessment is both feasible and already underway in at least one Member State.
The implication is clear: the serogroup-based criterion in Regulation 2073/2005, even if expanded to additional serogroups, would remain structurally unable to capture the full spectrum of STEC risk as defined by current scientific evidence. What determines pathogenic potential is the virulence gene profile — not the surface antigen.
The new genomic mandate — but only after cases emerge
Against this scientific backdrop, the EU has taken a significant step towards genomic surveillance of foodborne pathogens — though not in the direction of prevention. Implementing Regulation (EU) 2025/179, adopted on 31 January 2025 and applicable from 23 August 2026, mandates that Member States perform WGS on bacterial isolates associated with foodborne outbreak investigations and transmit the results to EFSA’s One Health WGS system [
5].
A critical and largely unremarked feature of this regulation is its scope regarding
E. coli: it mandates WGS for
Escherichia coli without any restriction by serogroup, serotype, or pathotype. The regulation applies to any
E. coli isolate associated or suspected to be associated with a foodborne outbreak, alongside
Salmonella enterica,
Listeria monocytogenes,
Campylobacter jejuni, and
C. coli [
5]. This represents an implicit but decisive departure from the serogroup-centred logic of Regulation 2073/2005.
The European Commission’s guidance on implementation reinforces this interpretation. The FAQ document issued in March 2025 explicitly states that the accumulated WGS data will enable authorities to differentiate “between highly pathogenic […] VTEC strains and less pathogenic ones” and to identify “persistence in the food production environment of certain foodborne bacterial isolates with specific virulence or antimicrobial resistance factors” [
11]. This is, in essence, virulence-based profiling — the same analytical paradigm that EFSA 2020 recommended and that Regulation 2073/2005 has yet to adopt for prevention.
However, the regulation is purely reactive in nature. WGS is triggered only after human cases have been identified and an outbreak investigation is underway. Food business operators are not required to perform WGS, although they must cooperate with competent authorities and provide isolates upon request [
11]. Voluntary submission of WGS data from routine monitoring is encouraged but not mandated.
The regulatory paradox
The EU now finds itself in an internally contradictory position regarding STEC control (
Figure 1). Three concurrent regulatory and scientific instruments operate under fundamentally different paradigms:
The preventive framework (Regulation 2073/2005) operates on serogroup-based testing, limited to six designations and a single food category.
The scientific evidence base (EFSA 2020) concludes that all STEC are pathogenic, that serogroup cannot predict clinical severity, and that virulence gene profiling is the appropriate basis for risk assessment.
The outbreak investigation mandate (Regulation 2025/179) requires WGS for any E. coli without serogroup restriction, with the stated aim of differentiating pathogenicity through genomic analysis.
The practical consequence of this misalignment is that a food product contaminated with a STEC strain belonging to a non-regulated serogroup — such as O91, O80, or O174 — but carrying a high-risk virulence profile (e.g. stx2a, eae-positive) would satisfy all EU food safety criteria. Should that product cause severe illness, the ensuing investigation would deploy WGS and identify the precise virulence determinants that the preventive criteria failed to detect. The information used to explain the outbreak would be the same information that, applied prospectively, could have prevented it.
Towards virulence-based prevention criteria
The implementation of Regulation 2025/179 from August 2026 will generate an unprecedented volume of STEC WGS data across EU Member States, linked through EFSA’s One Health system to human clinical isolates at ECDC. This infrastructure, built for reactive investigation, also provides the evidence base for a prospective revision of prevention criteria.
We propose that the European Commission consider four steps to close this gap. First, STEC-specific food safety criteria should be expanded beyond sprouts to other high-risk food matrices consistently identified by EFSA, including bovine meat and products thereof, raw milk dairy products, and fresh produce [
4]. Second, the transition from serogroup-based to virulence gene-based criteria should be initiated, using
stx subtype profiles and adhesion gene markers as primary risk determinants, consistent with the EFSA 2020 assessment and the ANSES four-group classification [
10]. Third, a tiered implementation approach should be adopted that accounts for the economic and technical capacity of food business operators: targeted PCR-based screening of key virulence genes (
stx subtypes,
eae) as a first-line tool accessible to small and medium-sized enterprises, with full WGS reserved for national reference laboratories, large operators, and confirmatory testing — leveraging the infrastructure and accredited laboratory capacity being built for compliance with Regulation 2025/179. Fourth, the ongoing revision of ISO/TS 13136 should be guided by the virulence-based paradigm rather than a simple expansion of the tested serogroup panel [
6].
Figure 2.
The EU STEC regulatory paradox: misalignment between prevention criteria, scientific evidence, and outbreak investigation tools. The three layers represent the current regulatory and scientific instruments governing STEC in the EU. The upper two layers (investigation and science) are aligned in their genomic and virulence-based paradigm. The lower layer (prevention) remains anchored in serogroup-based criteria applied to a single food category, creating a structural gap in public health protection.
Figure 2.
The EU STEC regulatory paradox: misalignment between prevention criteria, scientific evidence, and outbreak investigation tools. The three layers represent the current regulatory and scientific instruments governing STEC in the EU. The upper two layers (investigation and science) are aligned in their genomic and virulence-based paradigm. The lower layer (prevention) remains anchored in serogroup-based criteria applied to a single food category, creating a structural gap in public health protection.
These steps would not require entirely new regulatory architecture; they would extend the logic that the EU has already endorsed for investigation to the domain of prevention.