Submitted:
07 April 2026
Posted:
08 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methodology
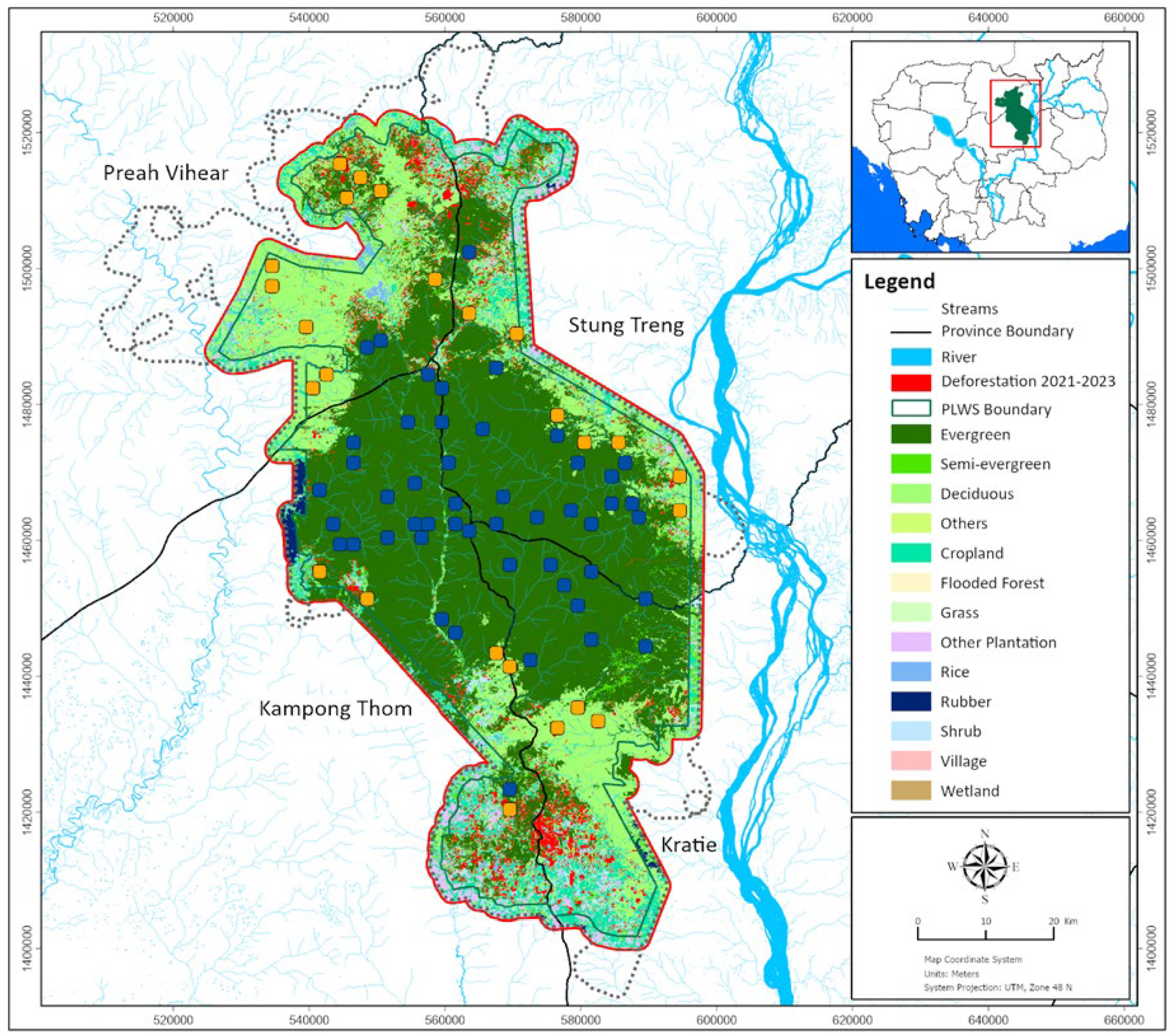
2.1. Study Site
2.2. Target Species
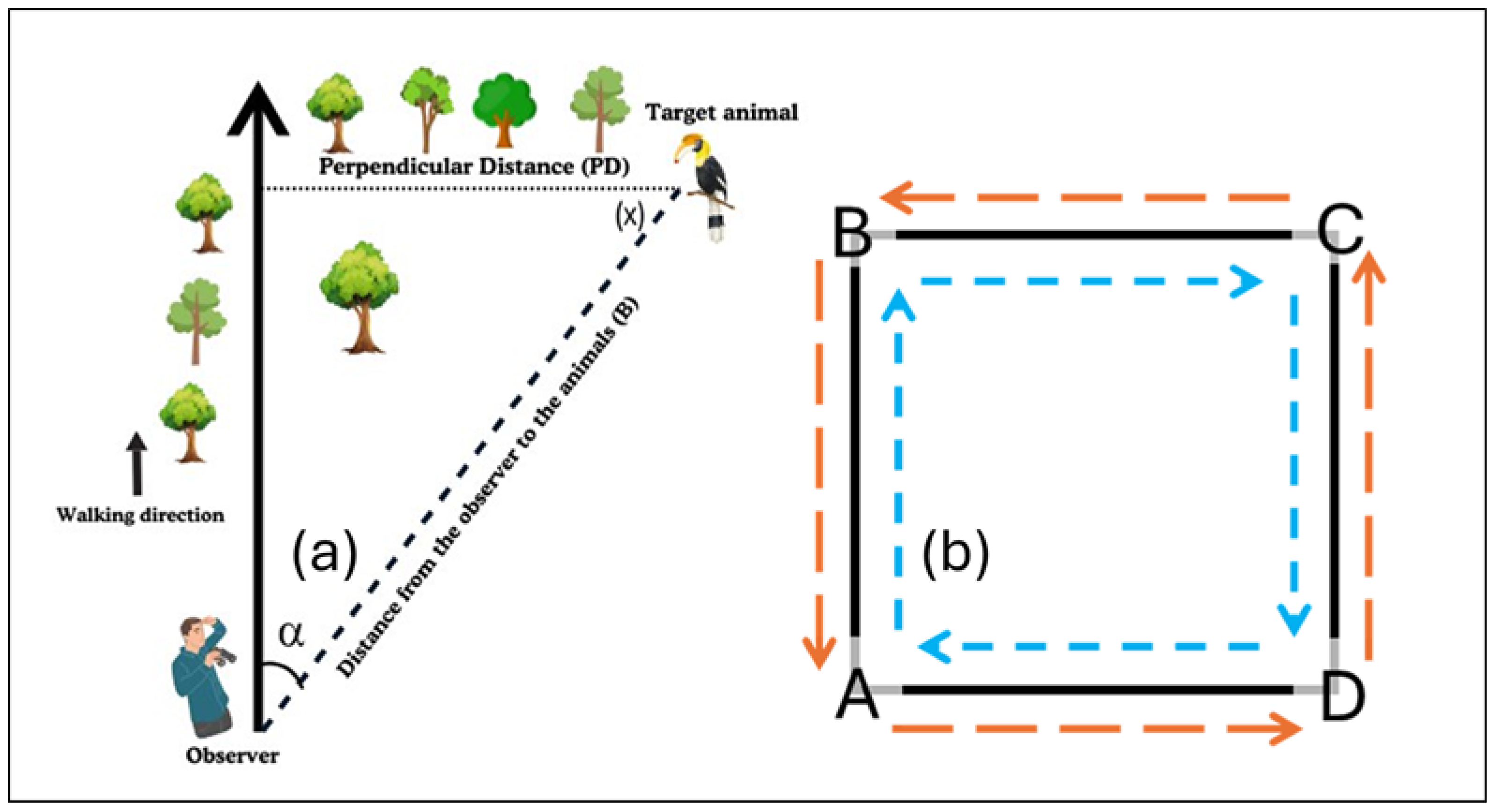
2.3. Transect Designs and Development
2.4. Data Collection, Processing, and Preparation
2.5. Forest Cover Proportion Calculation and Anthropogenic Pressure
2.6. Data Analysis
2.6.1. Species Distribution, Detection, and Diversity
2.6.2. Influences of Habitat Characteristics and Anthropogenic Pressure
3. Results
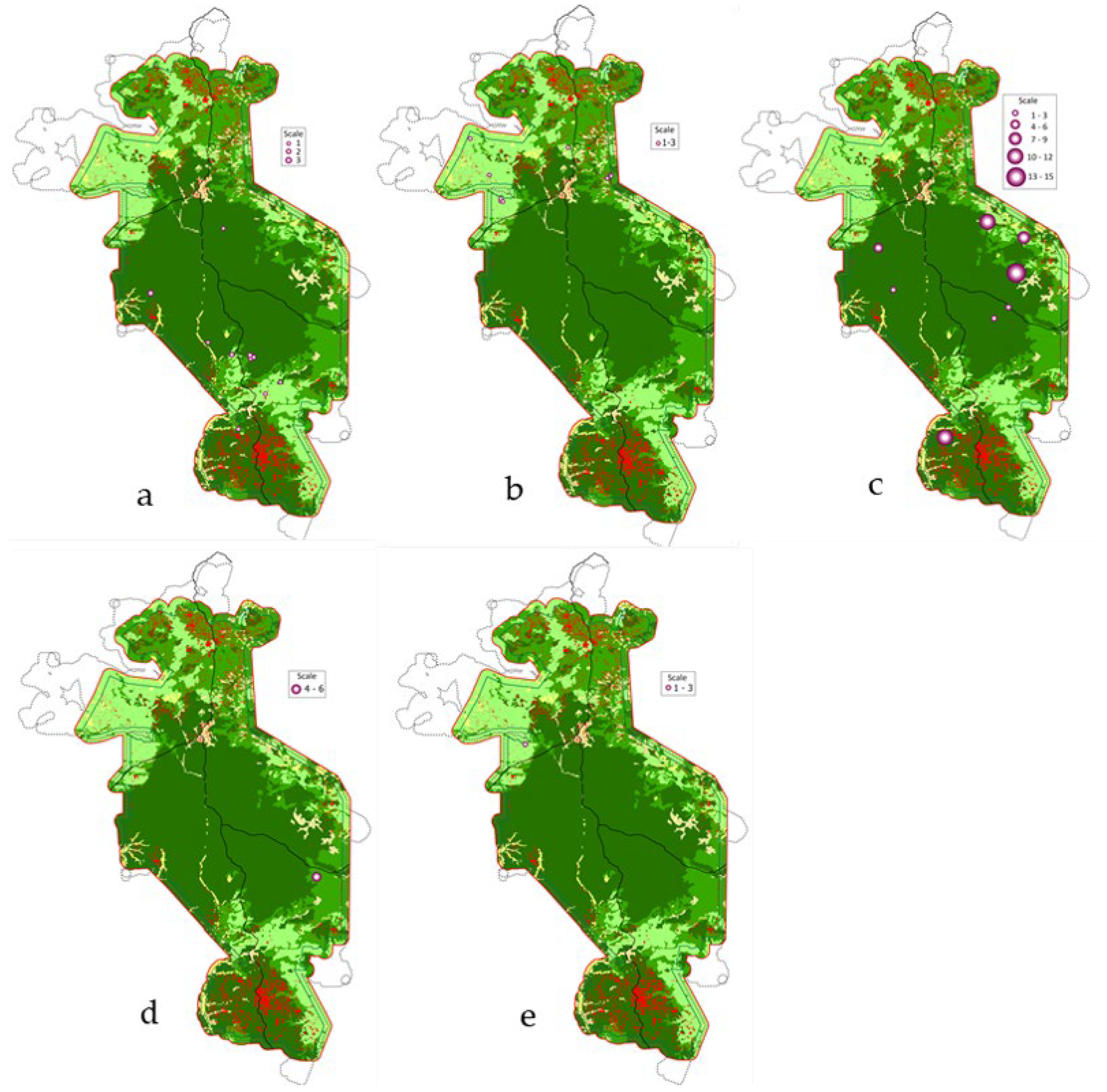
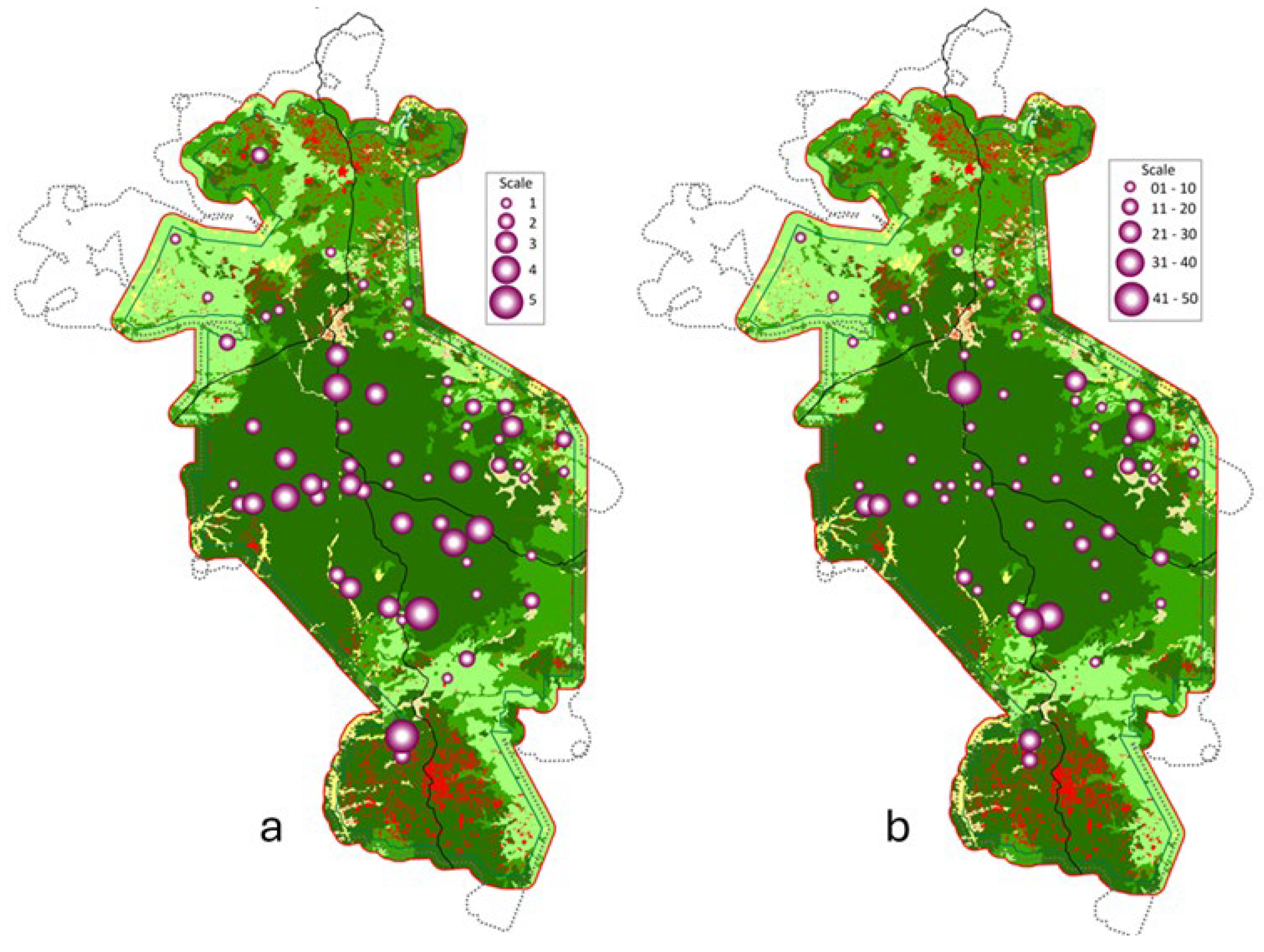
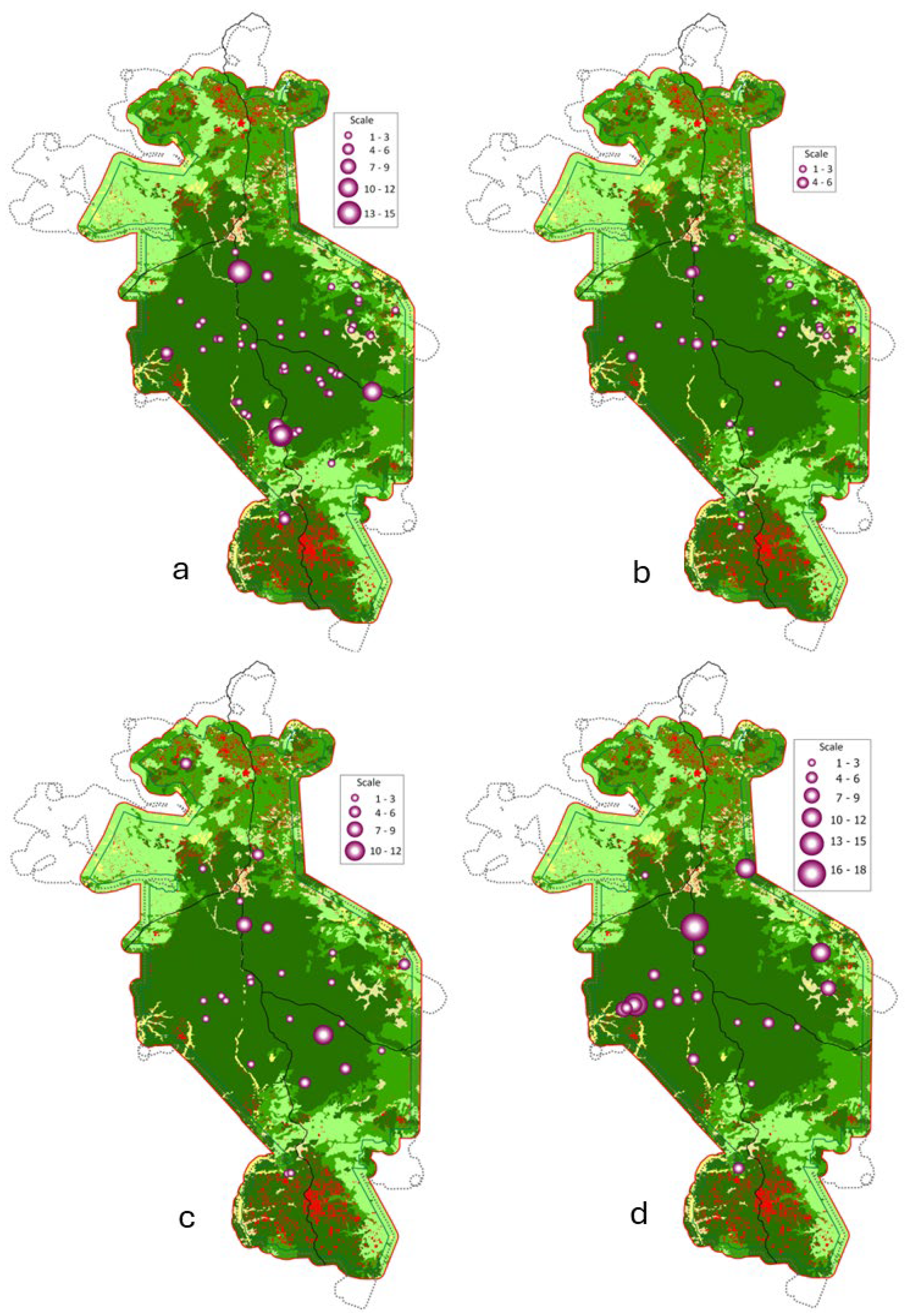
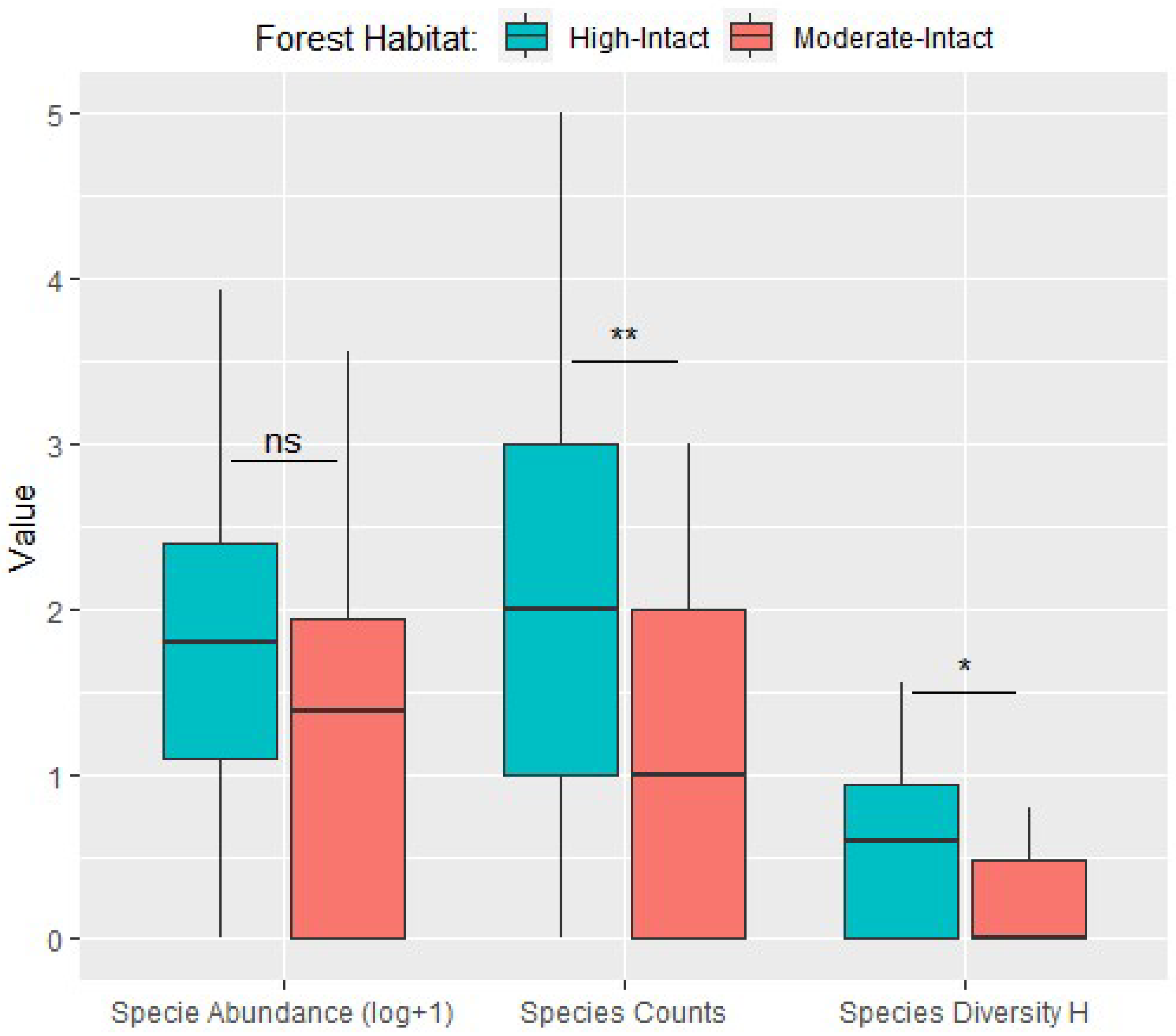
3.1. Distribution Patterns and Diversity
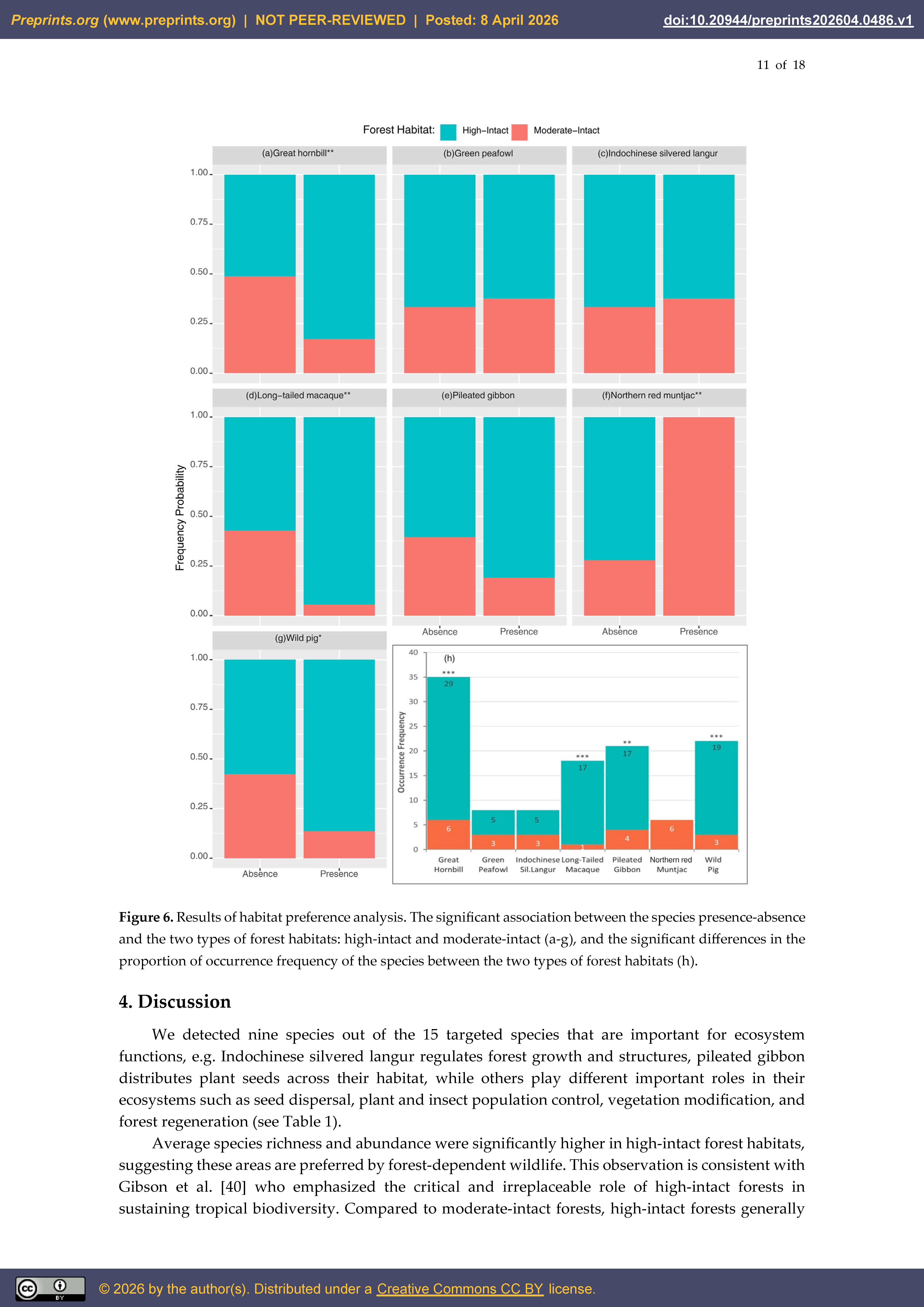
3.2. Habitat Preference and Anthropogenic Pressure
4. Discussion
5. Implication and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
Abbreviations
Appendix A
References
- Dirzo R, Young HS, Galetti M, Ceballos G, Isaac NJB, Collen B. Defaunation in the Anthropocene. Science (1979). 2014;345(6195):401–6.
- Yarrow G, others. Habitat requirements of wildlife: food, water, cover and space [Internet]. 2009. Available from: https://www.academia.edu/121106097/Habitat_Requirements_of_Wildlife_Food_Water_Cover_and_Space.
- Leimgruber P, Gagnon JB, Wemmer C, Kelly DS, Songer MA, Selig ER. Fragmentation of Asia’s remaining wildlands: implications for Asian elephant conservation. Anim Conserv. 2003;6(4):347–59.
- Duckworth JW, Steinmetz R, Timmins RJ, Pattanavibool A, Than Zaw DT, Hedges S. Bos gaurus. The IUCN Red List of Threatened Species 2008: e. T2891A9491805. 2008.
- Datta A, Rawat GS. Nest-site selection and nesting success of three hornbill species in Arunachal Pradesh, north-east India: Great Hornbill Buceros bicornis, Wreathed Hornbill Aceros undulatus and Oriental Pied Hornbill Anthracoceros albirostris. Bird Conserv Int. 2004;14(S1):S39–S52.
- Pin C, Ngoprasert D, Gray TNE, Savini T, Crouthers R, Gale GA. Utilization of waterholes by globally threatened species in deciduous dipterocarp forest of the Eastern Plains Landscape of Cambodia. Oryx. 2020;54(4):572–82.
- Theilade I, Phourin C, Schmidt L, Meilby H, van de Bult M, Friborg KG. Evergreen forest types of the central plains in Cambodia: floristic composition and ecological characteristics. Nord J Bot. 2022;2022(8):e03494.
- Auda E. Prey Lang Extended Landscape Camera Trap Survey. 2023.
- Olsson A, Emmett DE. A Floral and Faunal Biodiversity Assessment of Prey Long. 2007. Report.
- Gray TNE, Ou R, Huy K, Pin C, Maxwell AL. The status of large mammals in eastern Cambodia: a review of. Cambodian Journal of Natural History. 2012;42.
- Gray TNE, Chamnan H, Borey R, Collar NJ, Dolman PM. Habitat preferences of a globally threatened bustard provide support for community-based conservation in Cambodia. Biol Conserv. 2007;138(3–4):341–50.
- Gray TNE, Phan C. Habitat preferences and activity patterns of the larger mammal community in Phnom Prich Wildlife Sanctuary, Cambodia. Raffles Bull Zool. 2011;59(2):311–8.
- Theilade I, Schmidt L. REDD+ and Conservation of Prey Long Forest, Cambodia Summary of Scientific Findings 2007-2010 [Internet]. Forest & Landscape Working Papers no 66; 2011 [cited 2025 Feb 16]. Report. Available from: www.SL.life.ku.dk.
- SERVIR-Mekong. Biophysical M&E Dashboard Map Viewer [Internet]. 2025 [cited 2025 May 7]. Available from: https://cambodia-me-servir.adpc.net/mapviewer.
- Traeholt C, Bunthoen R, Rawson B, Samuth M, Vrak C, Vuthin S. Status Review of Pileated Gibbon, Hylobates Pileatus and Yellow-cheeked Crested Gibbon, Nomascus Gabriellae: In Cambodia. Fauna & Flora International Cambodia; 2005.
- Srivathsa A, Karanth KK, Jathanna D, Kumar NS, Karanth KU. On a dhole trail: Examining ecological and anthropogenic correlates of dhole habitat occupancy in the Western Ghats of India. PLoS One. 2014 Jun 3;9(6). https://doi.org/10.1371/journal.pone.0098803 PubMed PMID: 24893166.
- Ruikar S, Suryavanshi N, Nanaware J, Balasundaram H, Patil D, Pathade G. The Role of Wildlife in Ecosystem Health: Interactions and Impacts. REDVET-Revista electrónica de Veterinaria. 2024;25(1):275–95.
- Ong L, Tan WH, Davenport LC, McConkey KR, Mat Amin MKA bin, Campos-Arceiz A, et al. Asian elephants as ecological filters in Sundaic forests. Frontiers in Forests and Global Change. 2023;6. https://doi.org/10.3389/ffgc.2023.1143633.
- Hayes B, Khou EH, Neang T, Furey N, Chhin S, Holden J, et al. Biodiversity Assessment of Prey Lang–Kratie, Kampong Thom, Stung Treng and Preah Vihear Provinces. Conservation International, Winrock International, USAID, Phnom Penh, Cambodia. 2015.
- Chaichanathong S, Klinsawat W, Sukmak M, Sakulthai A, Wajjwalku W, Sripiboon S, et al. Genetic characterization of banteng (Bos javanicus) populations in Thailand for conservation. The Thai Journal of Veterinary Medicine. 2021 Dec 1;51(4):647–54. https://doi.org/10.56808/2985-1130.3162.
- Prayoon U, Suksavate W, Chaiyes A, Winitpornsawan S, Tunhikorn S, Angkaew C, et al. Importance of protected areas in conservation and recovery of gaur (Bos gaurus) in Thailand. 2022. Report.
- Gazagne E, Pitance JL, Savini T, Huynen MC, Poncin P, Brotcorne F, et al. Seed shadows of northern pigtailed macaques within a degraded forest fragment, Thailand. Forests. 2020 Nov 1;11(11):1–24. https://doi.org/10.3390/f11111184.
- Bunthoeun R, Rawson B, Samuth M, Virak C, Vuthin S. Status review of pileated gibbon, Hylobates pileatus and yellow-cheeked crested gibbon, Nomascus gabriellae, in Cambodia. 2005. Report.
- Hoang MD, Van BT, Le TH, Covert HH. Diet of Indochinese Silvered Langur (Trachypithecus germaini) in Kien Luong Karst Area, Vienam [Internet]. 2017. Report. Available from: https://www.researchgate.net/publication/282652802.
- Bernathirdin ANH, Wijayanto H, Kusindarta DL, Budipitojo T. Population Estimation and Identification of Long-Tailed Macaque (Macaca fascicularis) Group at Tlogo Muncar Tourism Area, Mount Merapi National Park, after Two Years Closure Caused by the Covid-19 Pandemic. In: IOP Conference Series: Earth and Environmental Science. Institute of Physics; 2023. https://doi.org/10.1088/1755-1315/1174/1/012028.
- Cuevas MF, Ojeda RA, Jaksic FM. Ecological strategies and impact of wild boar in phytogeographic provinces of Argentina with emphasis on aridlands. Mastozool Neotrop [Internet]. 2016;23(2):239–54. Available from: http://www.sarem.org.ar.
- Liang H, McConkey KR, Lim JY. From beak to fruit: An Asian hornbill database for frugivory and seed dispersal research. Glob Ecol Conserv. 2024 Jun 1;51. https://doi.org/10.1016/j.gecco.2024.e02879.
- HERNOWO JB, MARDIASTUTI ANI, ALIKODRA HS, KUSMANA CECEP. Behavior Ecology of the Javan Green Peafowl (Pavo muticus muticus Linnaeus 1758) in Baluran and Alas Purwo National Park, East Java. Hayati. 2011 Dec 1;18(4):164–76. https://doi.org/10.4308/hjb.18.4.164.
- Scotson L, Fredriksson G, Augeri D, Cheah C, Ngoprasert D, Wai- Ming W. IUCN Sun bear Helarctos malayaus Red list Assessment 2016 [Internet]. 2017 [cited 2025 Feb 6]. Available from: https://www.researchgate.net/publication/321682540.
- Steinmetz R, Garshelis DL, Chutipong W, Seuaturien N. Foraging ecology and coexistence of Asiatic black bears and sun bears in a seasonal tropical forest in Southeast Asia. J Mammal. 2013;94(1):1–18.
- Griffin O, Nuttall M. Status of Key Species in Keo Seima Wildlife Sanctuary 2010-2020 [Internet]. 2020 [cited 2025 Feb 8]. Report. Available from: https://doi.org/10.19121/2020.Report.38511.
- Nuñez-Penichet C, Cobos ME, Soberón J, Gueta T, Barve N, Barve V, et al. Selection of sampling sites for biodiversity inventory: Effects of environmental and geographical considerations. Methods Ecol Evol. 2022 Jul 1;13(7):1595–607. https://doi.org/10.1111/2041-210X.13869.
- Buckland ST, Rexstad EA, Marques TA, Oedekoven CS. Methods in Statistical Ecology Distance Sampling: Methods and Applications [Internet]. 2015. Report. Available from: http://www.springer.com/series/10235.
- Pavlacky DC, Green AW, George TL, Iovanna R, Bartuszevige AM, Correll MD, et al. Landscape-scale conservation mitigates the biodiversity loss of grassland birds. Ecological Applications. 2021. https://doi.org/10.5061/dryad.9zw3r22f3.
- Mittermeier RA, Mittermeier CG, Brooks TM, Pilgrim JD, Konstant WR, Da Fonseca GAB, et al. Wilderness and biodiversity conservation [Internet]. 2003. Report. Available from: www.pnas.org.
- Pramod AF, Kumara V, HT RG. Encounter rate of large mammals in Thanigebyle range of Bhadra wildlife sanctuary, Karnataka. Int Multidiscip Res J. 2012;2(9).
- Harrell FE. Regression Modeling Strategies: With Applications to Linear Models, Logistic and Ordinal Regression, and Survival Analysis [Internet]. 2nd ed. Springer; 2015. Available from: https://link.springer.com/book/10.1007/978-3-319-19425-7.
- Sor R, Park YS, Boets P, Goethals PLM, Lek S. Effects of species prevalence on the performance of predictive models. Ecol Modell. 2017;354:11–9.
- Zuur AF, Ieno EN, Walker NJ, Saveliev AA, Smith GM, others. Mixed effects models and extensions in ecology with R. Vol. 574. Springer; 2009.
- Gibson L, Lee TM, Koh LP, Brook BW, Gardner TA, Barlow J, et al. Primary forests are irreplaceable for sustaining tropical biodiversity. Nature. 2011;478(7369):378–81.
- Ghadiri Khanaposhtani M, Kaboli M, Karami M, Etemad V. Effect of habitat complexity on richness, abundance and distributional pattern of forest birds. Environ Manage. 2012;50(2):296–303.
- Spitzer HB, Meagher RK, Proudfoot KL. The impact of providing hiding spaces to farmed animals: A scoping review. PLoS One. 2022;17(11):e0277665.
- Miraldo A, Li S, Borregaard MK, Flórez-Rodríguez A, Gopalakrishnan S, Rizvanovic M, et al. An Anthropocene map of genetic diversity. Science (1979). 2016;353(6307):1532–5.
- Datta A. Hornbill abundance in unlogged forest, selectively logged forest and a forest plantation in Arunachal Pradesh, India. Oryx. 1998;32(4):285–94.
- Phoonjampa R, Koenig A, Brockelman WY, Borries C, Gale GA, Carroll JP, et al. Pileated gibbon density in relation to habitat characteristics and post-logging forest recovery. Biotropica. 2011;43(5):619–27.
- Fahrig L. Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Evol Syst. 2003;34(1):487–515.
- Gibbons MA, Harcourt AH. Biological correlates of extinction and persistence of primates in small forest fragments: a global analysis. Trop Conserv Sci. 2009;2(4):388–403.
- Sharma CM, Bhattacharjee S, Sharief A, Joshi BD, Thakur M, Sharma LK. A tapestry of habitats: exploring abundance and habitat preferences of the Northern Red Muntjac (Muntiacus vaginalis) across the Central Himalayan landscape. Mammalia. 2024;88(4):280–91.
- Brotcorne F, Maslarov C, Wandia IN, Fuentes A, Beudels-Jamar RC, Huynen MC. The role of anthropic, ecological, and social factors in sleeping site choice by long-tailed macaques (Macaca fascicularis). Am J Primatol. 2014;76(12):1140–50.
- Linkie M, Dinata Y, Nofrianto A, Leader-Williams N. Patterns and perceptions of wildlife crop raiding in and around Kerinci Seblat National Park, Sumatra. Anim Conserv. 2007;10(1):127–35.
- Delacour J. The pheasants of the world. (No Title). 1977.
- Saridnirun G, Meckvichai W, Dumrongrojwatthana P. Seasonal Distribution and Habitat Use of the Green Peafowl Pavo muticus, Linnaeus 1766, in Wiang Lor Wildlife Sanctuary, Phayao Province, Northern Thailand. Chiang Mai J Sci. 2017;44:824–38.
- Blake S, Hedges S. Sinking the flagship: the case of forest elephants in Asia and Africa. Conservation Biology. 2004;18(5):1191–202.
- Kamler JF. Extreme dogs: dholes [Internet]. 2013 [cited 2025 Apr 10]. Available from: https://www.researchgate.net/publication/277588881.
- Steinmetz R, Garshelis DL. Distinguishing Asiatic black bears and sun bears by claw marks on climbed trees. J Wildl Manage. 2008;72(3):814–21.
- Buckland ST, Rexstad EA, Marques TA, Oedekoven CS, others. Distance sampling: methods and applications. Vol. 431. Springer; 2015.
- Theilade I, Schmidt L. REDD+ and Conservation of Prey Long Forest, Cambodia. Forest & Landscape Denmark; 2011.
- Voet J. Conservation of Orangutans: reintroduction or habitat preservation? 2020.
- Svensson J, Bubnicki JW, Jonsson BG, Andersson J, Mikusiński G. Conservation significance of intact forest landscapes in the Scandinavian Mountains Green Belt. Landsc Ecol. 2020;35(9):2113–31.
| Common name | Scientific name | IUCN Status | Ecological roles | Previous records |
| Asian elephant | Elephas maximus | EN | Seed dispersal, habitat creation, and vegetation modification [18] | [8,9,19] |
| Banteng | Bos javanicus | EN | Seed dispersal [20] | [8,9,19] |
| Gaur | Bos gaurus | VU | Plant population control and main prey for large carnivores [21] | [9,19] |
| Dhole | Cuon alpinus | EN | Prey regulation [16] | [9,19] |
| Sambar deer | Rusa unicolor | VU | Seed dispersal and keystone herbivore (Quin, Morgan, & Murphy, 2023) | [8,9,19] |
| Northern pig-tailed macaque | Macaca leonina | VU | Seed dispersal and forest regeneration [22] | [9,19] |
| Pileated gibbon | Hylobates pileatus | EN | Seed dispersal [23] | [9,19] |
| Indochinese silvered langur | Trachypithecus germaini | EN | Regulate vegetation growth and forest structure [24] | [9,19] |
| Long-tailed macaque | Macaca fascicularis | VU | Seed dispersal and insect control through predation [25] | [8,9,19] |
| Northern red muntjac | Muntiacus vaginalis | LC | Herbivory, seed dispersal, and serving as prey species [50] | [8,9,19] |
| Wild pig | Sus scrofa | LC | Soil disturbance, seed dispersal, and maintaining ecological balance [26] | [8,9,19] |
| Great hornbill | Buceros bicornis | VU | Seed dispersal, forest regeneration, and ecological balance [27] | [9,19] |
| Green peafowl | Pavo muticus | EN | Feeding on plants and dispersing seeds [28] | [8,9,19] |
| Malayan sun bear | Helarctos malayanus | VU | Seed dispersal and maintenance of forest understory structure [29] | [8,9,19] |
| Asiatic black bear | Ursus thibetanus | VU | Seed dispersal and insect control [30] | [8,9,19] |
| No |
Common name |
Scientific name |
# Detections/ sightings |
Cluster Size (average) |
Encounter rate |
| 1 | Great hornbill | B. bicornis | 76 | 2.34 | 0.826 |
| 2 | Pileated gibbon | H. pileatus | 31 | 2.23 | 0.337 |
| 3 | Wild pig | S. scrofa | 25 | 2.88 | 0.272 |
| 4 | Long tailed macaque | M. fascicularis | 23 | 5.96 | 0.250 |
| 5 | Green peafowl | P. muticus | 13 | 1.62 | 0.141 |
| 6 | Northern red muntjac | M.vaginalis | 10 | 1 | 0.109 |
| 7 | Indochinese silvered langur | T. germaini | 8 | 8.13 | 0.087 |
| 8 | Gaur | B. gaurus | 1 | 1 | 0.011 |
| 9 | Sambar deer | R. unicolor | 1 | 1 | 0.011 |
| Wildlife Species | Important variables selected for the final model | AIC | ||||||
| EVE | DDF | OTP | CRL | RIF | DNV | Full model | Final model | |
| Great hornbill | 1.54 | - | - | - | - | - | 92.98 | 85.89 |
| Pileated gibbon | 0.69 | - | - | - | - | - | 96.74 | 87.64 |
| Indochinese silvered langur | - | - | - | - | -506.7 | - | 1103.3 | 49.62 |
| Northern red muntjac | -2.67 | - | 5.23 | - | - | -3.6 | 22 | 20.94 |
| Green peafowl | - | -798.56 | - | 30.22 | - | - | 48.41 | 34.66 |
| Long-tailed macaque | - | -1.01 | 0.46 | - | - | - | 91.42 | 80.32 |
| Wild pig | - | - | - | - | - | - | 107.7 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




