Submitted:
20 March 2026
Posted:
27 April 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Indications for Invasive Mediastinal Staging
- Tumors larger than 3 cm
- Centrally located tumors
- PET-positive mediastinal lymph nodes
- Enlarged lymph nodes on CT imaging
- Suspected N1 disease
Evolution of Mediastinal Staging
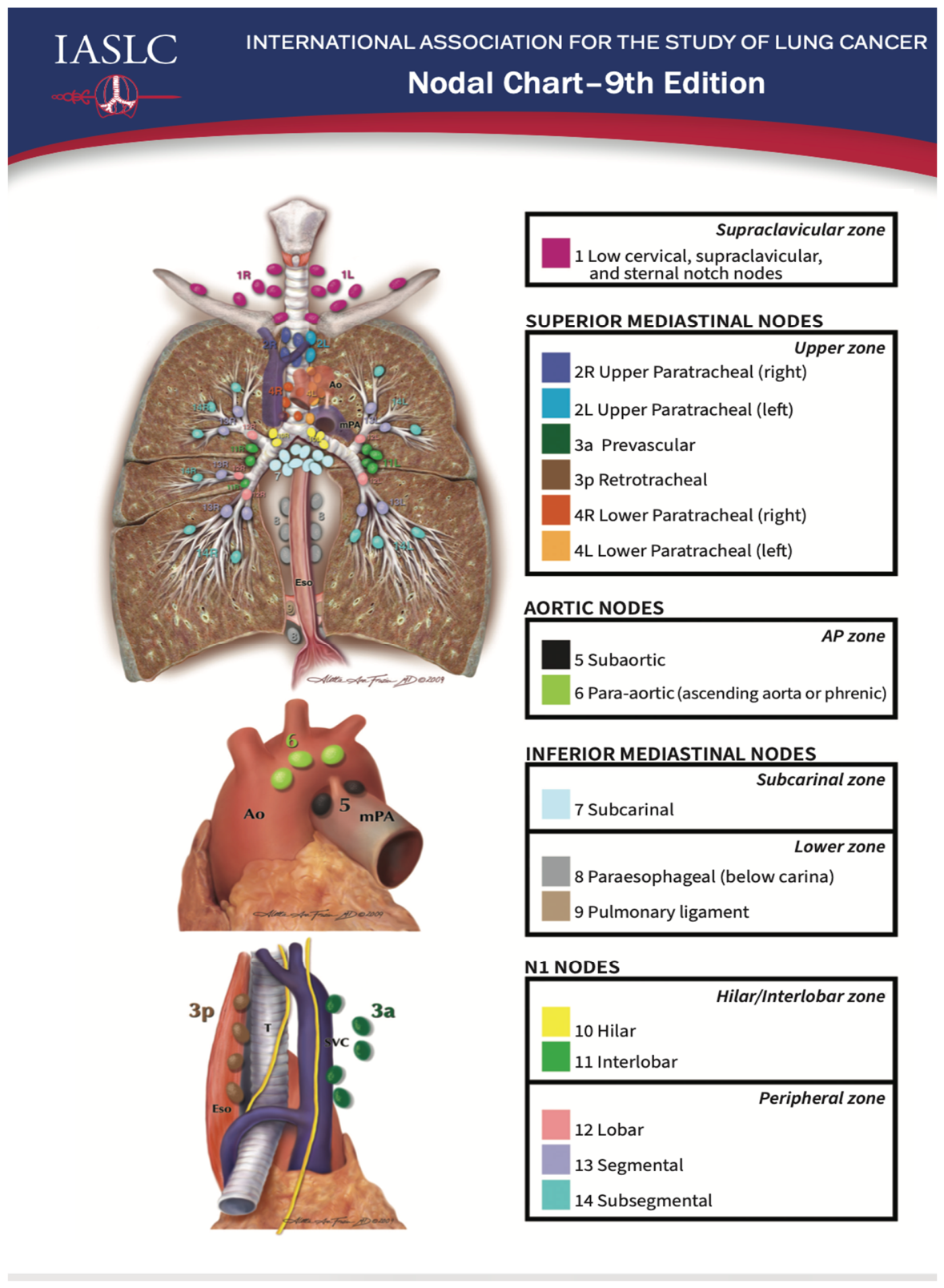
Mediastinal Nodal Mapping and Endosonographic Access
Stations Accessible by EBUS
Stations Accessible by EUS
Advantages of Combined Endosonographic Staging
Special Clinical Scenarios
Radiologically Normal Mediastinum
Restaging After Neoadjuvant Therapy
Stereotactic Body Radiation Therapy (SBRT) Planning
The Role of Endosonography in the Context of Molecular Markers
Technical Aspects of the Procedure
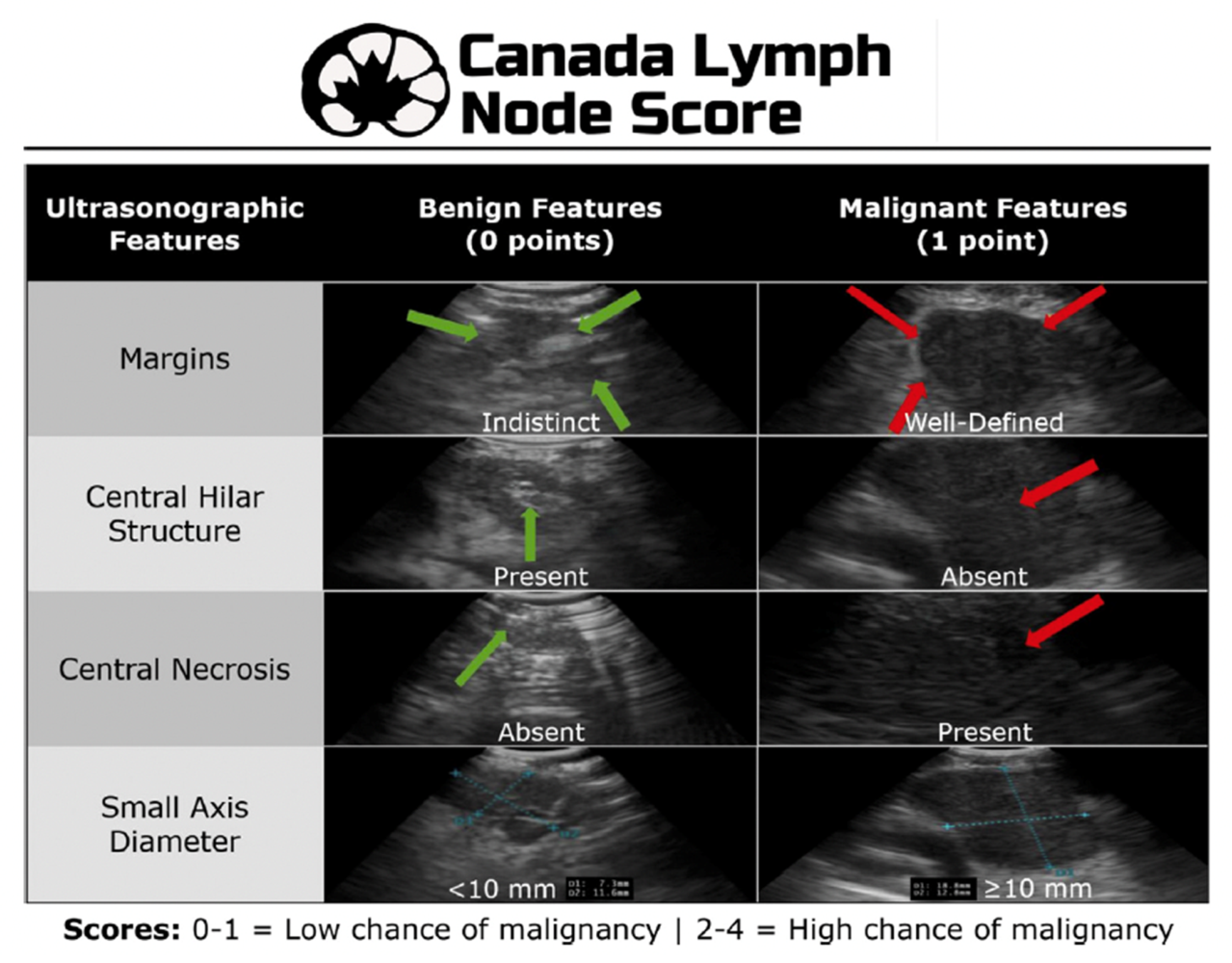
Procedure Competency
- Short-axis diameter ≥10 mm
- Well-defined margins
- Absence of central hilar structure
- Presence of central necrosis
Endosonography in Lymphoma, Sarcoidosis and Infections
Newer Thin Convex EBUS
Conclusion
Author Contributions
Data Availability Statement
References
- Peter Vilmann 1, Paul Frost Clementsen 2, Sara Colella 3, Mette Siemsen 4, Paul De Leyn 5, Jean-Marc Dumonceau 6, Felix J Herth 7, Alberto Larghi 8, Enrique Vazquez-Sequeiros 9, Cesare Hassan 8, Laurence Crombag 10et al. Combined endobronchial and esophageal endosonography for the diagnosis and staging of lung cancer ... Endoscopy 2015; 47: 545–559.
- Gerard A Silvestri 1, Anne V Gonzalez 2, Michael A Jantz 3, Mitchell L Margolis 4, Michael K Gould 5, Lynn T Tanoue 6, Loren J Harris 7, Frank C Detterbeck 8vMethods for staging non-small cell lung cancer: Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 2013;143(5): e211s-50s.
- W De Wever 1, S Ceyssens, L Mortelmans, S Stroobants, G Marchal, J Bogaert, J A Verschakelen Additional value of PET-CT in the staging of lung cancer: comparison with CT alone, PET alone and visual cor- relation of PET and CT. Eur Radiol 2007;17(1):23–32.
- Silvestri GA, Gould MK, Margolis ML. American College of Chest Physicians. Non-invasive staging of nonsmall cell lung cancer: ACCP evidenced based clinical practice guidelines (2nd edition). Chest 2007;132(3):178S–201S.
- Dales RE, Stark RM, Raman S. Computed tomography to stage lung cancer. Approaching a controversy using meta-analysis. Am Rev Respir Dis 1990;141(5.1):1096–101.
- Lardinois D, Weder W, Hany TF. Staging of non- small-cell lung cancer with integrated positron- emission tomography and computed tomography. N Engl J Med 2003;348(25):2500–7.
- Robert James Cerfolio 1, Ayesha S Bryant, Buddhiwardhan Ojha, Mohammad Eloubeidi. Improving the inaccuracies of clinical staging of patients with NSCLC: a prospective trial. Ann Thorac Surg 2005; 80(4):1207–13.
- McLoud TC, Bourgouin PM, Greenberg RW. Bronchogenic carcinoma: analysis of staging in the mediastinum with CT by correlative lymph node mapping and sampling. Radiology 1992;182(2): 319–23.
- K G Tournoy 1, S Maddens, R Gosselin, G Van Maele, J P van Meerbeeck, A Kelles. Integrated FDG-PET/CT does not make invasive staging of the intrathoracic lymph nodes in non- small cell lung cancer redundant: a prospective study. Thorax 2007;62(8):696–701.
- Toloza EM, Harpole L, McCrory DC. Noninvasive staging of non-small cell lung cancer: a review of the current evidence. Chest 2003;123(1):137S–46S.
- Silvestri GA, Hoffman B, Reed CE. Choosing between CT, positron emission tomography, endoscopic ultrasound with fine-needle aspiration, transbronchial needle aspiration, thoracoscopy, mediastinoscopy, and mediastinotomy for staging lung cancer. Chest 2003;123(2):333–5.
- Ping Gu 1, Yi-Zhuo Zhao, Li-Yan Jiang, Wei Zhang, Yu Xin, Bao-Hui Han Endobronchial ultrasound-guided transbronchial needle aspiration for staging of lung cancer: a systematic review and meta-analysis. Eur J Cancer 2009;45(8): 1389-96.
- Kazuhiro Yasufuku 1, Andrew Pierre, Gail Darling, Marc de Perrot, Thomas Waddell, Michael Johnston, Gilda da Cunha Santos, William Geddie, Scott Boerner, Lisa W Le, et al. A prospective controlled trial of endobronchial ultrasound-guided transbronchial needle aspiration compared with me- diastinoscopy for mediastinal lymph node staging of lung cancer. J Thorac Cardiovasc Surg 2011;142(6): 1393–400.
- Kazuhiro Yasufuku 1, Masako Chiyo, Yasuo Sekine, Prashant N Chhajed, Kiyoshi Shibuya, Toshihiko Iizasa, Takehiko Fujisawa. Real-time endobronchial ultrasound-guided transbronchial needle aspiration of mediastinal and hilar lymph no- des. Chest 2004;126(1):122–8.
- M Krasnik 1, P Vilmann, S S Larsen, G K Jacobsen Preliminary experience with a new method of endoscopic trans- bronchial real time ultrasound guided biopsy for diagnosis of mediastinal and hilar lesions. Thorax 2003;58(12):1083–6.
- R C Rintoul 1, K M Skwarski, J T Murchison, W A Wallace, W S Walker, I D Penman Endobronchial and endoscopic ultrasound-guided real-time fine-needle aspiration for mediastinal staging. Eur Respir J 2005;25(3):416–21.
- Kazuhiro Yasufuku 1, Masako Chiyo, Eitetsu Koh, Yasumitsu Moriya, Akira Iyoda, Yasuo Sekine, Kiyoshi Shibuya, Toshihiko Iizasa, Takehiko Fujisawa. Endobronchial ultrasound guided transbronchial needle aspiration for staging of lung cancer. Lung Cancer 2005; 50(3):347–54.
- F J F Herth 1, R Eberhardt, P Vilmann, M Krasnik, A Ernst. Real-time endobronchial ultrasound guided transbronchial needle aspiration for sampling mediastinal lymph nodes. Thorax 2006;61(9):795–8.
- Kazuhiro Yasufuku 1, Takahiro Nakajima, Ken Motoori, Yasuo Sekine, Kiyoshi Shibuya, Kenzo Hiroshima, Takehiko Fujisawa. Compari- son of endobronchial ultrasound, positron emission tomography, and CT for lymph node staging of lung cancer. Chest 2006;130(3):710–8.
- Felix J F Herth 1, Mark Krasnik, Nicolas Kahn, Ralf Eberhardt, Armin Ernst. Combined endoscopic-endobronchial ultrasound-guided fine-needle aspiration of mediastinal lymph nodes through a single bronchoscope in 150 patients with suspected lung cancer. Chest 2010; 138(4):790-4.
- Masahide Oki 1, Hideo Saka 2, Masahiko Ando 3, Rie Tsuboi 2, Masashi Nakahata 2, Saori Oka 2, Yoshihito Kogure 2, Chiyoe Kitagawa 2. Transbronchial versus transesophageal needle aspiration using an ultrasound bronchoscope for the diagnosis of mediastinal lesions: a randomized study. Chest 2014; 147(5): 1259-66.
- Bin Hwangbo 1, Geon-Kook Lee, Hee Seok Lee, Kun-Young Lim, Soo-Hyun Lee, Hyae-Young Kim, Hyun Sung Lee, Moon Soo Kim, Jong Mog Lee, Byung-Ho Nam, Jae Ill Zo. Transbronchial and transesophageal fine-needle aspiration using an ultrasound bronchoscope in mediastinal staging of potentially operable lung cancer. Chest 2010;138(4):795-802.
- Kurt G Tournoy 1, Frederic De Ryck, Lieve R Vanwalleghem, Frank Vermassen, Marleen Praet, Joachim G Aerts, Georges Van Maele, Jan P van Meerbeeck. Endoscopic ultrasound reduces surgical mediastinal staging in lung cancer: a randomized trial. Am J Respir Crit Care Med 2008;177(5): 531-5.
- Larsen SS, Krasnik M, Vilmann P. Endoscopic ultrasound guided biopsy of mediastinal lesions has a major impact on patient management. Thorax 2002;57: 98-103.
- Fritscher-Ravens A, Soehendra N, Schirrow L. Role of transesophageal endosonography-guided fine-needle aspiration in the diagnosis of lung cancer. Chest 2000;117:339-45.
- Wiersema MJ, Vazquez-Sequeiros E, Wiersema LM. Evaluation of mediastinal lymphadenopathy with endoscopic US-guided fine-needle aspiration biopsy. Radiology 2001;219:252-7.
- Wallace MB, Silvestri GA, Sahai AV. Endoscopic ultrasound-guided fine needle aspiration for staging patients with carcinoma of the lung. Ann Thorac Surg 2001;72:1861-7.
- M Giovannini 1, J F Seitz, G Monges, H Perrier, I Rabbia. Fine-needle aspiration cytology guided by endoscopic ultrasonography: Results in 141 patients. Endoscopy 1995;27:171-7.
- Micames CG, McCrory DC, Pavey DA. Endoscopic ultrasound-guided fine-needle aspiration for non-small cell lung cancer staging: A systematic review and metaanalysis. Chest 2007;131:539-48.
- Puli SR, Batapati Krishna Reddy J, Bechtold ML. Endoscopic ultrasound: It’s accuracy in evaluating mediastinal lymphadenopathy? A meta-analysis and systematic review. World J Gastroenterol 2008;14:3028-37.
- Frank C Detterbeck 1, Michael A Jantz, Michael Wallace, Johan Vansteenkiste, Gerard A Silvestri; . American College of Chest Physicians. Invasive mediastinal staging of lung cancer: ACCP evidence-based clinical practice guidelines (2nd edition). Chest 2007; 132(Suppl 3):202S–20S.
- Z T Hammoud 1, R C Anderson, B F Meyers, T J Guthrie, C L Roper, J D Cooper, G A Patterson. The current role of mediastinoscopy in the evaluation of thoracic disease. J Thorac Cardiovasc Surg 1999; 118(5):894–9.
- Anthony Lemaire 1, Ivana Nikolic, Thomas Petersen, Jack C Haney, Eric M Toloza, David H Harpole Jr, Thomas A D’Amico, William R Burfeind. Nine-year single center experience with cervical mediastinoscopy complications and false negative rate. Ann Thorac Surg 2006;82(4):1185–9.
- Sahajal Dhooria 1, Ashutosh N Aggarwal 1, Dheeraj Gupta 1, Digambar Behera 1, Ritesh Agarwal 2. Utility and Safety of Endoscopic Ultrasound With Bronchoscope-Guided Fine-Needle Aspiration in Mediastinal Lymph Node Sampling: Systematic Review and Meta-Analysis. Respir Care. 2015 Jul; 60(7):1040-50.
- Christophe Dooms 1, Kurt G Tournoy 2, Olga Schuurbiers 3, Herbert Decaluwe 4, Frédéric De Ryck 5, Ad Verhagen 6, Roel Beelen 7, Erik van der Heijden 8, Paul De Leyn 4. Endosonography for mediastinal nodal staging of clinical N1 non-small cell lung cancer: a prospective multicenter study. Chest. 2015 Jan; 147(1):209-215.
- Herth FJ, Eberhardt R, Krasnik M, Ernst A. Endobronchial ultrasound-guided transbronchial needle aspiration of lymph nodes in the radiologically and positron emission tomography-normal mediastinum in patients with lung cancer. Chest. 2008 Apr;133(4):887-91.
- Herth FJ, Ernst A, Eberhardt R, Vilmann P, Dienemann H, Krasnik M. Endobronchial ultrasound-guided transbronchial needle aspiration of lymph nodes in the radiologically normal mediastinum. Eur Respir J. 2006 Nov; 28(5):910-4.
- Ong P, Grosu H, Eapen GA, Rodriguez M, Lazarus D, Ost D, Jimenez CA, Morice R, Bandi V, Tamara L, Cornwell L, Green L, Zhu A, Casal RF. Endobronchial ultrasound-guided transbronchial needle aspiration for systematic nodal staging of lung cancer in patients with N0 disease by computed tomography and integrated positron emission tomography-computed tomography. Ann Am Thorac Soc. 2015 Mar; 12(3):415-9.
- Felix J F Herth 1, Jouke T Annema, Ralf Eberhardt, Kazuhiro Yasufuku, Armin Ernst, Mark Krasnik, Robert C Rintoul. Endobronchial ultrasound with transbronchial needle aspiration for restaging the mediastinum in lung cancer. J Clin Oncol 2008; 26(20):3346-50.
- Cetinkaya E, Usluer O, Yılmaz A, Tutar N, Çam E, Özgül MA, Demirci NY. Is endobronchial ultrasound-guided transbronchial needle aspiration an effective diagnostic procedure in restaging of non-small cell lung cancer patients? Endosc Ultrasound. 2017 May-Jun;6(3):162-167.
- Martin B von Bartheld 1, Michel I M Versteegh, Jerry Braun, Luuk N A Willems, Klaus F Rabe, Jouke T Annema. Transesophageal ultrasound-guided fine-needle aspiration for the mediastinal restaging of non-small cell lung cancer. J Thorac Oncol 2011;6:1510-15.
- Devadatta Sarwate 1, Saiyad Sarkar, William S Krimsky, Constantine M Burgan, Kalpesh Patel, Ruth Evans, Daniel P Harley. Optimization of mediastinal staging in potential candidates for stereotactic radiosurgery of the chest. J Thorac Cardiovasc Surg 2012; 144(1):81-6.
- Vial MR, Khan KA, O’Connell O, Peng SA, Gomez DR, Chang JY, Rice DC, Mehran R, Jimenez CJ, Grosu HB, Ost DE, Eapen GA. Vial MR, Khan KA, O’Connell O, Peng SA, Gomez DR, Chang JY, Rice DC, Mehran R, Jimenez CJ, Grosu HB, Ost DE, Eapen GA. Endobronchial Ultrasound-Guided Transbronchial Needle Aspiration in the Nodal Staging of Stereotactic Ablative Body Radiotherapy Patients.Ann Thorac Surg. 2017 May;103(5):1600-1605.
- Folch E, Yamaguchi N, Vanderlaan PA. Adequacy of lymph node transbronchial needle aspirates using convex probe endobronchial ultrasound for multiple tumor genotyping techniques in non-small-cell lung cancer. J Thorac Oncol 2013;8:1438-44.
- Nakajima T, Yasufuku K, Suzuki M. Assessment of epidermal growth factor receptor mutation by endobronchial ultrasound-guided transbronchial needle aspiration. Chest 2007;132:597-602.
- Garcia-Olive I, Monso E, Andreo F. Endobronchial ultrasound-guided transbronchial needle aspiration for identifying EGFR mutations. Eur Respir J 2010;35:391-5.
- Schuurbiers OC, Looijen-Salamon MG, Ligtenberg MJ, van der Heijden HF.A brief retrospective report on the feasibility of epidermal growth factor receptor and KRAS mutation analysis in transesophageal ultrasound- and endobronchial ultrasound-guided fine needle cytological aspirates. J Thorac Oncol 2010;5:1664-7.
- Ronald van Eijk 1, Jappe Licht, Melanie Schrumpf, Mehrdad Talebian Yazdi, Dina Ruano, Giusi I Forte, Petra M Nederlof, Maud Veselic, Klaus F Rabe, Jouke T Annema, Vincent Smit, Hans Morreau, Tom van Wezel. Rapid KRAS, EGFR, BRAF and PIK3CA mutation analysis of fine needle aspirates from non-small-cell lung cancer using allele-specific qPCR. PLoS One 2011;6:e17791.
- George Santis 1, Roger Angell, Guillermina Nickless, Alison Quinn, Amanda Herbert, Paul Cane, James Spicer, Ronan Breen, Emma McLean, Khalid Tobal. Screening for EGFR and KRAS mutations in endobronchial ultrasound derived transbronchial needle aspirates in non-small cell lung cancer using COLD-PCR. PLoS One 2011;6:e25191.
- Takahiro Nakajima 1, Kazuhiro Yasufuku 2, Akira Nakagawara 3, Hideki Kimura 4, Ichiro Yoshino 5. Multigene mutation analysis of metastatic lymph nodes in non-small cell lung cancer diagnosed by endobronchial ultrasound-guided transbronchial needle aspiration. Chest 2011;140:1319-24.
- Julissa Jurado 1, Anjali Saqi 2, Roger Maxfield 3, Alexis Newmark 4, Matt Lavelle 4, Matthew Bacchetta 4, Lyall Gorenstein 4, Frank Dovidio 4, Mark E Ginsburg 4, Joshua Sonett 4, et al. The efficacy of EBUS-guided transbronchial needle aspiration for molecular testing in lung adenocarcinoma. Ann Thorac Surg 2013; 96:1196-202. 10.1016/j.athoracsur.2013.05.066.
- R J José 1, P Shaw, M Taylor, D R Lawrence, P J George, S M Janes, N Navani. Impact of EBUS-TBNA on modalities for tissue acquisition in patients with lung cancer. QJM 2014;107:201-6. 10.1093/qjmed/hct233.
- Abiramy Jeyabalan 1, Nidhi Bhatt 2, Martin J Plummeridge 1, Andrew R L Medford 1. Adequacy of endobronchial ultrasound-guided transbronchial needle aspiration samples processed as histopathological samples for genetic mutation analysis in lung adenocarcinoma. Mol Clin Oncol 2016; 4:119-25.
- Schuurbiers OC, Looijen-Salamon MG, Ligtenberg MJ, van der Heijden HF. A brief retrospective report on the feasibility of epidermal growth factor receptor and KRAS mutation analysis in transesophageal ultrasound- and endobronchial ultrasound-guided fine needle cytological aspirates. J Thorac Oncol. 2010 Oct;5(10):1664-7.
- Sakakibara R, Inamura K, Tambo Y et al. EBUS-TBNA as a promising method for the evaluation of tumor PD-L1 expression in lung cancer. Clinical Lung Cancer (2017). [CrossRef]
- Nasir BS, Edwards M, Tiffault V, Kazakov J, Khereba M, Ferraro P, Liberman M. Transesophageal pulmonary nodule biopsy using endoscopic ultrasonography. J Thorac Cardiovasc Surg. 2014 Sep;148(3):850-5.
- Wallace MB, Pascual JM, Raimondo M,et al. Minimally invasive endoscopic staging of suspected lung cancer. JAMA 2008; 299(5):540-6.
- Crombag LMMJ, Szlubowski A, Stigt JA, Schuurbiers O, Korevaar DA, Bonta PI, Annema JT. EUS-B-FNA vs conventional EUS-FNA for left adrenal gland analysis in lung cancer patients. Lung Cancer. 2017 Jun;108:38-44.
- Liberman M, Sampalis J, Duranceau A, et al. Endosonographic mediastinal lymph node staging of lung cancer. Chest 2014; 146(2):389-97.
- Toloza EM, Harpole L, McCrory DC. Noninvasive staging of non-small cell lung cancer: a review of the current evidence. Chest 2003;123(1):137S–46S.
- Silvestri GA, Hoffman B, Reed CE. Choosing between CT, positron emission tomography, endoscopic ultrasound with fine-needle aspiration, transbronchial needle aspiration, thoracoscopy, mediastinoscopy, and mediastinotomy for staging lung cancer. Chest 2003;123(2):333–5.
- Robert James Cerfolio 1, Ayesha S Bryant, Buddhiwardhan Ojha, Mohammad Eloubeidi. Improving the inaccuracies of clinical staging of patients with NSCLC: a prospective trial. Ann Thorac Surg 2005; 80(4):1207–13.
- McLoud TC, Bourgouin PM, Greenberg RW. Bronchogenic carcinoma: analysis of staging in the mediastinum with CT by correlative lymph node mapping and sampling. Radiology 1992;182(2): 319–23.
- Z T Hammoud 1, R C Anderson, B F Meyers, T J Guthrie, C L Roper, J D Cooper, G A Patterson. The current role of mediastinoscopy in the evaluation of thoracic disease. J Thorac Cardiovasc Surg 1999; 118(5):894–9.
- Anthony Lemaire 1, Ivana Nikolic, Thomas Petersen, Jack C Haney, Eric M Toloza, David H Harpole Jr, Thomas A D’Amico, William R Burfeind. Nine-year single center experience with cervical mediastinoscopy complications and false negative rate. Ann Thorac Surg 2006;82(4):1185–9.
- Von Bartheld MB, van Breda A, Annema JT. Complication rate of endosonography (endobronchial and endoscopic ultrasound): a systematic review. Respiration 2014;87(4):343-51.
- Aerts JG. EUS-FNA of enlarged necrotic lymph nodes may cause infectious mediastinitis. J Thorac Oncol 2008;3: 1191-3.
- Gamrekeli A. Infection of a bronchogenic cyst after ultrasonography-guided fine needle aspiration. Ann Thorac Surg 2013;95: 2154-5.
- Huang CT. A rare constellation of empyema, lung abscess, and mediastinal abscess as a complication of endobronchial ultrasound-guided transbronchial needle aspiration. Eur J Cardiothorac Surg 2011;40:264-5.
- Wildi SM. Diagnosis of benign cysts of the mediastinum: the role and risks of EUS and FNA. Gastrointest Endosc 2003;58:362-8.
- Von Bartheld MB1, Annema JT. Endosonography-related mortality and morbidity for pulmonary indications: a nationwide survey in the Netherlands. Gastrointest Endosc. 2015 Dec; 82(6):1009-15.
- Oliveira RL, Liberman M. Endosonographic Mediastinal Lymph Node Staging in Non-Small Cell Lung Cancer: How I Teach It. Ann Thorac Surg. 2017 Jul; 104(1):18-21.
- Kurt G Tournoy 1, Jouke T Annema, Mark Krasnik, Felix J F Herth, Jan P van Meerbeeck. Endoscopic and endobronchial ultrasonography according to the proposed lymph node map definition in the seventh edition of the tumor, node, and metastasis classification for lung cancer. J Thorac Oncol 2009; 4:1576–1584.
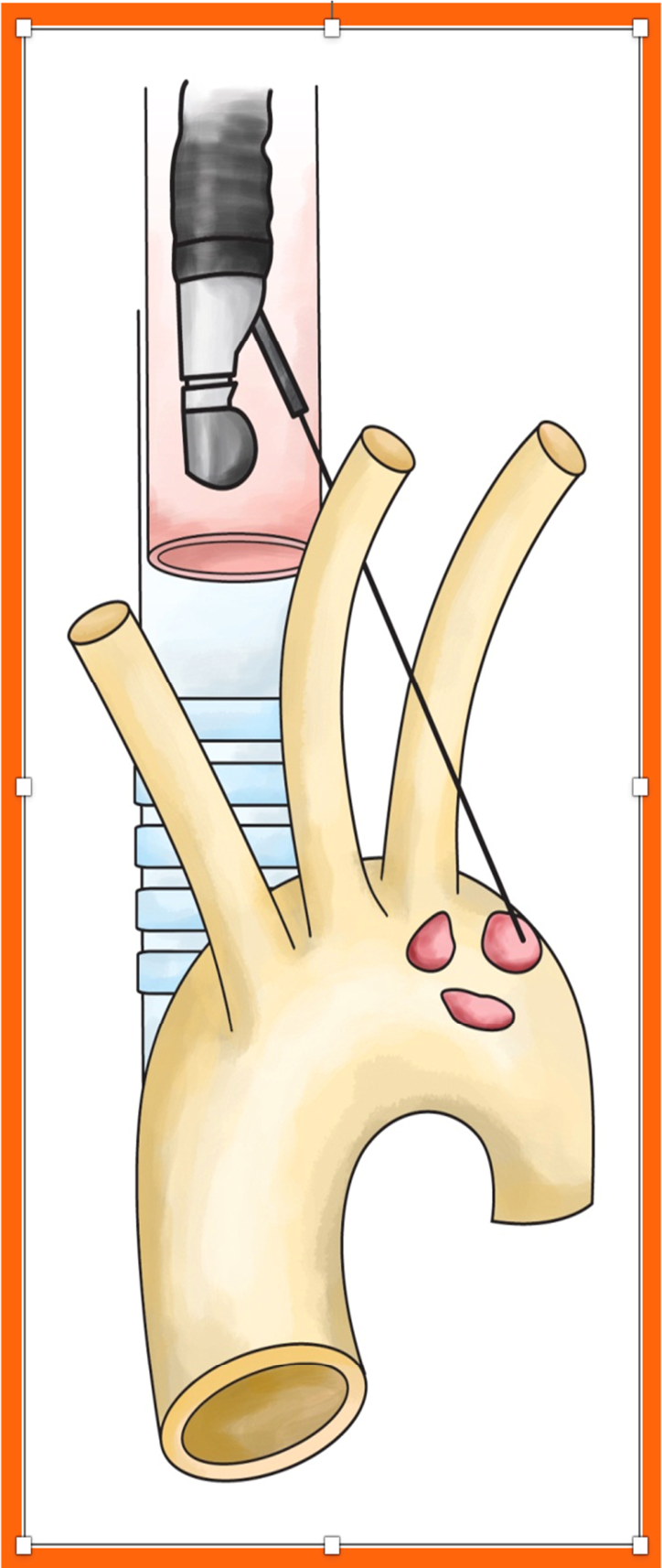
- Bendzsak A, Oliveira R, Goudie E, Thiffault V, Jouquan A, Ferraro P, Liberman M. Evaluation of the Mediastinum: Differentiating Between Stations 4L, 5, and 6 Using EBUS and EUS. Ann Thorac Surg. 2017 Feb;103(2):e219-e221.
- Moishe Liberman 1, André Duranceau, Etienne Grunenwald, Jocelyne Martin, Vicky Thiffault, Mohamed Khereba, Pasquale Ferraro. New technique performed by using EUS access for biopsy of paraaortic (station 6) mediastinal lymph nodes without traversing the aorta (with video).Gastrointest Endosc 2011;73(5):1048-51.
- Kazakov J, Hegde P, Tahiri M, Thiffault V, Ferraro P, Liberman M. Endobronchial and Endoscopic Ultrasound-Guided Transvascular Biopsy of Mediastinal, Hilar, and Lung Lesions. Ann Thorac Surg. 2017 Mar; 103(3):951-955.
- Christian Jenssen 1, Jouke Tabe Annema 1, Paul Clementsen 1, Xin-Wu Cui 1, Mathias Maximilian Borst 1, Christoph Frank Dietrich 1. Ultrasound techniques in the evaluation of the mediastinum, part 2: mediastinal lymph node anatomy and diagnostic reach of ultrasound techniques, clinical work up of neoplastic and inflammatory mediastinal lymphadenopathy using ultrasound techniques and how to learn mediastinal endosonography. J Thorac Dis 2015; 7:E439–E458.
- Stather DR, Maceachern P, Chee A. Trainee impact on advanced diagnostic bronchoscopy: an analysis of 607 consecutive procedures in an interventional pulmonary practice. Respirology. 2013 Jan; 18(1):179-84.
- Lars Konge 1, Paul Frost Clementsen 2, Charlotte Ringsted 3, Valentina Minddal 2, Klaus Richter Larsen 4, Jouke T Annema 5. Simulator training for endobronchial ultrasound: a randomised controlled trial. Eur Respir J 2015; 46:1140–1149.Randomized controlled trial investigating the impact of simulator-based training in EBUS.
- Konge L, Colella S, Vilmann P, Clementsen PF. How to learn and to perform endoscopic ultrasound and endobronchial ultrasound for lung cancer staging: a structured guide and review. Endosc Ultrasound 2015; 4:4–9.
- Medford AR. Learning curve for endobronchial ultrasound-guided transbronchial needle aspiration [letter]. Chest 2012;141:1643.
- Alberto Fernández-Villar 1, Virginia Leiro-Fernández 2, Maribel Botana-Rial 2, Cristina Represas-Represas 2, Manuel Núñez-Delgado 2. The endobronchial ultrasound-guided transbronchial needle biopsy learning curve for mediastinal and hilar lymph node diagnosis. Chest 2012;141:278-9.
- Ernst A, Silvestri GA, Johnstone D. Interventional pulmonary procedures: guidelines from the American College of Chest Physicians. Chest 2003;123:1693-717.
- Bolliger CT, Mathur PN, Beamis JF. ERS/ ATS statement on interventional pulmonology. European Respiratory Society/American Thoracic Society. Eur Respir J 2002;19:356-73.
- Block MI. Endobronchial ultrasound for lung cancer staging: how many stations should be sampled? Ann Thorac Surg 2010;89:1582-7.
- Miller RJ, Mudambi L, Vial MR, Hernandez M, Eapen GA. Miller RJ, Mudambi L, Vial MR, Hernandez M, Eapen GA. Ann Am Thorac Soc. 2017 Apr 11.
- Trisolini R, Cancellieri A, Tinelli C. Rapid onsite evaluation of transbronchial aspirates in the diagnosis of hilar and mediastinal adenopathy: a randomized trial. Chest 2011;139:395-401.
- Oki M, Saka H, Kitagawa C. Rapid onsite cytologic evaluation during endobronchial ultrasound-guided transbronchial needle aspiration for diagnosing lung cancer: a randomized study. Respiration 2013; 85: 486-92.
- Brian T Collins 1, Alexander C Chen, Jeff F Wang, Cory T Bernadt, Souzan Sanati. Improved laboratory resource utilization and patient care with the use of rapid on site evaluation for endobronchial ultrasound fine-needle aspiration biopsy. Cancer Cytopathol 2013;121:544-51.
- Rotolo N, Cattoni M, Crosta G, Nardecchia E, Poli A, Moretti F, Conti V, La Rosa S, Dominioni L, Imperatori A. Comparison of multiple techniques for endobronchial ultrasound-transbronchial needle aspiration specimen preparation in a single institution experience. J Thorac Dis. 2017 May;9(Suppl 5): S381-S385.
- Lonny B Yarmus 1, Jason Akulian 2, Noah Lechtzin 2, Faiza Yasin 2, Biren Kamdar 2, Armin Ernst 3, David E Ost 4, Cynthia Ray 5, Sarah R Greenhill 6, Carlos A Jimenez 4, et al. Comparison of 21-gauge and 22-gauge aspiration needle in endobronchial ultrasound-guided transbronchial needle aspiration: results of the american college of chest physicians quality improvementregistry, education, and evaluation registry.Chest 2013;143(4):1036-43.
- Jeyabalan A, Shelley-Fraser G, Medford AR. Impact of needle gauge on characterization of endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) histology samples. Respirology. 2014 Jul;19(5):735-9.
- Nagula S, Pourmand K, Aslanian H, Bucobo JC, Gonda T, Gonzalez S, Goodman A, Gross SA, Ho S, DiMaio CJ, Kim M, Pais S, Poneros J, Robbins D, Schnoll-Sussman F, Sethi A, Buscaglia JM. Comparing EUS-Fine Needle Aspiration and EUS-Fine Needle Biopsy for Solid Lesions: A Multicenter, Randomized Trial. Clin Gastroenterol Hepatol. 2017 Jun 14. pii: S1542-3565(17)30719-X.
- Tyan C, Patel P, Czarnecka K, Gompelmann D, Eberhardt R, Fortin M, MacEachern P, Hergott CA, Dumoulin E, Tremblay A, Kemp SV, Shah PL, Herth FJF, Yasufuku K. Flexible 19-Gauge Endobronchial Ultrasound-Guided Transbronchial Needle Aspiration Needle: First Experience. Respiration. 2017;94(1):52-57.
- Roberto F Casal 1, Gregg A Staerkel 2, David Ost 3, Francisco A Almeida 4, Mateen H Uzbeck 3, George A Eapen 3, Carlos A Jimenez 3, Graciela M Nogueras-Gonzalez 5, Mona Sarkiss 6, Rodolfo C Morice 3. Randomized. Chest 2012;142(3):568-73.
- Lee HS, Lee GK, Lee HS, Kim MS, Lee JM, Kim HY, Nam BH, Zo JI, Hwangbo B. Real-time endobronchial ultrasound-guided transbronchial needle aspiration in mediastinal staging of non-small cell lung cancer: how many aspirations per target lymph node station? Chest 2008 Aug;134(2):368-374.
- Fujiwara T, Yasufuku K, Nakajima T, Chiyo M, Yoshida S, Suzuki M, Shibuya K, Hiroshima K, Nakatani Y, Yoshino I. The utility of sonographic features during endobronchial ultrasound-guided transbronchial needle aspiration for lymph node staging in patients with lung cancer: a standard endobronchial ultrasound image classification system. Chest. 2010 Sep;138(3):641-7.
- Schmid-Bindert G, Jiang H, Kähler G, Saur J, Henzler T, Wang H, Ren S, Zhou C, Pilz LR. Predicting malignancy in mediastinal lymph nodes by endobronchial ultrasound: a new ultrasound scoring system. Respirology. 2012 Nov;17(8):1190-8.
- Casal RF. Endobronchial ultrasound images may predict malignant involvement of mediastinal lymph nodes: is tissue still the issue? Respirology. 2012 Nov;17(8):1155-6.
- Nosotti M, Palleschi A, Tosi D, Mendogni P, Righi I, Carrinola R, Rosso L. Nosotti M, Palleschi A, Tosi D, Mendogni P, Righi I, Carrinola R, Rosso L. J Thorac Dis. 2017 May;9(Suppl 5): S376-S380.
- Sun J, Zheng X, Mao X, Wang L, Xiong H, Herth FJF, Han B. Sun J, Zheng X, Mao X, Wang L, Xiong H, Herth FJF, Han B. Respiration. 2017;93(5):327-338. [CrossRef]
- Erer OF, Erol S, Anar C, Aydogdu Z, Ozkan SA. Diagnostic yield of EBUS-TBNA for lymphoma and review of the literature. Endosc Ultrasound. 2016 Apr 28. [CrossRef]
- Steinfort DP, Conron M, Tsui A. Endobronchial ultrasound-guided transbronchial needle aspiration for the evaluation of suspected lymphoma. J Thorac Oncol 2010;5:804-9.
- Kennedy MP, Jimenez CA, Bruzzi JF. Endobronchial ultrasound-guided transbronchial needle aspiration in the diagnosis of lymphoma. Thorax 2008;63: 360-5.
- Kheir F, Itani A, Assasa O, Alraiyes AH. The utility of endobronchial ultrasound-transbronchial needle aspiration in lymphoma. Endosc Ultrasound. 2016 Jan-Feb; 5(1):43-8.
- Grosu HB, Iliesiu M, Caraway NP, Medeiros LJ, Lei X, Jimenez CA, Morice RC, Casal RF, Ost D, Eapen GA. Endobronchial Ultrasound-Guided Transbronchial Needle Aspiration for the Diagnosis and Subtyping of Lymphoma. Ann Am Thorac Soc. 2015 Sep;12(9):1336-44.
- Talebian Yazdi M, von Bartheld MB, Waaijenborg FG, Nabers J, Venmans BJ, Veselic-Charvat M, Jansen PM, Marijt EW, Annema JT. Endosonography for the diagnosis of malignant lymphoma presenting with mediastinal lymphadenopathy. J Bronchology Interv Pulmonol. 2014 Oct;21(4):298-305.
- Rocco Trisolini 1, Luigi Lazzari Agli, Carmine Tinelli, Annalisa De Silvestri, Valeria Scotti, Marco Patelli. Endobronchial ultrasound-guided transbronchial needle aspiration for diagnosis of sarcoidosis in clinically unselected study populations. Respirology. 2015 Feb; 20(2):226-34.
- von Bartheld MB, Dekkers OM, Szlubowski A, Eberhardt R, Herth FJ, in ‘t Veen JC, de Jong YP, van der Heijden EH, Tournoy KG, Claussen M, van den Blink B, Shah PL, Zoumot Z, Clementsen P, Porsbjerg C, Mauad T, Bernardi FD, van Zwet EW, Rabe KF, Annema JT. Endosonography vs conventional bronchoscopy for the diagnosis of sarcoidosis: the GRANULOMA randomized clinical trial. JAMA. 2013 Jun 19; 309(23):2457-64.
- Wang Y, Zhu H, Yang S, Wang K, Tian PW, Shi JY, Han QB, Luo YX, Luo FM, Li WM. The Diagnostic Value of Endobronchial Ultrasound-guided Needle Aspiration Biopsy for Lung or Mediastinal Lymph Node Cancer and Tuberculosis. Sichuan Da Xue Xue Bao Yi Xue Ban. 2017 May; 48(3):347-351.
- Sodhi A, Supakul R, Williams GW, Tolley EA, Headley AS, Murillo LC1, Kadaria D. Role of Transbronchial Needle Aspiration (Conventional and EBUS Guided) in the Diagnosis of Histoplasmosis in Patients Presenting with Mediastinal Lymphadenopathy. South Med J. 2017 Jan; 110(1):33-36.
- Sánchez-Cabral O1, Martínez-Mendoza D, Fernandez-Bussy S, Aspuru-García E, Rivera-Rosales R, Luna-Rivero C, Martínez-Orozco JA, Reyes-Terán G. Usefulness of Endobronchial Ultrasound in Patients with Human Immunodeficiency Virus Infection and Mediastinal Lymphadenopathy. Respiration. 2017; 93(6):424-429.
- Patel P, Wada H, Hu HP, Hirohashi K, Kato T, Ujiie H, Ahn JY, Lee D, Geddie W, Yasufuku K. Patel P, Wada H, Hu HP, Hirohashi K, Kato T, Ujiie H, Ahn JY, Lee D, Geddie W, Yasufuku K. Ann Thorac Surg. 2017 Apr;103(4):1158-1164.
- Basil S. Nasir, MBBCh, Marcel Edwards, MD, Vicky Tiffault, RN, Jordan Kazakov, MD, Mohammed Khereba, MD, Pasquale Ferraro, MD, and Moishe Liberman, MD, PhD. Transesophageal pulmonary nodule biopsy usingendoscopic ultrasonography september 2014.
- Rita Bianchi 1, Alessandra Dubini 2, Silvia Asioli 2, Claudia Ravaglia 3, Sara Tomassetti 3, Silvia Puglisi 3, Sara Piciucchi 4, Christian Gurioli 3, Carlo Gurioli 3, Roberto Fiocca 1, et al. Transbronchial cryobiopsy: an effective tool in the diagnosis of lymphoproliferative disorders of the lung. ERJ Open Research. 2020.
- Gabriel Dayan 1, Stephan Soder 1, Anny Godin 1, Antonio Maietta 2, Philippe Stephenson 2, Bernard Lemieux 3, Moishe Liberman 4. Endosonography-Guided Biopsy as a First Test in the Diagnosis of Lymphoma. Seminars in Thoracic and Cardiovascular Surgery. 2021.
- Danielle A Hylton 1, Simon Turner 2, Biniam Kidane 3, Jonathan Spicer 4, Feng Xie 1, Forough Farrokhyar 1, Kazuhiro Yasufuku 5, John Agzarian 6, Waël C Hanna 7; . The Canada Lymph Node Score for prediction of malignancy in mediastinal lymph nodes during endobronchial ultrasound. Journal of Thoracic and Cardiovascular Surgery. 2020.
| T Category | Descriptor | N0 | N1 | N2a | N2b | N3 |
| T1a | ≤1 cm | IA1 | IIA | IIB | IIIA | IIIB |
| T1b | >1to ≤2 cm | IA2 | IIA | IIB | IIIA | IIIB |
| T1c | >2 to ≤ 3 cm | IA3 | IIA | IIB | IIIA | IIIB |
| T2a | Visceral pleura / central invasion | IB | IIB | IIIA | IIIB | IIIB |
| T2a | >3 to ≤4 cm | IB | IIB | IIIA | IIIB | IIIB |
| T2b | >4 to ≤5 cm | IIA | IIB | IIIA | IIIB | IIIB |
| T3 | >5 to ≤ 7 cm | IIB | IIIA | IIIA | IIIB | IIIC |
| T3 | Invasion | IIB | IIIA | IIIA | IIIB | IIIC |
| T3 | Same lobe separate nodules | IIB | IIIA | IIIA | IIIB | IIIC |
| T4 | >7 cm | IIIA | IIIA | IIIB | IIIB | IIIC |
| T4 | Invasion | IIIA | IIIA | IIIB | IIIB | IIIC |
| T4 | Ipsilateral separate tumor nodules | IIIA | IIIA | IIIB | IIIB | IIIC |
| M1a | Contralateral tumor nodules | IVA | IVA | IVA | IVA | IVA |
| M1a | Pleural/pericardial effusion or nodules | IVA | IVA | IVA | IVA | IVA |
| M1b | Single extrathoracic metastasis | IVA | IVA | IVA | IVA | IVA |
| M1c1 | Multiple metastases in one organ | IVB | IVB | IVB | IVB | IVB |
| M1c2 | Multiple metastases in >1 organ | IVB | IVB | IVB | IVB | IVB |
|
T: Primary Tumor | |
| Category | Definition |
| Tx | Primary tumor cannot be assessed |
| T0 | No evidence of primary tumor |
| Tis | Carcinoma in situ |
| T1 | Tumor surrounded by lung or visceral pleura, or in a lobar or more peripheral bronchus |
| T1mi | Minimally invasive adenocarcinoma |
| T1a | Tumor ≤1 cm in greatest dimension |
| T1b | Tumor >1 cm but ≤2 cm |
| T1c | Tumor >2 cm but ≤3 cm |
| T2 | Tumor with any of the following features |
| T2a | >3 cm but ≤4 cm; or visceral pleura invasion; or involves main bronchus; or associated with atelectasis/obstructive pneumonitis |
| T2b | >4 cm but ≤5 cm |
| T3 | >5 cm but ≤7 cm; or chest wall invasion; or pericardium/phrenic nerve involvement; or same-lobe tumor nodules |
| T4 | >7 cm; or invasion of mediastinum, heart, great vessels, trachea, esophagus, diaphragm; or tumor nodules in a different ipsilateral lobe |
|
N: Regional Lymph Nodes | |
| Category | Definition |
| NX | Regional lymph nodes cannot be assessed |
| N0 | No regional lymph node metastasis |
| N1 | Ipsilateral peribronchial and/or hilar lymph node involvement |
| N2 | Ipsilateral mediastinal and/or subcarinal lymph nodes |
| N2a | Single-station N2 involvement |
| N2b | Multi-station N2 involvement |
| N3 | Contralateral mediastinal or hilar, or supraclavicular lymph nodes |
|
M: Distant Metastasis | |
| Category | Definition |
| M0 | No distant metastasis |
| M1 | Distant metastasis present |
| M1a | Pleural/pericardial disease or contralateral lung nodules |
| M1b | Single extrathoracic metastasis |
| M1c | Multiple extrathoracic metastases |
| M1c1 | Multiple metastases in a single organ |
| M1c2 | Multiple metastases in multiple organs |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




