Submitted:
07 April 2026
Posted:
09 April 2026
You are already at the latest version
Abstract
Keywords:
Background
Neurobiological Basis of Phytocompound Modulation in Autism
Part 1: Potential Role of Aberrant Synaptic Plasticity
Part 2: Cannabinoid–Terpene Interactions with Plasticity and Excitatory-Inhibitory Systems
Conclusion
Abbreviations
Acknowledgments
References
- Hirota, T.; King, B. H. Autism Spectrum Disorder: A Review. JAMA 2023, 329(2), 157–168. [Google Scholar] [CrossRef] [PubMed]
- Maskey, M.; Warnell, F.; Parr, J. R.; Le Couteur, A.; McConachie, H. Emotional and behavioural problems in children with autism spectrum disorder. Journal of autism and developmental disorders 2013, 43(4), 851–859. [Google Scholar] [CrossRef]
- McCracken, J. T.; McGough, J.; Shah, B.; Cronin, P.; Hong, D.; Aman, M. G.; Arnold, L. E.; Lindsay, R.; Nash, P.; Hollway, J.; McDougle, C. J.; Posey, D.; Swiezy, N.; Kohn, A.; Scahill, L.; Martin, A.; Koenig, K.; Volkmar, F.; Carroll, D.; Lancor, A. Research Units on Pediatric Psychopharmacology Autism Network (2002). Risperidone in children with autism and serious behavioral problems. The New England journal of medicine 347(5), 314–321. [CrossRef] [PubMed]
- Correll, C. U.; Manu, P.; Olshanskiy, V.; Napolitano, B.; Kane, J. M.; Malhotra, A. K. Cardiometabolic risk of second-generation antipsychotic medications during first-time use in children and adolescents. JAMA 2009, 302(16), 1765–1773. [Google Scholar] [CrossRef] [PubMed]
- McPheeters, M. L.; Warren, Z.; Sathe, N.; Bruzek, J. L.; Krishnaswami, S.; Jerome, R. N.; Veenstra-Vanderweele, J. A systematic review of medical treatments for children with autism spectrum disorders. Pediatrics 2011, 127(5), e1312–e1321. [Google Scholar] [CrossRef]
- Adler, B. A.; Wink, L. K.; Early, M.; Shaffer, R.; Minshawi, N.; McDougle, C. J.; Erickson, C. A. Drug-refractory aggression, self-injurious behavior, and severe tantrums in autism spectrum disorders: a chart review study. Autism: the international journal of research and practice 2015, 19(1), 102–106. [Google Scholar] [CrossRef]
- Höfer, J.; Hoffmann, F.; Bachmann, C. Use of complementary and alternative medicine in children and adolescents with autism spectrum disorder: A systematic review. Autism: the international journal of research and practice 2017, 21(4), 387–402. [Google Scholar] [CrossRef]
- Bilge, S.; Ekici, B. CBD-enriched cannabis for autism spectrum disorder: an experience of a single center in Turkey and reviews of the literature. Journal of cannabis research 2021, 3(1), 53. [Google Scholar] [CrossRef]
- Silva, E. A. D.; Junior; Medeiros, W. M. B.; Torro, N.; Sousa, J. M. M.; Almeida, I. B. C. M.; Costa, F. B. D.; Pontes, K. M.; Nunes, E. L. G.; Rosa, M. D. D.; Albuquerque, K. L. G. D. Cannabis and cannabinoid use in autism spectrum disorder: a systematic review. Trends in psychiatry and psychotherapy 44 2022, e20200149. [Google Scholar] [CrossRef]
- Silva, E. A. D.; Junior; Medeiros, W. M. B.; Santos, J. P. M. D.; Sousa, J. M. M.; Costa, F. B. D.; Pontes, K. M.; Borges, T. C.; Espínola, C.; Neto Segundo; Andrade E Silva, A. H.; Nunes, E. L. G.; Alves, N. T.; Rosa, M. D. D.; Albuquerque, K. L. G. D. Evaluation of the efficacy and safety of cannabidiol-rich cannabis extract in children with autism spectrum disorder: randomized, double-blind, and placebo-controlled clinical trial. Trends in psychiatry and psychotherapy 2024, 46, e20210396. [Google Scholar] [CrossRef]
- Aran, A.; Cassuto, H.; Lubotzky, A.; Wattad, N.; Hazan, E. Brief Report: Cannabidiol-Rich Cannabis in Children with Autism Spectrum Disorder and Severe Behavioral Problems- A Retrospective Feasibility Study. Journal of autism and developmental disorders 2019, 49(3), 1284–1288. [Google Scholar] [CrossRef] [PubMed]
- Bar-Lev Schleider, L.; Mechoulam, R.; Saban, N.; Meiri, G.; Novack, V. Real life Experience of Medical Cannabis Treatment in Autism: Analysis of Safety and Efficacy. Scientific reports 2019, 9(1), 200. [Google Scholar] [CrossRef] [PubMed]
- Fleury-Teixeira, P.; Caixeta, F. V.; Ramires da Silva, L. C.; Brasil-Neto, J. P.; Malcher-Lopes, R. Effects of CBD-Enriched Cannabis sativa Extract on Autism Spectrum Disorder Symptoms: An Observational Study of 18 Participants Undergoing Compassionate Use. Frontiers in neurology 10 2019, 1145. [Google Scholar] [CrossRef] [PubMed]
- Barchel, D.; Stolar, O.; De-Haan, T.; Ziv-Baran, T.; Saban, N.; Fuchs, D. O.; Koren, G.; Berkovitch, M. Oral Cannabidiol Use in Children With Autism Spectrum Disorder to Treat Related Symptoms and Co-morbidities. Frontiers in pharmacology 2019, 9, 1521. [Google Scholar] [CrossRef]
- Mostafavi, M.; Gaitanis, J. Autism Spectrum Disorder and Medical Cannabis: Review and Clinical Experience. Seminars in pediatric neurology 35 2020, 100833. [Google Scholar] [CrossRef]
- Fusar-Poli, L.; Cavone, V.; Tinacci, S.; Concas, I.; Petralia, A.; Signorelli, M. S.; Díaz-Caneja, C. M.; Aguglia, E. Cannabinoids for People with ASD: A Systematic Review of Published and Ongoing Studies. Brain sciences 2020, 10(9), 572. [Google Scholar] [CrossRef]
- Abu-Sawwa, R.; Scutt, B.; Park, Y. Emerging Use of Epidiolex (Cannabidiol) in Epilepsy. The journal of pediatric pharmacology and therapeutics: JPPT: the official journal of PPAG 2020, 25(6), 485–499. [Google Scholar] [CrossRef]
- Velayudhan, L.; McGoohan, K.; Bhattacharyya, S. Safety and tolerability of natural and synthetic cannabinoids in adults aged over 50 years: A systematic review and meta-analysis. PLoS medicine 2021, 18(3), e1003524. [Google Scholar] [CrossRef]
- ElSohly, M. A.; Radwan, M. M.; Gul, W.; Chandra, S.; Galal, A. Phytochemistry of Cannabis sativa L. Progress in the chemistry of organic natural products 2017, 103, 1–36. [Google Scholar] [CrossRef]
- Hanuš, L. O.; Meyer, S. M.; Muñoz, E.; Taglialatela-Scafati, O.; Appendino, G. Phytocannabinoids: a unified critical inventory. Natural product reports 2016, 33(12), 1357–1392. [Google Scholar] [CrossRef]
- Rosenberg, E. C.; Tsien, R. W.; Whalley, B. J.; Devinsky, O. Cannabinoids and Epilepsy. Neurotherapeutics: the journal of the American Society for Experimental NeuroTherapeutics 2015, 12(4), 747–768. [Google Scholar] [CrossRef]
- McPartland, J. M.; Russo, E. B. Cannabis and cannabis extracts: Greater than the sum of their parts? Journal of Cannabis Therapeutics 2001, 1(3–4), 103–132. [Google Scholar] [CrossRef]
- Russo, E. B. The Case for the Entourage Effect and Conventional Breeding of Clinical Cannabis: No “Strain,” No Gain. Frontiers in plant science 2019, 9, 1969. [Google Scholar] [CrossRef] [PubMed]
- Pamplona, F. A.; da Silva, L. R.; Coan, A. C. Potential Clinical Benefits of CBD-Rich Cannabis Extracts Over Purified CBD in Treatment-Resistant Epilepsy: Observational Data Meta-analysis. Frontiers in neurology 2018, 9, 759. [Google Scholar] [CrossRef] [PubMed]
- Maayah, Z. H.; Takahara, S.; Ferdaoussi, M.; Dyck, J. R. B. The molecular mechanisms that underpin the biological benefits of full-spectrum cannabis extract in the treatment of neuropathic pain and inflammation. Biochimica et biophysica acta. Molecular basis of disease 2020, 1866(7), 165771. [Google Scholar] [CrossRef] [PubMed]
- Mechoulam, R.; Ben-Shabat, S. From gan-zi-gun-nu to anandamide and 2-arachidonoylglycerol: the ongoing story of cannabis. Natural product reports 1999, 16(2), 131–143. [Google Scholar] [CrossRef]
- Ben-Shabat, S.; Fride, E.; Sheskin, T.; Tamiri, T.; Rhee, M. H.; Vogel, Z.; Bisogno, T.; De Petrocellis, L.; Di Marzo, V.; Mechoulam, R. An entourage effect: inactive endogenous fatty acid glycerol esters enhance 2-arachidonoyl-glycerol cannabinoid activity. European journal of pharmacology 1998, 353(1), 23–31. [Google Scholar] [CrossRef]
- Carlini, E. A.; Karniol, I. G.; Renault, P. F.; Schuster, C. R. Effects of marihuana in laboratory animals and in man. British journal of pharmacology 1974, 50(2), 299–309. [Google Scholar] [CrossRef]
- Mayford, M.; Siegelbaum, S. A.; Kandel, E. R. Synapses and memory storage. Cold Spring Harbor perspectives in biology 2012, 4(6), a005751. [Google Scholar] [CrossRef]
- Hitti, F. L.; Siegelbaum, S. A. The hippocampal CA2 region is essential for social memory. Nature 2014, 508(7494), 88–92. [Google Scholar] [CrossRef]
- Davis, M. H.; Gaskell, M. G. A complementary systems account of word learning: neural and behavioural evidence. Philosophical transactions of the Royal Society of London. Series B, Biological sciences 2009, 364(1536), 3773–3800. [Google Scholar] [CrossRef] [PubMed]
- Duff, M. C.; Brown-Schmidt, S. The hippocampus and the flexible use and processing of language. Frontiers in Human Neuroscience 6 2012, 69. [Google Scholar] [CrossRef] [PubMed]
- Karni, A.; Meyer, G.; Jezzard, P.; Adams, M. M.; Turner, R.; Ungerleider, L. G. Functional MRI evidence for adult motor cortex plasticity during motor skill learning. Nature 1995, 377(6545), 155–158. [Google Scholar] [CrossRef] [PubMed]
- Doyon, J.; Benali, H. Reorganization and plasticity in the adult brain during learning of motor skills. Current opinion in neurobiology 2005, 15(2), 161–167. [Google Scholar] [CrossRef]
- Yashiro, K.; Riday, T. T.; Condon, K. H.; Roberts, A. C.; Bernardo, D. R.; Prakash, R.; Weinberg, R. J.; Ehlers, M. D.; Philpot, B. D. Ube3a is required for experience-dependent maturation of the neocortex. Nature neuroscience 2009, 12(6), 777–783. [Google Scholar] [CrossRef]
- Hutsler, J. J.; Zhang, H. Increased dendritic spine densities on cortical projection neurons in autism spectrum disorders. Brain research 1309 2010, 83–94. [Google Scholar] [CrossRef]
- Martínez-Cerdeño, V. Dendrite and spine modifications in autism and related neurodevelopmental disorders in patients and animal models. Developmental neurobiology 2017, 77(4), 393–404. [Google Scholar] [CrossRef]
- Huber, K. M.; Gallagher, S. M.; Warren, S. T.; Bear, M. F. Altered synaptic plasticity in a mouse model of fragile X mental retardation. Proceedings of the National Academy of Sciences of the United States of America 2002, 99(11), 7746–7750. [Google Scholar] [CrossRef]
- Harlow, E. G.; Till, S. M.; Russell, T. A.; Wijetunge, L. S.; Kind, P.; Contractor, A. Critical period plasticity is disrupted in the barrel cortex of FMR1 knockout mice. Neuron 2010, 65(3), 385–398. [Google Scholar] [CrossRef]
- Tropea, D.; Giacometti, E.; Wilson, N. R.; Beard, C.; McCurry, C.; Fu, D. D.; Flannery, R.; Jaenisch, R.; Sur, M. Partial reversal of Rett Syndrome-like symptoms in MeCP2 mutant mice. Proceedings of the National Academy of Sciences of the United States of America 2009, 106(6), 2029–2034. [Google Scholar] [CrossRef]
- Asaka, Y.; Jugloff, D. G.; Zhang, L.; Eubanks, J. H.; Fitzsimonds, R. M. Hippocampal synaptic plasticity is impaired in the Mecp2-null mouse model of Rett syndrome. Neurobiology of disease 2006, 21(1), 217–227. [Google Scholar] [CrossRef]
- Ehninger, D.; Han, S.; Shilyansky, C.; Zhou, Y.; Li, W.; Kwiatkowski, D. J.; Ramesh, V.; Silva, A. J. Reversal of learning deficits in a Tsc2+/- mouse model of tuberous sclerosis. Nature medicine 2008, 14(8), 843–848. [Google Scholar] [CrossRef]
- von der Brelie, C.; Waltereit, R.; Zhang, L.; Beck, H.; Kirschstein, T. Impaired synaptic plasticity in a rat model of tuberous sclerosis. The European journal of neuroscience 2006, 23(3), 686–692. [Google Scholar] [CrossRef] [PubMed]
- Wegener, S.; Buschler, A.; Stempel, A. V.; Kang, S. J.; Lim, C. S.; Kaang, B. K.; Shoichet, S. A.; Manahan-Vaughan, D.; Schmitz, D. Defective Synapse Maturation and Enhanced Synaptic Plasticity in Shank2 Δex7-/- Mice. eNeuro 2018, 5(3), ENEURO.0398–17. [Google Scholar] [CrossRef] [PubMed]
- Morimura, N.; Yasuda, H.; Yamaguchi, K.; Katayama, K. I.; Hatayama, M.; Tomioka, N. H.; Odagawa, M.; Kamiya, A.; Iwayama, Y.; Maekawa, M.; Nakamura, K.; Matsuzaki, H.; Tsujii, M.; Yamada, K.; Yoshikawa, T.; Aruga, J. Autism-like behaviours and enhanced memory formation and synaptic plasticity in Lrfn2/SALM1-deficient mice. Nature communications 8 2017, 15800. [Google Scholar] [CrossRef] [PubMed]
- LeBlanc, J. J.; Fagiolini, M. Autism: a “critical period” disorder? In Neural plasticity, 2011; 2011; p. 921680. [Google Scholar] [CrossRef]
- Chung, L.; Bey, A. L.; Jiang, Y. H. Synaptic plasticity in mouse models of autism spectrum disorders. The Korean journal of physiology & pharmacology: official journal of the Korean Physiological Society and the Korean Society of Pharmacology 2012, 16(6), 369–378. [Google Scholar] [CrossRef]
- Muellerleile, J.; Blistein, A.; Rohlmann, A.; Scheiwe, F.; Missler, M.; Schwarzacher, S. W.; Jedlicka, P. Enhanced LTP of population spikes in the dentate gyrus of mice haploinsufficient for neurobeachin. Scientific reports 2020, 10(1), 16058. [Google Scholar] [CrossRef]
- Rigby, M. J.; Orefice, N. S.; Lawton, A. J.; Ma, M.; Shapiro, S. L.; Yi, S. Y.; Dieterich, I. A.; Frelka, A.; Miles, H. N.; Pearce, R. A.; Yu, J. P. J.; Li, L.; Denu, J. M.; Puglielli, L. Increased expression of SLC25A1/CIC causes an autistic-like phenotype with altered neuron morphology. Brain: a journal of neurology 2022, 145(2), 500–516. [Google Scholar] [CrossRef]
- Simões de Oliveira, L.; O’Leary, H. E.; Nawaz, S.; Loureiro, R.; Davenport, E. C.; Baxter, P.; Louros, S. R.; Dando, O.; Perkins, E.; Peltier, J.; Trost, M.; Osterweil, E. K.; Hardingham, G. E.; Cousin, M. A.; Chattarji, S.; Booker, S. A.; Benke, T. A.; Wyllie, D. J. A.; Kind, P. C. Enhanced hippocampal LTP but normal NMDA receptor and AMPA receptor function in a rat model of CDKL5 deficiency disorder. Molecular autism 2024, 15(1), 28. [Google Scholar] [CrossRef]
- Mohammadkhani, R.; Ghahremani, R.; Salehi, I.; Safari, S.; Karimi, S. A.; Zarei, M. Impairment in social interaction and hippocampal long-term potentiation at perforant pathway-dentate gyrus synapses in a prenatal valproic acid-induced rat model of autism. Brain communications 2022, 4(5), fcac221. [Google Scholar] [CrossRef]
- Rinaldi, T.; Kulangara, K.; Antoniello, K.; Markram, H. Elevated NMDA receptor levels and enhanced postsynaptic long-term potentiation induced by prenatal exposure to valproic acid. Proceedings of the National Academy of Sciences of the United States of America 2007, 104(33), 13501–13506. [Google Scholar] [CrossRef] [PubMed]
- Martin, H. G.; Manzoni, O. J. Late onset deficits in synaptic plasticity in the valproic acid rat model of autism. Frontiers in cellular neuroscience 2014, 8, 23. [Google Scholar] [CrossRef] [PubMed]
- Soda, T.; Mapelli, L.; Locatelli, F.; Botta, L.; Goldfarb, M.; Prestori, F.; D’Angelo, E. Hyperexcitability and Hyperplasticity Disrupt Cerebellar Signal Transfer in the IB2 KO Mouse Model of Autism. The Journal of neuroscience: the official journal of the Society for Neuroscience 2019, 39(13), 2383–2397. [Google Scholar] [CrossRef] [PubMed]
- Oberman, L.; Eldaief, M.; Fecteau, S.; Ifert-Miller, F.; Tormos, J. M.; Pascual-Leone, A. Abnormal modulation of corticospinal excitability in adults with Asperger’s syndrome. The European journal of neuroscience 2012, 36(6), 2782–2788. [Google Scholar] [CrossRef]
- Benthall, K. N.; Cording, K. R.; Agopyan-Miu, A. H. C. W.; Wong, C. D.; Chen, E. Y.; Bateup, H. S. Loss of Tsc1 from striatal direct pathway neurons impairs endocannabinoid-LTD and enhances motor routine learning. Cell reports 2021, 36(6), 109511. [Google Scholar] [CrossRef]
- Wang, W.; Li, C.; Chen, Q.; van der Goes, M. S.; Hawrot, J.; Yao, A. Y.; Gao, X.; Lu, C.; Zang, Y.; Zhang, Q.; Lyman, K.; Wang, D.; Guo, B.; Wu, S.; Gerfen, C. R.; Fu, Z.; Feng, G. Striatopallidal dysfunction underlies repetitive behavior in Shank3-deficient model of autism. The Journal of clinical investigation 2017, 127(5), 1978–1990. [Google Scholar] [CrossRef]
- Mink, J. W. The basal ganglia: focused selection and inhibition of competing motor programs. Progress in neurobiology 1996, 50(4), 381–425. [Google Scholar] [CrossRef]
- Gipson, C. D.; Olive, M. F. Structural and functional plasticity of dendritic spines - root or result of behavior? Genes, brain, and behavior 2017, 16(1), 101–117. [Google Scholar] [CrossRef]
- Sala, C.; Segal, M. Dendritic spines: the locus of structural and functional plasticity. Physiological reviews 2014, 94(1), 141–188. [Google Scholar] [CrossRef]
- Feenstra, M. G.; Botterblom, M. H.; van Uum, J. F. Novelty-induced increase in dopamine release in the rat prefrontal cortex in vivo: inhibition by diazepam. Neuroscience letters 1995, 189(2), 81–84. [Google Scholar] [CrossRef]
- Yamasaki, M.; Takeuchi, T. Locus Coeruleus and Dopamine-Dependent Memory Consolidation. Neural plasticity 2017, 2017, 8602690. [Google Scholar] [CrossRef]
- Tritsch, N. X.; Sabatini, B. L. Dopaminergic modulation of synaptic transmission in cortex and striatum. Neuron 2012, 76(1), 33–50. [Google Scholar] [CrossRef]
- Takeuchi, T.; Duszkiewicz, A. J.; Sonneborn, A.; Spooner, P. A.; Yamasaki, M.; Watanabe, M.; Smith, C. C.; Fernández, G.; Deisseroth, K.; Greene, R. W.; Morris, R. G. Locus coeruleus and dopaminergic consolidation of everyday memory. Nature 2016, 537(7620), 357–362. [Google Scholar] [CrossRef] [PubMed]
- Jay, T. M. Dopamine: a potential substrate for synaptic plasticity and memory mechanisms. Progress in neurobiology 2003, 69(6), 375–390. [Google Scholar] [CrossRef] [PubMed]
- Pavăl, D. A Dopamine Hypothesis of Autism Spectrum Disorder. Developmental neuroscience 2017, 39(5), 355–360. [Google Scholar] [CrossRef] [PubMed]
- Klanker, M.; Feenstra, M.; Denys, D. Dopaminergic control of cognitive flexibility in humans and animals. Frontiers in neuroscience 2013, 7, 201. [Google Scholar] [CrossRef]
- van Holstein, M.; Aarts, E.; van der Schaaf, M. E.; Geurts, D. E.; Verkes, R. J.; Franke, B.; van Schouwenburg, M. R.; Cools, R. Human cognitive flexibility depends on dopamine D2 receptor signaling. Psychopharmacology 2011, 218(3), 567–578. [Google Scholar] [CrossRef]
- Buzsáki, G. Hippocampal sharp wave-ripple: A cognitive biomarker for episodic memory and planning. Hippocampus 2015, 25(10), 1073–1188. [Google Scholar] [CrossRef]
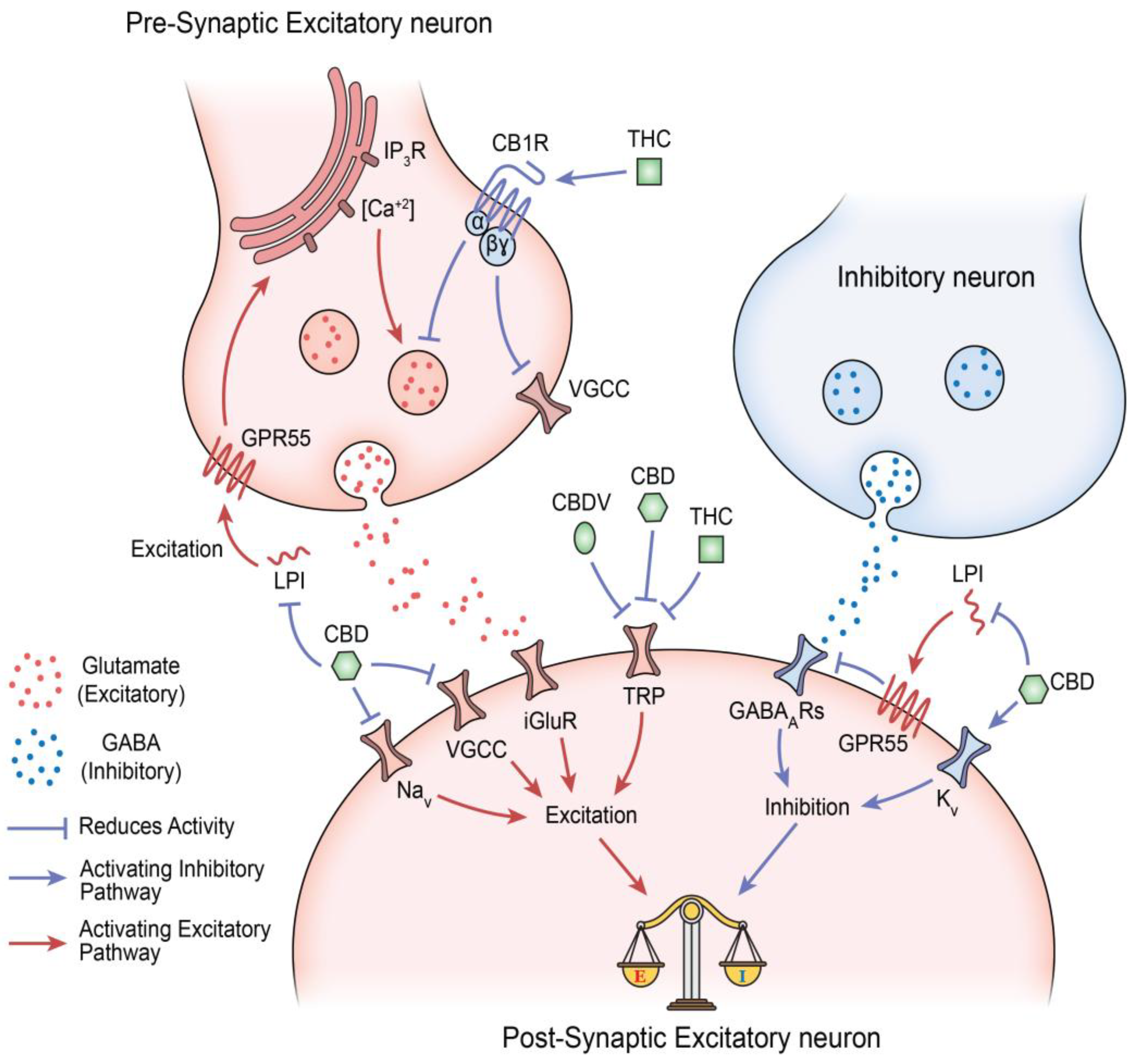
- Rosenberg, E. C.; Chamberland, S.; Bazelot, M.; Nebet, E. R.; Wang, X.; McKenzie, S.; Jain, S.; Greenhill, S.; Wilson, M.; Marley, N.; Salah, A.; Bailey, S.; Patra, P. H.; Rose, R.; Chenouard, N.; Sun, S. E. D.; Jones, D.; Buzsáki, G.; Devinsky, O.; Woodhall, G.; Tsien, R. W. Cannabidiol modulates excitatory-inhibitory ratio to counter hippocampal hyperactivity. Neuron 2023, 111(8), 1282–1300.e8. [Google Scholar] [CrossRef]
- Sylantyev, S.; Jensen, T. P.; Ross, R. A.; Rusakov, D. A. Cannabinoid- and lysophosphatidylinositol-sensitive receptor GPR55 boosts neurotransmitter release at central synapses. Proceedings of the National Academy of Sciences of the United States of America 2013, 110(13), 5193–5198. [Google Scholar] [CrossRef]
- Malenka, R. C.; Bear, M. F. LTP and LTD: an embarrassment of riches. Neuron 2004, 44(1), 5–21. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A. Long-Term Potentiation at CA3-CA1 Hippocampal Synapses with Special Emphasis on Aging, Disease, and Stress. Frontiers in aging neuroscience 2011, 3, 7. [Google Scholar] [CrossRef]
- Huganir, R. L.; Nicoll, R. A. AMPARs and synaptic plasticity: the last 25 years. Neuron 2013, 80(3), 704–717. [Google Scholar] [CrossRef] [PubMed]
- Niswender, C. M.; Conn, P. J. Metabotropic glutamate receptors: physiology, pharmacology, and disease. Annual review of pharmacology and toxicology 2010, 50, 295–322. [Google Scholar] [CrossRef] [PubMed]
- Sawzdargo, M.; Nguyen, T.; Lee, D. K.; Lynch, K. R.; Cheng, R.; Heng, H. H.; George, S. R.; O’Dowd, B. F. Identification and cloning of three novel human G protein-coupled receptor genes GPR52, PsiGPR53 and GPR55: GPR55 is extensively expressed in human brain. Brain research. Molecular brain research 1999, 64(2), 193–198. [Google Scholar] [CrossRef]
- Marichal-Cancino, B. A.; Fajardo-Valdez, A.; Ruiz-Contreras, A. E.; Mendez-Díaz, M.; Prospero-García, O. Advances in the Physiology of GPR55 in the Central Nervous System. Current neuropharmacology 2017, 15(5), 771–778. [Google Scholar] [CrossRef]
- Rubenstein, J. L.; Merzenich, M. M. Model of autism: increased ratio of excitation/inhibition in key neural systems. Genes, brain, and behavior 2003, 2(5), 255–267. [Google Scholar] [CrossRef]
- De Petrocellis, L.; Ligresti, A.; Moriello, A. S.; Allarà, M.; Bisogno, T.; Petrosino, S.; Stott, C. G.; Di Marzo, V. Effects of cannabinoids and cannabinoid-enriched Cannabis extracts on TRP channels and endocannabinoid metabolic enzymes. British journal of pharmacology 2011, 163(7), 1479–1494. [Google Scholar] [CrossRef]
- Bisogno, T.; Hanus, L.; De Petrocellis, L.; Tchilibon, S.; Ponde, D. E.; Brandi, I.; Moriello, A. S.; Davis, J. B.; Mechoulam, R.; Di Marzo, V. Molecular targets for cannabidiol and its synthetic analogues: effect on vanilloid VR1 receptors and on the cellular uptake and enzymatic hydrolysis of anandamide. British journal of pharmacology 2001, 134(4), 845–852. [Google Scholar] [CrossRef]
- Etemad, L.; Karimi, G.; Alavi, M. S.; Roohbakhsh, A. Pharmacological effects of cannabidiol by transient receptor potential channels. Life sciences 2022, 300, 120582. [Google Scholar] [CrossRef]
- Russo, E. B.; Burnett, A.; Hall, B.; Parker, K. K. Agonistic properties of cannabidiol at 5-HT1a receptors. Neurochemical research 2005, 30(8), 1037–1043. [Google Scholar] [CrossRef] [PubMed]
- Ahrens, J.; Demir, R.; Leuwer, M.; de la Roche, J.; Krampfl, K.; Foadi, N.; Karst, M.; Haeseler, G. The nonpsychotropic cannabinoid cannabidiol modulates and directly activates alpha-1 and alpha-1-Beta glycine receptor function. Pharmacology 2009, 83(4), 217–222. [Google Scholar] [CrossRef] [PubMed]
- De Petrocellis, L.; Vellani, V.; Schiano-Moriello, A.; Marini, P.; Magherini, P. C.; Orlando, P.; Di Marzo, V. Plant-derived cannabinoids modulate the activity of transient receptor potential channels of ankyrin type-1 and melastatin type-8. The Journal of pharmacology and experimental therapeutics 2008, 325(3), 1007–1015. [Google Scholar] [CrossRef] [PubMed]
- Ross, H. R.; Napier, I.; Connor, M. Inhibition of recombinant human T-type calcium channels by Delta9-tetrahydrocannabinol and cannabidiol. The Journal of biological chemistry 2008, 283(23), 16124–16134. [Google Scholar] [CrossRef]
- Ghovanloo, M. R.; Shuart, N. G.; Mezeyova, J.; Dean, R. A.; Ruben, P. C.; Goodchild, S. J. Inhibitory effects of cannabidiol on voltage-dependent sodium currents. The Journal of biological chemistry 2018, 293(43), 16546–16558. [Google Scholar] [CrossRef]
- Zhang, H. B.; Heckman, L.; Niday, Z.; Jo, S.; Fujita, A.; Shim, J.; Pandey, R.; Al Jandal, H.; Jayakar, S.; Barrett, L. B.; Smith, J.; Woolf, C. J.; Bean, B. P. Cannabidiol activates neuronal Kv7 channels. eLife 2022, 11, e73246. [Google Scholar] [CrossRef]
- Frodella, C. M.; Pruett, S. B.; Ross, M. K.; Kaplan, B. L. F. Oxytocin and Vasopressin Gene Expression in the Brain as Potential Biomarkers for Cannabidiol Therapeutic Efficacy. Biomedicines 2024, 12(6), 1273. [Google Scholar] [CrossRef]
- Leweke, F. M.; Piomelli, D.; Pahlisch, F.; Muhl, D.; Gerth, C. W.; Hoyer, C.; Klosterkötter, J.; Hellmich, M.; Koethe, D. Cannabidiol enhances anandamide signaling and alleviates psychotic symptoms of schizophrenia. Translational psychiatry 2012, 2(3), e94. [Google Scholar] [CrossRef]
- Rakhshan, F.; Day, T. A.; Blakely, R. D.; Barker, E. L. Carrier-mediated uptake of the endogenous cannabinoid anandamide in RBL-2H3 cells. The Journal of pharmacology and experimental therapeutics 2000, 292(3), 960–967. [Google Scholar] [CrossRef]
- Marsicano, G.; Lutz, B. Expression of the cannabinoid receptor CB1 in distinct neuronal subpopulations in the adult mouse forebrain. The European journal of neuroscience 1999, 11(12), 4213–4225. [Google Scholar] [CrossRef]
- Castillo, P. E.; Younts, T. J.; Chávez, A. E.; Hashimotodani, Y. Endocannabinoid signaling and synaptic function. Neuron 2012, 76(1), 70–81. [Google Scholar] [CrossRef] [PubMed]
- Jensen, K. R.; Berthoux, C.; Nasrallah, K.; Castillo, P. E. Multiple cannabinoid signaling cascades powerfully suppress recurrent excitation in the hippocampus. Proceedings of the National Academy of Sciences of the United States of America 2021, 118(4), e2017590118. [Google Scholar] [CrossRef] [PubMed]
- Katona, I.; Urbán, G. M.; Wallace, M.; Ledent, C.; Jung, K. M.; Piomelli, D.; Mackie, K.; Freund, T. F. Molecular composition of the endocannabinoid system at glutamatergic synapses. The Journal of neuroscience: the official journal of the Society for Neuroscience 2006, 26(21), 5628–5637. [Google Scholar] [CrossRef] [PubMed]
- Hashimotodani, Y.; Nasrallah, K.; Jensen, K. R.; Chávez, A. E.; Carrera, D.; Castillo, P. E. LTP at Hilar Mossy Cell-Dentate Granule Cell Synapses Modulates Dentate Gyrus Output by Increasing Excitation/Inhibition Balance. Neuron 2017, 95(4), 928–943.e3. [Google Scholar] [CrossRef]
- Muller, C.; Morales, P.; Reggio, P. H. Cannabinoid Ligands Targeting TRP Channels. Frontiers in molecular neuroscience 2019, 11, 487. [Google Scholar] [CrossRef]
- Qin, N.; Neeper, M. P.; Liu, Y.; Hutchinson, T. L.; Lubin, M. L.; Flores, C. M. TRPV2 is activated by cannabidiol and mediates CGRP release in cultured rat dorsal root ganglion neurons. The Journal of neuroscience: the official journal of the Society for Neuroscience 2008, 28(24), 6231–6238. [Google Scholar] [CrossRef]
- Abood, M. E.; Rizvi, G.; Sallapudi, N.; McAllister, S. D. Activation of the CB1 cannabinoid receptor protects cultured mouse spinal neurons against excitotoxicity. Neuroscience letters 2001, 309(3), 197–201. [Google Scholar] [CrossRef]
- van der Stelt, M.; Veldhuis, W. B.; Bär, P. R.; Veldink, G. A.; Vliegenthart, J. F.; Nicolay, K. Neuroprotection by Delta9-tetrahydrocannabinol, the main active compound in marijuana, against ouabain-induced in vivo excitotoxicity. The Journal of neuroscience: the official journal of the Society for Neuroscience 2001, 21(17), 6475–6479. [Google Scholar] [CrossRef]
- El-Remessy, A. B.; Khalil, I. E.; Matragoon, S.; Abou-Mohamed, G.; Tsai, N. J.; Roon, P.; Caldwell, R. B.; Caldwell, R. W.; Green, K.; Liou, G. I. Neuroprotective effect of (-)Delta9-tetrahydrocannabinol and cannabidiol in N-methyl-D-aspartate-induced retinal neurotoxicity: involvement of peroxynitrite. The American journal of pathology 2003, 163(5), 1997–2008. [Google Scholar] [CrossRef]
- Mechoulam, R.; Panikashvili, D.; Shohami, E. Cannabinoids and brain injury: therapeutic implications. Trends in molecular medicine 2002, 8(2), 58–61. [Google Scholar] [CrossRef]
- Gilbert, G. L.; Kim, H. J.; Waataja, J. J.; Thayer, S. A. Delta9-tetrahydrocannabinol protects hippocampal neurons from excitotoxicity. Brain research 2007, 1128(1), 61–69. [Google Scholar] [CrossRef]
- Nagayama, T.; Sinor, A. D.; Simon, R. P.; Chen, J.; Graham, S. H.; Jin, K.; Greenberg, D. A. Cannabinoids and neuroprotection in global and focal cerebral ischemia and in neuronal cultures. The Journal of neuroscience: the official journal of the Society for Neuroscience 1999, 19(8), 2987–2995. [Google Scholar] [CrossRef] [PubMed]
- Zani, A.; Braida, D.; Capurro, V.; Sala, M. Delta9-tetrahydrocannabinol (THC) and AM 404 protect against cerebral ischaemia in gerbils through a mechanism involving cannabinoid and opioid receptors. British journal of pharmacology 2007, 152(8), 1301–1311. [Google Scholar] [CrossRef] [PubMed]
- Hampson, A. J.; Grimaldi, M.; Axelrod, J.; Wink, D. Cannabidiol and Delta9-tetrahydrocannabinol are neuroprotective antioxidants. Proceedings of the National Academy of Sciences of the United States of America 1998, 95(14), 8268–8273. [Google Scholar] [CrossRef] [PubMed]
- Vigli, D.; Cosentino, L.; Raggi, C.; Laviola, G.; Woolley-Roberts, M.; De Filippis, B. Chronic treatment with the phytocannabinoid Cannabidivarin (CBDV) rescues behavioural alterations and brain atrophy in a mouse model of Rett syndrome. Neuropharmacology 140 2018, 121–129. [Google Scholar] [CrossRef]
- Zamberletti, E.; Gabaglio, M.; Piscitelli, F.; Brodie, J. S.; Woolley-Roberts, M.; Barbiero, I.; Tramarin, M.; Binelli, G.; Landsberger, N.; Kilstrup-Nielsen, C.; Rubino, T.; Di Marzo, V.; Parolaro, D. Cannabidivarin completely rescues cognitive deficits and delays neurological and motor defects in male Mecp2 mutant mice. Journal of psychopharmacology (Oxford, England) 2019, 33(7), 894–907. [Google Scholar] [CrossRef]
- Zamberletti, E.; Gabaglio, M.; Woolley-Roberts, M.; Bingham, S.; Rubino, T.; Parolaro, D. Cannabidivarin Treatment Ameliorates Autism-Like Behaviors and Restores Hippocampal Endocannabinoid System and Glia Alterations Induced by Prenatal Valproic Acid Exposure in Rats. Frontiers in cellular neuroscience 13 2019, 367. [Google Scholar] [CrossRef]
- Deiana, S.; Watanabe, A.; Yamasaki, Y.; Amada, N.; Arthur, M.; Fleming, S.; Woodcock, H.; Dorward, P.; Pigliacampo, B.; Close, S.; Platt, B.; Riedel, G. Plasma and brain pharmacokinetic profile of cannabidiol (CBD), cannabidivarine (CBDV), Δ9-tetrahydrocannabivarin (THCV) and cannabigerol (CBG) in rats and mice following oral and intraperitoneal administration and CBD action on obsessive-compulsive behaviour. Psychopharmacology 2012, 219(3), 859–873. [Google Scholar] [CrossRef]
- Hill, A. J.; Weston, S. E.; Jones, N. A.; Smith, I.; Bevan, S. A.; Williamson, E. M.; Stephens, G. J.; Williams, C. M.; Whalley, B. J. Δ9-Tetrahydrocannabivarin suppresses in vitro epileptiform and in vivo seizure activity in adult rats. Epilepsia 2010, 51(8), 1522–1532. [Google Scholar] [CrossRef]
- Angley, N. Medical marijuana and autism: ‘I’m getting my boy back’ mom says. CNN. 2021. Available online: https://www.cnn.com/2021/12/19/health/autism-medical-marijuana-cbd-weed-documentary/index.html.
- Pretzsch, C. M.; Voinescu, B.; Lythgoe, D.; Horder, J.; Mendez, M. A.; Wichers, R.; Ajram, L.; Ivin, G.; Heasman, M.; Edden, R. A. E.; Williams, S.; Murphy, D. G. M.; Daly, E.; McAlonan, G. M. Effects of cannabidivarin (CBDV) on brain excitation and inhibition systems in adults with and without Autism Spectrum Disorder (ASD): a single dose trial during magnetic resonance spectroscopy. Translational psychiatry 2019, 9(1), 313. [Google Scholar] [CrossRef]
- Huizenga, M. N.; Sepulveda-Rodriguez, A.; Forcelli, P. A. Preclinical safety and efficacy of cannabidivarin for early life seizures. Neuropharmacology 148 2019, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Nachnani, R.; Raup-Konsavage, W. M.; Vrana, K. E. The Pharmacological Case for Cannabigerol. The Journal of pharmacology and experimental therapeutics 2021, 376(2), 204–212. [Google Scholar] [CrossRef] [PubMed]
- Hill, A. J.; Jones, N. A.; Smith, I.; Hill, C. L.; Williams, C. M.; Stephens, G. J.; Whalley, B. J. Voltage-gated sodium (NaV) channel blockade by plant cannabinoids does not confer anticonvulsant effects per se. Neuroscience letters 2014, 566, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Li, W.; Malhi, N. K.; Huang, J.; Li, Q.; Zhou, Z.; Wang, R.; Peng, J.; Yin, T.; Wang, H. Cannabigerol (CBG): A Comprehensive Review of Its Molecular Mechanisms and Therapeutic Potential. Molecules (Basel, Switzerland) 2024, 29(22), 5471. [Google Scholar] [CrossRef]
- Cascio, M. G.; Gauson, L. A.; Stevenson, L. A.; Ross, R. A.; Pertwee, R. G. Evidence that the plant cannabinoid cannabigerol is a highly potent alpha2-adrenoceptor agonist and moderately potent 5HT1A receptor antagonist. British journal of pharmacology 2010, 159(1), 129–141. [Google Scholar] [CrossRef]
- Valdeolivas, S.; Navarrete, C.; Cantarero, I.; Bellido, M. L.; Muñoz, E.; Sagredo, O. Neuroprotective properties of cannabigerol in Huntington’s disease: studies in R6/2 mice and 3-nitropropionate-lesioned mice. Neurotherapeutics: the journal of the American Society for Experimental NeuroTherapeutics 2015, 12(1), 185–199. [Google Scholar] [CrossRef]
- Borrelli, F.; Fasolino, I.; Romano, B.; Capasso, R.; Maiello, F.; Coppola, D.; Orlando, P.; Battista, G.; Pagano, E.; Di Marzo, V.; Izzo, A. A. Beneficial effect of the non-psychotropic plant cannabinoid cannabigerol on experimental inflammatory bowel disease. Biochemical pharmacology 2013, 85(9), 1306–1316. [Google Scholar] [CrossRef]
- Hill, A. J.; Williams, C. M.; Whalley, B. J.; Stephens, G. J. Phytocannabinoids as novel therapeutic agents in CNS disorders. Pharmacology & therapeutics 2012, 133(1), 79–97. [Google Scholar] [CrossRef]
- Karler, R.; Cely, W.; Turkanis, S. A. The anticonvulsant activity of cannabidiol and cannabinol. Life sciences 1973, 13(11), 1527–1531. [Google Scholar] [CrossRef]
- Rhee, M. H.; Vogel, Z.; Barg, J.; Bayewitch, M.; Levy, R.; Hanus, L.; Breuer, A.; Mechoulam, R. Cannabinol derivatives: binding to cannabinoid receptors and inhibition of adenylylcyclase. Journal of medicinal chemistry 1997, 40(20), 3228–3233. [Google Scholar] [CrossRef]
- Ghovanloo, M. R.; Effraim, P. R.; Tyagi, S.; Zhao, P.; Dib-Hajj, S. D.; Waxman, S. G. Functionally-selective inhibition of threshold sodium currents and excitability in dorsal root ganglion neurons by cannabinol. Communications biology 2024, 7(1), 120. [Google Scholar] [CrossRef]
- Walsh, K. B.; McKinney, A. E.; Holmes, A. E. Minor Cannabinoids: Biosynthesis, Molecular Pharmacology and Potential Therapeutic Uses. Frontiers in pharmacology 2021, 12, 777804. [Google Scholar] [CrossRef] [PubMed]
- Ligresti, A.; Moriello, A. S.; Starowicz, K.; Matias, I.; Pisanti, S.; De Petrocellis, L.; Laezza, C.; Portella, G.; Bifulco, M.; Di Marzo, V. Antitumor activity of plant cannabinoids with emphasis on the effect of cannabidiol on human breast carcinoma. The Journal of pharmacology and experimental therapeutics 2006, 318(3), 1375–1387. [Google Scholar] [CrossRef] [PubMed]
- Udoh, M.; Santiago, M.; Devenish, S.; McGregor, I. S.; Connor, M. Cannabichromene is a cannabinoid CB2 receptor agonist. British journal of pharmacology 2019, 176(23), 4537–4547. [Google Scholar] [CrossRef] [PubMed]
- Sepulveda, D. E.; Vrana, K. E.; Kellogg, J. J.; Bisanz, J. E.; Desai, D.; Graziane, N. M.; Raup-Konsavage, W. M. The Potential of Cannabichromene (CBC) as a Therapeutic Agent. The Journal of pharmacology and experimental therapeutics 2024, 391(2), 206–213. [Google Scholar] [CrossRef]
- de Sousa, D. P.; Nóbrega, F. F.; Santos, C. C.; de Almeida, R. N. Anticonvulsant activity of the linalool enantiomers and racemate: investigation of chiral influence. Natural product communications 2010, 5(12), 1847–1851. [Google Scholar] [CrossRef]
- Koutroumanidou, E.; Kimbaris, A.; Kortsaris, A.; Bezirtzoglou, E.; Polissiou, M.; Charalabopoulos, K.; Pagonopoulou, O. Increased seizure latency and decreased severity of pentylenetetrazol-induced seizures in mice after essential oil administration. Epilepsy research and treatment 2013, 2013, 532657. [Google Scholar] [CrossRef]
- Guzmán-Gutiérrez, S. L.; Bonilla-Jaime, H.; Gómez-Cansino, R.; Reyes-Chilpa, R. Linalool and β-pinene exert their antidepressant-like activity through the monoaminergic pathway. Life sciences 2015, 128, 24–29. [Google Scholar] [CrossRef]
- Mallmann, M. P.; Mello, F. K.; Neuberger, B.; da Costa Sobral, K. G.; Fighera, M. R.; Royes, L. F. F.; Furian, A. F.; Oliveira, M. S. Beta-caryophyllene attenuates short-term recurrent seizure activity and blood-brain-barrier breakdown after pilocarpine-induced status epilepticus in rats. Brain research 2022, 1784, 147883. [Google Scholar] [CrossRef]
- Weston-Green, K.; Clunas, H.; Jimenez Naranjo, C. A Review of the Potential Use of Pinene and Linalool as Terpene-Based Medicines for Brain Health: Discovering Novel Therapeutics in the Flavours and Fragrances of Cannabis. Frontiers in psychiatry 2021, 12, 583211. [Google Scholar] [CrossRef]
- Zhang, L. L.; Yang, Z. Y.; Fan, G.; Ren, J. N.; Yin, K. J.; Pan, S. Y. Antidepressant-like Effect of Citrus sinensis (L.) Osbeck Essential Oil and Its Main Component Limonene on Mice. Journal of agricultural and food chemistry 2019, 67(50), 13817–13828. [Google Scholar] [CrossRef]
- Vieira, A. J.; Beserra, F. P.; Souza, M. C.; Totti, B. M.; Rozza, A. L. Limonene: Aroma of innovation in health and disease. Chemico-biological interactions 2018, 283, 97–106. [Google Scholar] [CrossRef]
- Surendran, S.; Qassadi, F.; Surendran, G.; Lilley, D.; Heinrich, M. Myrcene-What Are the Potential Health Benefits of This Flavouring and Aroma Agent? Frontiers in nutrition 2021, 8, 699666. [Google Scholar] [CrossRef] [PubMed]
- Perry, N. S.; Houghton, P. J.; Theobald, A.; Jenner, P.; Perry, E. K. In-vitro inhibition of human erythrocyte acetylcholinesterase by salvia lavandulaefolia essential oil and constituent terpenes. The Journal of pharmacy and pharmacology 2000, 52(7), 895–902. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Upadhyay, S.; Erdogan Orhan, I.; Kumar Jugran, A.; L D Jayaweera, S.; A Dias, D.; Sharopov, F.; Taheri, Y.; Martins, N.; Baghalpour, N.; Cho, W. C.; Sharifi-Rad, J. Therapeutic Potential of α- and β-Pinene: A Miracle Gift of Nature. Biomolecules 2019, 9(11), 738. [Google Scholar] [CrossRef] [PubMed]
- Russo, E. B. Taming THC: potential cannabis synergy and phytocannabinoid-terpenoid entourage effects. British journal of pharmacology 2011, 163(7), 1344–1364. [Google Scholar] [CrossRef]
- Elisabetsky, E.; Brum, L. F.; Souza, D. O. Anticonvulsant properties of linalool in glutamate-related seizure models. Phytomedicine: international journal of phytotherapy and phytopharmacology 1999, 6(2), 107–113. [Google Scholar] [CrossRef]
- El Alaoui, C.; Chemin, J.; Fechtali, T.; Lory, P. Modulation of T-type Ca2+ channels by Lavender and Rosemary extracts. PloS one 2017, 12(10), e0186864. [Google Scholar] [CrossRef]
- de Oliveira, C. C.; de Oliveira, C. V.; Grigoletto, J.; Ribeiro, L. R.; Funck, V. R.; Grauncke, A. C.; de Souza, T. L.; Souto, N. S.; Furian, A. F.; Menezes, I. R.; Oliveira, M. S. Anticonvulsant activity of β- caryophyllene against pentylenetetrazol-induced seizures. Epilepsy & behavior: E&B 2016, 56, 26–31. [Google Scholar] [CrossRef]
- Gertsch, J.; Leonti, M.; Raduner, S.; Racz, I.; Chen, J. Z.; Xie, X. Q.; Altmann, K. H.; Karsak, M.; Zimmer, A. Beta-caryophyllene is a dietary cannabinoid. Proceedings of the National Academy of Sciences of the United States of America 2008, 105(26), 9099–9104. [Google Scholar] [CrossRef]
- Yang, H.; Woo, J.; Pae, A. N.; Um, M. Y.; Cho, N. C.; Park, K. D.; Yoon, M.; Kim, J.; Lee, C. J.; Cho, S. α-Pinene, a Major Constituent of Pine Tree Oils, Enhances Non-Rapid Eye Movement Sleep in Mice through GABAA-benzodiazepine Receptors. Molecular pharmacology 2016, 90(5), 530–539. [Google Scholar] [CrossRef] [PubMed]
- Kessler, A.; Sahin-Nadeem, H.; Lummis, S. C.; Weigel, I.; Pischetsrieder, M.; Buettner, A.; Villmann, C. GABA(A) receptor modulation by terpenoids from Sideritis extracts. Molecular nutrition & food research 2014, 58(4), 851–862. [Google Scholar] [CrossRef]
- Poorthuis, R. B.; Enke, L.; Letzkus, J. J. Cholinergic circuit modulation through differential recruitment of neocortical interneuron types during behaviour. The Journal of physiology 2014, 592(19), 4155–4164. [Google Scholar] [CrossRef] [PubMed]
- Martinello, K.; Mascia, A.; Casciato, S.; Di Gennaro, G.; Esposito, V.; Zoli, M.; Gotti, C.; Fucile, S. α4β2* nicotinic acetylcholine receptors drive human temporal glutamate/GABA balance toward inhibition. The Journal of physiology 2025, 603(6), 1645–1662. [Google Scholar] [CrossRef]
- Takács, V. T.; Cserép, C.; Schlingloff, D.; Pósfai, B.; Szőnyi, A.; Sos, K. E.; Környei, Z.; Dénes, Á.; Gulyás, A. I.; Freund, T. F.; Nyiri, G. Co-transmission of acetylcholine and GABA regulates hippocampal states. Nature communications 2018, 9(1), 2848. [Google Scholar] [CrossRef]
- Vargas, D. L.; Nascimbene, C.; Krishnan, C.; Zimmerman, A. W.; Pardo, C. A. Neuroglial activation and neuroinflammation in the brain of patients with autism. Annals of neurology 2005, 57(1), 67–81. [Google Scholar] [CrossRef]
- Stellwagen, D.; Malenka, R. C. Synaptic scaling mediated by glial TNF-alpha. Nature 2006, 440(7087), 1054–1059. [Google Scholar] [CrossRef]
- Beattie, E. C.; Stellwagen, D.; Morishita, W.; Bresnahan, J. C.; Ha, B. K.; Von Zastrow, M.; Beattie, M. S.; Malenka, R. C. Control of synaptic strength by glial TNFalpha. Science (New York, N.Y.) 2002, 295(5563), 2282–2285. [Google Scholar] [CrossRef]
- Yirmiya, R.; Goshen, I. Immune modulation of learning, memory, neural plasticity and neurogenesis. Brain, behavior, and immunity 2011, 25(2), 181–213. [Google Scholar] [CrossRef]
- Riazi, K.; Galic, M. A.; Kentner, A. C.; Reid, A. Y.; Sharkey, K. A.; Pittman, Q. J. Microglia-dependent alteration of glutamatergic synaptic transmission and plasticity in the hippocampus during peripheral inflammation. The Journal of neuroscience: the official journal of the Society for Neuroscience 2015, 35(12), 4942–4952. [Google Scholar] [CrossRef]
- Mahan, A. L.; Ressler, K. J. Fear conditioning, synaptic plasticity and the amygdala: implications for posttraumatic stress disorder. Trends in neurosciences 2012, 35(1), 24–35. [Google Scholar] [CrossRef] [PubMed]
- Suvrathan, A.; Bennur, S.; Ghosh, S.; Tomar, A.; Anilkumar, S.; Chattarji, S. Stress enhances fear by forming new synapses with greater capacity for long-term potentiation in the amygdala. Philosophical transactions of the Royal Society of London. Series B, Biological sciences 2013, 369(1633), 20130151. [Google Scholar] [CrossRef] [PubMed]
- Rauch, S. L.; Shin, L. M.; Phelps, E. A. Neurocircuitry models of posttraumatic stress disorder and extinction: human neuroimaging research--past, present, and future. Biological psychiatry 2006, 60(4), 376–382. [Google Scholar] [CrossRef] [PubMed]
- Roozendaal, B.; McEwen, B. S.; Chattarji, S. Stress, memory and the amygdala. Nature reviews. Neuroscience 2009, 10(6), 423–433. [Google Scholar] [CrossRef]
- Lamanna, J.; Ferro, M.; Spadini, S.; Racchetti, G.; Malgaroli, A. The Dysfunctional Mechanisms Throwing Tics: Structural and Functional Changes in Tourette Syndrome. Behavioral sciences (Basel, Switzerland) 2023, 13(8), 668. [Google Scholar] [CrossRef]
- Brandt, V. C.; Niessen, E.; Ganos, C.; Kahl, U.; Bäumer, T.; Münchau, A. Altered synaptic plasticity in Tourette’s syndrome and its relationship to motor skill learning. PloS one 2014, 9(5), e98417. [Google Scholar] [CrossRef]
- Ahmari, S. E.; Spellman, T.; Douglass, N. L.; Kheirbek, M. A.; Simpson, H. B.; Deisseroth, K.; Gordon, J. A.; Hen, R. Repeated cortico-striatal stimulation generates persistent OCD-like behavior. Science (New York, N.Y.) 2013, 340(6137), 1234–1239. [Google Scholar] [CrossRef]
- Gillan, C. M.; Robbins, T. W. Goal-directed learning and obsessive-compulsive disorder. Philosophical transactions of the Royal Society of London. Series B, Biological sciences 2014, 369(1655), 20130475. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



