Submitted:
03 April 2026
Posted:
07 April 2026
You are already at the latest version
Abstract
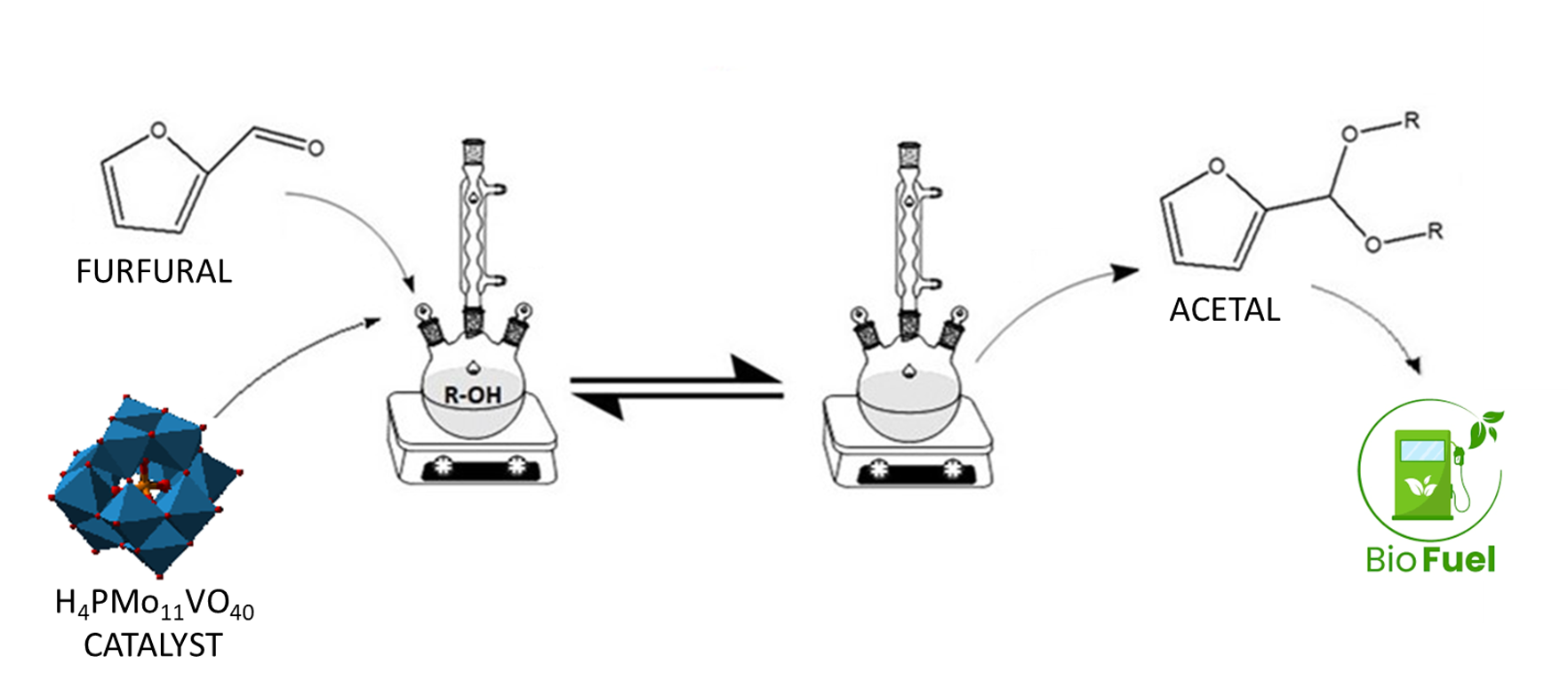
Keywords:
1. Introduction
2. Materials and Methods
2.1. Synthesis of the H4PMo11VO40
2.2. Synthesis of H5PMo10V2O40
2.3. Synthesis of H6PMo9V3O40
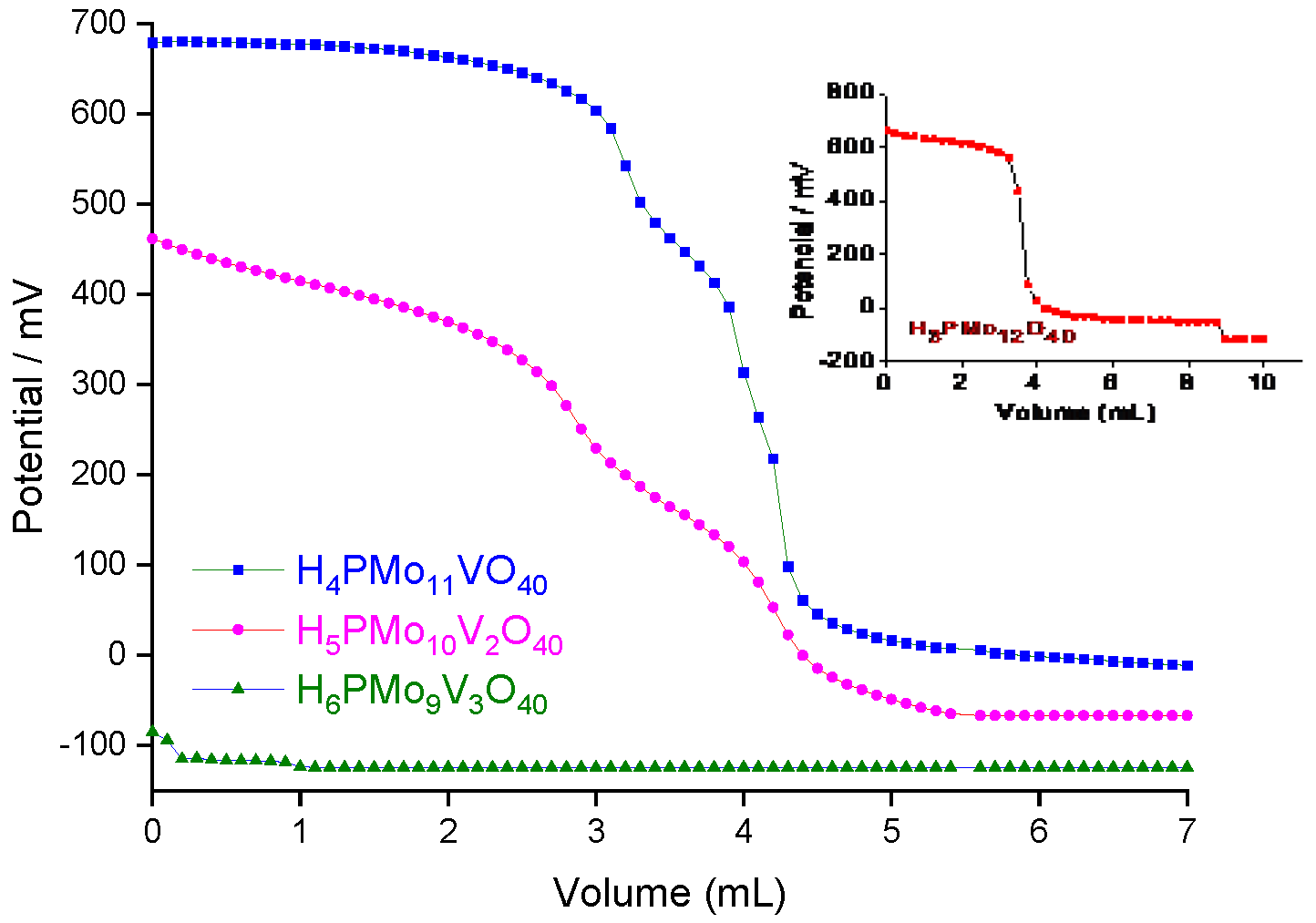
2.4. Characterization of the Catalysts
2.5. Identification of The Main Reaction Products
2.6. Catalytic Tests
3. Results and Discussion
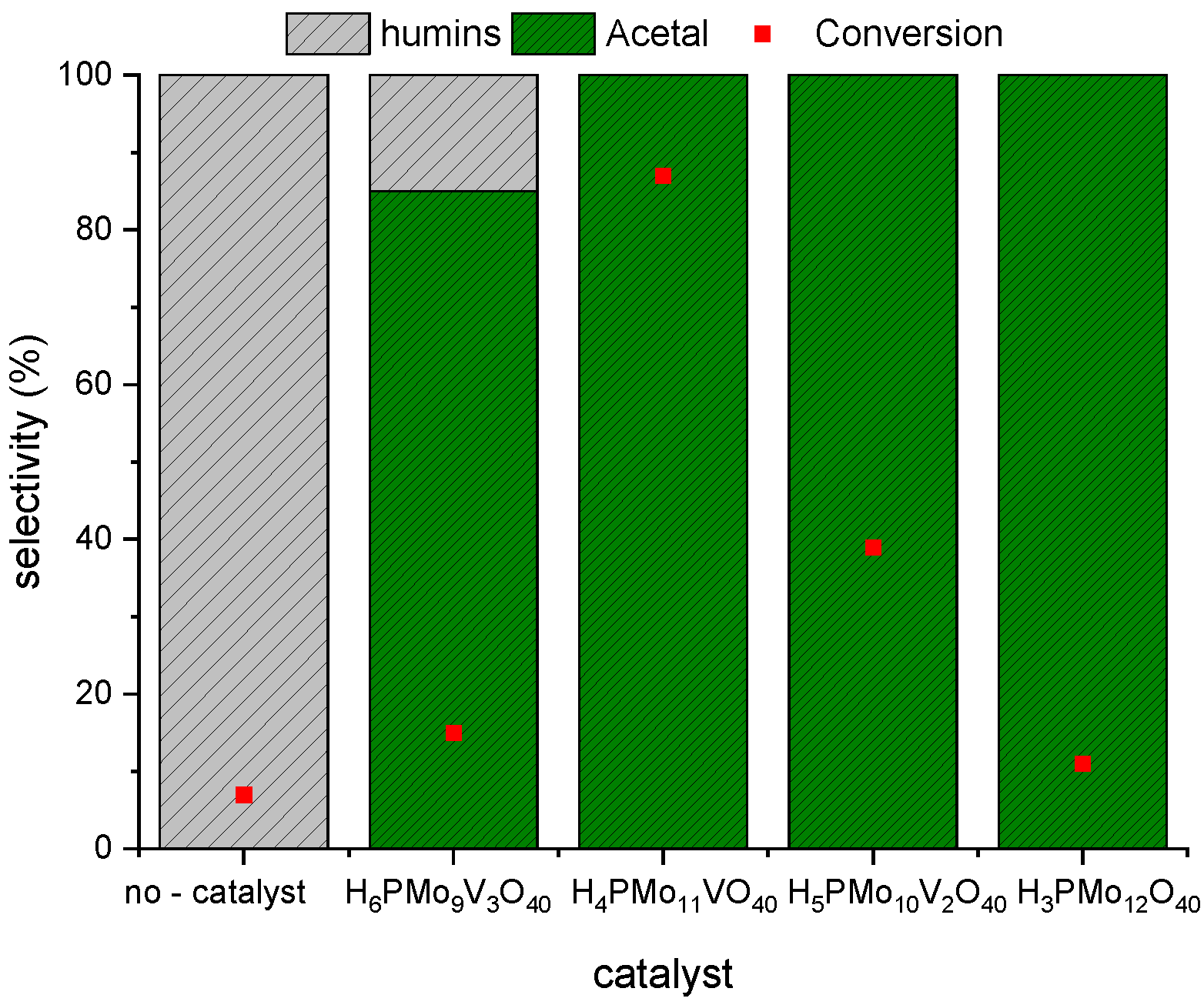
3.1. Effect of Vanadium Doping on the Conversion and Selectivity of the Furfural Acetalization Reaction with Methyl Alcohol Catalyzed by Phosphomolybdic Acid

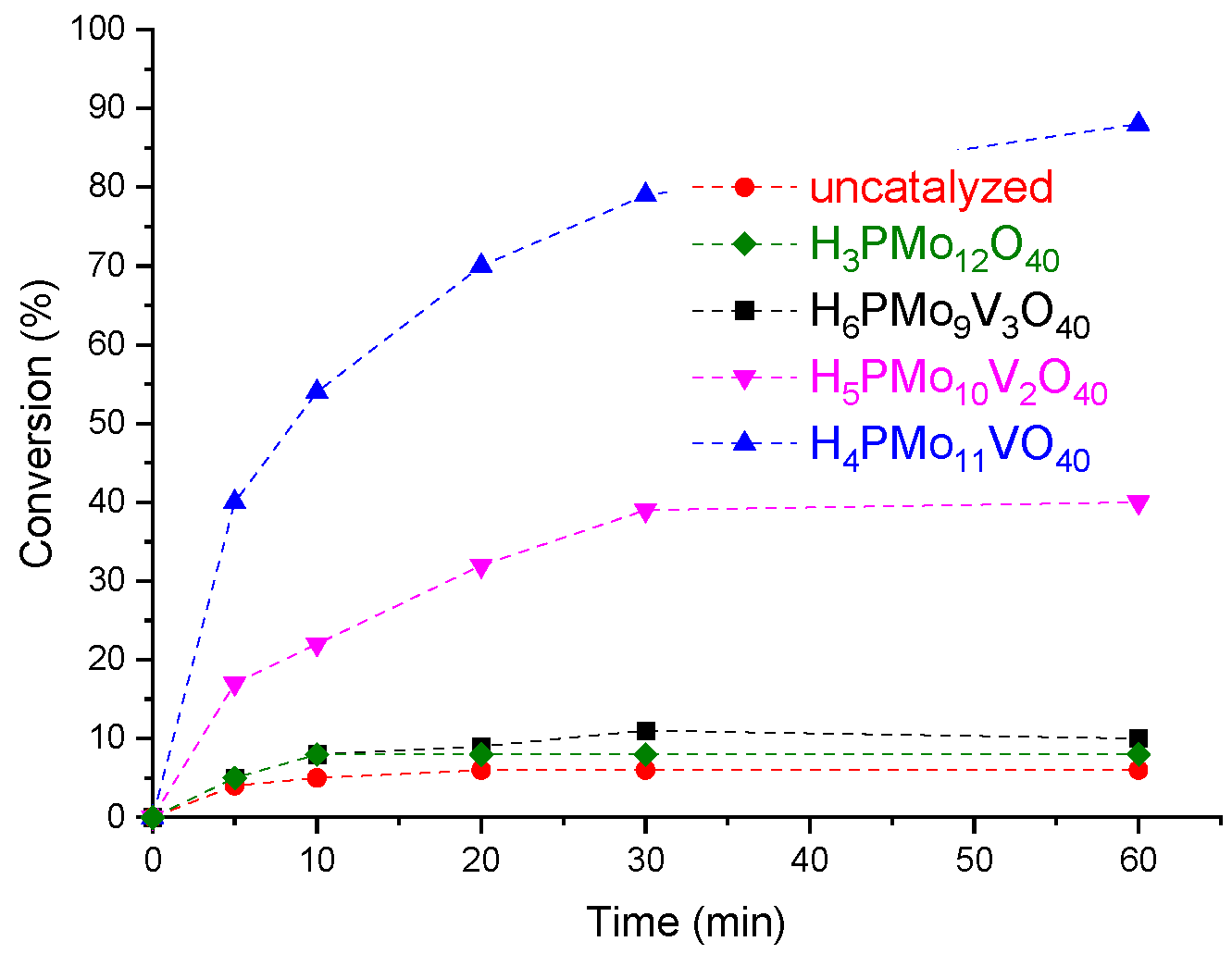
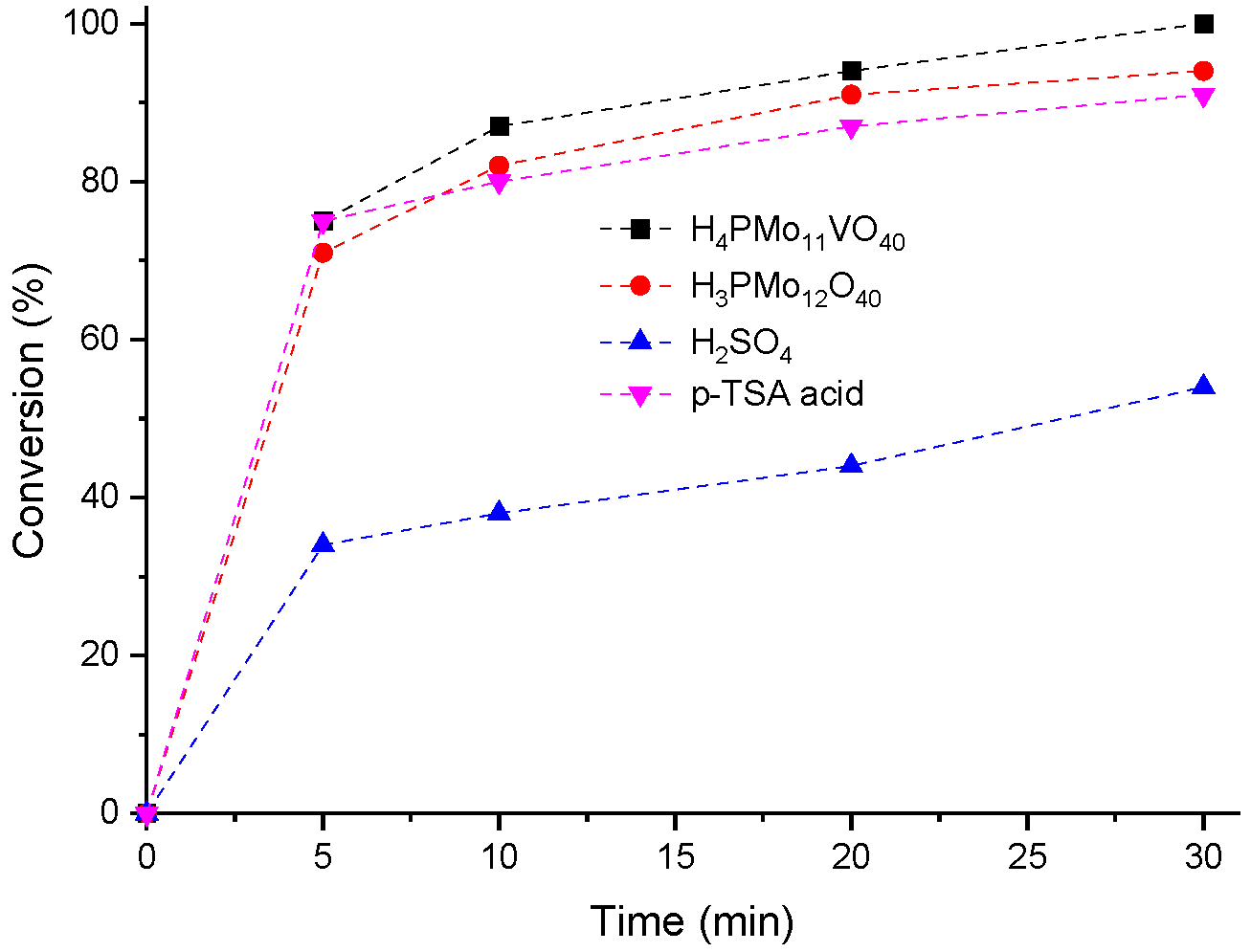
3.2. Assessment the Activity of other Brønsted acid Catalysts
3.3. Impacts of H4PMo11VO40 Catalyst Load on The Reaction Conversions of Condensation of Furfural with Methyl Alcohol
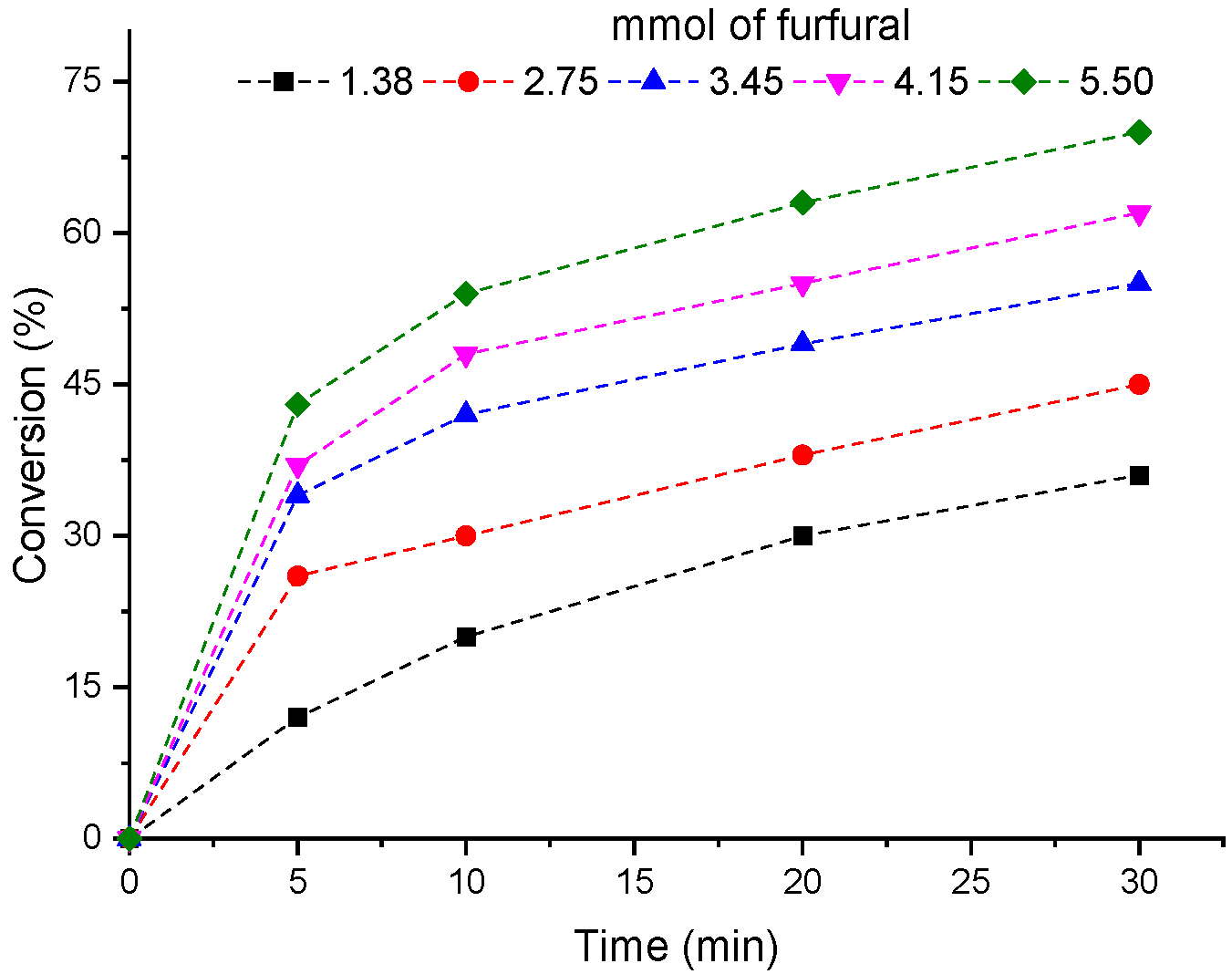
3.4. Influence of Furfural Load on the H4PMo11VO40-Catalyzed Condensation Reactions With Methyl Alcohol
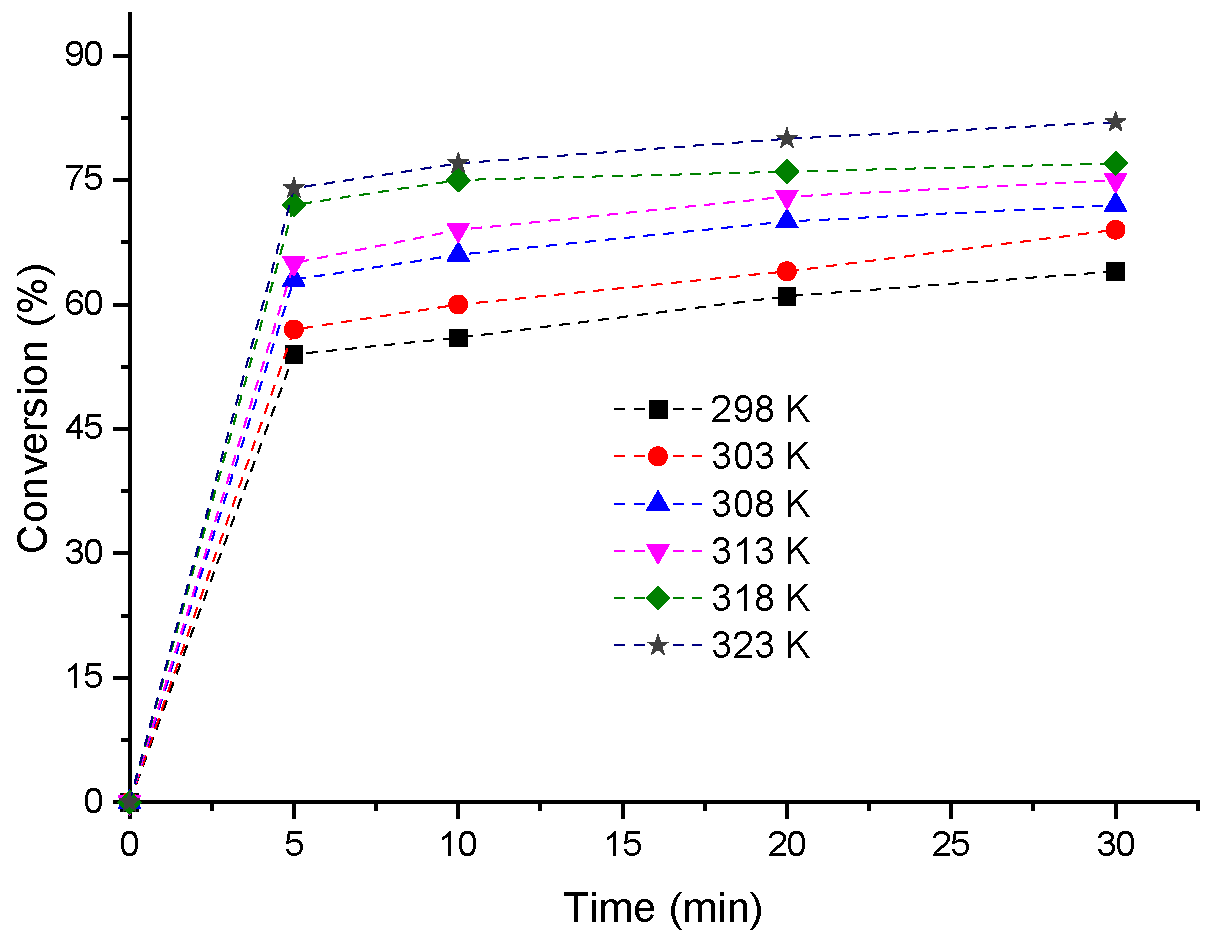
3.5. Effects of Temperature on the H4PMo11VO40-Catalyzed Condensation Reactions of Furfural with Methyl Alcohol
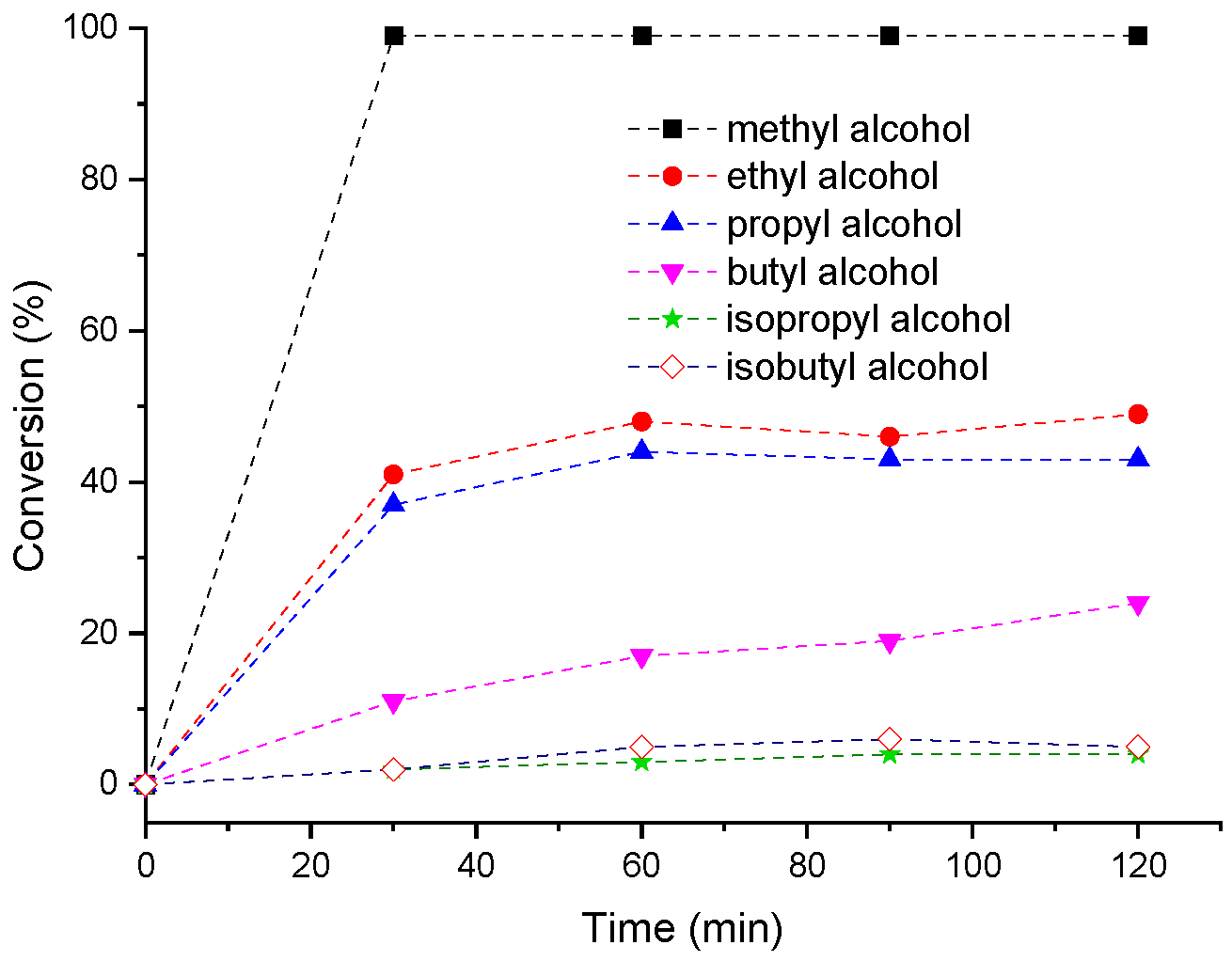
3.6. Effects of Alkyl Alcohol on the H4PMo11VO40-Catalyzed Condensation Reactions of Furfural
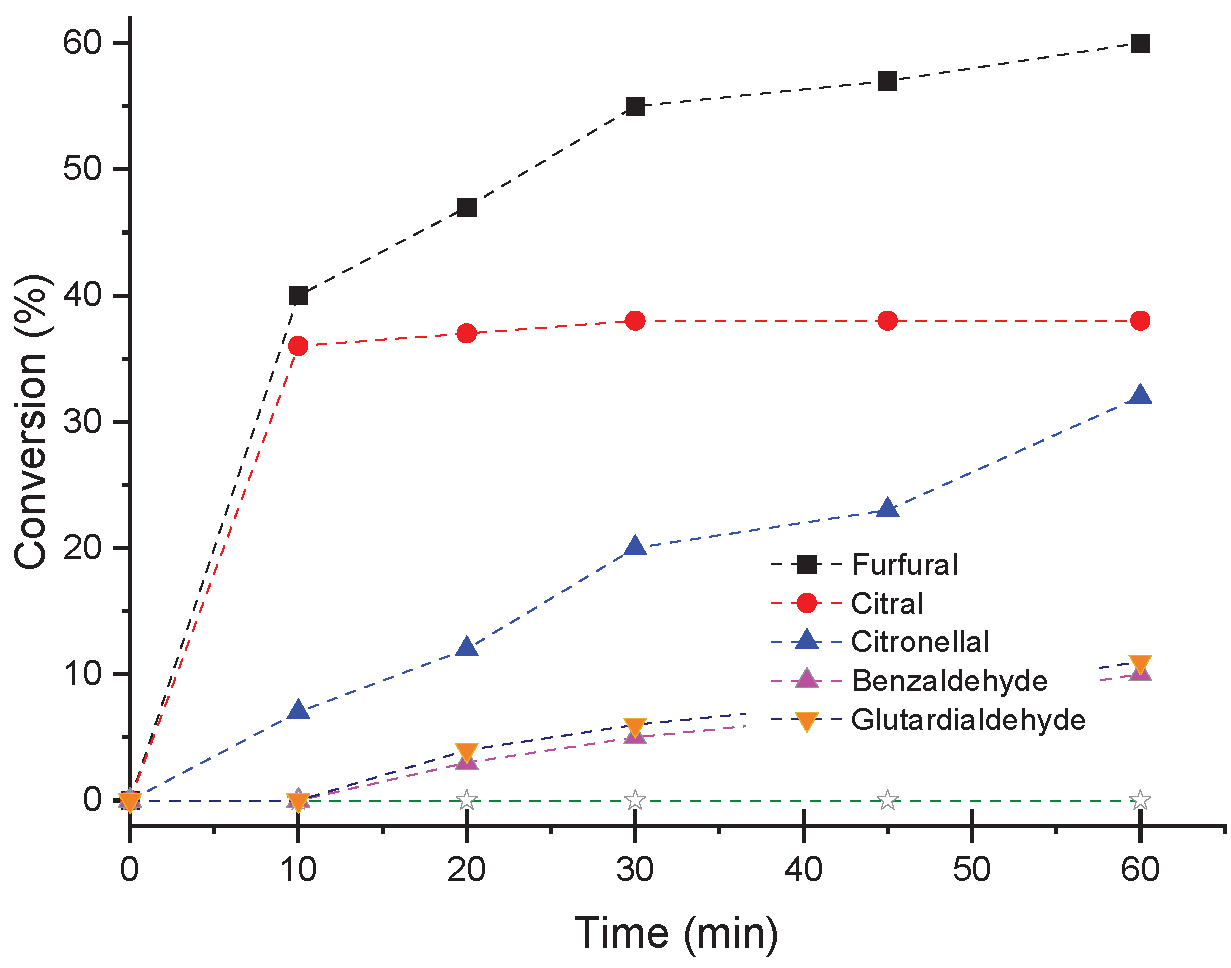
3.7. Influence of Aldehyde on the H4PMo11VO40-Catalyzed Condensation Reactions with Methyl Alcohol
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Malode, S. J.; Prabhu, K. P.; Mascarenhas, R. J.; Shetti, N. P.; Aminabhavi, T. M. Recent advances and viability in biofuel production. Energy Convers. Manag., 2020, 10,100070-. [CrossRef]
- Agarwal, A.; Rana, M.; Park, J-H. Advancement in technologies for the depolymerization of lignin, Fuel Process. Technol., 2018, 181, 115-132. [CrossRef]
- Liguori, F.; Moreno-Marrodan, C.; Barbaro, P. Biomass-derived chemical substitutes for bisphenol A: Recent advancements in catalytic synthesis. Chem. Soc. Rev., 2020, 49, 6329-6363. [CrossRef]
- Katahira, R.; Mittal, A.; Mckinney, K.; Chen, X.; Tucker, M. P.; Johnson, D. K.; Beckham, G. T. Base-Catalyzed Depolymerization of Biorefinery Lignins. ACS Sustainable Chem. Eng., 2016, 3, 1474-1486. [CrossRef]
- Xu, C.; Paone, E.; Rodríguez-Padrón, D.; Lugue, R.; Mauriello, F. Recent catalytic routes for the preparation and the upgrading of biomass derived furfural and 5-hydroxymethylfurfural. Chem. Soc. Rev., 2020, 49, 4273-4306. [CrossRef]
- Aldureid, A.; Montané, D.; Liorca, J.; Medina, F. Ni-Mg/Al Mixed Oxides Prepared from Layered Double Hydroxides as Catalysts for the Conversion of Furfural to Tetrahydrofurfuryl Alcohol. Chem., 2023, 5, 571-588. [CrossRef]
- Hoang, A. T.; Pham, V. V.; 2-Methylfuran (MF) as a potential biofuel: A thorough review on the production pathway from biomass, combustion progress, and application in engines. Renew. Sust. Energy Rev., 2021, 148, 111265. [CrossRef]
- Zhao, Y.; Lu, K.; Xu, H.; Zhu, L.; Wang, S. A critical review of recent advances in the production of furfural and 5-hydroxymethylfurfural from lignocellulosic biomass through homogeneous catalytic hydrothermal conversion. Renew. Sust. Energy Rev., 2021, 139, 110706. [CrossRef]
- da Silva, M.J., Lopes, N.P.G.; Bruziquesi, C.G.O. Furfural acetalization over Keggin heteropolyacid salts at room temperature: Effect of cesium doping. Reac. Kinet. Mech. Cat., 2021, 133, 913-931. [CrossRef]
- Rubio-Cabellera, J. M.; Saravanamurugan, S.; Maireles-Torres, P.; Riisager, A. Acetalization of furfural with zeolites under benign reaction conditions. Catal Today, 2014, 234, 233-236. [CrossRef]
- da Silva, M. J.; Chaves, D. M.; Júlio, A. A.; Rodrigues, F. A.; Bruziquesi, C. G. Sn(II)-Exchanged Keggin Silicotungstic Acid-Catalyzed Etherification of Glycerol and Ethylene Glycol with Alkyl Alcohols. Ind. Eng. Chem. Res., 2020, 59, 9858-9868. [CrossRef]
- Raut, S. U.; Bhagat, P. R. Efficient photocatalytic acetalization of furfural to biofuel components using carboxyl-functionalized porphyrin photocatalyst, under visible light irradiations. Biomass Conv. Bioref., 2021. [CrossRef]
- Morone, A.; Apte, M.; Pandey, R.A. Levulinic acid production from renewable waste resources: Bottlenecks, potential remedies, advancements and applications. Renew Sustain Energy Rev 2015, 51, 548–65.
- Ratthiwal, J.; Lazaro, N.; Reyes, A. A. R.; Osman, S. M.; Reubroycharoen, P.; Luque, R. Batch and continuous-flow room temperature furfural acetalization with ethanol over aluminophosphate (APAl) catalysts for biofuels production. Fuel, 2023, 332, 126049. [CrossRef]
- Appaturi, J. N.; Ramalingam, R. J.; Al-Lohedan, H. A.; Khoerunnisa, F.; Ling, T. C.; Ng, E-P. Selective synthesis of dioxolane biofuel additive via acetalization of glycerol and furfural enhanced by MCM-41-alanine bifunctional catalyst. Fuel, 2021, 288, 119573. [CrossRef]
- Teixeira, M.G.; Natalino, R.; da Silva, M. J. A kinetic study of heteropolyacid-catalyzed furfural acetalization with methanol at room temperature via ultraviolet spectroscopy. Catal. Today, 2020, 344, 143-149. [CrossRef]
- Silveira, C. C.; Mendes, S. R.; Ziembowicz, F. I.; Lenardão, E. J.; Perin, G. The use of anhydrous CeCl3 as a recyclable and selective catalyst for the acetalization of aldehydes and ketones. J. Braz. Chem. Soc. 2010, 21, 371-374.
- Kanai, S.; Nagahara, I.; Kita, Y.; Kamata, K.; Hara, M. A bifunctional cerium phosphate catalyst for chemoselective acetalization. Chem. Sci. 2017, 8, 3146-3153. [CrossRef]
- da Silva, M.J.; Teixeira, M.G.; Natalino, R., Highly selective synthesis under benign reaction conditions of furfural dialkyl acetal using SnCl2 as a recyclable catalyst, N. J. Chem. 2019, 43, 8606-8612. [CrossRef]
- Song, H.; Jin, F.; Liu, Q.; Liu, H. Zeolite-catalyzed acetalization reaction of furfural with alcohol under solvent-free conditions. Mol. Catal., 2021, 513, 111752. [CrossRef]
- da Silva, M.J.; Chaves, D. M.; Ferreira, S. O.; da Silva, R. C.; Filho, J. B. G.; Bruziquesi, C. G. O.; Al-Rabiah, A. A. Impacts of Sn(II) doping on the Keggin heteropolyacid-catalyzed etherification of glycerol with tert-butyl alcohol, Chem. Eng. Sci., 2022, 247, 116913. [CrossRef]
- da Silva, M.J.; Julio, A.A.; Ferreira, S.O.; da Silva, R.C.; Chaves, D.M., Tin(II) phosphotungstate heteropoly salt: An efficient solid catalyst to synthesize bioadditives ethers from glycerol, Fuel, 2019, 254, 115606-115617. [CrossRef]
- da Silva, M. J.; Temóteo, R.L. Trivalent metal cation exchanged-phosphotungstic acid salts: Efficient catalysts for the oxidation of geraniol with hydrogen peroxide. Catal. Lett., 2026 156, 84 – 98.
- Ribeiro, C. J. A.; Pereira, M. M.; Kozhevnikova, E. F.; Kozhevnikov, I. V.; Gusevskaya, E. V.; Kelly Rocha, K. A. S. Heteropoly acid catalysts in upgrading of biorenewables: Synthesis of para-menthenic fragrance compounds from α-pinene oxide, Catal. Today, 2020, 344, 166-170. [CrossRef]
- da Silva, M. J.; Teixeira, M. G. Assessment on the double role of the transition metal salts on the acetalization of furfural: Lewis and Brønsted acid catalysts. Mol. Catal., 2018, 40-47. [CrossRef]
- Vilanculo, C. B.; da Silva, M.J. Na3+xPMo12-xVxO40-catalyzed one-pot oxidative esterification of benzaldehyde with hydrogen peroxide. RSC Adv., 2021, 11, 34979 – 34987.
- Fendg. C.; Li, Q.; Wu, P.; Liu, B.; Hu, H.; Xue, G. Phosphotungstic Acid Supported on Magnetic Mesoporous Tantalum Pentoxide Microspheres: Efficient Heterogeneous Catalysts for Acetalization of Benzaldehyde with Ethylene Glycol. Catal. Lett., 2020, 150, 1204-1217. [CrossRef]
- Castanheiro, J. E.; Vital, J.; Fonseca, I. M.; Ramos, A. M. Glycerol conversion into biofuel additives by acetalization with pentanal over heteropolyacids immobilized on zeolites. Catal. Today, 2020, 346, 76-80. [CrossRef]
- da Silva, M. J.; Lopes, N. P. G.; Rodrigues, A. A. Biodiesel Additives Synthesis Using Solid Heteropolyacid Catalysts. Energies 2023, 16, 1332. [CrossRef]
- da Silva, M. J.; Rodrigues, A. A.; Lopes, N. P. G. Cesium Heteropolyacid Salts: Synthesis, Characterization and Activity of the Solid and Versatile Heterogeneous Catalysts. Chemistry 2023, 5, 662-690. [CrossRef]
- da Silva, M. J.; Liberto N. A., Soluble and Solid Supported Keggin Heteropolyacids as Catalysts in Reactions for Biodiesel Production: Challenges and Recent Advances, Curr. Org. Chem., 2016, 20 (12), 2016,1263 – 1283. [CrossRef]
- Vilanculo, C.B.; da Silva, M.J.; Teixeira, M.G.; Villarreal, J.A. One-pot synthesis at room temperature of epoxides and linalool derivative pyrans in monolacunary Na7PW11O39-catalyzed oxidation reactions by hydrogen peroxide, RSC Adv., 2020, 10, 7691–7697. [CrossRef]
- da Silva, M.J.; Andrade,P.H.S.; Sampaio, V.F.C.; Transition metal-substituted potassium silicotungstate salts as catalysts for oxidation of terpene alcohols with hydrogen peroxide, Catal. Lett., 2021, 151, 2094–2106 . [CrossRef]
- Rodrigues, A.A.; da Silva, M.J.; Ferreira, S.O.; da Silva, R.C.; Silva, T.A.; de Araújo, E.N.D.; Assessment of the metal exchanged phosphomolybdic acid salt-catalyzed nerol oxidation reactions with hydrogen peroxide, Mol. Catal. 2023, 545 113221-113235. [CrossRef]
- da Silva, M. J.; Ribeiro, C. J. A.; Rodrigues, A. A. H4PMo11VO40-catalyzed β-citronellal condensation reactions. Catal. Lett. 2023. [CrossRef]
- Vilanculo, C.B.; da Silva, M.J; Rodrigues, A.A.; Ferreira, S.O.; da Silva, R.C.; Vanadium-doped sodium phosphomolybdate salts as catalysts in the terpene alcohols oxidation with hydrogen peroxide, RSC Adv., 2021, 11, 24072–24085. [CrossRef]
- Barteau, K. P.; Lyons, J. E.; Song, I. K.; Barteau, M. A. UV–visible spectroscopy as a probe of heteropolyacid redox properties: Application to liquid phase oxidations. Top. Catal., 2006, 41, 55-62. [CrossRef]
- Vilanculo, C. B.; da Silva, M.J. How the content of protons and vanadium affects the activity of H3+nPMo12-nVnO40 (n = 0, 1, 2, or 3) catalysts on the oxidative esterification of benzaldehyde with hydrogen peroxide. Catal. Lett., 2022, 153, 2045–2056. [CrossRef]
- Jing F.; Katryniok B.; Dumeignil F.; Bordes-Richard E. Catalytic selective oxidation of isobutane to methacrylic acid on supported (NH4)3HPMo11VO40 catalysts. J Catal. 2014, 309, 121–135.
- Tsigdinos, G. A., Hallada, C. Molybdovanadophosphoric acids and their salts. I. Investigation of methods of preparation and characterization. J. Inorg. Chem., 1968, 7, 437-441. [CrossRef]
- Vilanculo, C. B.; da Silva, M. J. Na4PMo11VO40-Catalyzed one-pot oxidative esterification of benzaldehyde with hydrogen peroxide. RSC Adv., 2021, 11, 34979-34987. [CrossRef]
- Rao, K. T.V.; Rao, P. S. N.; Nagaraju, P.; Prasad, P. S. S.; Lingaiah, N., Room temperature selective oxidation of toluene over vanadium substituted polyoxometalate catalysts, J. Mol. Catal., A, 2009, 303, 84-89. [CrossRef]
- Timofeeva, M.N., Maksimov, G.M. & Likholobov, V.A. Acidity of solutions of heteropoly acids with various structures and compositions. Kinet. Catal., 2001, 42, 30-34. [CrossRef]
- Serwicka, E.M., Bruckman, K., Haber, J., Paukshtis, E.A., Yurchenko, E.N. Appl Catal A, 1991, 73:153–163. [CrossRef]
- Villabrille, P., Romanelli, G., Vázquez, P., Cáceres. C., Appl Catal A 2004, A 270:101–111. [CrossRef]
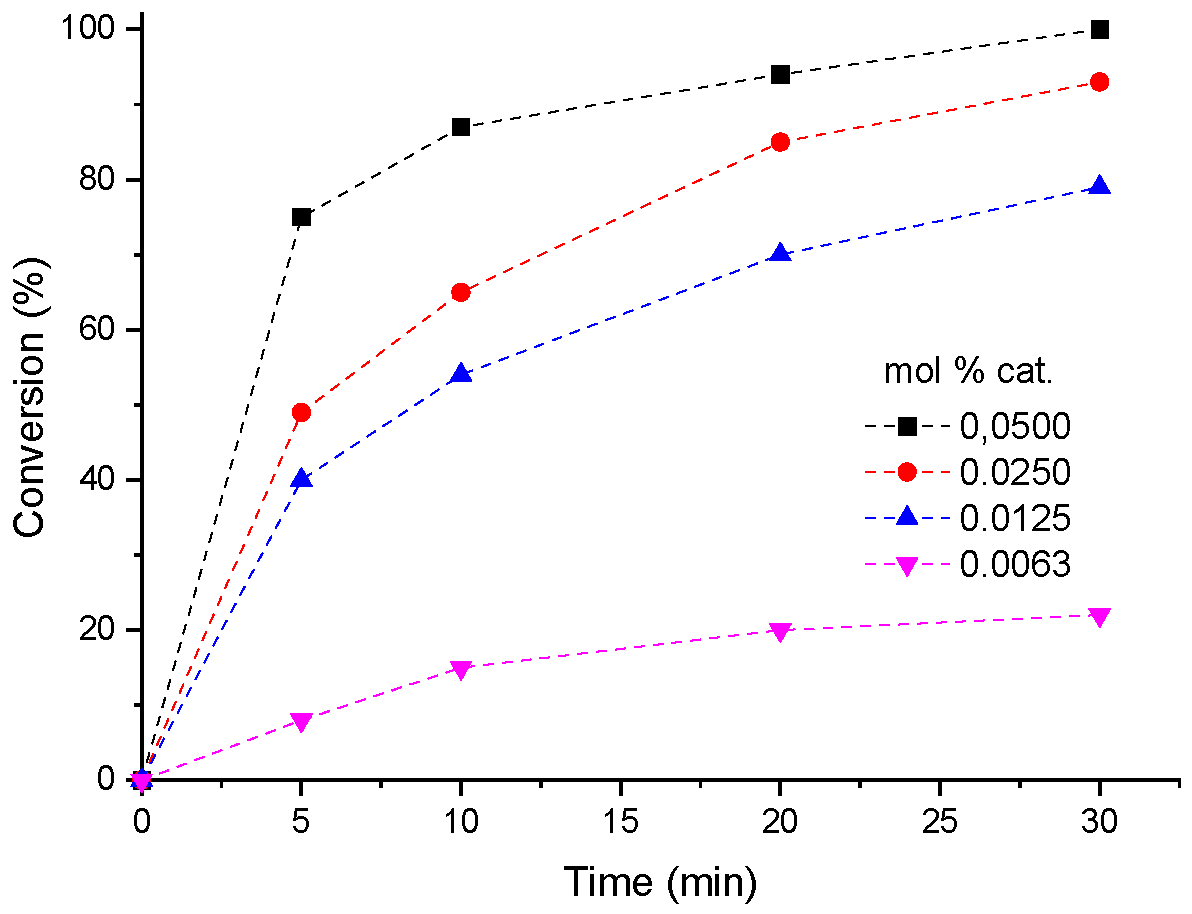
| Entry | Load (mol %) | mmol catalyst x 103- | converted mmol furfural |
TON |
|---|---|---|---|---|
| 1 | 0.0500 | 2.750 x 10-3 | 5.50 | 2014 |
| 2 | 0.0250 | 1.375 x 10-3 | 5.06 | 3680 |
| 3 | 0.0125 | 6.875x 10-4 | 4.40 | 6400 |
| 4 | 0.0063 | 3.437x 10-4 | 1.37 | 4000 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




