Submitted:
03 April 2026
Posted:
07 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Training Program
2.3. Cardiorespiratory Fitness Assessment
2.4. Push-Up Test
2.5. Muscular Performance Assessment
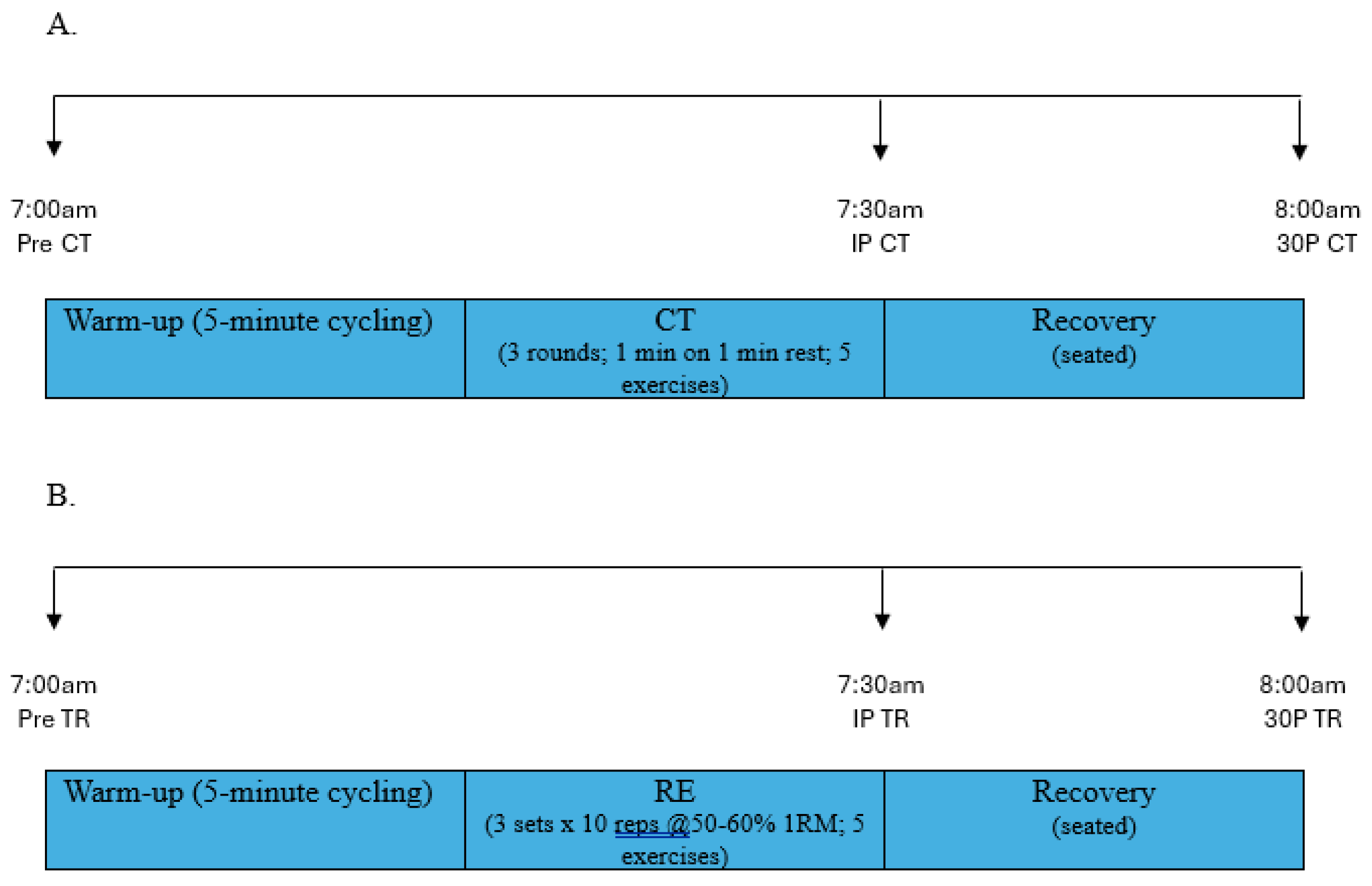
2.6. Exercise Protocol
2.7. Blood Sampling and Biochemical Assays
2.8. Statistical Analyses
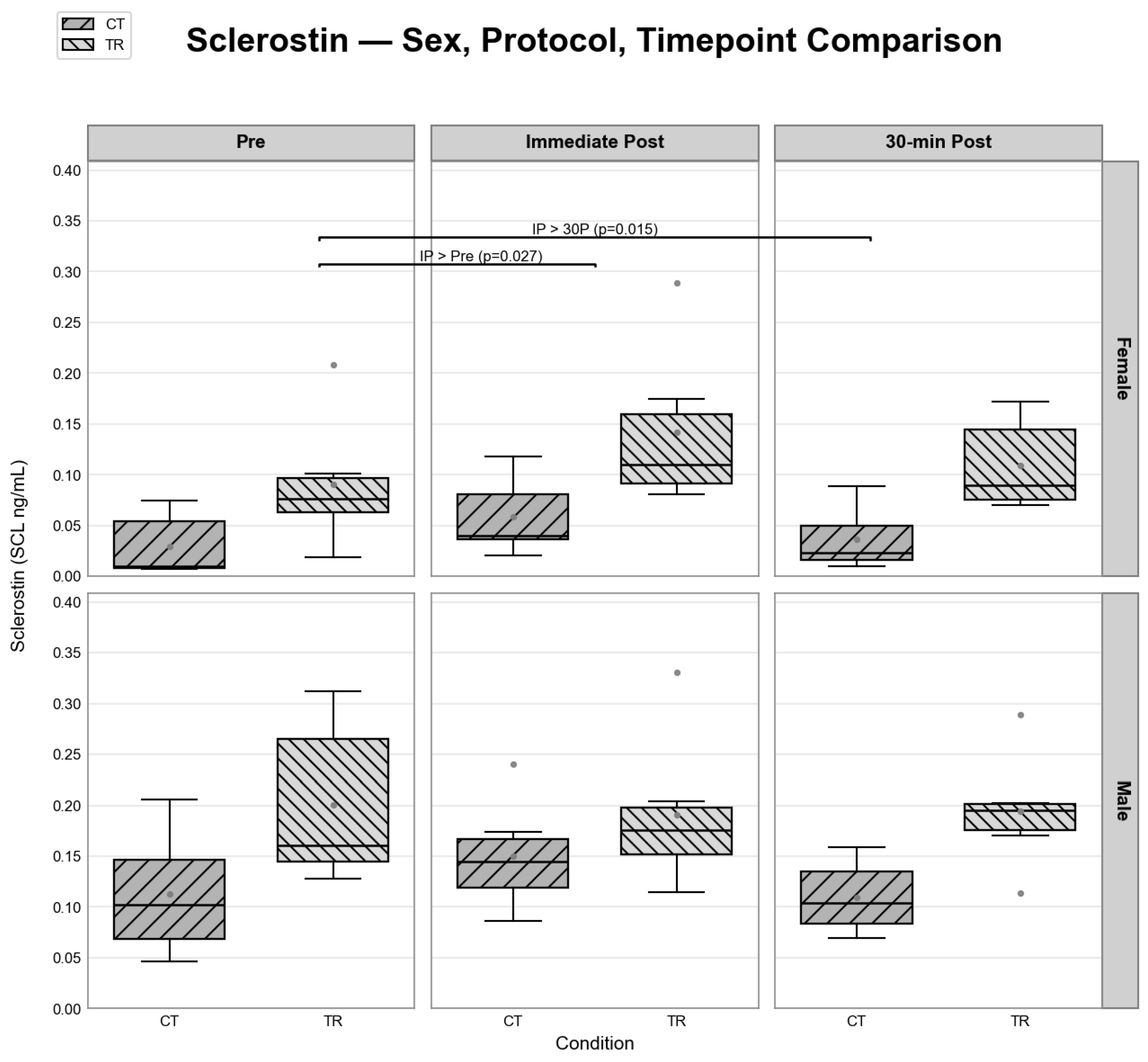
3. Results
3.1. Participant Characteristics, Body Composition Variables, and Cardiorespiratory Fitness Assessment
3.2. Lower and Upper Body Muscle Performance Assessment
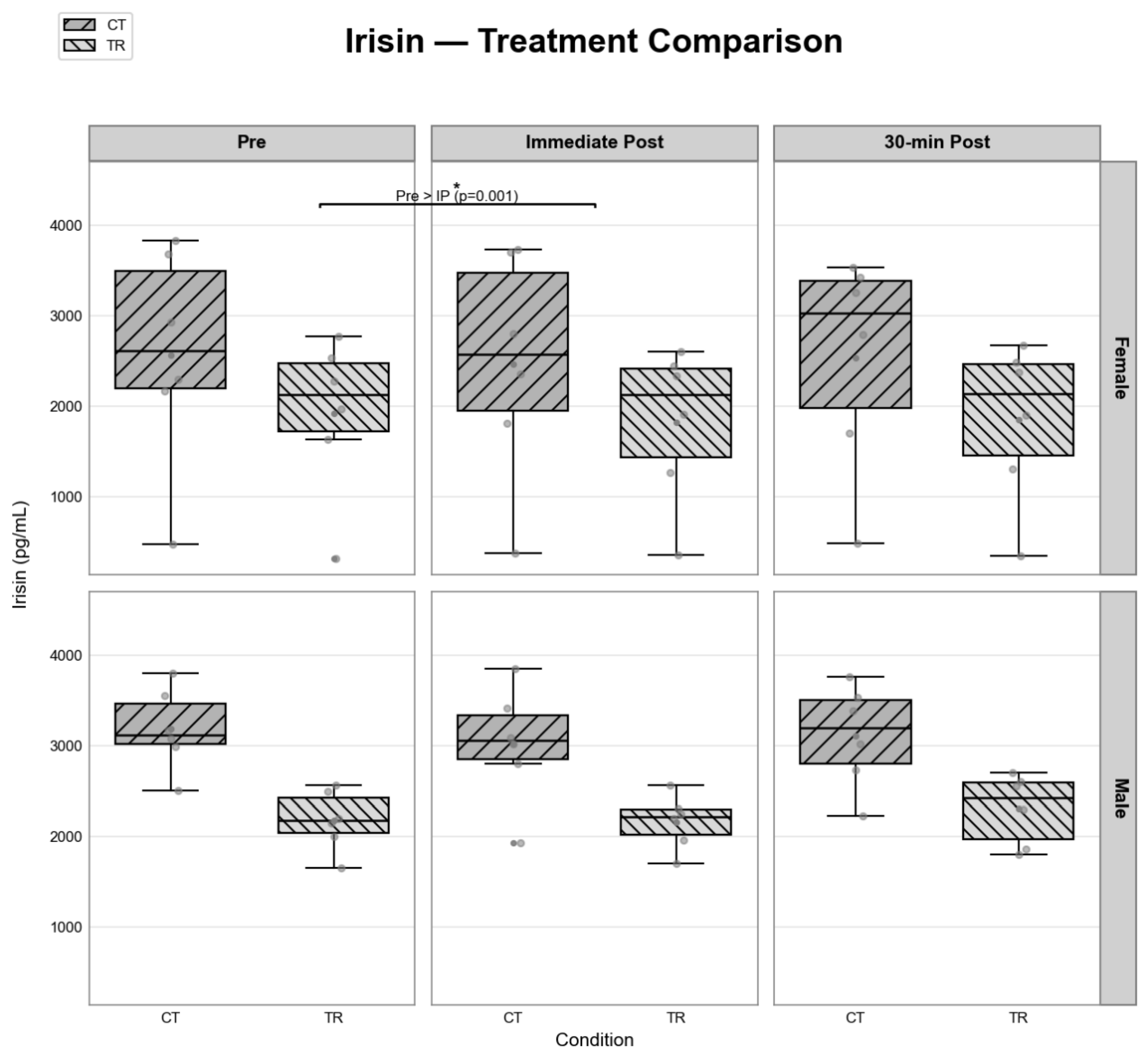
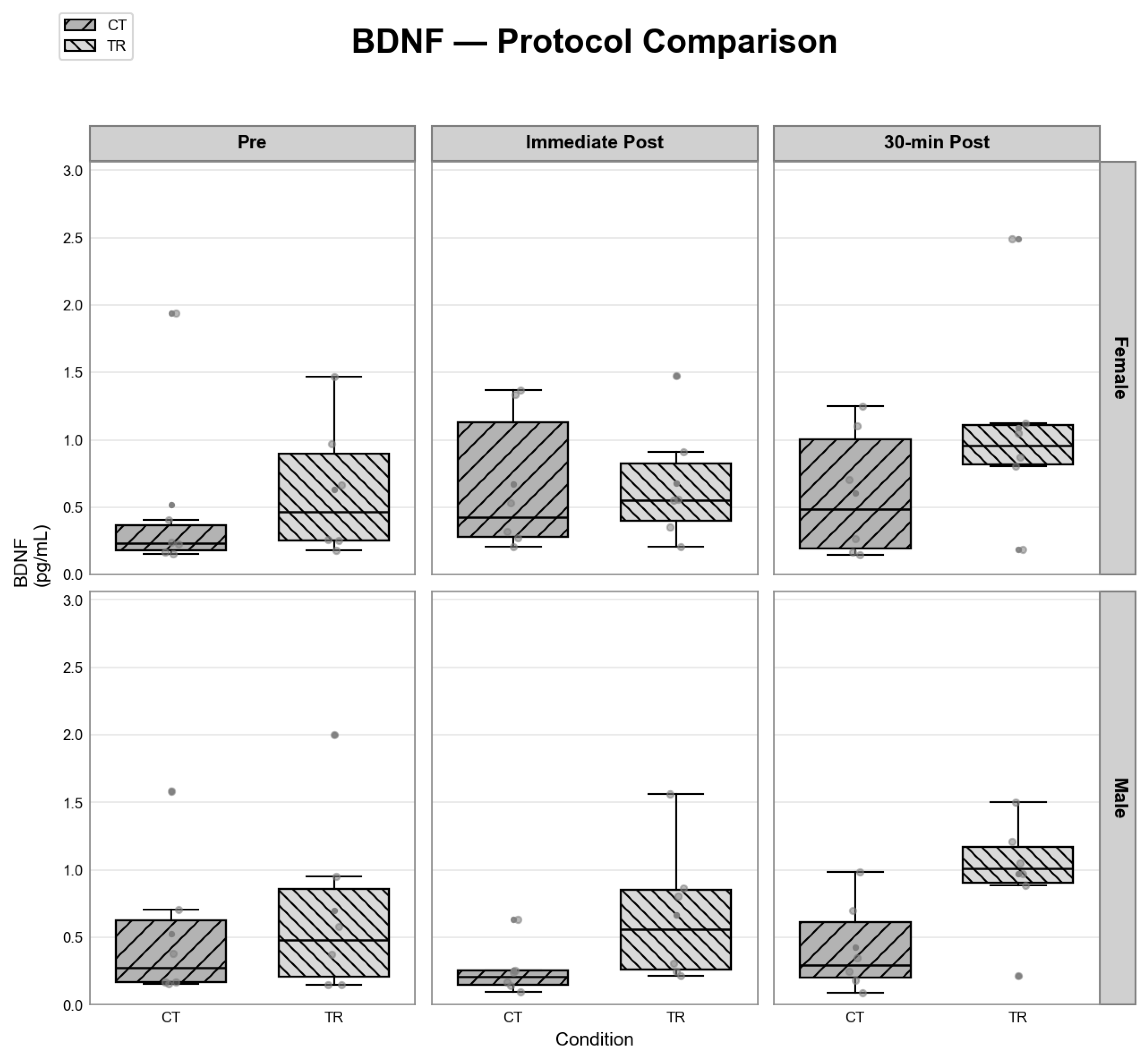
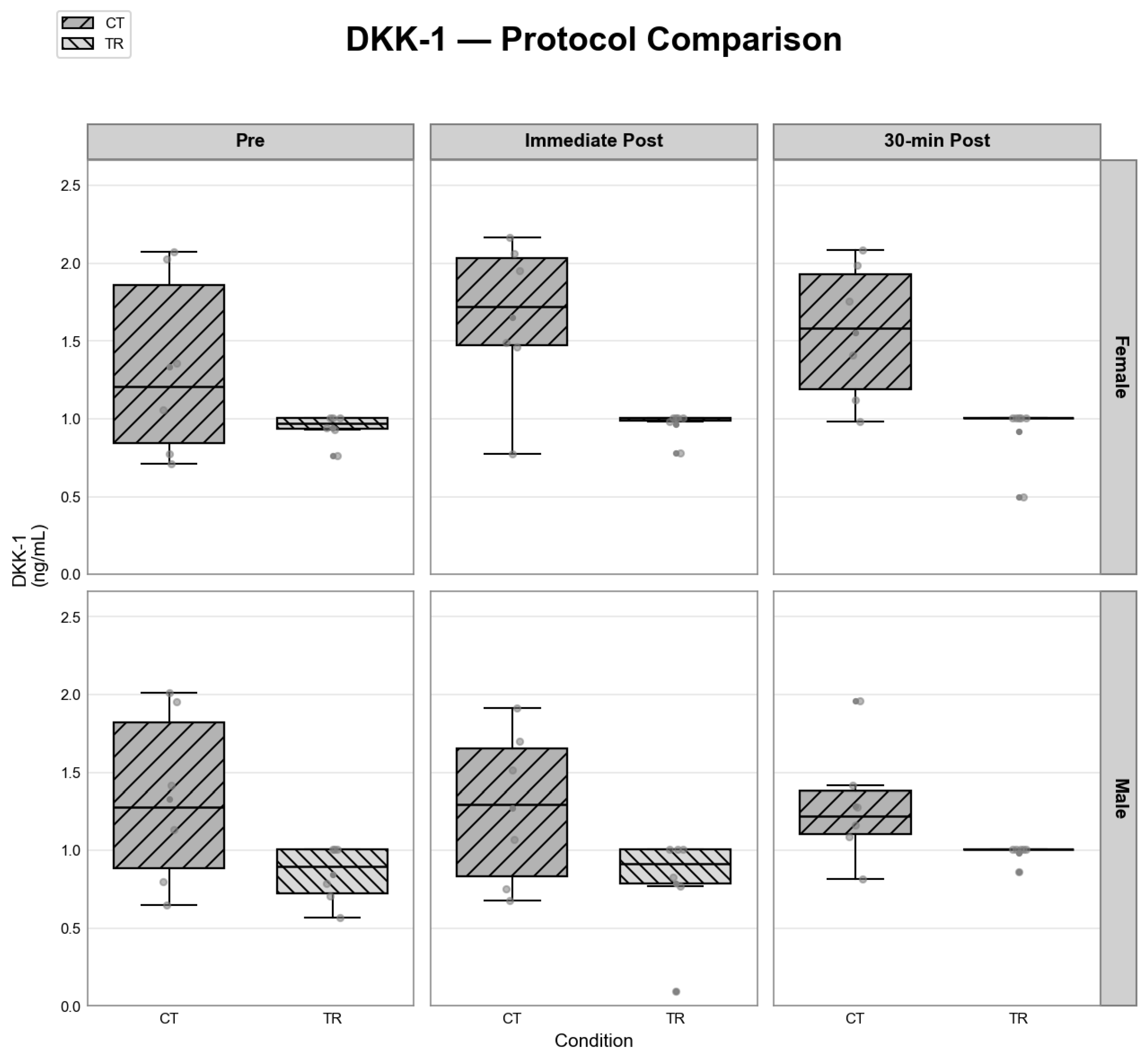
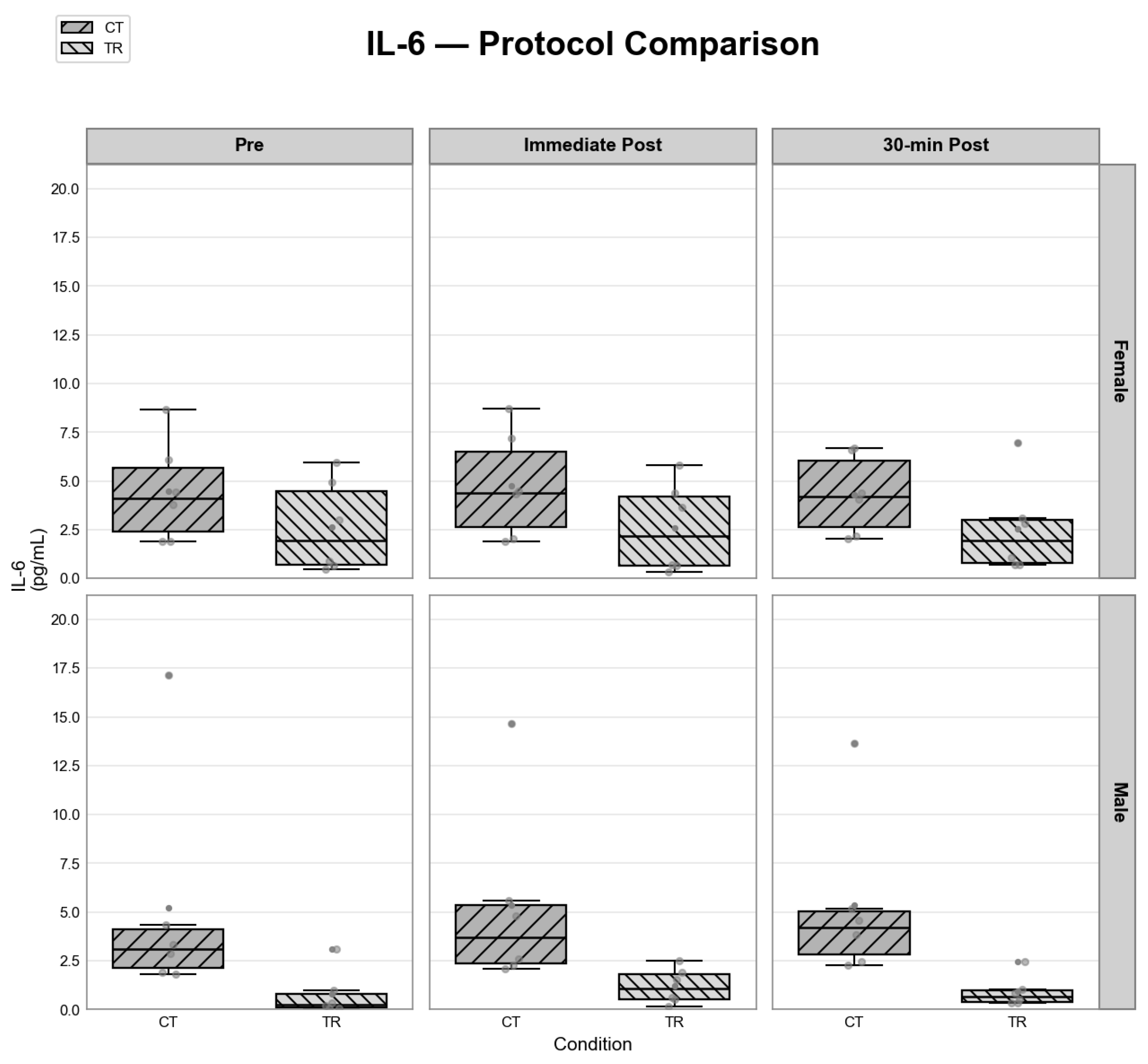
3.3. Exerkines Responses Across Sex, Protocol, and Time Point
4. Discussions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Booth, F. W.; Roberts, C. K.; Laye, M. J. Lack of exercise is a major cause of chronic diseases. Compr. Physiol. 2012, 2, 1143–1211. [Google Scholar] [CrossRef] [PubMed]
- Dhuli, K.; Naureen, Z.; Medori, M. C.; Fioretti, F.; Caruso, P.; Perrone, M. A.; et al. Physical activity for health. J. Prev. Med. Hyg. 2022, 63, E150. [Google Scholar] [PubMed]
- Chow, L.S.; Gerszten, R.E.; Taylor, J.M.; Pedersen, B.K.; Van Praag, H.; Trappe, S.; Febbraio, M.A.; Galis, Z.S.; Gao, Y.; Haus, J.M.; Lanza, I.R. Nat. Rev. Endocrinol. 2022, 18, 273–289. [CrossRef] [PubMed]
- Novelli, G.; Calcaterra, G.; Casciani, F.; Pecorelli, S.; Mehta, J. L. “Exerkines”: A comprehensive term for the factors produced in response to exercise. Biomedicines 2024, 12, 1975. [Google Scholar] [CrossRef]
- Kirk, B.; Feehan, J.; Lombardi, G.; Duque, G. Muscle, bone, and fat crosstalk: the biological role of myokines, osteokines, and adipokines. Curr. Osteoporos. Rep. 2020, 18, 388–400. [Google Scholar] [CrossRef]
- Liu, J. J.; Wong, M. D.; Toy, W. C.; Tan, C. S.; Liu, S.; Ng, X. W.; et al. Lower circulating irisin is associated with type 2 diabetes mellitus. J. Diabetes Complicat. 2013, 27, 365–369. [Google Scholar] [CrossRef]
- Lu, X.; Chen, Y.; Shi, Y.; Shi, Y.; Su, X.; Chen, P.; Wu, D.; Shi, H. Exercise and exerkines: Mechanisms and roles in anti-aging and disease prevention. Exp. Gerontol. 2025, 200, 112685. [Google Scholar] [CrossRef]
- Robbins, J. M.; Gerszten, R. E. Exercise, exerkines, and cardiometabolic health: From individual players to a team sport. J. Clin. Investig. 2023, 133, e168121. [Google Scholar] [CrossRef]
- Dethlefsen, C.; Lillelund, C.; Midtgaard, J.; Andersen, C.; Pedersen, B. K.; Christensen, J. F.; Hojman, P. Exercise regulates breast cancer cell viability: Systemic training adaptations versus acute exercise responses. Breast cancer Res. Treat. 2016, 159, 469–479. [Google Scholar] [CrossRef]
- Bettariga, F.; Taaffe, D.R.; Galvão, D.A.; Lopez, P.; Bishop, C.; Markarian, A.M.; Natalucci, V.; Kim, J.-S.; Newton, R.U. Exercise training mode effects on myokine expression in healthy adults: A systematic review with meta-analysis. J. Sport Health Sci. 2024, 13, 764–779. [Google Scholar] [CrossRef]
- Zhu, C.; Ding, X.; Chen, M.; Feng, J.; Zou, J.; Zhang, L. Exercise-mediated skeletal muscle-derived IL-6 regulates bone metabolism: A new perspective on muscle–bone crosstalk. Biomolecules 2025, 15, 893. [Google Scholar] [CrossRef]
- Jung, M. E.; Bourne, J. E.; Little, J. P. Where does HIT fit? An examination of the affective response to high-intensity intervals in comparison to continuous moderate-and continuous vigorous-intensity exercise in the exercise intensity-affect continuum. PloS ONE 2014, 9, e114541. [Google Scholar] [CrossRef]
- Kim, K. H.; Lee, H. B. Effects of circuit training interventions on bone metabolism markers and bone density of old women with osteopenia. J. Exerc. Rehabil. 2019, 15, 302. [Google Scholar] [CrossRef]
- A'Naja, M. N.; Batrakoulis, A.; Camhi, S. M.; McAvoy, C.; Sansone, J. S.; Reed, R. 2025 ACSM worldwide fitness trends: Future directions of the health and fitness industry. ACSM's Health Fit. J. 2024, 28, 11–25. [Google Scholar]
- Sharma-Ghimire, P.; Chen, Z.; Sherk, V.; Bemben, M.; Bemben, D. Sclerostin and parathyroid hormone responses to acute whole-body vibration and resistance exercise in young women. J. Bone Miner. Metab. 2019, 37, 358–367. [Google Scholar] [CrossRef] [PubMed]
- Green, G. K.; Stone, W. J.; Tolusso, D. V.; Schafer, M. A.; Lyons, T. S. A VO2max protocol for Young, apparently healthy Adults. Int. J. Exerc. Sci. 2023, 16, 1257–1268. [Google Scholar] [CrossRef]
- Kellner, P.; Neubauer, J.; Kellnerova, Z.; Zahradnicek, P.; Novotny, V.; Wawrzacz, L.; Puncochar, M. The reasoning behind assessing push-up tests: An in-depth analysis. J. Phys. Educ. Sport 2023, 23, 1706–1713. [Google Scholar]
- Grgic, J.; Lazinica, B.; Schoenfeld, B. J.; Pedisic, Z. Test-retest reliability of the one-repetition maximum (1RM) strength assessment: A Systematic review. Sports Med. Open 2020, 6, 31. [Google Scholar] [CrossRef] [PubMed]
- Koltun, K. J.; Sterczala, A. J.; Sekel, N. M.; Krajewski, K. T.; Martin, B. J.; Lovalekar, M.; Connaboy, C.; Flanagan, S. D.; Wardle, S. L.; O'Leary, T. J.; et al. Effect of acute resistance exercise on bone turnover in young adults before and after concurrent resistance and interval training. Physiol. Rep. 2024, 12, e15906. [Google Scholar] [CrossRef]
- Bonewald, L. F.; Johnson, M. L. Osteocytes, mechanosensing and Wnt signaling. Bone 2008, 42, 606–615. [Google Scholar] [CrossRef]
- Drake, M. T.; Srinivasan, B.; Mödder, U. I.; Peterson, J. M.; McCready, L. K.; Riggs, B. L.; Dwyer, D.; Stolina, M.; Kostenuik, P.; khosla, S. Effects of parathyroid hormone treatment on circulating sclerostin levels in postmenopausal women. J. Clin. Endocrinol. Metab. 2010, 95, 5056–5062. [Google Scholar] [CrossRef]
- Amrein, K.; Amrein, S.; Drexler, C.; Dimai, H. P.; Dobnig, H.; Pfeifer, K.; Tomaschitz, A.; Pieber, T. R.; Fahrleitner-Pammer, A. Sclerostin and its association with physical activity, age, gender, body composition, and bone mineral content in healthy adults. J. Clin. Endocrinol. Metab. 2012, 97, 148–154. [Google Scholar] [CrossRef]
- Grasso, D.; Corsetti, R.; Lanteri, P.; Di Bernardo, C.; Colombini, A.; Graziani, R.; et al. Bone-muscle unit activity, salivary steroid hormones profile, and physical effort over a 3-week stage race. Scand. J. Med. Sci. Sports 2015, 25, 70–80. [Google Scholar] [CrossRef]
- Lombardi, G.; Lanteri, P.; Colombini, A.; Mariotti, M.; Banfi, G. Sclerostin concentrations in athletes: Role of load and gender. J. Biol. Regul. Homeost. Agents 2012, 26, 157–163. [Google Scholar]
- Zhao, D.; Hua, R.; Riquelme, M. A.; Cheng, H.; Guda, T.; Xu, H.; Gu, S.; Jiang, J. X. Osteocytes regulate bone anabolic response to mechanical loading in male mice via activation of integrin α5. Bone Res. 2022, 49. [Google Scholar] [CrossRef]
- Kouvelioti, R.; LeBlanc, P.; Falk, B.; Ward, W. E.; Josse, A. R.; Klentrou, P. Effects of high-intensity interval running versus cycling on sclerostin, and markers of bone turnover and oxidative stress in young men. Calcif. Tissue Int. 2019, 104, 582–590. [Google Scholar] [CrossRef] [PubMed]
- Pickering, M. E.; Simon, M.; Sornay-Rendu, E.; Chikh, K.; Carlier, M. C.; Raby, A. L.; Szulc, P.; Confavreux, C. B. Serum sclerostin increases after acute physical activity. Calcif. Tissue Int. 2017, 101, 170–173. [Google Scholar] [CrossRef] [PubMed]
- Prowting, J. L.; Skelly, L. E.; Kurgan, N.; Fraschetti, E. C.; Klentrou, P.; Josse, A. R. Acute effects of milk vs. carbohydrate on bone turnover biomarkers following loading exercise in young adult females. Front. Nutr. 2022, 9, 840973. [Google Scholar] [CrossRef]
- Dekker, J.; Nelson, K.; Kurgan, N.; Falk, B.; Josse, A.; Klentrou, P. Wnt signaling-related osteokines and transforming growth factors before and after a single bout of plyometric exercise in Child and adolescent females. Pediatr. Exerc.Sci. 2017, 29, 504–512. [Google Scholar] [CrossRef]
- Staab, J. S.; Lutz, L. J.; Foulis, S. A.; Gaffney-Stomberg, E.; Hughes, J. M. Load carriage aerobic exercise stimulates a transient rise in biochemical markers of bone formation and resorption. J. Appl. Physiol. 2023, 134, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Ghardashi-Afousi, A.; Davoodi, M.; Hesamabadi, B. K.; Asvadi-Fard, M.; Bigi, M. A. B.; Izadi, M. R.; Gaeini, A. A. Improved carotid intima-media thickness-induced high-intensity interval training associated with decreased serum levels of Dkk-1 and sclerostin in type 2 diabetes. J. Diabetes Complicat. 2020, 34, 107469. [Google Scholar] [CrossRef]
- Sharma-Ghimire, P.; Buchanan, S.; Bemben, M. G.; Knehans, A.; Campbell, J.; Bemben, D. A. Sclerostin and Dickkopf-1 characteristics according to age and physical activity Levels in premenopausal women. J. Clin. Densitom. 2022, 25, 168–177. [Google Scholar] [CrossRef]
- Lara-Castillo, N.; Kim-Weroha, N. A.; Kamel, M. A.; Javaheri, B.; Ellies, D. L.; Krumlauf, R. E.; Thiagarajan, G.; Johnson, M. L. In vivo mechanical loading rapidly activates β-catenin signaling in osteocytes through a prostaglandin- mediated mechanism. Bone 2015, 76, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, T.; Nazarali, P.; Hedayati, M.; Alizadeh, R. The effect of eight weeks of high intensity interval training on osteopontin and some bone mineral indices in young women. J. Phys. Educ. Sport 2018, 18, 532–535. [Google Scholar]
- Wang, Y.; Hong, L.; Jiang, J.; Zhang, X.; Chen, J.; Diao, H. Osteopontin may Improve postinjury muscle repair via matrix metalloproteinases and TGF-β activation in regular exercise. Int. J. Med. Sci. 2023, 20, 1202–1211. [Google Scholar] [CrossRef] [PubMed]
- You, J. S.; Ji, H. I.; Chang, K. J.; Yoo, M. C.; Yang, H. I.; Jeong, I. K.; Kim, K. S. Serum osteopontin concentration is decreased by exercise-induced fat loss but is not correlated with body fat percentage in obese humans. Mol. Med. Rep. 2013, 8, 579–584. [Google Scholar] [CrossRef]
- Raman, A.; Peiffer, J. J.; Hoyne, G. F.; Lawler, N. G.; Currie, A.; Fairchild, T. J. Exercise-induced responses in matrix metalloproteinases and osteopontin are not moderated by exercise format in males with overweight or obesity. Eur. J. Appl. Physiol. 2023, 123, 1115–1124. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Shou, P.; Zhang, L.; Xu, C.; Zheng, C.; Han, Y.; Li, W.; Huang, Y.; Zhang, X.; Shao, C.; et al. An osteopontin-integrin interaction plays a critical role in directing adipogenesis and osteogenesis by mesenchymal stem cells. Stem cells 2014, 32, 327–337. [Google Scholar] [CrossRef]
- Uaesoontrachoon, K.; Wasgewatte Wijesinghe, D. K.; Mackie, E. J.; Pagel, C. N. Osteopontin deficiency delays inflammatory infiltration and the onset of muscle regeneration in a mouse model of muscle injury. Dis. Model. Mech. 2013, 6, 197–205. [Google Scholar]
- Gassner, M.; Balaskovits, P.; Melchior, P.; Wagner, M.; Wagner, K. H.; König, D. Reactive oxygen species and IL-6 responses to resistance-based HIIT: A time-dependent analysis. Free Radic. Biol. Med. 2025, 242, 250–257. [Google Scholar] [CrossRef]
- Petersen, A. M.; Pedersen, B. K. The anti-inflammatory effect of exercise. J. Appl. Physiol. 2005, 98, 1154–1162. [Google Scholar] [CrossRef]
- Mezil, Y. A.; Allison, D.; Kish, K.; Ditor, D.; Ward, W. E.; Tsiani, E.; Klentrou, P. Response of bone turnover markers and cytokines to high-intensity low-impact exercise. Med. Sci. Sports Exerc. 2015, 47, 1495–1502. [Google Scholar] [CrossRef]
- Kim, J. Y.; Kim, H. J.; Kim, C. S. Effects of 12-week combined exercise on RANKL/RANK/OPG signaling and bone-resorption cytokines in healthy college females. J. Exerc. Nutrition. Biochem. 2019, 23, 13–20. [Google Scholar] [CrossRef]
- Marques, E. A.; Wanderley, F.; Machado, L.; Sousa, F.; Viana, J. L.; Moreira-Gonçalves, D.; Moreira, P.; Mota, J.; Carvalho, J. Effects of resistance and aerobic exercise on physical function, bone mineral density, OPG and RANKL in older women. Exp. Gerontol. 2011, 46, 524–532. [Google Scholar] [CrossRef]
- Marques, E. A.; Mota, J.; Viana, J. L.; Tuna, D.; Figueiredo, P.; Guimarães, J. T.; Carvalho, J. Response of bone mineral density, inflammatory cytokines, and biochemical bone markers to a 32-week combined loading exercise programme in older men and women. Arch. Gerontology. Geriatr. 2013, 57, 226–233. [Google Scholar] [CrossRef]
- Onal, M.; Xiong, J.; Chen, X.; Thostenson, J. D.; Almeida, M.; Manolagas, S. C.; O'Brien, C. A. Receptor activator of nuclear factor κB ligand (RANKL) protein expression by B lymphocytes contributes to ovariectomy-induced bone loss. J. Biol. Chem. 2012, 287, 29851–29860. [Google Scholar] [CrossRef] [PubMed]
- Yarrow, J. F.; White, L. J.; McCoy, S. C.; Borst, S. E. Training augments resistance exercise-induced elevation of circulating brain derived neurotrophic factor (BDNF). Lett. 2010, 479, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, C.; Antunes, B. M.; Giacon, T. R.; Vanderlei, L. C. M.; Campos, E. Z.; Peres, F. P.; Clark, N. W.; Panissa, V. L. G.; Lira, F. S. Influence of acute and chronic high-intensity intermittent aerobic plus strength exercise on BDNF, Lipid and Autonomic Parameters. J. Sports Sci. Med. 2019, 18, 359–368. [Google Scholar] [PubMed]
- Arazi, H.; Babaei, P.; Moghimi, M.; Asadi, A. Acute effects of strength and endurance exercise on serum BDNF and IGF-1 levels in older men. BMC Geriatr. 2021, 21(1), 50. [Google Scholar] [CrossRef]
- Piepmeier, A. T.; Etnier, J. L. Brain-derived neurotrophic factor (BDNF) as a potential mechanism of the effects of acute exercise on cognitive performance. J. Sport Health Sci. 2015, 4, 14–2. [Google Scholar] [CrossRef]
- Zoladz, J. A.; Pilc, A.; Majerczak, J.; Grandys, M.; Zapart-Bukowska, J.; Duda, K. Endurance training increases plasma brain-derived neurotrophic factor concentration in young healthy men. J. Physiolo. Pharmacol. 2008, 59, 119–132. [Google Scholar]
- Zhang, J.; Zhu, L.; Zhou, J.; Yu, Q.; Yang, G.; Luo, C.; Meng, J.; Xing, S.; Liu, J.; Mou, D.; Yang, X. BDNF alleviates senescence and enhances osteogenic differentiation in bone marrow mesenchymal stem cells via the TrkB/PI3K/AKT pathway. Tissue Cell 2025, 96, 102972. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Huang, J.; Hu, M.; Cui, X.; Leng, L.; Wang, K.; et al. Acute high-intensity interval exercise is superior to moderate-intensity continuous exercise in enhancing endothelial function and its associated biomarkers in sedentary young individuals: The possible involvement of lactate. J. Exerc. Sci. Fit. 2025, 23, 60–68. [Google Scholar] [CrossRef]
- Kabak, B.; Belviranli, M.; Okudan, N. Irisin and myostatin responses to acute high-intensity interval exercise in humans. Horm. Mol. Biol. Clin.Investig 2018, 35(3). [Google Scholar] [CrossRef]
- Fox, J.; Rioux, B. V.; Goulet, E. D.; Johanssen, N. M.; Swift, D. L.; Bouchard, D. R.; et al. Effect of an acute exercise bout on immediate post-exercise irisin concentration in adults: A meta-analysis. Scand. J. Med. Sci. Sports 2018, 28, 16–28. [Google Scholar] [CrossRef]
- Roh, H. T.; Cho, S. Y.; So, W. Y. Effects of regular taekwondo intervention on oxidative stress biomarkers and myokines in overweight and obese adolescents. Int. J. Environ. Res. Public Health 2020, 17, 2505. [Google Scholar] [CrossRef]
- Norheim, F.; Langleite, T.M.; Hjorth, M.; Holen, T.; Kielland, A.; Stadheim, H.K.; Gulseth, H.L.; Birkeland, K.I.; Jensen, J.; Drevon, C.A. The effects of acute and chronic exercise on PGC-1α, irisin and browning of subcutaneous adipose tissue in humans. FEBS J. 2014, 281, 739–749. [Google Scholar] [CrossRef] [PubMed]
- Contrepois, K.; Wu, S.; Moneghetti, K.J.; Hornburg, D.; Ahadi, S.; Tsai, M.S.; Metwally, A.A.; Wei, E.; Lee-McMullen, B.; Quijada, J.V.; Chen, S.; Christle, J.W.; Ellenberger, M.; Balliu, B.; Taylor, S.; Knowles, D.A.; Mishra, T.; Tang, H.; Snyder, M.P. Molecular choreography of acute exercise. Cell 2020, 181, 1112–1130. [Google Scholar] [CrossRef]
| Variable | Male (n = 6) | Female (n = 6) |
| Age (yrs) | 20.67 + 1.02 | 20.17 + 0.54 |
| Height (cm) | 169.76 + 2.22 | 162.77 + 3.32 |
| Weight (kg) | 83.58 + 7.87 | 66.66 + 7.37 |
| Calcium Intake (mg/day) | 527.67 + 139.10 | 557.67 + 135.75 |
| pBPAQ Score | 51.11 + 9.80 | 29.99 + 8.52 |
| cBPAQ | 5.89 + 2.17 | 2.84 + 1.08 |
| tBPAQ | 28.50 + 4.92 | 16.42 + 4.12 |
| BMI | 27.73 + 3.20 | 24.68 + 1.94 |
| Body Fat % | 20.98 + 5.74 | 32.02 + 4.05 |
| Fat Mass (kg) | 19.91 + 6.87 | 22.61 + 4.72 |
| Fat Free Mass (kg) | 38.01 + 1.63 | 27.05 + 2.53 |
| pBPAQ = past Bone Specific Physical Activity Questionnaire; cBPAQ = current Bone Specific Physical Activity Questionnaire; tBPAQ = total Bone Specific Physical Activity Questionnaire. | ||
| Variable | Male (n = 6) | Female (n = 6) |
|---|---|---|
| Push-up Score | 30.33 + 4.29 | 20.33 + 1.52 |
| RHR | 72.00 + 7.03 | 73.17 + 3.25 |
| RBP Systolic (mmHg) | 119.17 + 9.30 | 110.17 + 3.12 |
| RBP Diastolic (mmHg) | 66.17 + 3.24 | 66.00 + 3.34 |
| PEHR | 129.00 + 13.37 | 129.17 + 5.40 |
| PEBP Systolic (mmHg) | 145.33 + 13.30 | 150.33 + 9.14 |
| PEBP Diastolic (mmHg)* | 60.17 + 5.01 | 85.33 + 7.26 |
| VO2max (ml/kg/min) | 50.74 + 3.47 | 43.41 + 2.84 |
| Variables | Male (n = 6) | Female (n = 6) |
| RHG** | 39.89 + 2.19 | 27.58 + 2.57 |
| LHG | 37.55 + 4.47 | 26.38 + 2.63 |
| TIA (s) | 0.59 + 0.04 | 0.50 + 0.03 |
| VJ Height (in) | 17.85 + 2.73 | 12.6 + 1.59 |
| Velocity | 1.30 + 0.09 | 1.18 +0.05 |
| Power (watts) | 1,023.94 + 83.99 | 755.6 + 86.68 |
| Relative Power (kg/watts) | 12.41 + 0.84 | 11.37 + 0.51 |
| Variable | Male (n = 6) | Female (n = 6) |
| Leg Press | 514.00 + 68.65 | 361.67 + 66.60 |
| Seated Cable Rows** | 175.00 + 9.22 | 98.33 + 9.37 |
| Bench Press** | 180.00 + 8.47 | 64.17 + 6.38 |
| Deadlifts** | 98.33 + 4.22 | 59.17 + 5.39 |
| Seated Shoulder Press** | 56.67 + 2.47 | 25.83 + 3.00 |
| Variables. | Male CT | Female CT | Male TR | Female TR | Significant Effects | p |
| SCL (pg/ml) |
||||||
| Pre | 0.11 ± 0.02 | 0.03 ± 0.02 | 0.21 ± 0.02 | 0.07 ± 0.02 | Sex, Protocol, Time Point | 0.008, 0.001, 0.04 |
| IP | 0.14 ± 0.02 | 0.06 ± 0.02 | 0.22 ± 0.02 | 0.13 ± 0.02 | IP>Pre, 30P |
0.027, 0.015 |
| 30P | 0.10 ± 0.01 | 0.04 ± 0.02 | 0.17 ± 0.02 | 0.12 ± 0.02 | ||
| OPN (pg/ml) |
||||||
| Pre | 19.32 ± 1.8 | 19.39 ± 1.80 | 17.23 ± 1.43 | 19.40 ± 1.43 | Time Point Pre>30P |
0.014 (0.04) |
| IP | 14.98 ± 2.4 | 18.11 ± 2.42 | 15.62 ± 2.5 | 14.40 ± 2.56 | ||
| 30P | 16.31 ± 2.6 | 16.55 ± 2.60 | 13.05 ± 1.7 | 16.85 ± 1.70 | ||
| RANKL (pg/ml) |
||||||
| Pre | 0.35 ± 0.08 | 0.43 ± 0.08 | 0.31 ± 0.07 | 0.42 ± 0.07 | Time Point |
|
| IP | 0.28 ± 0.07 | 0.35 ± 0.07 | 0.40 ± 0.08 | 0.29 ± 0.08 | 30P>Pre, IP |
0.02, 0.001 |
| 30P | 0.49 ± 0.08 | 0.51 ± 0.08 | 0.71 ± 0.12 | 0.59 ± 0.12 | ||
| Irisin (pg/ml) |
||||||
| Pre | 3018.65 ± 399.52 | 2734.61 ± 399.52 | 2091.57 ± 278.94 | 2002.11 ± 278.94 | Protocol and time point | 0.001 |
| IP | 2805.48 ± 430.28 | 2674.75 ± 430.28 | 2007.62 ± 275.32 | 1971.56 ± 275.32 | Pre> IP | 0.01 |
| 30P | 2830.34 ± 408.03 | 2819.86 ± 408.03 | 2094.98 ± 297.85 | 2053.43 ± 297.85 |
| Marker | Sex | Treatment | Timepoint | N | Median (Q1, Q3) |
| BDNF (pg/ml) | Male | CT | IP | 6 | 0.207 (0.141, 0.258) |
| Male | CT | Post 30 | 6 | 0.297 (0.184, 0.702) | |
| Male | CT | Pre | 6 | 0.276 (0.166, 0.707) | |
| Male | TR | IP | 6 | 0.559 (0.245, 0.868) | |
| Male | TR | Post 30 | 6 | 1.011 (0.885, 1.211) | |
| Male | TR | Pre | 6 | 0.479 (0.148, 0.955) | |
| Female | CT | IP | 6 | 0.429 (0.271, 1.333) | |
| Female | CT | Post 30 | 6 | 0.486 (0.168, 1.101) | |
| Female | CT | Pre | 6 | 0.233 (0.166, 0.406) | |
| Female | TR | IP | 6 | 0.554 (0.354, 0.911) | |
| Female | TR | Post 30 | 6 | 0.960 (0.802, 1.127) | |
| Female | TR | Pre | 6 | 0.464 (0.251, 0.972) | |
| DKK-1 (pg/ml) | Male | CT | IP | 6 | 1.294 (0.750, 1.697) |
| Male | CT | Post 30 | 6 | 1.220 (1.090, 1.418) | |
| Male | CT | Pre | 6 | 1.276 (0.799, 1.955) | |
| Male | TR | IP | 6 | 0.917 (0.770, 1.006) | |
| Male | TR | Post 30 | 6 | 1.006 (1.006, 1.006) | |
| Male | TR | Pre | 6 | 0.897 (0.705, 1.006) | |
| Female | CT | IP | 6 | 1.723 (1.465, 2.063) | |
| Female | CT | Post 30 | 6 | 1.581 (1.120, 1.987) | |
| Female | CT | Pre | 6 | 1.208 (0.774, 2.026) | |
| Female | TR | IP | 6 | 1.006 (0.985, 1.006) | |
| Female | TR | Post 30 | 6 | 1.006 (1.006, 1.006) | |
| Female | TR | Pre | 6 | 0.974 (0.933, 1.006) | |
| IL-6 (pg/ml) | Male | CT | IP | 6 | 3.689 (2.282, 5.576) |
| Male | CT | Post 30 | 6 | 4.214 (2.476, 5.187) | |
| Male | CT | Pre | 6 | 3.108 (1.905, 4.348) | |
| Male | TR | IP | 6 | 1.073 (0.502, 1.894) | |
| Male | TR | Post 30 | 6 | 0.674 (0.352, 1.022) | |
| Male | TR | Pre | 6 | 0.263 (0.083, 0.972) | |
| Female | CT | IP | 6 | 4.396 (2.051, 7.211) | |
| Female | CT | Post 30 | 6 | 4.204 (2.178, 6.603) | |
| Female | CT | Pre | 6 | 4.103 (1.923, 6.099) | |
| Female | TR | IP | 6 | 2.159 (0.679, 4.385) | |
| Female | TR | Post 30 | 6 | 1.957 (0.712, 3.094) | |
| Female | TR | Pre | 6 | 1.936 (0.637, 4.947) |
| Variables | Effect | Test valuea | Df | p-value | Adj. p-value (FDRb) |
| BDNF (ng/ml) | Sex | 0.3354737 | 1 | 0.562 | 0.594 |
| Protocol | 7.1491876 | 1 | 0.0075* | 0.01* | |
| Time point | 4.1801622 | 2 | 0.124 | 0.247 | |
| Sex: Protocol | 0.1852342 | 1 | 0.667 | 0.667 | |
| Sex: Timepoint | 0.7422852 | 2 | 0.69 | 0.94 | |
| Protocol: Timepoint | 2.1499369 | 2 | 0.341 | 0.934 | |
| Sex: Protocol: Timepoint | 0.3303485 | 2 | 0.848 | 0.937 | |
| DKK-1 (ng/ml) | Sex | 2.7546041 | 1 | 0.097 | 0.388 |
| Protocol | 23.6687421 | 1 | <0.001** | <0.001** | |
| Timepoint | 1.5050720 | 2 | 0.471 | 0.617 | |
| Sex: Protocol | 1.7444637 | 1 | 0.187 | 0.667 | |
| Sex: Timepoint | 1.5087329 | 2 | 0.47 | 0.94 | |
| Protocol: Timepoint | 1.0482078 | 2 | 0.592 | 0.934 | |
| Sex: Protocol: Timepoint | 2.3590369 | 2 | 0.307 | 0.615 | |
|
IL-6 (ng/ml) |
Sex | 0.5438761 | 1 | 0.461 | 0.594 |
| Protocol | 54.5026553 | 1 | <0.001** | <0.001** | |
| Timepoint | 0.9664192 | 2 | 0.617 | 0.617 | |
| Sex: Protocol | 0.7270522 | 1 | 0.394 | 0.667 | |
| Sex: Timepoint | 0.5292637 | 2 | 0.767 | 0.94 | |
| Protocol: Timepoint | 0.1374544 | 2 | 0.934 | 0.934 | |
| Sex: Protocol: Timepoint | 0.1292577 | 2 | 0.937 | 0.937 |
| Dependent Variable | Predictor Variables | β | SEE | R2 | p |
|
Pre CT SCL |
Push-ups* BDNF C** Left HGS** |
0.006 0.045 0.002 |
0.04 0.03 0.02 |
0.68 0.86 0.94 |
0.043 0.002 <0.001 |
| Pre TR SCL | JT TIA* 1RM BB BP** |
0.729 0.001 |
0.05 0.04 |
0.67 0.84 |
0.019 0.006 |
|
Pre TR RANKL |
JT Velocity Trad DB DL* CI** JT TIA** |
0.603 -0.009 0.000 -0.803 |
0.15 0.07 0.06 0.04 |
0.38 0.86 0.92 0.98 |
0.245 0.035 0.005 <0.001 |
| Pre CT OPN | Right HGS** | 0.215 | 1.92 | 0.85 | <0.001 |
| Pre TR OPN | tBPAQ | -0.104 | 1.63 | 0.85 | 0.363 |
| Pre CT BDNF | CI | 0.001 | 0.42 | 0.55 | 0.363 |
|
Pre TR IL-6 |
% BF CI* cBPAQ** |
0.121 0.002 0.228 |
1.23 0.99 0.74 |
0.64 0.79 0.90 |
0.124 0.01 0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




