Submitted:
02 April 2026
Posted:
07 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
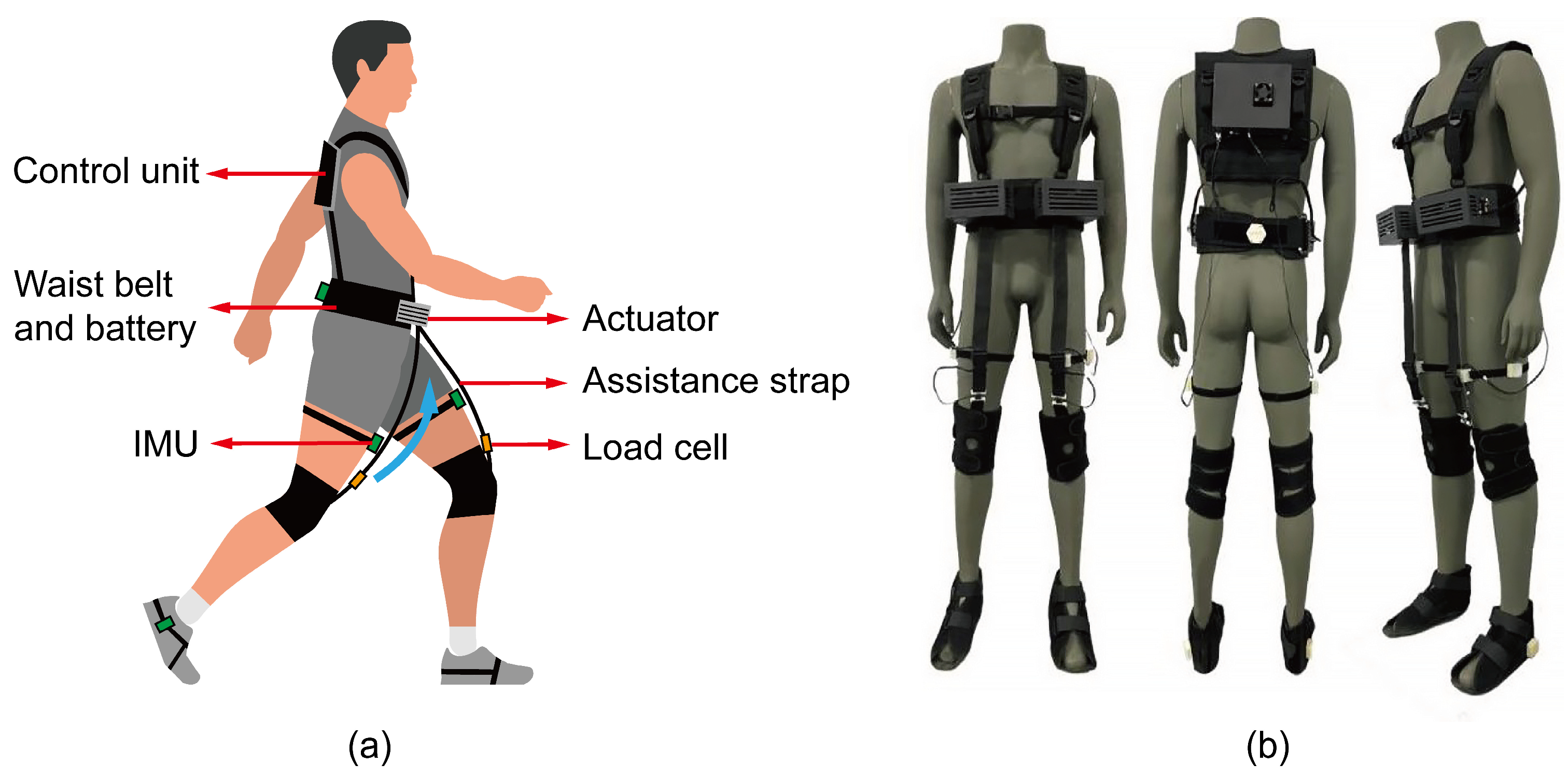
2.1. The Soft Lower-Limb Exoskeleton Platform
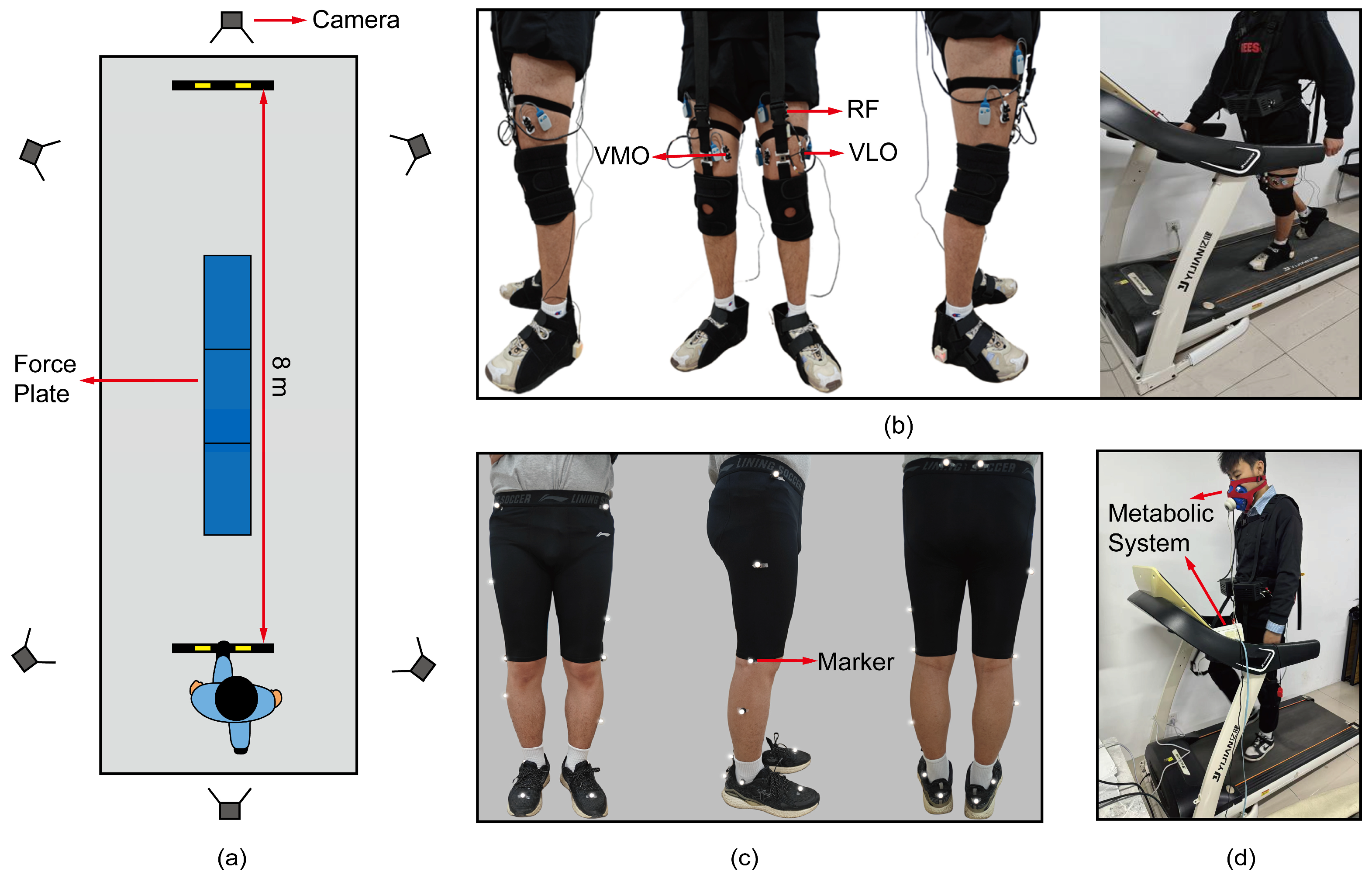
2.2. Participants and Experimental Protocol
2.2. PDFM
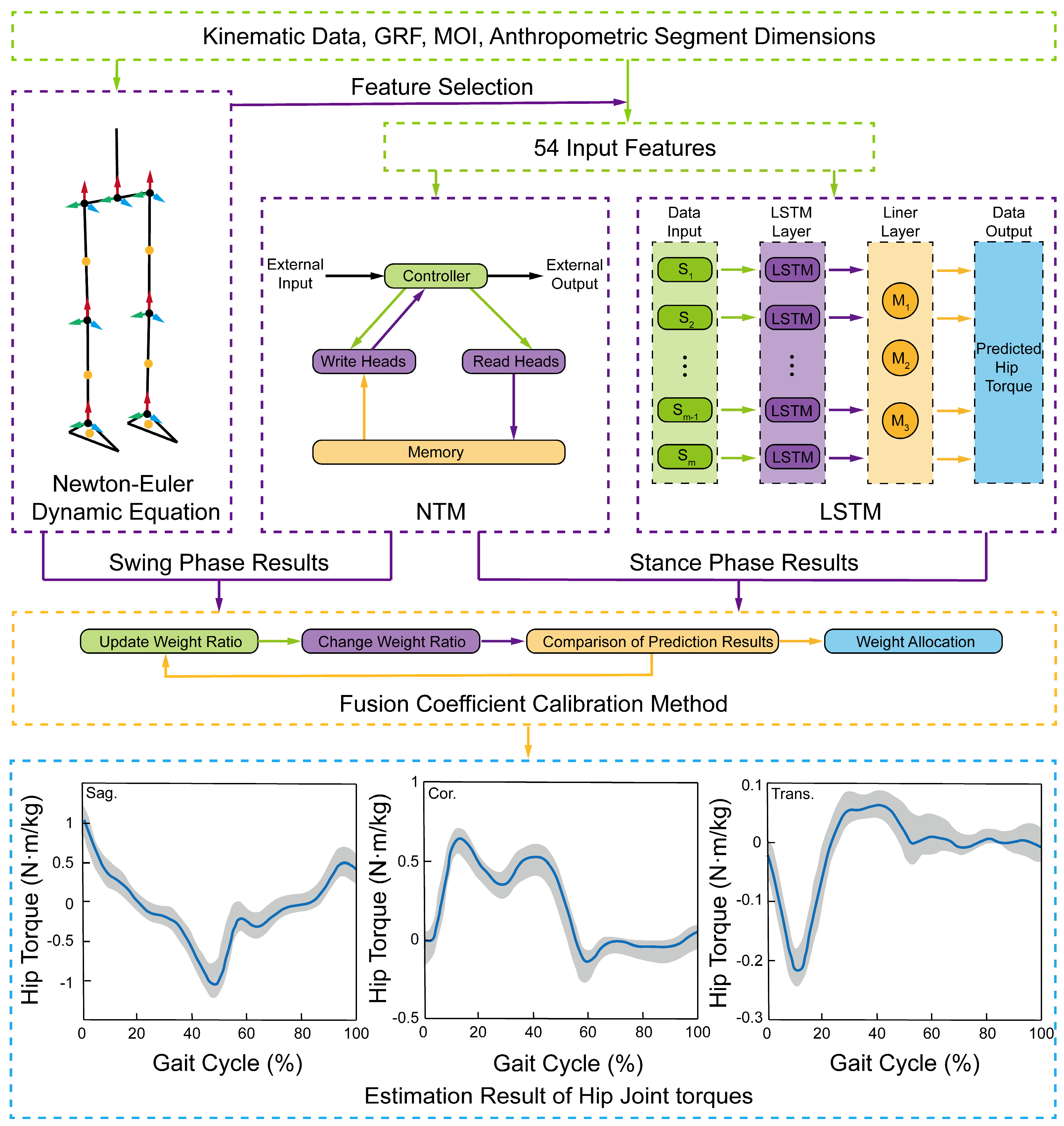
2.3.1. Newton–Euler Inverse Dynamics Model
2.3.2. Selection of Neural Network Models
2.3.3. Construction of the PDFM
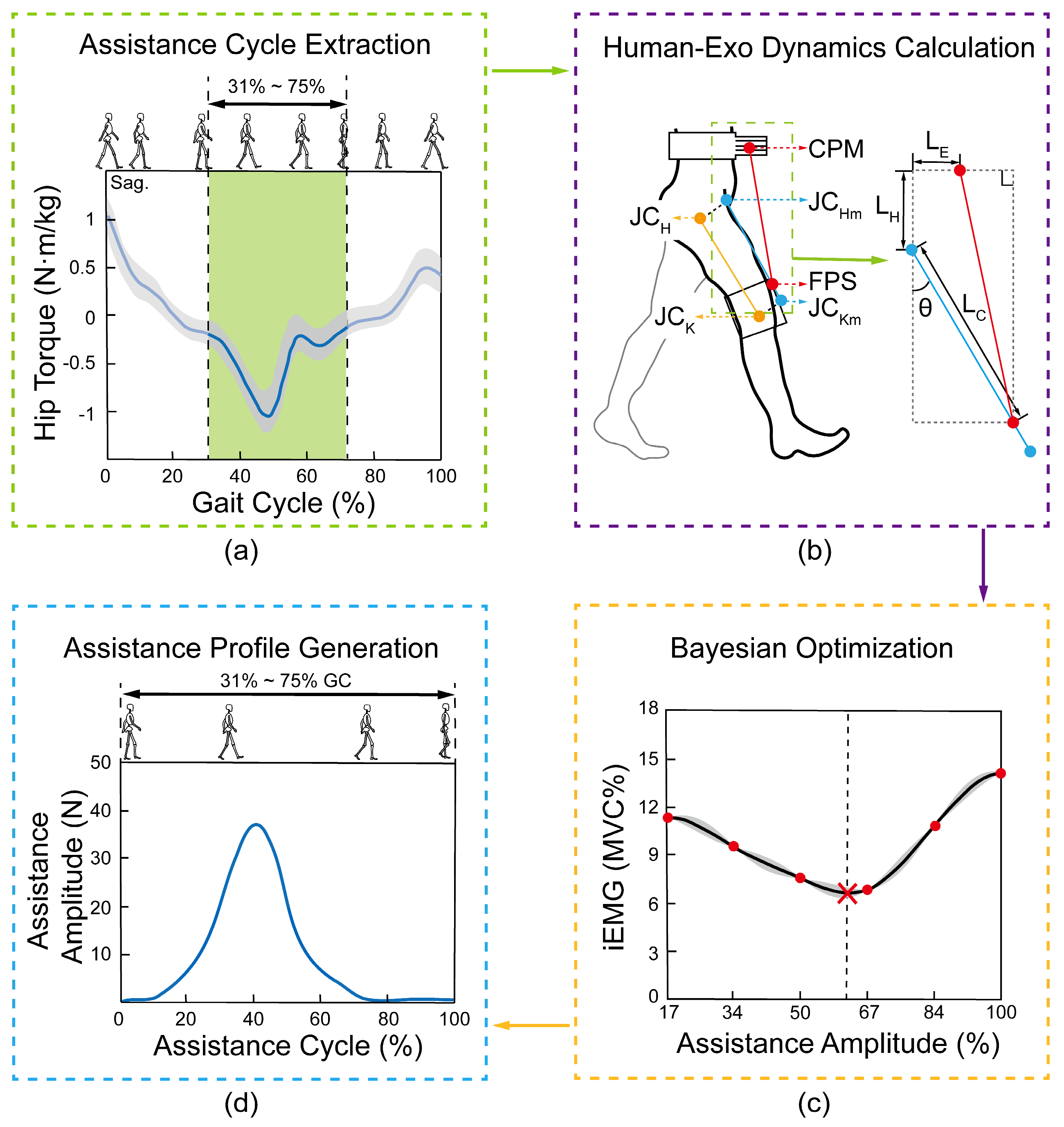
2.4. Exoskeleton Assistance Profile Planning
2.4.1. Assistance Gait Cycle Planning
2.4.2. Human-Exoskeleton Dynamics Model
2.4.3. Exoskeleton Assistance Function
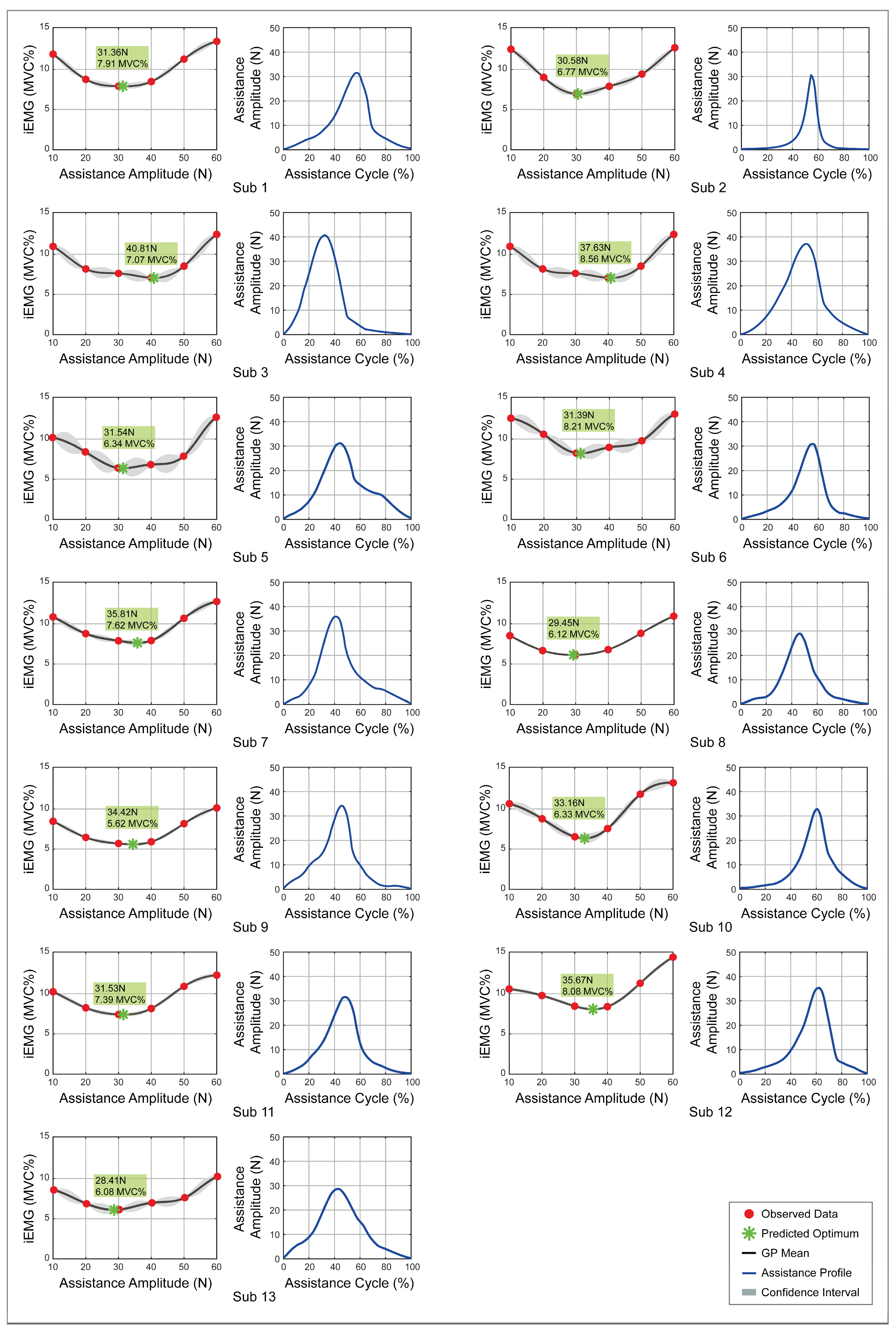
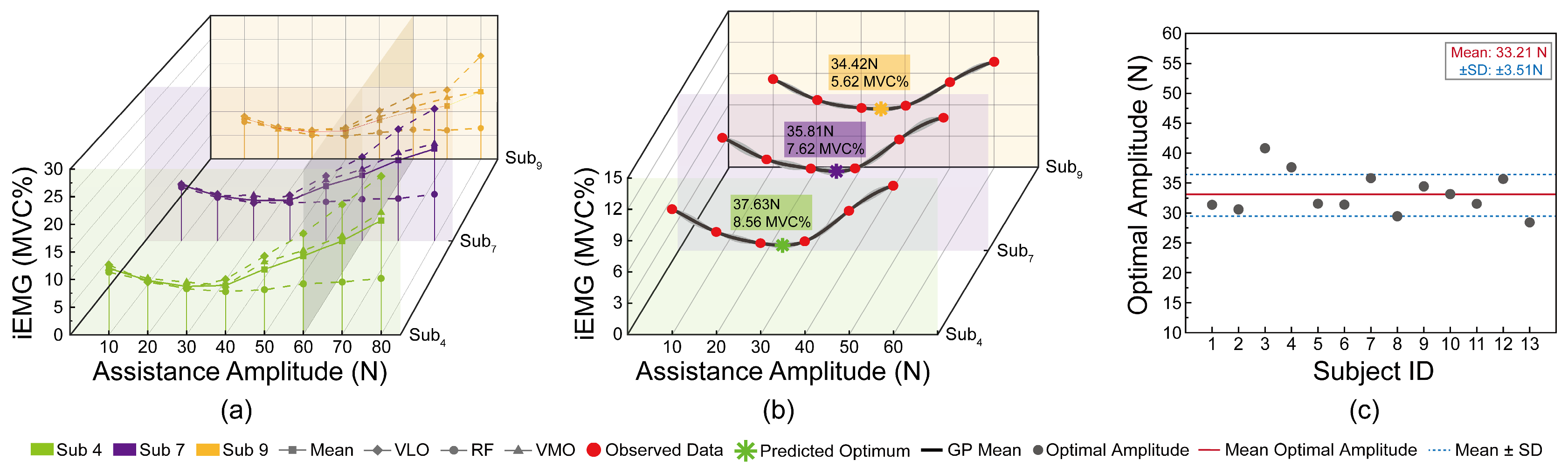
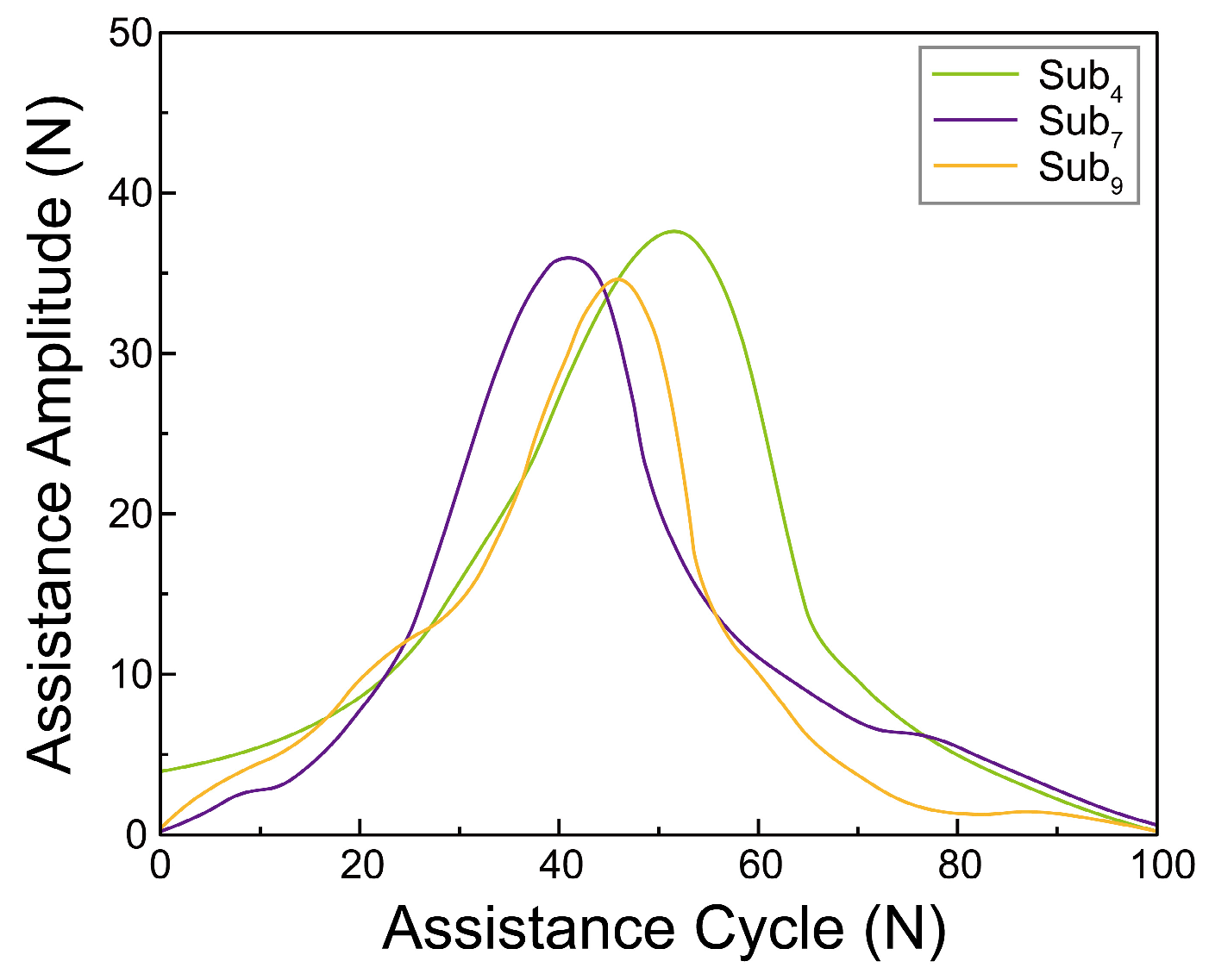
2.4.4. Optimization of Assistance Parameters
2.5. Statistical Analysis
3. Result
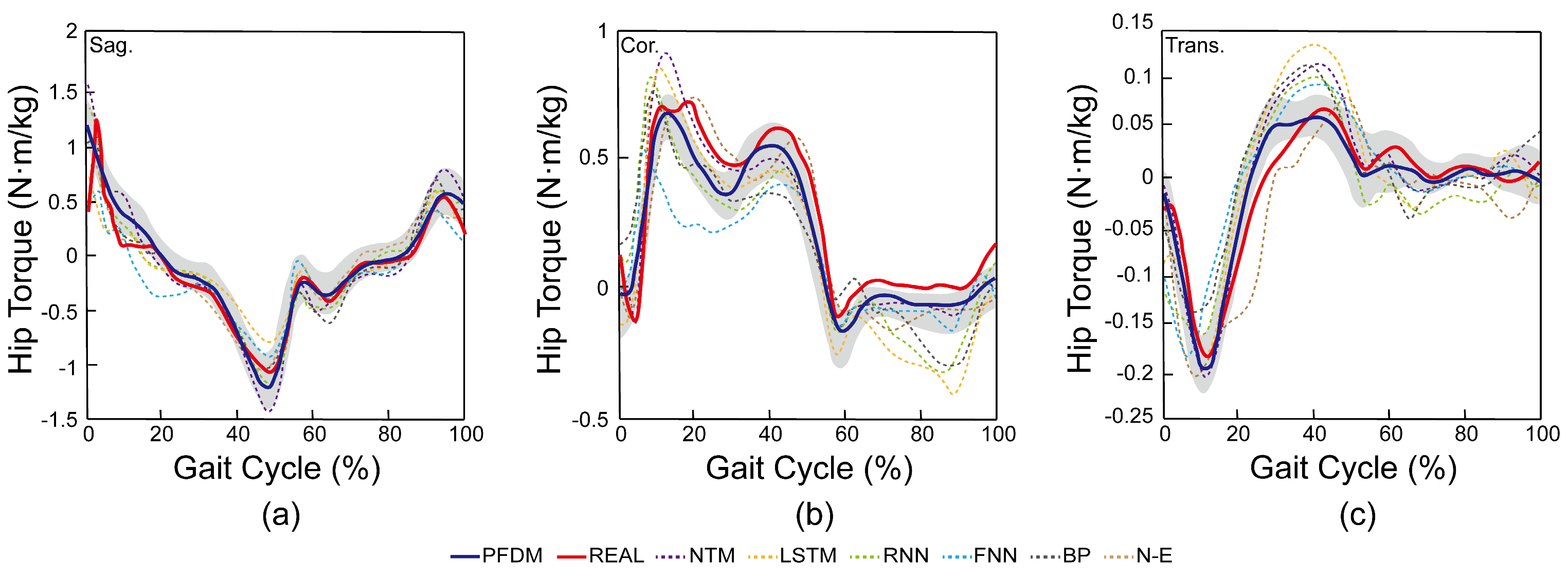
3.1. Accuracy of Hip Moment Output by PDFM
3.2. Bayesian Optimization Outcomes
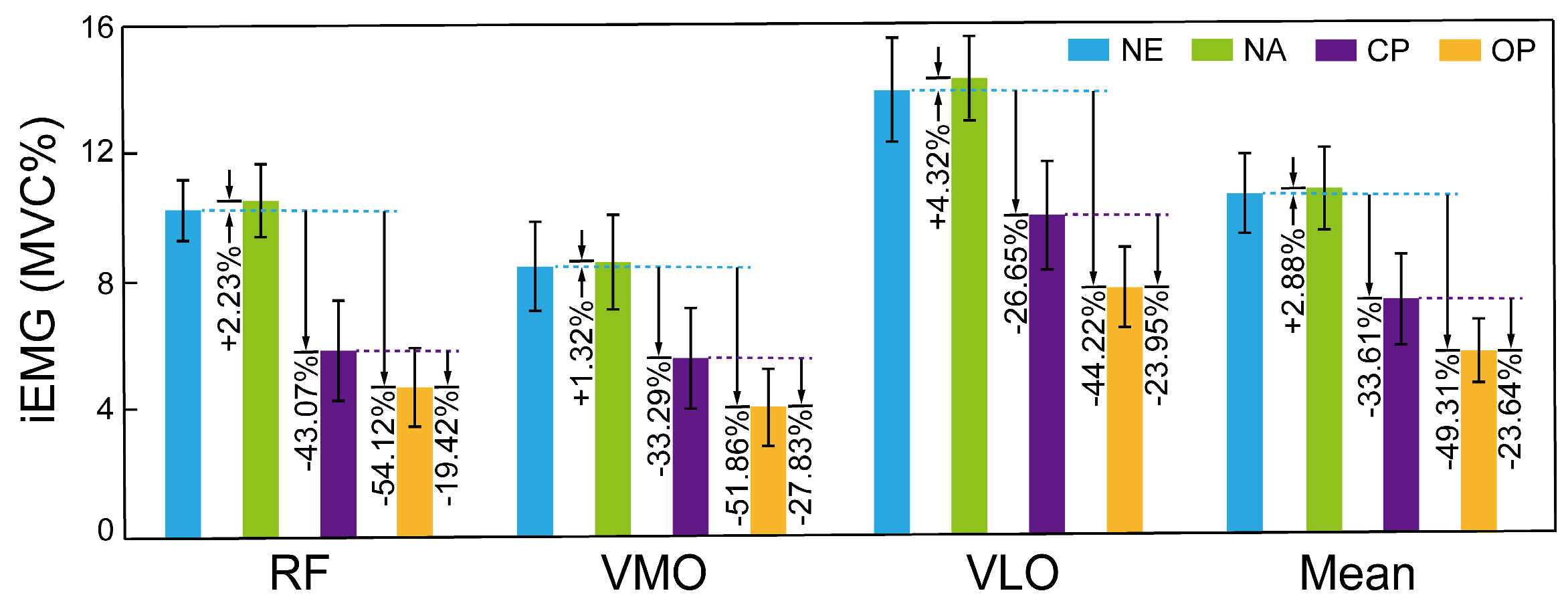
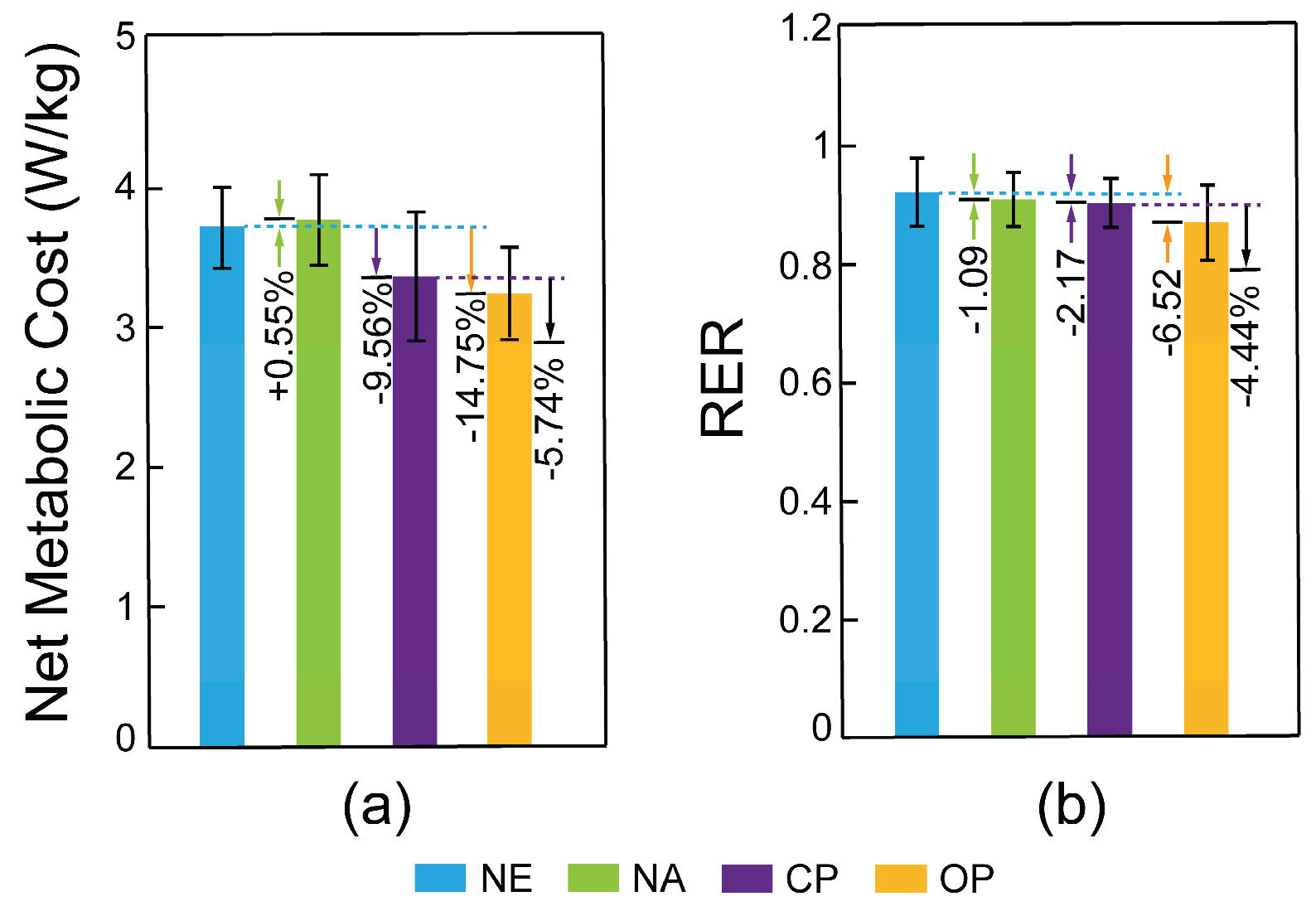
3.3. Electromyographic and Metabolic Effect of the Assistance
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PDFM | Physics-guided Dynamic Fusion Model |
| PINN | Physics-informed Neural Networks |
| N–E | Newton–Euler |
| EMG | Electromyography |
| GRF | Ground Reaction Forces |
| IMUs | Inertial Measurement Units |
| LSTM | Long Short-term Memory |
| RMSE | Root Mean Square Error |
| NTM | Neural Turing Machines |
| sEMG | surface Electromyography |
| iEMG | integrated Electromyography |
| RF | Rectus Femoris |
| VMO | Vastus Medialis Obliquus |
| VLO | Vastus Lateralis Obliquus |
| MVC | Maximum Voluntary Contraction |
| NE | No-exo |
| NA | Non-assisted |
| CP | Conventional Profile |
| OP | Optimized Profile |
| BPNN | Backpropagation Neural Network |
| FNN | Feedforward Neural Network |
| RNN | Recurrent Neural Network |
| LOSO | Leave-one-subject-out |
| FCCM | Fusion Coefficient Calibration Method |
| SSV | Stratified Safety Validation |
| GP | Gaussian Process |
| ANOVA | One-way Repeated-measures Analysis of Variance |
| RER | Respiratory Exchange Ratio |
Appendix A
| Participant No. | Sex | Age (years) | Height (m) | Body mass (kg) |
|---|---|---|---|---|
| 1 | M | 25 | 1.78 | 72.6 |
| 2 | M | 26 | 1.72 | 68.3 |
| 3 | M | 25 | 1.83 | 78.6 |
| 4 | M | 27 | 1.86 | 80.4 |
| 5 | M | 25 | 1.85 | 65.1 |
| 6 | M | 31 | 1.75 | 78.2 |
| 7 | M | 26 | 1.69 | 65.5 |
| 8 | M | 28 | 1.83 | 84.4 |
| 9 | F | 32 | 1.68 | 62.1 |
| 10 | F | 34 | 1.67 | 71.4 |
| 11 | F | 27 | 1.72 | 63.4 |
| 12 | F | 31 | 1.57 | 56.5 |
| 13 | F | 28 | 1.69 | 60.6 |
| Mean ± SD | 8M/5F | 28.08 ± 3.12 | 1.74 ± 0.09 | 69.78 ± 8.56 |
| Model | Hyperparameters & Training Details |
|---|---|
| BPNN | • Architecture: Input + FC + Output • Hidden layers: 3 FC layers (64-128-64 units) • Activation: ReLU • Optimizer: Adam • Learning rate: 5×10-5 • Regularization: L2 penalty (λ=1×10-5) • Batch size: 16 |
| FNN | • Architecture: Input + FNN + Output • Hidden layers: 2 FC layers (128-64 units) • Activation: ReLU • Optimizer: Adam • Learning rate: 5×10-5 • Regularization: L2 penalty (λ=1×10-5) • Batch size: 16 |
| LSTM | • Architecture: Input + LSTM + FC (linear) + Output • LSTM hidden units: 128 • FC layer units: 3 units • Optimizer: Adam • Learning rate: 5×10-5 • Dropout: 0.4 • Batch size: 16 |
| RNN | • Architecture: Input + RNN + FC + Output • Hidden units: 128 • FC layer units: 3 • Optimizer: Adam • Learning rate: 5×10-5 • Dropout: 0.3 • Batch size: 16 |
| NTM | • Architecture: Input + NTM + FC (linear) + Output • Controller: LSTM (128 hidden units) • Memory matrix: 128 × 40 (content-addressable) • FC layer units: 3 units • Read/Write heads: 1 head • Optimizer: Adam • Learning rate: 5×10-5 • Batch size: 16 |
References
- Kim, E; Lee, S.H.; Kim, D; Yu, J; Lee, H.J.; Kim, Y.H. A wearable hip exoskeleton for anaerobic exercise in healthy adults. Sci Rep 2024, 14, 26424. [Google Scholar] [CrossRef]
- Lee, D; Lee, S.H.; Young, A.J. AI-driven universal lower-limb exoskeleton system for community ambulation. Sci Adv 2024, 10, eadq0288. [Google Scholar] [CrossRef]
- Kim, H.S.; Park, J.H.; Lee, H.S.; Lee, J.Y.; Jung, J.W.; Park, S.B.; Hyun, D.J.; Park, S.; Yoon, J.; Lim, H. Effects of wearable powered exoskeletal training on functional mobility, physiological health and quality of life in non-ambulatory spinal cord injury patients. J Korean Med Sci 2021, 36, e80. [Google Scholar] [CrossRef]
- Chen, W.B.; Wu, S.; Zhou, T.C.; Xiong, C.H. On the biological mechanics and energetics of the hip joint muscle-tendon system assisted by passive hip exoskeleton. Bioinspir Biomim 2019, 14, 016012. [Google Scholar] [CrossRef]
- Hao, M.; Chen, K.; Fu, C.L. Effects of hip torque during step-to-step transition on center-of-mass dynamics during human walking examined with numerical simulation. J Biomech 2019, 90, 33–39. [Google Scholar] [CrossRef]
- Ishmael, M.K.; Gunnell, A.; Pruyn, K.; Creveling, S.; Hunt, G.; Hood, S.; Archangeli, D.; Foreman, K.B.; Lenzi, T. Powered hip exoskeleton reduces residual hip effort without affecting kinematics and balance in individuals with above-knee amputations during walking. IEEE Trans Biomed Eng 2023, 70, 1162–1171. [Google Scholar] [CrossRef]
- Lee, H.J.; Lee, S.; Chang, W.H.; Seo, K.; Shim, Y.; Choi, B.O.; Ryu, G.H.; Kim, Y.H. A wearable hip assist robot can improve gait function and cardiopulmonary metabolic efficiency in elderly adults. IEEE Trans Neural Syst Rehabil Eng 2017, 25, 1549–1557. [Google Scholar] [CrossRef] [PubMed]
- Gordon, D.F.N.; McGreavy, C.; Christou, A.; Vijayakumar, S. Human-in-the-loop optimization of exoskeleton assistance via online simulation of metabolic cost. IEEE Trans Robot 2022, 38, 1410–1429. [Google Scholar] [CrossRef]
- Zhou, T.C.; Xiong, C.H.; Zhang, J.J.; Hu, D.; Chen, W.B.; Huang, X.L. Reducing the metabolic energy of walking and running using an unpowered hip exoskeleton. J Neuroeng Rehabil 2021, 18, 95. [Google Scholar] [CrossRef] [PubMed]
- Young, A.J.; Gannon, H.; Ferris, D.P. A biomechanical comparison of proportional electromyography control to biological torque control using a powered hip exoskeleton. Front Bioeng Biotechnol 2017, 5, 37. [Google Scholar] [CrossRef]
- Farris, D.J.; Robertson, B.D.; Sawicki, G.S. Elastic ankle exoskeletons reduce soleus muscle force but not work in human hopping. J Appl Physiol 2013, 115, 579–585. [Google Scholar] [CrossRef]
- Pandy, M.G.; Berme, N. A numerical method for simulating the dynamics of human walking. J Biomech 1988, 21, 1043–1051. [Google Scholar] [CrossRef] [PubMed]
- Gui, K.; Liu, H.H.; Zhang, D.G. A practical and adaptive method to achieve EMG-based torque estimation for a robotic exoskeleton. IEEE-ASME Trans Mechatron 2019, 24, 483–494. [Google Scholar] [CrossRef]
- Maideen, A.; Mohanarathinam, A. Deep learning-based torque and joint angle estimation from electromyography signals: a hybrid DNN-LSTM approach. KSII Trans Internet Inf Syst 2025, 19, 2529–2547. [Google Scholar]
- Zhang, L.B.; Soselia, D.; Wang, R.L.; Gutierrez-Farewik, E.M. Lower-limb joint torque prediction using LSTM neural networks and transfer learning. IEEE Trans Neural Syst Rehabil Eng 2022, 30, 600–609. [Google Scholar] [CrossRef]
- Zhang, L.B.; Soselia, D.; Wang, R.L.; Gutierrez-Farewik, E.M. Estimation of joint torque by EMG-driven neuromusculoskeletal models and LSTM networks. IEEE Trans Neural Syst Rehabil Eng 2023, 31, 3722–3731. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.S.; Chen, Z.L.; Zhan, H.R.; Zhang, J.Y.; Wu, X.L.; Jiang, D.; Guo, Q. Lower limb joint torque prediction using long short-term memory network and gaussian process regression. Sensors 2023, 23, 9576. [Google Scholar] [CrossRef]
- Kim, J.; Quinlivan, B.T.; Deprey, L.A.; Revi, D.A.; Eckert-Erdheim, A.; Murphy, P.; Orzel, D.; Walsh, C.J. Reducing the energy cost of walking with low assistance levels through optimized hip flexion assistance from a soft exosuit. Sci Rep 2022, 12, 11004. [Google Scholar] [CrossRef]
- Witte, K.A.; Fiers, P.; Sheets, A.L.; Collins, S.H. Improving the energy economy of human running with powered and unpowered ankle exoskeleton assistance. Sci Robot 2020, 5, eaay9108. [Google Scholar] [CrossRef]
- Zhang, B.; Jiang, W.Z.; Tan, X.W.; Su, J.H.; Zhao, Y.W.; Zhao, X.A. Modular soft exoskeleton design and control for assisting movements in multiple lower limb joint configurations. IEEE Trans Autom Sci Eng 2025, 22, 20799–20813. [Google Scholar] [CrossRef]
- Guo, S.; Xiang, Q.; Hashimoto, K.; Jin, S. Assistive force of a belt-type hip assist suit for lifting the swing leg during walking. In Proceedings of the 2020 IEEE International Conference on Robotics and Automation (ICRA), France, 31 May 2020 - 31 August 2020. [Google Scholar]
- Molina-Rueda, F.; Fernández-González, P.; Cuesta-Gómez, A.; Koutsou, A.; Carratalá-Tejada, M.; Miangolarra-Page, J.C. Test-retest reliability of a conventional gait model for registering joint angles during initial contact and toe-off in healthy subjects. Int J Environ Res Public Health 2021, 18, 1343. [Google Scholar] [CrossRef]
- Xiang, Q.; Wang, J.X.; Liu, Y.; Guo, S.J.; Liu, L. Gait recognition and assistance parameter prediction determination based on kinematic information measured by inertial measurement units. Bioengineering-Basel 2024, 11, 275. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Lu, W.; Wang, W.; Wang, H.; Xue, M.M.; Zhang, X.Y. A soft hip exoskeleton biomimetic assistance method incorporating dual-pretension and biomechanics-based force modeling. Med Biol Eng Comput 2026, PMID 7704869. [Google Scholar] [CrossRef] [PubMed]
- Winter, D.A. Human balance and posture control during standing and walking. Gait Posture 1995, 3, 193–214. [Google Scholar] [CrossRef]
- Neptune, R.R.; McGowan, C.P. Muscle contributions to whole-body sagittal plane angular momentum during walking. J Biomech 2011, 44, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Romijn, J.A.; Coyle, E.F.; Sidossis, L.S.; Gastaldelli, A.; Horowitz, J.F.; Endert, E.; Wolfe, R.R. Regulation of endogenous fat and carbohydrate metabolism in relation to exercise intensity and duration. Am J Physiol 1993, 265, E380–E391. [Google Scholar] [CrossRef]
| Assistance Function Order | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| rRMSE | 21.8% | 7.49% | 2.58% | 0.53% | 0.19% | 0.18% | 0.18% | 0.17% |
| R2 | 0.554 | 0.949 | 0.994 | 0.999 | 1 | 1 | 1 | 1 |
| Model | R | RMSE (N·m/kg) | Correlation Coefficient | |||||||
| Sag. | Cor. | Trans. | Sag. | Cor. | Trans. | Sag. | Cor. | Trans. | ||
| BPNN | 88.31% (6.2%) |
65.22% (8.2%) |
67.53% (10.5%) |
0.242 (0.072) |
0.161 (0.037) |
0.045 (0.018) |
0.867 (0.046) |
0.813 (0.033) |
0.817 (0.032) |
|
| FNN | 86.77% (5.4%) |
64.35% (5.6%) |
68.24% (9.7%) |
0.263 (0.081) |
0.189 (0.049) |
0.053 (0.019) |
0.871 (0.032) |
0.801 (0.049) |
0.767 (0.072) |
|
| LSTM | 89.64% (7.7%) |
71.23% (7.5%) |
74.29% (10.2%) |
0.181 (0.052) |
0.175 (0.036) |
0.047 (0.016) |
0.879 (0.036) |
0.895 (0.032) |
0.871 (0.033) |
|
| NTM | 84.72% (8.7%) |
85.46% (8.1%) |
86.44% (7.1%) |
0.242 (0.062) |
0.149 (0.033) |
0.032 (0.018) |
0.889 (0.035) |
0.904 (0.031) |
0.915 (0.038) |
|
| RNN | 89.26% (5.6%) |
66.71% (15.8%) |
66.72% (9.2%) |
0.191 (0.048) |
0.177 (0.048) |
0.041 (0.015) |
0.891 (0.034) |
0.849 (0.048) |
0.787 (0.065) |
|
| N–E | 85.91% (4.6%) |
88.43% (4.2%) |
79.06% (9.7%) |
0.175 (0.043) |
0.118 (0.023) |
0.034 (0.012) |
0.935 (0.031) |
0.939 (0.022) |
0.897 (0.032) |
|
| PDFM | 92.51% (4.1%) |
86.86% (5.1%) |
88.15% (6.1%) |
0.157 (0.041) |
0.126 (0.026) |
0.028 (0.012) |
0.938 (0.028) |
0.924 (0.024) |
0.929 (0.029) |
|
| Dimensions | Initial contact | Loading response | Mid stance | TerminalStance | Pre-swing | Swing | ||||||||||
| wLSTM | kNTM | wLSTM | kNTM | wLSTM | kNTM | wLSTM | kNTM | wLSTM | kNTM | wN–E | kNTM | |||||
| Sag. | 0.06 (0.02) |
0.71 (0.05) |
0.88 (0.09) |
0.46 (0.07) |
0.29 (0.11) |
0.93 (0.14) |
0.95 (0.17) |
0.44 (0.11) |
0.41 (0.06) |
0.62 (0.13) |
0.89 (0.09) |
0.25 (0.08) |
||||
| Cor. | 0.22 (0.05) |
0.92 (0.17) |
0.07 (0.02) |
0.74 (0.07) |
0.77 (0.08) |
0.12 (0.06) |
0.85 (0.09) |
0.37 (0.04) |
0.22 (0.05) |
0.91 (0.07) |
0.61 (0.12) |
0.28 (0.03) |
||||
| Trans. | 0.11 (0.04) |
0.69 (0.08) |
0.05 (0.04) |
0.98 (0.16) |
0.09 (0.04) |
0.85 (0.12) |
0.13 (0.05) |
0.56 (0.13) |
0.07 (0.02) |
0.86 (0.06) |
0.62 (0.14) |
0.87 (0.07) |
||||
| Condition | RF (%) | VMO (%) | VLO (%) | Mean (%) | Metabolic Cost (W/kg) |
RER |
|---|---|---|---|---|---|---|
| NE | 10.31 (1.35) |
8.35 (1.42) |
13.66 (1.63) |
10.77 (1.19) |
3.66 (0.28) |
0.92 (0.05) |
| NA | 10.54 (1.13) |
8.46 (1.49) |
14.25 (1.32) |
11.08 (1.26) |
3.68 (0.32) |
0.91 (0.04) |
| CP | 5.87 (1.58) |
5.57 (1.58) |
10.02 (1.71) |
7.15 (1.37) |
3.31 (0.44) |
0.90 (0.04) |
| OP | 4.73 (1.19) |
4.02 (1.22) |
7.62 (1.22) |
5.46 (1.01) |
3.12 (0.36) |
0.86 (0.06) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).